Abstract
There is increasing evidence in some malignancies that the tumor clone is heterogeneous in regard to proliferation and differentiation. The cancer stem cell hypothesis implies that not all the cells in the tumor have the same capacity to proliferate and maintain the growth of the tumor. Only a relatively small fraction of cells in the tumor, termed cancer stem cells (CSCs), possess the ability to proliferate and self-renew extensively. In the past decade, several groups have reported the existence of a CSC population in different human brain tumors from both children and adults. We report here the identification of a CSC population from a Boxer dog with glioblastoma multiforme (GBM) that possesses a great capacity for proliferation, self-renewal, and differentiation. This cloned cell line is aneuploid, forms neurospheres in culture, possesses CSC markers, and reproduces the original dog GBM when inoculated into the nude mouse brain.
Keywords
Introduction
Brain tumor stem cells are resistant to conventional radiation and pharmacologic treatments. 2, 27 Development of more effective therapies for glial malignancies would be facilitated by the existence of in vitro and in vivo models that faithfully recapitulate the stem cell component of these lesions. The cancer stem cell hypothesis implies that tumors contain a minority of cells with stem-like properties, which have the ability to self-renew and sustain the growth of the tumor. 9 Tumor stem cells were first identified in hematopoietic malignancies and more recently were also demonstrated in various solid tumors, including cancers of the brain such as glioblastomas, medulloblastomas, and ependymomas. 2, 22– 26 Brain tumor stem cells in gliomas are defined by 1) the capacity to self-renew, 2) the ability to initiate brain tumors upon orthotopic implantation, and 3) multipotency; that is, the capacity to differentiate into cells with a neuronal, astrocytic, or oligodendroglial phenotype. 37, 50 However, multilineage differentiation is not a requirement of the cancer stem cell (CSC). It is more important that CSCs regenerate the cell types observed in the original tumor. 52 In addition, brain tumor stem cells are characterized by the expression of neural stem cell antigens and the ability to grow as nonadherent spheres termed “neurospheres” when cultured in the presence of epidermal growth factor (EGF) and fibroblast growth factor (FGF) under serum-free conditions. Thus, stem-like cells in brain tumors share many characteristics with normal neural stem cells, supporting the hypothesis that brain tumors can arise from neural stem or progenitor cells. 37, 50 Recent studies further show that glioblastoma cells cultured under neural stem cell conditions can display heterogeneous growth characteristics and molecular profiles, suggesting that they may either arise from different cell types or from similar cells that have acquired different genetic alterations. 3, 14 Whether CSCs arise from normal stem cells, progenitor cells, or differentiated cells is not known at the present time. Nevertheless, CSCs in gliomas express CD133 and nestin that mark neural and progenitor cells. 14, 45 The factors governing glioma cell differentiation and migratory potential, and therefore tumor phenotype, are not completely understood. On the basis of gene expression profiling and by neurobiologic criteria, 2 subtypes among human glioblastoma cultures established under neural stem cell conditions were identified. One cluster of CSCs expressed neurodevelopmental genes and displayed a full stem-like phenotype. The second cluster displayed an expression signature for extracellular matrix (ECM)–related genes and only a restricted stem-like phenotype, fulfilling only, in part, the criteria considered typical of glioblastoma stem cells. 16 Whether these differences reflect variations in stem-like cells present between individual glioblastomas and/or distinct emerging lineages remains to be established.
There is recent evidence generated from preliminary studies in solid tumors suggesting that CSCs reside in a niche. This hypothesis derives from reports on normal stem cells from various tissues, showing that stem cells exist within protective niches that are composed of a number of differentiated cell types. 13, 36 This cellular microenvironment provides direct cell contacts and secreted factors that maintain stem cells in a quiescent state. Histologic observations of mouse tissues suggest that neural stem cells lie within a vascular niche in which endothelial cells regulate stem cell self-renewal. 39 In gliomas, it is hypothesized that vascular endothelial cells provide such a niche for the glioma CSCs similar to the situation with normal neuronal stem cells. 8 Disruption of this niche by anti–vascular endothelial growth factor (VEGF) treatment resulted in depletion of the CD133+ cells in a mouse model. 8 If the notion that niches protect CSCs proves correct, then targeting these microenvironments could prove to be an efficient treatment for gliomas. However, the CSC niche hypothesis is new, and more evidence is needed to advance this field.
In veterinary medicine there is a considerable literature that describes the occurrence of spontaneous tumors of the central nervous system in animals. 1– 42 Astrocytoma, of which glioblastoma multiforme (GBM) is the most malignant form, is one of the most common neoplasms of the central nervous system in animals. Of the domesticated animal species, most examples are seen in dogs, with much lower occurrence in other species. 42, 43 Intracranial neoplasia occurs more frequently in dogs than in humans (14.5 per year per 100,000 canine population at risk compared with 4 to 5 per 100,000 humans). 15, 19
Canine intracranial glioblastoma occurs most commonly in brachycephalic breeds, in particular the Boxer and Boston Terrier. No sex predilection has been reported, and they are recognized with greater incidence in animals over 6 years of age. The neoplasm is most commonly located in the cerebrum and diencephalon. Clinical signs caused by astrocytomas may vary with tumor location, and neurologic deficits reflect direct nervous tissue involvement as well as secondary effects of peritumor edema, necrosis, hemorrhage, compression, herniation, or obstructive hydrocephalus. 43 Computed tomography (CT) and magnetic resonance imaging (MRI) are routinely available to veterinary practitioners for diagnosis and localization of intracranial lesions. CT characteristics of some canine brain tumors have been reported to be similar to those in humans. 46, 47
GBM, one of the most malignant and devastating forms of glioma, appears to have the highest incidence in dog. 43 The highest incidence (30%) of this tumor occurs in the Boxer. Despite progress in research on the molecular aspects of GBMs, the prognosis of these brain tumors continues to be dismal in humans and animal species. One reason for the lack of clinical advances is ignorance of the cellular origin of this disease combined with a lack of understanding of the mechanisms of glioma cell migration and dispersal, which results in delays in the application of molecular analyses to treatment and impairs anticipation of tumor biologic behavior.
In the present study we demonstrate that GBM isolated from a Boxer dog (D-GBM) has cells with phenotypic characteristics of CSCs. As a proof of concept, we characterized this cell line's morphophenotype and its specific stem cell markers such as nestin and CD133. CSCs demonstrated glial and neuronal differentiation when exposed to differentiation growth factors in vitro. An intracranial orthotopic model using nude mice was used for in vivo evaluation of D-GBM tumorigenicity. Immunohistochemistry results of nude mouse xenografts also demonstrated glial and neuronal differentiation. This is the first report demonstrating the presence of CSCs in dog GBM. Our data also demonstrate the similarity between human and dog GBM and emphasize the importance of studying dog spontaneous brain tumors in order to elucidate the mechanism(s) of tumor dispersal, postsurgical reoccurrence, and resistance to therapy.
