Abstract
Burkholderia pseudomallei is the cause of melioidosis in humans and other animals. Disease occurs predominately in Asia and Australia. It is rare in North America, and affected people and animals typically have a history of travel to (in human cases) or importation from (in animal cases) endemic areas. We describe the gross and histopathologic features and the microbiologic, molecular, and immunohistochemical diagnoses of a case of acute meningoencephalomyelitis and focal pneumonia caused by B. pseudomallei infection in a pigtail macaque that was imported from Indonesia to the United States for research purposes. This bacterium has been classified as a Tier 1 overlap select agent and toxin; therefore, recognition of pathologic features, along with accurate and timely confirmatory diagnostic testing, in naturally infected research animals is imperative to protect animals and personnel in the laboratory animal setting.
Melioidosis is a multisystemic disease caused by the Gram-negative bacterium Burkholderia pseudomallei. The disease is endemic in parts of Southeast Asia, northern Australia, and the Indian subcontinent; sporadic cases have also been reported from Central and South America. The bacterium is a facultative anaerobe found in soil and water in these regions. It readily infects humans and many other animals, resulting in a wide range of outcomes, from asymptomatic infection to disseminated fatal disease in up to 40% of cases. 28 Infection may occur through a variety of means, including inhalation, contamination of skin wounds, or accidental ingestion from the environment. 5,17,18,25 Direct transmission from infected to naive individuals and sexual and vertical transmission are considered rare. 1,6,7,18,24,27 The incubation period is highly variable and may be days to decades long. Longer incubations are often associated with subclinical infections, with fulminant disease developing during periods of host immunosuppression. 2,12,17,19,28
Clinical signs of melioidosis in all species are variable and depend on the route and dose of infection, virulence of the infecting bacterial strain, and perhaps most importantly, the immune status of the host and presence of concurrent chronic diseases. 6,11,12,28 The most common disease manifestations in people are pneumonia and septicemia with systemic abscess formation. Localized cutaneous abscesses at the site of bacterial inoculation, meningoencephalitis, osteomyelitis, and arthritis also occur commonly. 12 In nonhumans, the disease is best described in ruminants and swine. As with humans, the clinical signs vary depending on the site(s), severity, and stage of infection. Overall, they are similar to those in humans and are clinically indistinguishable from other disease entities, particularly bacterial septicemias. 24 Characteristic but nonspecific pathologic findings include abscesses with suppurative or caseous exudates in multiple tissues. 20,24,25
Asian macaques are widely imported from melioidosis-endemic regions for use in biomedical research; therefore, subclinically infected macaques may represent a possible vector for the spread of this potentially deadly bacterium into research facilities in nonendemic areas. Given the bacterium's potential for rapid and easy transmission among many different species in contaminated environments, introduction of a single infected animal could have potentially devastating effects on an animal colony. Furthermore, it could result in introduction into the human population through interactions of animal caretakers with infected animals and/or their contaminated environments if personal protective equipment is not used.
Because the clinical presentation of melioidosis is highly variable and may mimic other infectious diseases, recognizing the characteristic pathology and following up with appropriate diagnostic testing at the time of necropsy are essential in identifying this disease within research animal colonies. Herein, we describe the gross and histopathologic features, along with the microbiologic, molecular, and immunohistochemical diagnoses, of B. pseudomallei meningoencephalomyelitis and pneumonia in a captive-bred pigtail macaque imported to the United States from Indonesia for research purposes.
Case Report
History
An adult female pigtail macaque (Macaca nemestrina) was humanely euthanized after a 2-day course of sudden-onset neurologic signs involving multiple cranial nerve deficits and generalized tremors and stiffness, which progressed despite supportive care. Clinical signs referable to organ systems other than the central nervous system were not observed. The animal had been captive-bred in Indonesia and imported via the Philippines to the United States for research use approximately 6 months prior to presentation. She had not yet been actively involved in any research studies at the time of euthanasia. Of note, at the time of introduction to the facility, she had a cutaneous abscess over the right stifle. Bacterial culture revealed coagulase-negative Staphylococcus sp and a Gram-negative bacillus that could not be identified. The abscess resolved with antibiotic treatment.