Materials and Methods
Tumor specimen and primary tumor cultures
The primary tumor cells were isolated from an 8-year-old male Boxer dog that was diagnosed with an intracranial tumor by CT scan at the College of Veterinary Medicine and Biomedical Sciences at Texas A&M University. The owner refused any treatment or surgical removal of tumor and by his approval the dog was euthanatized and necropsy was performed immediately. The brain was removed, and an intracranial tumor obliterating the lateral ventricle and compressing the adjacent areas of the brain parenchyma was detected (Figs. 1,2). A frozen section from a tumor section stained with HE confirmed the diagnosis of GBM, Grade IV (WHO classification). Tumor tissues were excised and saved in sterile Petri dishes with Hank's balanced salt solution (Invitrogen, Carlsbad, CA). Samples were cut into 1-mm3 fragments and digested with 1 mg/ml collagenase/dispase (Worthington Biochemical, Lakewood, NJ) for 30 minutes at 37°C. Digested fragments were filtered through a 70-mm cell mesh (Sigma Aldrich, St. Louis, MO), and the isolated cells were seeded into T25 flasks at 5,000 cells/cm2. The culture medium (neurosphere medium) consisted of neurobasal medium (Invitrogen) with B27 supplement (20 ml/ml, Invitrogen), Glutamax (10 ml/ml, Invitrogen), fibroblast growth factor-2 (FGF-2, 20 ng/ml; Peprotech, Rocky Hill, NJ), and epidermal growth factor (EGF, 20 ng/ml, Peprotech). Growth factors were renewed twice weekly. Spheres were split by mechanical dissociation when they reached a size of 500 µm. Isolated tumor cells were also cultured in Dulbecco's modified Eagle medium (DMEM) supplemented with 20% horse serum, 100 IU/ml penicillin, and 100 mg/ml streptomycin. After the primary cell culture was established, some cells were maintained in DMEM supplemented with 10% fetal bovine serum (FBS) and some in neurobasal medium. In all experiments, cells were maintained at 37°C in a humidified incubator at 5% CO2. A cloned line was obtained by limiting dilution assay.
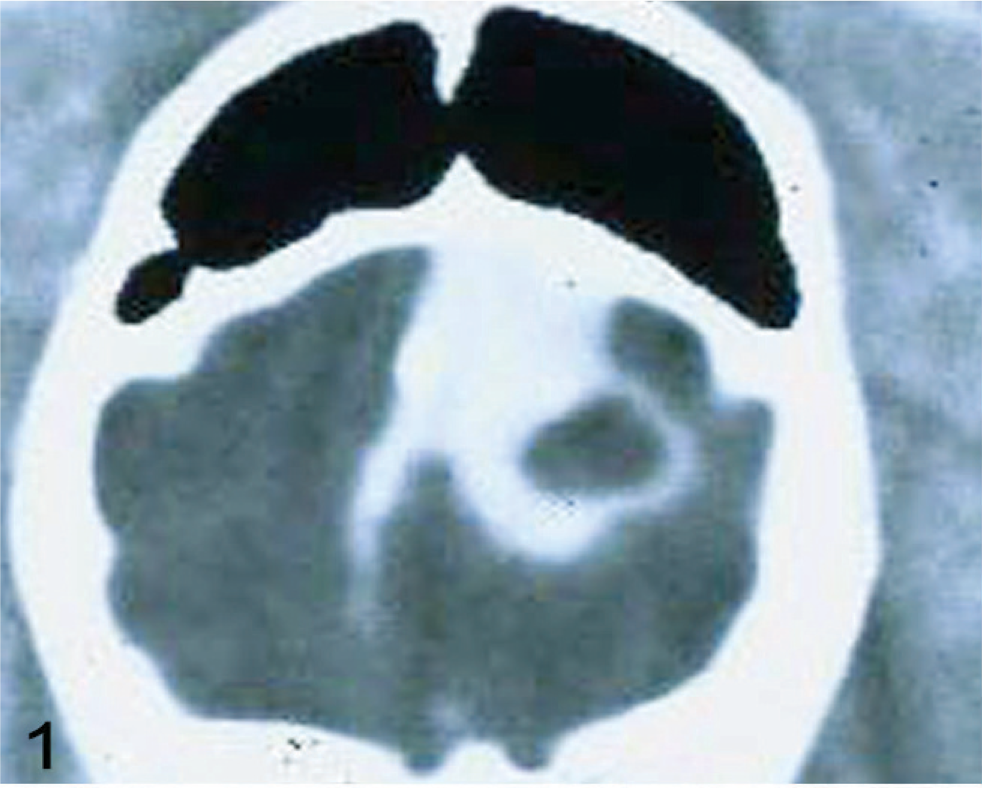
CT image of GBM from a Boxer dog.
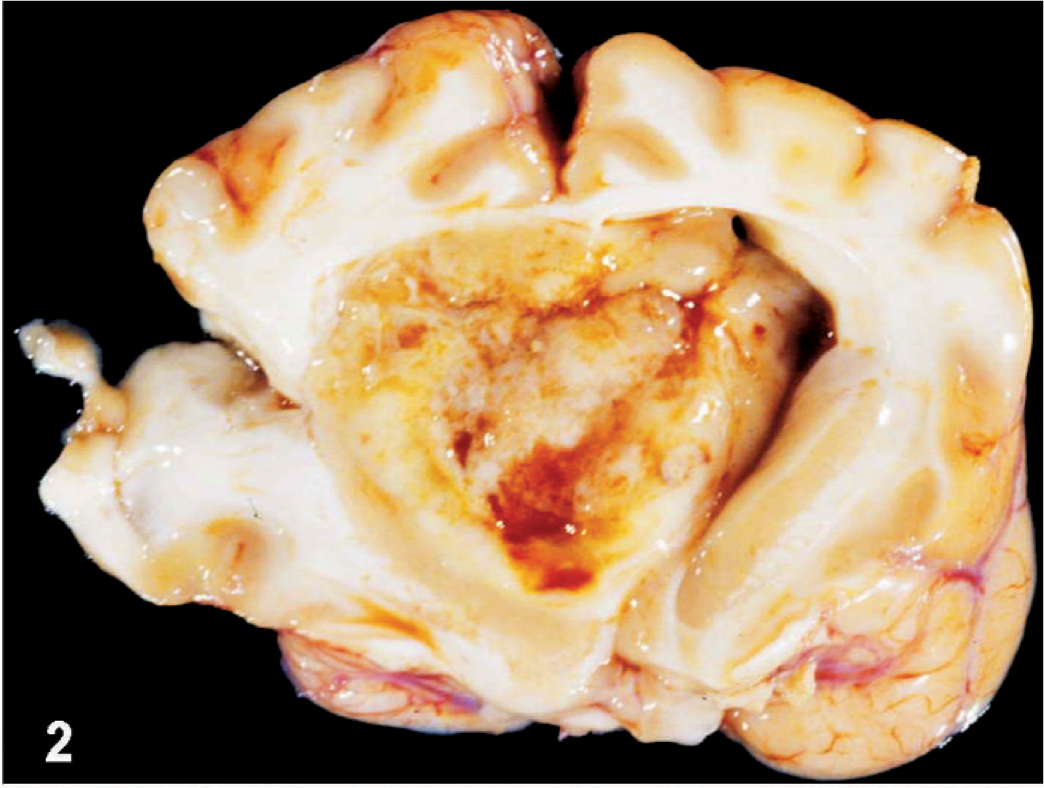
Gross appearance of an intraventricular GBM.
MRI diagnosed a second glioma at our institution from a 6-year-old female Boston Terrier in 2008. The dog was presented with clinical diagnosis of epileptic seizures and at the owner's request the dog was euthanatized. The necropsy was performed immediately postmortem and a well-circumscribed, 1.5-cm in diameter tumor was located in the right frontal lobe. A small fragment of the tumor was taken for tissue culture as described above. Histopathologic examination confirmed a pilocytic astrocytoma, PCA, (grade I, WHO classification). A primary culture consisting of pilocytic (piliform) astrocytes was obtained.
Flow cytometric analyses
Neurosphere and adherent D-GBM cells cultured in neurobasal and DMEM medium were collected and incubated with phycoerythrin (PE)-conjugated anti-mouse Prominin I (Prominin I, CD133, AC133) antibody (eBioscience, San Diego, CA) for 30 minutes at a concentration of 1 µg per million cells. Cells incubated in phosphate buffered saline (PBS) served as a negative control. Cell analysis for the expression of CD133 was performed using a BD FACSAria cell sorter (BD Biosciences, San Jose, CA). Forty-thousand cells were acquired with relevant controls using DIVA 6.0 (BD Biosciences) software to quantitatively estimate the percentage of cells expressing CD133 mAb. Postacquisition was performed using analytical software (Floro fo, Tree Star, Ashland, OR).
Assessment of self-renewal
Self-renewal is a critical feature of neural stem cells and CSCs. To demonstrate the self-renewal ability of glioblastoma cells in culture, single colony formation analysis was performed. The procedure was done as previously described. 53 In brief, the cells were harvested, and the suspension centrifuged at 1,000 × g for 5 minutes to collect the cells. The cell pellet was resuspended in fresh serum-containing medium. Cell viability was determined using a trypan blue exclusion assay, and the number of cells was determined using a hemocytometer. The formation of spheres or colonies from single cells was analyzed using a limiting dilution assay. Dissociated cells were seeded into 96-well plates at a theoretical density of 1 cell per well in neurosphere medium. After overnight culture, microscopic observation was used to identify wells that contained a single cell. Wells containing no cell or more than one cell were excluded, and those with only one were marked and checked daily under a microscope to count the number of cell clones. Colony formation was scored 14 days after initial seeding. The clones were then removed and cultured similarly in new 96-well plates to generate subclones.
Invasion assay
In vitro invasiveness was evaluated, as previously described, 7 using Boyden invasion chambers (BD Biosciences). In brief, each Boyden chamber consists of a BD Falcon TC Companion Plate with Falcon Cell Culture inserts containing an 8-µm pore polyethylene terephthalate (PET) membrane with a thin layer of Matrigel basement membrane matrix. The interior of the inserts was first rehydrated for 2 hours with warm (37°C) bicarbonate-based culture medium. The upper chambers were filled with 0.5 ml of D-GBM grade IV cells (1.25 × 105 cells/ml) in serum-free DMEM medium. DMEM with 10% FBS was placed in the lower chambers as a chemoattractant. The chambers were incubated at 37°C in a humidified atmosphere of 5% CO2 in air or in hypoxic conditions of 5% CO2, and 95% N2 for 24 hours. Nonmigrating cancer cells on the upper surface of the inserts were removed by wiping with a cotton swab; migrating cells on the lower surface were fixed and stained with Insta Stain 3 Step (S&K Reagent, Denver, CO). The invasive potential was quantified by counting the total number of cells on the lower surface of the inserts using a light microscope. Three random visual fields were counted for each assay. Each invasion experiment was carried out in triplicate.
Immunohistochemistry
Five-micron (5-µm) paraffin-embedded sections were used for immunohistochemical study. After deparaffinization, the sections were subjected to an antigen retrieval protocol by heating them in 10-mM citrate buffer (pH 6.0) for 10 minutes. Potential nonspecific binding sites were blocked with 5% normal goat or rabbit serum in PBS. After blocking, the sections were incubated with primary antibody for 1 hour at room temperature to overnight at 4°C. The primary antibodies VEGF-A, VEGFR2, and CD 34 were purchased from Santa Cruz Biotechnology (Santa Cruz, CA) and a 1 : 50 dilution was used. Nestin and beta III tubulin antibodies were purchased from Millipore (Billerica, MA). CD 133 antibodies were purchased from eBioscience; GFAP antibodies were purchased from DakoCytomation (Carpinteria, CA). Factor VIII (Von Willebrand) antibody was purchased from (Abcam, Cambridge, MA) For double-labeling immunostaining, sections were pretreated with anti–Factor VIII antibody as described above. Next, tissue was incubated with anti-VEGF antibody overnight at 4°C. After incubation with the secondary antibody, sections were treated with alkaline phosphatase-avidin-biotin substrate and red chromogen (Vector red; Vector Laboratories, Burlingame, CA), for color development. Following primary antibody reaction, sections were washed and incubated with either biotin-conjugated anti-rabbit or anti-goat IgG (Vector Laboratories) at a dilution of 1/200. A Vector-ABC streptavidin-peroxidase kit with a benzidine substrate for color and streptavidin-phosphatase kit (KPL, Gaithersburg, MD) with a HistoMark Red substrate (KPL) were used for color development. Counter-staining was done with diluted hematoxylin. Sections that were not incubated with primary antibody served as negative controls. For immunofluorescence experiments, we used the same Beta III, CD133, and VEGFR2 antibodies. Following primary antibody reaction, sections were washed and incubated with goat anti-rabbit IgG-FITC (Santa Cruz) or goat anti-mouse IgG (Invitrogen, Carlsbad, CA) at a dilution of 1 : 100. The samples were then washed and incubated with DAPI (Santa Cruz), at a concentration of 1 mg/ml for 30 minutes at room temperature. Sections that were not incubated with primary antibody served as negative control.
Total RNA extraction and semiquantitative reverse transcription polymerase chain reaction
Total RNA was extracted from a D-GBM grade-IV cell culture, and a dog GBM grade I (pilocytic astrocytoma) primary cell culture isolated from a Boston Terrier using an SV RNA extraction kit (Promega, Madison, WI) according to the manufacturer's directions. RNA was quantified by absorbance at 260 nm. Using a Super Script III First Strand Synthesis System (Invitrogen), 100 ng of total RNA was amplified by reverse transcription–polymerase chain reaction (RT-PCR) and the cDNAs were amplified in a 25-µl reaction. PCR primers were as follows:
Alpha-5 primers: (forward, 5′-CATTTCCGAGTCTGGGCCAA-3′; reverse, 5′-TGGAGGGGGGGCTTGAGCTGAGCTT-3′) 17
Alpha-1 primers: (forward, 5′-GTGCCAAGGAGGGGAGTTA-3′; reverse, 5′-ACAGTTCCGTTCCAGTCATAGG-3′) 17
Beta-3 primers: (forward, 5′-GGGGACTGCCCTGTGT-3′; reverse, 5′-CTTTTTCGGTCGTGGGATGGTG-3′) 17
Nestin primers (forward, 5′-GAGAACCAGGAGCAAGTGAA-3′; reverse, 5′-TTTCCAGAGGCTTCAGTGTC-3′) 49
CD133 primers (forward, 5′-GGACACAAAAGCCAACAATC-3′; reverse, 5′-ATCTTGACCCATTGCAGGTA-3′) 49
βIII tubulin primers (forward, 5′-GCACACTGCTCATCAACAAG-3′; reverse, 5′-TCTTGCTCTCCTTCATGGA-3′) 49
GFAP (forward, 5′-CGAGTTACCAGGAGGCACTA-3′; reverse, 5′-TCCACGGTCTTTACCACAAT-3′) 49
β-actin primers (forward, 5′-ATGTACGTAAGCCAGGC-3′; reverse, 5′-AAGGAACTGGAAAAGAGC-3′) 31
VEGF primers (forward, 5′-GACCCTGGTGGACATCTTCCAGGA-3′; reverse, 5′-GGTGAGAGGTCTAGTTCCCGA-3′) 52
VEGFR2 primers (forward, 5′-GCCAATGAAGGGGAACTGAAGA-3′; reverse, 5′-CTCTGACTGCTGGTGATGCTGTC-3′) 18
Expected size for the alpha-5 integrin PCR product was 323 base pairs (bp), 414 bp for the alpha-1 integrin, and 260 bp for the beta-3 integrin. PCR conditions for alpha-5, alpha-1, and beta-3 were carried out with an initial denaturation at 94°C for 2 minutes, followed by 35 cycles at a denaturation temperature of 94°C for 15 seconds, annealing at 55°C for 30 seconds, and extension at 72°C for 1 minute. A final extension of 5 minutes at 72°C was carried out. The expected size for the nestin PCR product was 328 bp, 315 bp for CD133, 357 bp for βIII-tubulin, and 277 bp for GFAP. PCR conditions for βIII-tubulin, CD133, and GFAP were carried out with an initial denaturation at 94°C for 2 minutes, followed by 35 cycles at a denaturation temperature of 94°C for 30 seconds, annealing at 58°C for 30 seconds, and extension at 72°C for 1 minute. PCR conditions for nestin were carried out with an initial denaturation at 94°C for 2 minutes, followed by 35 cycles at a denaturation temperature of 94°C for 30 seconds, annealing at 60°C for 30 seconds, and extension at 72°C for 1 minute. A final extension of 5 minutes at 72°C was carried out. To demonstrate the integrity of the RNA samples used in the RT-PCR reactions, parallel amplifications with oligonucleotide primers for mouse β-actin were performed. The expected size for the β-actin PCR product was 403 bp. PCR fragments were analyzed on 1.5% agarose gels stained with ethidium bromide.