Necropsy
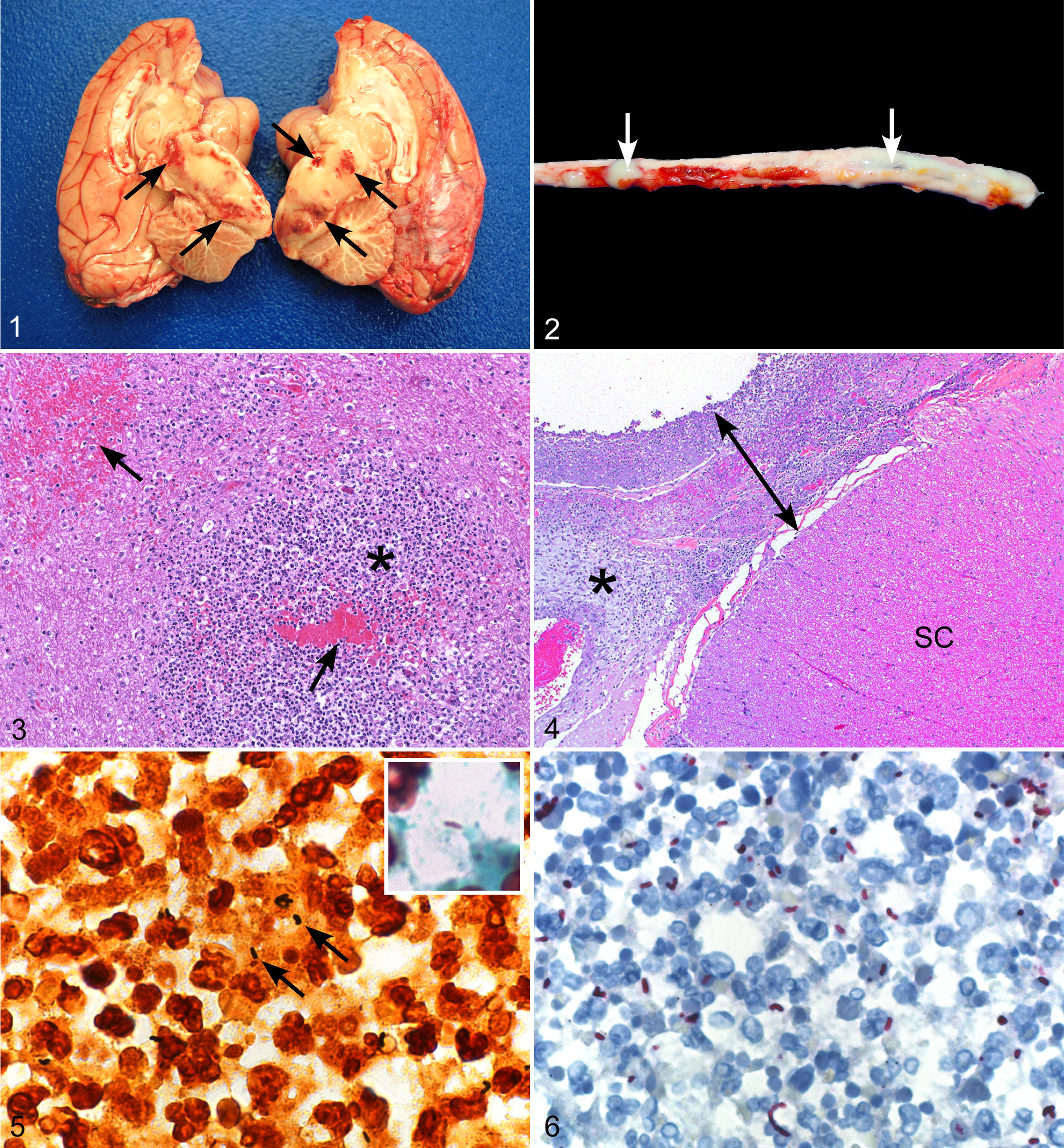
Gross findings were limited to the central nervous system and lungs. The meninges over the parietal and occipital lobes of the cerebrum were congested. Suppurative exudate was present between the ventral surface of the right rostral brainstem and underlying sphenoid bone. The brainstem, pons, and midbrain contained multiple poorly demarcated regions of malacia, cavitation, and yellow discoloration with scattered foci of hemorrhage (Fig. 1). The thoracolumbar spinal meninges were diffusely expanded by a highly viscous, tan suppurative exudate (Fig. 2). A 1-cm area of the right cranioventral lung lobe was firmly adhered to the thoracic wall; a small amount of suppurative exudate oozed from the cut surface. At the time of necropsy, no evidence of the previous stifle abscess remained, and all other tissues were grossly unremarkable. A complete set of tissues was collected and fixed in 10% neutral buffered formalin at room temperature for 7 days.
Histopathology
Tissues were processed for routine paraffin histology, sectioned at a thickness of 4 microns, and routinely stained with hematoxylin-eosin, Warthin-Starry, and Lillie-Twort Gram stains.
Histopathologic evaluation of the brain revealed bilateral lesions in the brainstem, pons, midbrain, and, to a lesser extent, thalamus. The cerebral cortex was spared. Lesions consisted of variably defined, nonencapsulated abscesses and microabscesses comprising necrotic foci with dense neutrophilic infiltrates. Acute hemorrhage was also multifocally present (Fig. 3). In multiple sections of thoracic and lumbar spinal cord, the leptomeninges were markedly thickened by a neutrophilic and lymphoplasmacytic infiltrate mixed with fibrinonecrotic debris and granulation tissue. The spinal cord itself was spared of notable inflammation or degenerative changes (Fig. 4). In distal lumbar sections, the inflammatory exudate extensively involved spinal nerves and those of the cauda equina and extended out into the epidural adipose tissue. In a section from the right cranioventral lung lobe from the site of gross pleural adhesion, there was a well-defined, nonencapsulated focus of acute pneumonia characterized by destruction of alveolar walls and pooling of viable and degenerate neutrophils, cellular debris, and hemorrhage. Numerous microabscesses with similar features to those in the brain were scattered throughout multiple sections of liver. Other than mild lymphoplasmacytic enterocolitis, other tissues (heart, thyroid gland, spleen, adrenal gland, kidney, urinary bladder, ovary, uterus, and stomach) were histologically unremarkable.
Warthin-Starry stain revealed numerous bacilli in the pneumonic lung, as well as moderate and low numbers of bacilli within the thoracolumbar meningeal exudate and microabscesses of the brain, respectively (Fig. 5). Bacilli were weakly Gram negative (Fig. 5, inset). Bacteria were not identified with special stains in liver sections.
Bacterial Culture
Swabs from brainstem abscesses and spinal meningeal exudates were submitted to the Georgia Laboratory Animal Diagnostic Service (GLADS) for bacterial aerobic culture. Inoculations were carried out in a biosafety cabinet. Blood and MacConkey agars (Remel; Thermo Scientific, Lenexa, KS) were inoculated and incubated at 35 ± 5°C overnight in a tight-sealed container. Pure growth of a non–lactose-fermenting, oxidase-positive, Gram-negative rod was observed. Identification was attempted using the BIOLOG (Gen III) system, which yielded an identification of Burkholderia ambifaria, 63% probability. This was considered below the limit of acceptability, and the isolate was recommended for 16S rRNA sequencing for definitive identification.
Polymerase Chain Reaction
Bacterial identification through nucleic acid sequencing is routinely carried out at GLADS, where 1500 bp of the 16S ribosomal RNA are amplified and sequenced using primers described by Relman. 23 DNA was extracted from whole-cell suspensions with the MoBio Ultraclean Microbial DNA Kit (MoBio, Carlsbad, CA). Each polymerase chain reaction (PCR; 10 μl total) contained 2 mM MgCl2 PCR buffer (Idaho Technologies, Salt Lake City, UT), 0.05U Taq polymerase (Roche, Pleasanton, CA), 0.2 mM dNTPs (Roche), and 0.1 μM of each primer. The PCR product was purified (Millipore Microcon, Billerica, MA) and sequencing was performed by Seqwrite (Houston, TX). A contiguous sequence of 1391 bp was generated and assembled using Sequencher 5.0 (Gene Codes Corporation, Ann Arbor, MI). The resulting sequence was found to have >99% similarity with B. pseudomallei (BLAST; National Center for Biotechnology Information, Bethesda, MD).