For VEGF, one set of primers, which amplified 2 splicing variants of VEGF mRNA (VEGF164 and VEGF188) was used. The PCR profile consisted of initial denaturation at 94°C for 7 minutes, followed by 35 cycles of denaturation at 94°C for 30 seconds, annealing at 58°C for 30 seconds, extension at 72°C for 90 seconds, and extension at 72°C for 7 minutes. The expected length of the PCR products was 462 bp for VEGF164, and 514 bp for VEGF188. Expected size for VEGFR2 PCR product was 537 base pairs. PCR conditions for VRGFR2 were carried out with an initial denaturation of 94°C for 3 minutes, followed by 30 cycles at a denaturation of 94°C for 30 seconds, annealing at 60°C for 30 seconds, and extension at 72°C for 30 seconds. A final extension of 5 minutes at 72°C was carried out.
Intracranial tumor cell injection into nude mice
For evaluation of tumorigenicity, cells cultured in DMEM and neurobasal medium were resuspended in 7 µl of Hank's balanced salt solution and injected stereotactically into the brains of mature nude mice (Harlan, Indianapolis, IN) under anesthesia Ketamine/xylazine cocktail (53.7 mg/ml Ketamine, 9.26 mg/ml xylazine, delivered at 1 ml/kg). Coordinates for stereotactical injections into the brain were 3-mm lateral and 0.5-mm posterior of the bregma and 2.5-mm deep to the cortical surface of the brain. The inoculated mice were kept in the animal facility and euthanatized when they developed clinical neurologic signs because of tumor burden. Mice were anesthetized with a Ketamine/xylazine cocktail and perfused with 4% paraformaldehyde. The evaluation of the tumor within the brain was performed with HE staining.
Statistical analysis
Quantification of semiquantitative RT-PCR bands density was performed on a Macintosh computer using the public domain National Institutes of Health (NIH) Image program (developed at the US National Institutes of Health and available at http://rsb.info.nih.gov/nih-image). Data from invasion assay experiments are presented as mean ± standard deviation (SD), and statistical comparisons were made using Student's t-test. A P-value of <.05 was considered statistically significant.
Results
Glioblastoma in Boxer: morphologic presentation and similarity with human glioblastoma
An intracranial neoplasm was diagnosed by CT scan of an 8-year-old male Boxer dog. The necropsy revealed an intracranial tumor mass, which obliterated the left lateral ventricle (Figs. 1,2). A GBM was diagnosed on histopathologic examination performed on frozen sections and formalin-fixed, paraffin-embedded tissue. The tumor consisted of a heterogeneous glial cell population, multifocal areas of necrosis with perinecrotic cellular pallisading, and marked neovascularization with glomeruloid pattern (Figs. 3–6). The dog glioblastoma cellular heterogeneity consisted of areas showing oligodendroglial, gemistocytic, or fibroblastic astroglial differentiation, but an estimated one third of the tumor cells were undifferentiated. A disrupted and fragmented ependymal lining of the lateral ventricle was located at the periphery of the tumor mass. These morphologic features are characteristic and similar to human glioblastoma grade IV classification. 29 In addition, this GBM showed intratumoral microglial infiltration and peritumoral astroglial activation.
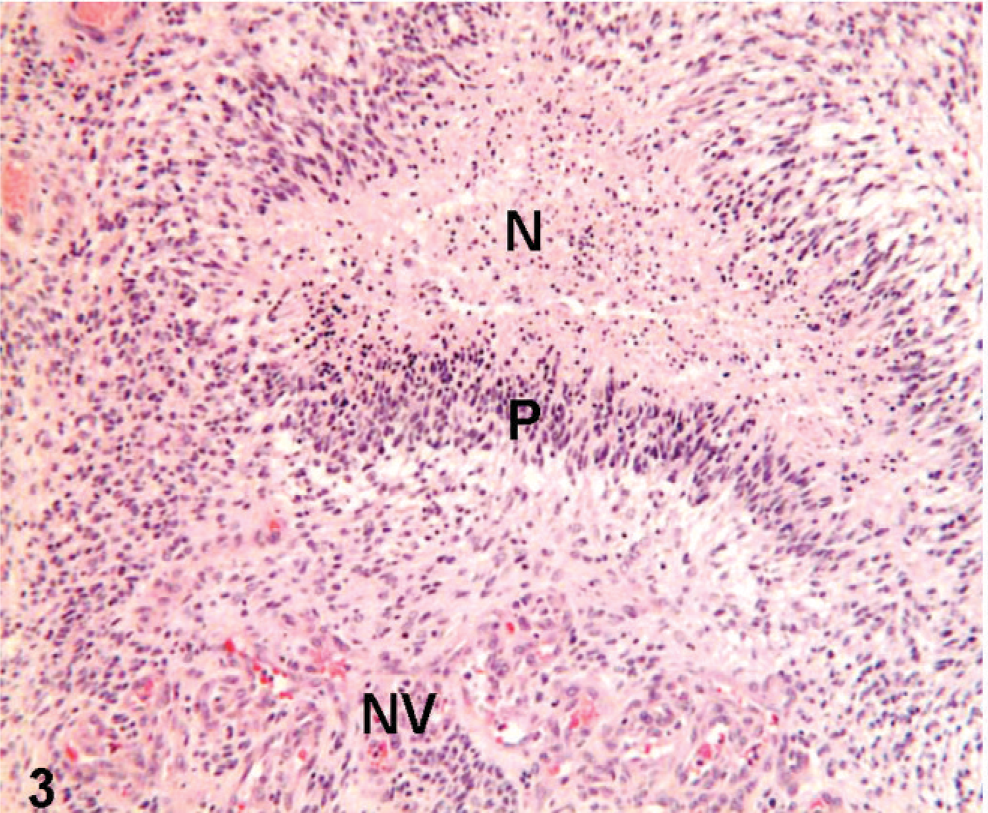
Histologic appearance of Boxer dog GBM. Necrosis (N), palisading (P), and neovascularization (NV) are evident. These are also the pathognomonic features of human GBM.