Immunohistochemistry
Specimens were evaluated by immunohistochemistry using 2 rabbit polyclonal anti–B. pseudomallei antibodies: one raised against the whole organism and one raised against the bacterial flagellum (National Kaohsiung Normal University, Kaohsiung City, Taiwan). Immunohistochemical testing was performed using an alkaline phosphatase (AP) polymer detection system (Thermo Scientific). Four-micron sections of formalin-fixed paraffin embedded tissue were deparaffinized and rehydrated using gradations (100%, 95%, and 70%) of ethanol. Proteinase K (Roche) digestion was performed for 15 minutes at room temperature (RT), after which Ultra V Block (Thermo Scientific) was applied for 10 minutes at RT. Tissues were then incubated with the rabbit polyclonal anti–B. pseudomallei antibody (30 minutes at RT), followed by Primary Antibody Enhancer (Thermo Scientific; 10 minutes at RT), AP Polymer (Thermo Scientific; 15 minutes at RT), and Naphthol Phosphate Substrate/Fast Red Tablet (Thermo Scientific; 20 minutes at RT). Sections were counterstained in Mayer’s Modified Hematoxylin (Poly Scientific, Bay Shore, NY). Appropriate positive and negative controls were run in parallel. Tissues from previously diagnosed cases of human melioidosis were used as positive controls. For negative controls, the primary antibodies were replaced with normal rabbit serum (Centers for Disease Control and Prevention, Atlanta, GA).
For both anti–B. pseudomallei antibodies, extensive labeling of bacilli was observed within the spinal meningeal exudate, brain abscesses (Fig. 6), and the pneumonic lung. Scattered bacterial labeling was also seen within necrotic foci in the liver. Bacilli were not identified by immunohistochemistry in sections of spleen, kidney, adrenal gland, stomach, or intestine.
Discussion
Melioidosis has historically been identified in the tropical regions of Southeast Asia, northern Australia, the Indian subcontinent, and, to a lesser extent, Central and South America, but more recently has emerged in regions peripheral and distant to these areas. 8,17,25 The causative bacterium, B. pseudomallei, has been classified as a Tier 1 overlap select agent and toxin (formerly a category B bioterrorism agent) by the US Department of Health and Human Services, because of its intrinsic antibiotic resistance and potential for easy dissemination with high mortality among humans and many other animal species. 6,22
Because Asian macaques are extensively used in biomedical research, they represent a potential portal of entry for melioidosis into nonendemic areas. Several cases of melioidosis have been reported in macaques and other nonhuman primate species associated with such facilities. These include rhesus macaques, pigtail macaques, stumptail macaques, a chimpanzee, and an orangutan. Among these species, infections have been associated with fight or puncture wounds or with surgical implantation of foreign materials during research procedures and have resulted in cutaneous and visceral abscesses, lymphadenitis, pneumonia, septicemia, and osteomyelitis. Disease was recognized in these animals ranging from 3 days to 10 years after importation. 3,4,13 –15,19,21,26 The animal in this report had a stifle abscess at the time of arrival to our facility. Although Burkholderia was not definitively identified from this lesion, this may have been the initial site of infection, with progression to central nervous system and lung involvement 6 months later.
To the authors’ knowledge, this is the first report of naturally acquired melioidosis predominately involving the central nervous system in a research monkey in the United States. In Australia but rarely in other endemic areas, 3% to 5% of human melioidosis cases have neurologic disease, which manifests as cranial nerve deficits and/or myelitis with peripheral motor weakness. 9,10,27,28 It is these patients who are at highest risk of fatal infection. 17 Lesions in such cases include meningoencephalitis with microabscesses in the brainstem and spinal cord, similar to those observed in this macaque. 6,17 Neurologic melioidosis primarily involving the brainstem has also been observed in naturally infected horses and experimentally infected mice and sheep. 9
Because animal melioidosis is almost never encountered in the United States, it was not initially considered in the differential for this animal’s clinical disease or pathologic findings. Routine laboratory testing initiated at the time of necropsy ultimately led to identification of the organism. This highlights the fact that lesions of melioidosis are nonspecific and cannot be distinguished clinically or by gross or histopathology from those of other bacterial infections. Identification of B. pseudomallei within lesions is therefore imperative for definitive diagnosis. The diagnosis was confirmed in this macaque by culture, 16S rRNA sequencing, and immunohistochemistry. Samples for culture should be handled under biosafety level 3 conditions when Burkholderia is suspected. 25 Furthermore, any fresh biologic sample from nonhuman primates should always be handled in a biosafety cabinet since these animals could be carriers of many other zoonotic diseases that may not be suspected at the time of sample submission. B. pseudomallei can reliably be isolated from abscess material and may also be cultured from feces or swabs from the throat, sputum, blood, or urine. 25 It grows readily on routine culture media within 24 to 48 hours at 37°C and selectively on Ashdown’s medium, which contains crystal violet, glycerol, and gentamicin. 25,27
Although biochemical tests and colony and cell morphology are used for diagnosis in countries where melioidosis is endemic, B. pseudomallei is not present in the identification database of most bacterial identification systems in nonendemic regions. Therefore, in countries where its isolation is very rare, reliance on such methods may result in misidentification of the organism if not isolated in a laboratory with appropriate expertise and knowledge of the agent. A complete history and country of origin for the submitted sample are crucial to aid the laboratory in its diagnosis.
16S rRNA sequencing is an effective way to determine bacterial genus and, in many cases, bacterial species and was used to confirm the identity of the causative agent in this case. As sequencing technology has become more economical and more widely accessible, its use has been increasing. Sequencing of nearly the full 16S ribosomal RNA is a reliable method to distinguish among the most common Burkholderia spp. 16 Immunohistochemistry using anti–B. pseudomallei–specific antibodies provides another means of diagnosis in formalin-fixed paraffin embedded tissues; however, because highly sensitive and specific antibodies are limited in their commercial availability, this testing often can be performed only at specialized reference laboratories. 25
In conclusion, B. pseudomallei is an uncommon infection outside of endemic areas and is essentially always associated with travel to (in human cases) or importation from (in animal cases) endemic areas. Because nonhuman primates used in biomedical research are frequently imported from Asia, melioidosis should be considered in the differential diagnosis for these animals when they present with 1 or more abscesses or with nonspecific symptoms consistent with infectious disease, regardless of the time frame since importation. Necropsy and histopathology, coupled with microbiological, molecular, and immunohistochemical assays, aid in the rapid diagnosis of melioidosis and therefore can facilitate prompt implementation of appropriate biosecurity measures regarding reporting, containment, and elimination of the infection in research animal colonies.
Footnotes
Acknowledgements
We thank Dr Robyn Engel and Dr Sharon Dietz of the Animal Resources Branch (ARB), Division of Scientific Resources, Centers for Disease Control and Prevention (CDC), for their assistance during necropsy, as well as Dr Crystal Johnson and Dr Brianna Skinner of ARB for providing information about this animal’s clinical signs and veterinary care. We thank Dr Wun-Ju Shieh, Dr Christopher Paddock, and Dr Atis Muehlenbachs of IDPB for collaboration on the histopathologic evaluation of the case, as well as Dr Ya-Lei Chen, Professor, Department of Microbiology at National Kaohsiung Normal University, Kaohsiung, Taiwan, for providing the primary antibodies used for immunohistochemistry. We thank Dr Henry Walke, Dr David Blaney, Dr Alex Hoffmaster, and Mindy Glass Elrod from the Bacterial Special Pathogens Branch, Division of High-Consequence Pathogens and Pathology, CDC, for their expertise and guidance regarding biosafety issues concerning this case. We also thank the bacteriology and molecular biology staff at the Athens Veterinary Diagnostic Laboratory for their help and expertise.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