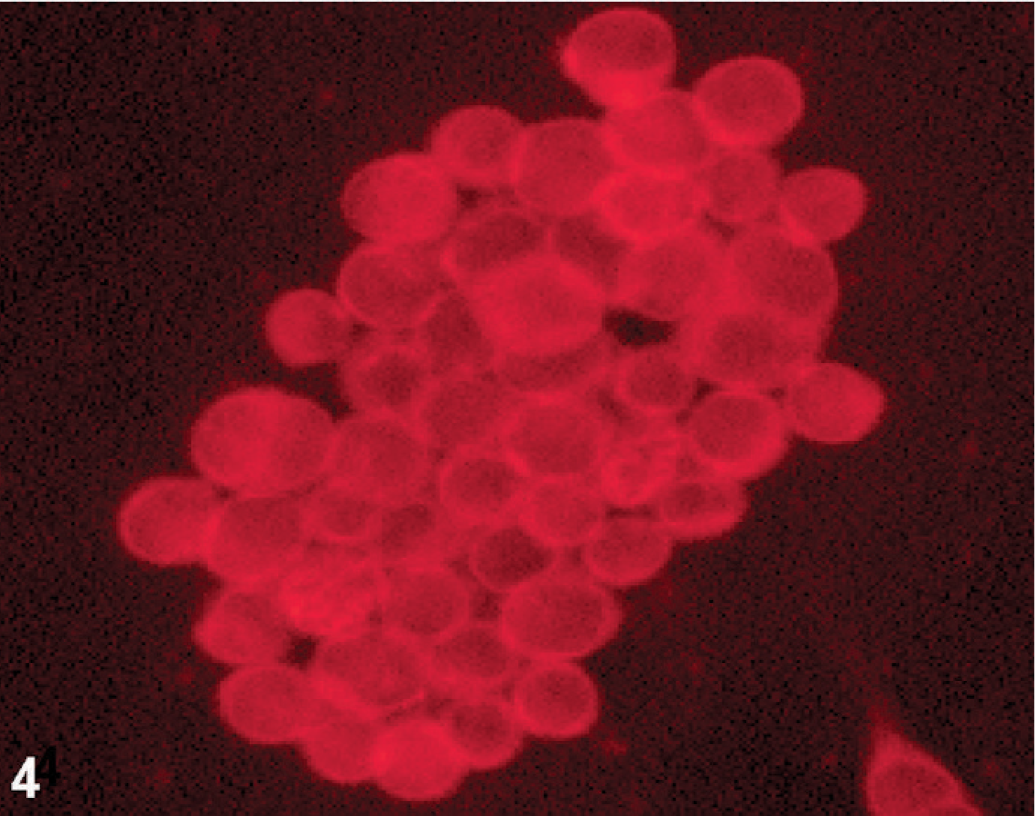
Nestin immunofluorescence of cultured D-GBM cells (neurosphere cultured in neurobasal medium, passage 3) derived from a Boxer dog. Notice the spheroid pattern of growth that characterizes this glioblastoma.
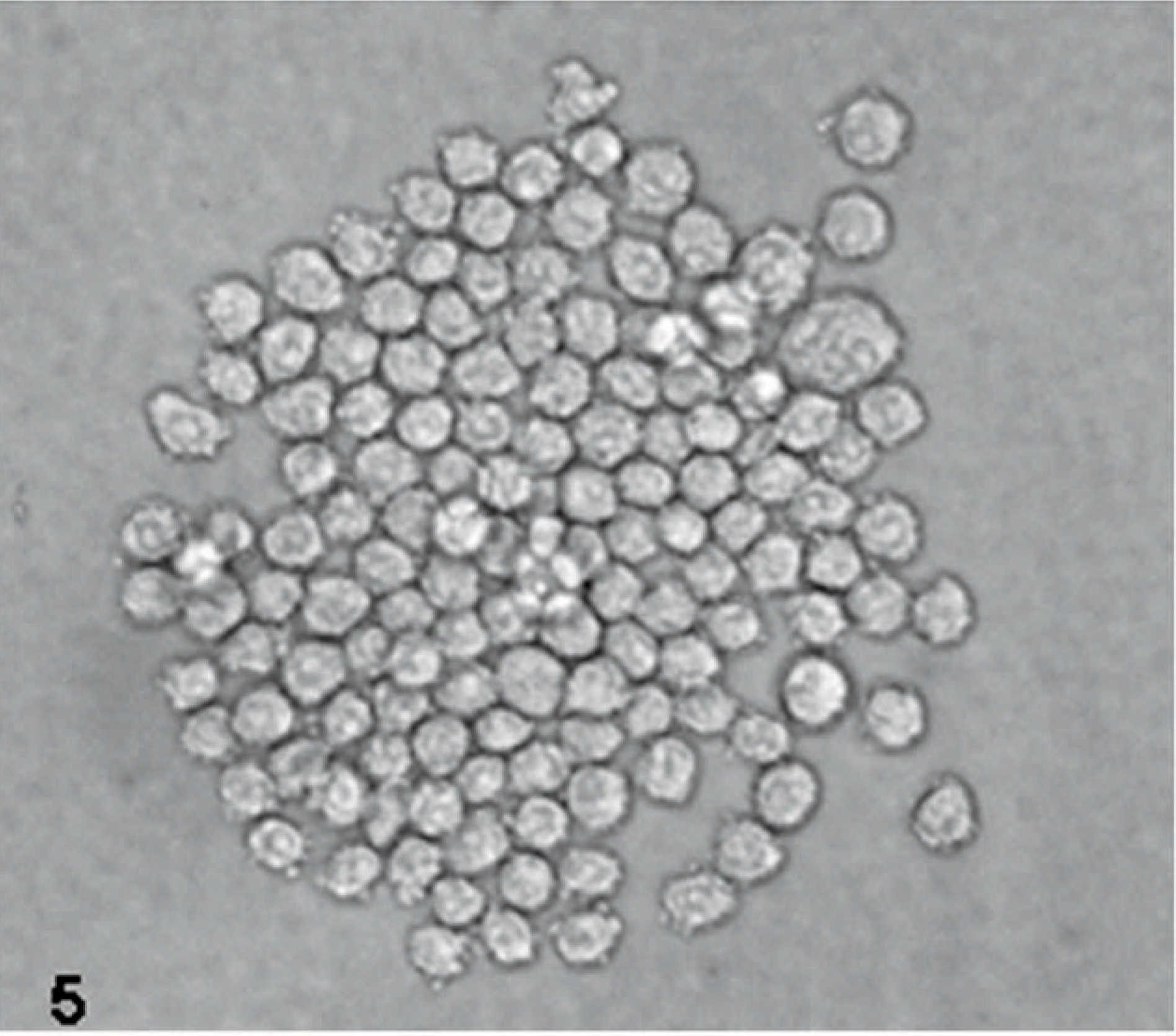
Inverted light microscopy image of a neurosphere arising from D-GBM cultured in neurobasal medium supplemented with EGF and FGF.
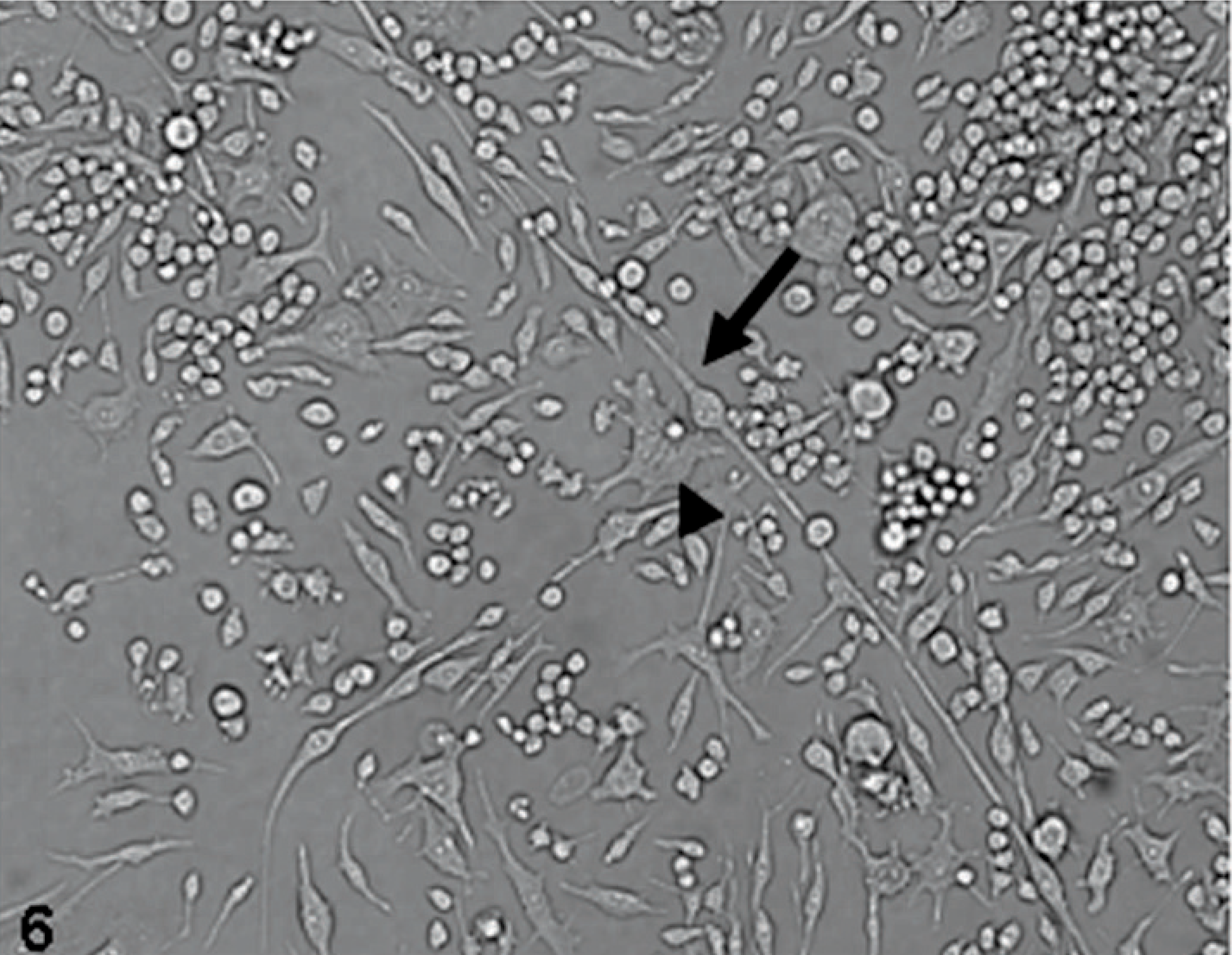
Inverted light microscopy appearance of D-GBM cultured in DMEM medium supplemented with 10% FBS, passage 3. Notice neural cell differentiation of D-GBM; bipolar astrocytes (arrow) and neurons, some multinucleated (arrowhead).
Pilocytic astrocytoma from a Boston Terrier histopathology was characterized by low cellularity and predominantly bipolar, elongated (piloid, hair-like) cells as previously described 42 (Figs. 7,8).
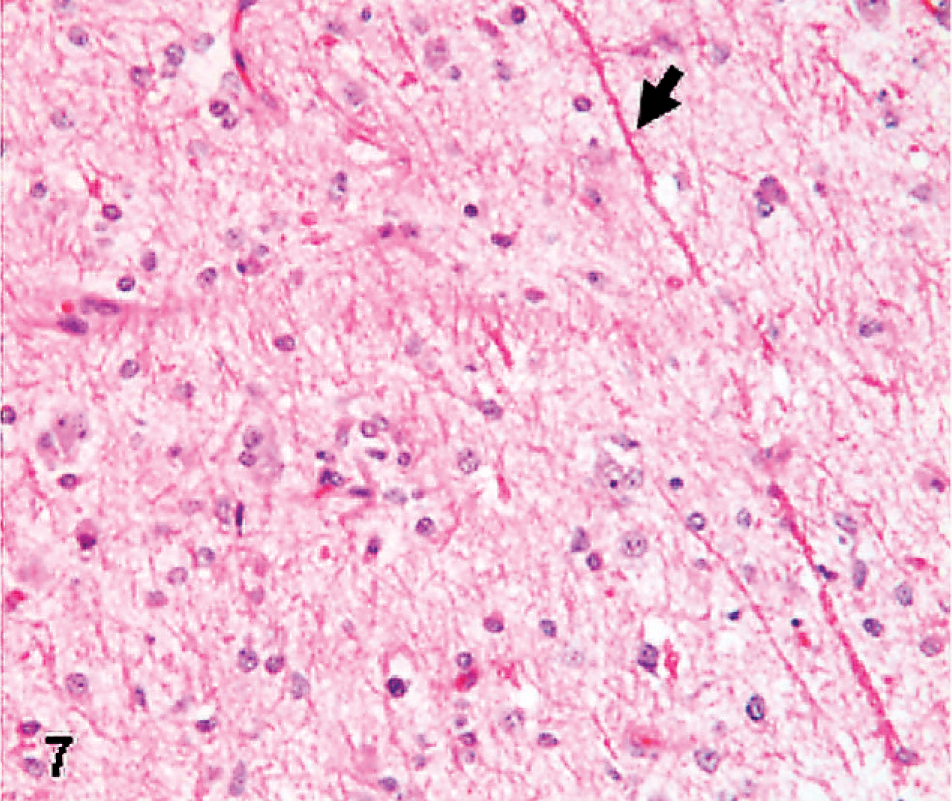
Histopathologic appearance of a pilocytic astrocytoma from a Boston Terrier. Notice the hypocellularity and piliform type of astrocytes and Rosenthal bodies (arrow) features that characterize this grade-I glioma. HE.
Pilocytic astrocytoma in primary tissue culture showing a bipolar piliform astrocytic phenotype.
In-vitro culture of dog GBM and its characterization
Necropsy was performed 1 hour posteuthanasia, and a small segment of tumor was taken and processed for tissue culture as outlined in Materials and Methods section. Our quest was to determine if dog gliomas possess CSC-like properties as previously reported in human gliomas. We were able to obtain glioma cultures (D-GBM) from a grade-IV GBM isolated from a Boxer dog (Figs. 5,6). Cultured D-GBM cells demonstrated neurosphere formation in both neurobasal media supplemented with FGF and EGF and also in DMEM supplemented with 10% FBS. All primary D-GBM cultures were composed of single cells, which gave rise to clonal spheres within a week. The nonattached neurospheres that grew in neurobasal medium supplemented with EGF and FGF were passed by simple mechanical dissociation. The tumor cells that grew in DMEM supplemented with 10% FBS formed neurospheres, which partially adhered to the culture flask. They were lightly attached and we passed them by mechanical manipulation or light trypsinization. A cloned cell line was established from this dog GBM, which has been passed 20 times to the present time. All the experiments using this D-GBM were performed with cells of early passage (up to 5). There were no differences in marker expression due to passage number, and cells maintained a similar phenotype up to passage 20. We were not able to obtain a primary cell line from Boston Terrier pilocytic astrocytoma (PCA) tumor. We used the primary culture of PCA grade I for comparative study with grade-IV glioblastoma (Figs. 7,8).
Flow cytometric analysis of glioblastoma stem-like cells
We analyzed cultured cells for their cell surface expression of markers for the CSC phenotype using a flow cytometric method. Dog GBM cells isolated from a Boxer dog expressed the CSC surface marker CD133 (see Fig. 9), as has been reported in some human glioma counterparts, hematopoietic malignancies, and other solid tumors. Mechanically dissociated, undifferentiated cells were incubated with antibodies against CD133, and the percentage of immunoreactive cells was analyzed. The procedure was performed using cells maintained in neurobasal and DMEM medium. There were no significant differences in the number of CD133-labeled cells from neurospheres harvested from both types of medium. There was a lower expression of labeled cells for CD133 in the partly adherent cells maintained in DMEM when compared with floating neurospheres (see Fig. 9).
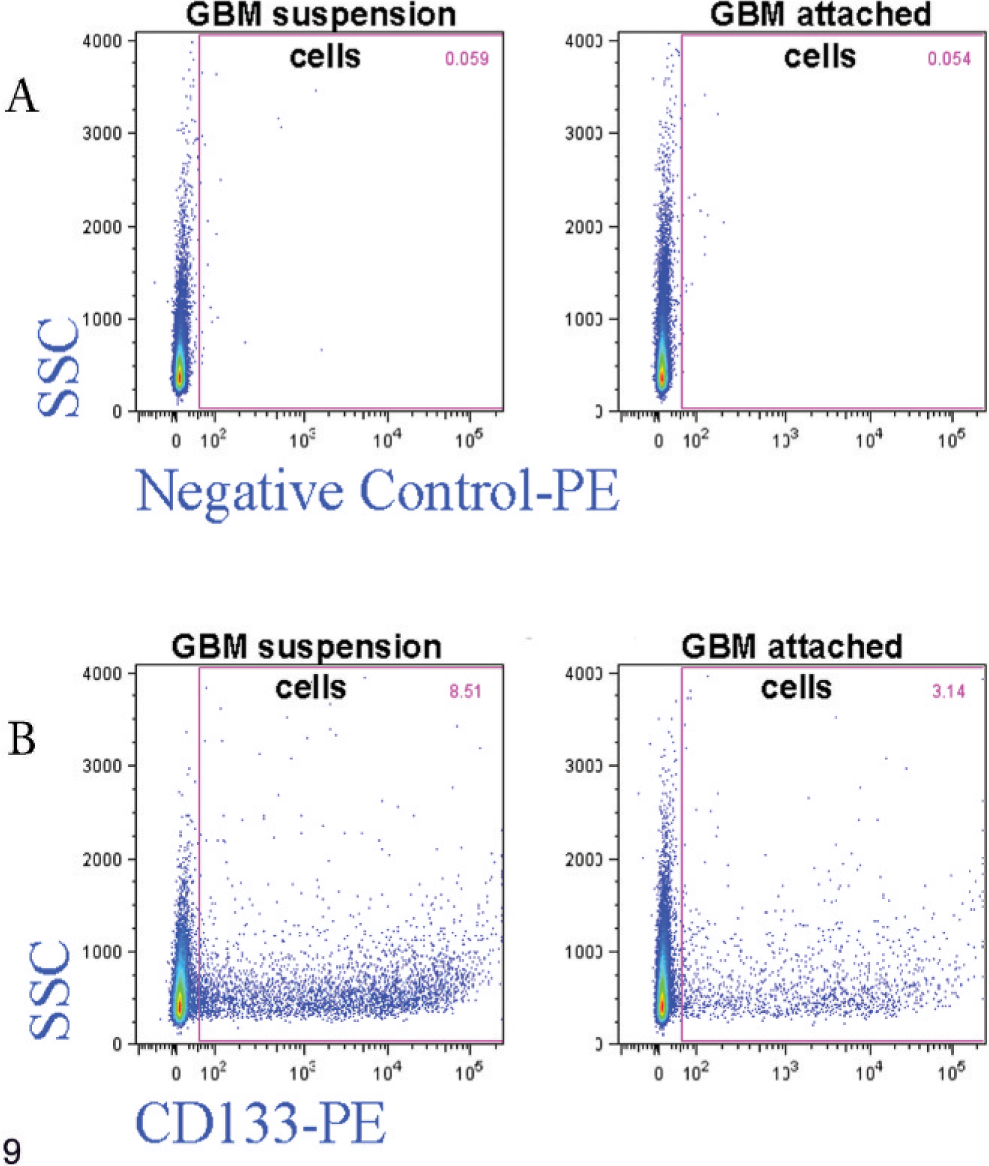
Flow cytometric analysis of D-GBM (grade IV isolated from a Boxer dog) cells expressing CD133. The relative distribution of cells expressing CD133 was determined in D-GBM suspension cells and adherent cells. (A) Control (unstained) samples. (B) CD133 stained sample. D-GBM suspension cells 8.52% of cells are positive for CD133, and in GBM attached cells only 3.14% of cells are positive for CD133.
Self-renewal of D-GBM cells
Self-renewal is a critical feature of neural stem cells and CSCs. 37, 50 We performed limiting dilution assays to analyze whether single D-GBM cells have the capacity to form new spheres or colonies. Subsphere formation was observed for the nonadherent and for the partly adherent D-GBM cells. In this serial dilution assay, 53 clones, each consisting of 500 to 800 cells, grew out of the 53 original single cells. The clone formation rate was 100%. The primary clones were then dissociated and plated into new 96-well microplates to generate subclones. The subclone formation rate was 100%, with 122 subclones from 122 single cells.
In vivo growth of glioma neurospheres
To address whether D-GBM cells are tumorigenic in vivo, cells were xenografted into the striatum of nude mice. Initially, three nude mice were inoculated intracranially with 1 × 106 D-GBM cells from dissociated neurospheres to assess the tumorigenicity potential. We euthanatized these mice at 10 days postinoculation (dpi) due to severe neurologic deficit. Large, invasive tumors were detected invading the cortex, corpus callosum, striatum, and lateral ventricles upon histopathologic examination (Fig. 10). Next, for each cell line maintained in neurobasal and DMEM medium (adherent and partly adherent), 3 animals were injected with 200,000 cells and at least 3 with 1,000 cells. All mice that received 200,000 cells developed neurologic clinical signs and were euthanatized at 25 dpi with tumors. The mice inoculated intracranially with 1,000 cells also developed tumors after a longer time (60 dpi). The tumor xenografts demonstrated the cardinal signature of the original GBM: cellular heterogeneity, marked neovascularization, and necrosis (Figs. 10–13). The morphology of tumors clearly reproduced the phenotype of the original dog tumor from which these cells were isolated. There was no morphologic difference between the xenografts derived from neurosphere cells maintained in neurobasal or DMEM medium. The nude mouse xenograft was harvested and recultured in FGF- and EGF-supplemented neurobasal medium and also in DMEM containing 10% FBS. The cultured xenografts formed nonattached neurospheres in growth factor–supplemented neurobasal medium. Nude mouse xenografts cultured in DMEM with serum formed nonattached neurospheres, but there were also many attached, piled up colonies, which were loosely adherent and showed neural cell differentiation (Figs. 14,15).
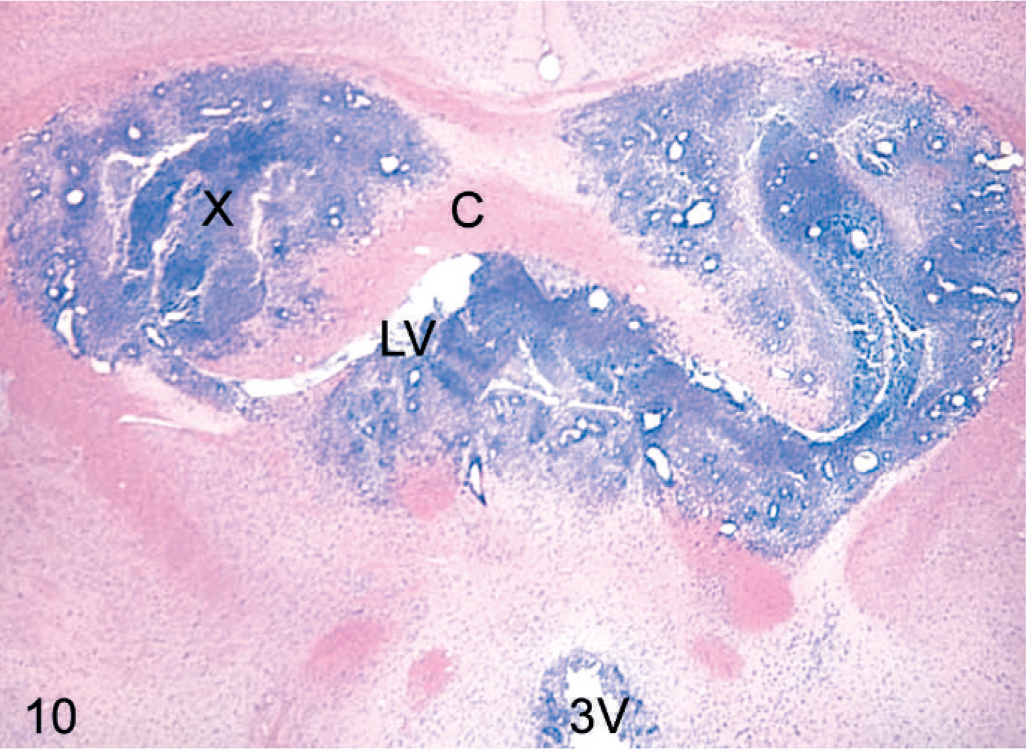
Brain coronal section: the intracranial orthotopic inoculation of D-GBM cells in a nude mouse (1×106) euthanatized at 10 dpi. Tumor xenograft (X) invading the putamen, corpus callosum (C) and lateral ventricle (LV), and third ventricle (3V). HE. Original magnification ×2.
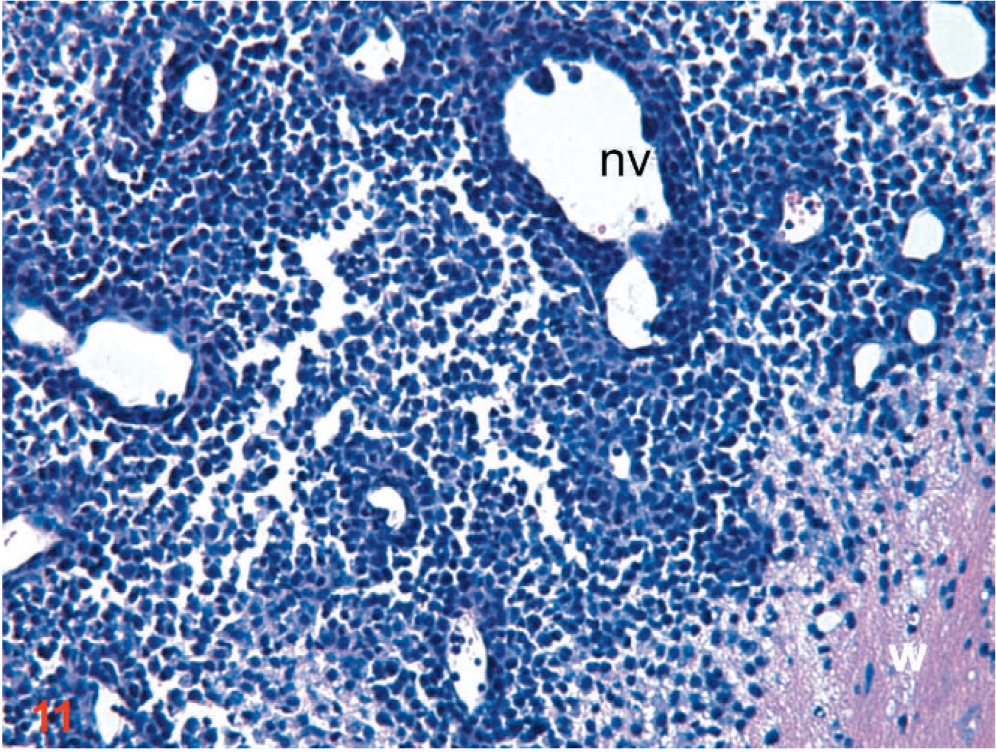
Coronal section: the intracranial orthotopic xenograft of D-GBM cells in a nude mouse (1×106) euthanatized at 10 dpi. Intense neovascularization (nv) within the fast growing tumor. Tumor vessels appear tortuous, hyperdilated, and irregularly shaped. Notice also tumor cells infiltrating into the white matter (w). HE. Original magnification ×20.
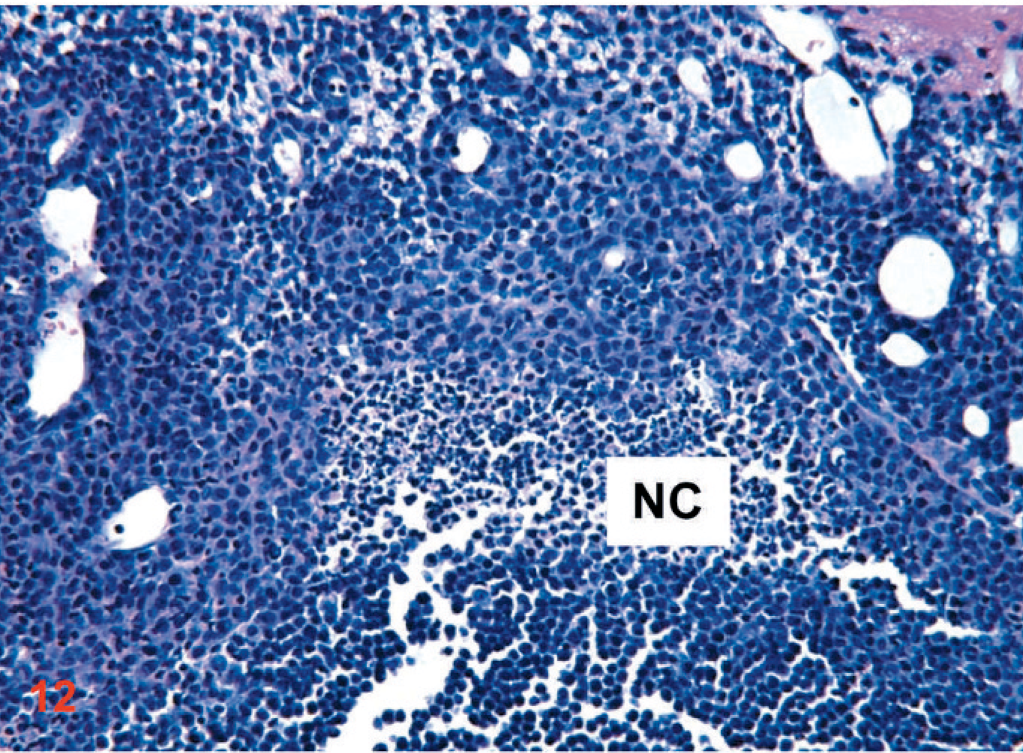
Coronal section: the intracranial orthotopic D-GBM cells xenograft in a nude mouse (1×106) euthanatized at 10 dpi. A necrotic center (NC) is evident within the tumor core and an abundance of neovessels. HE. Original magnification ×20.
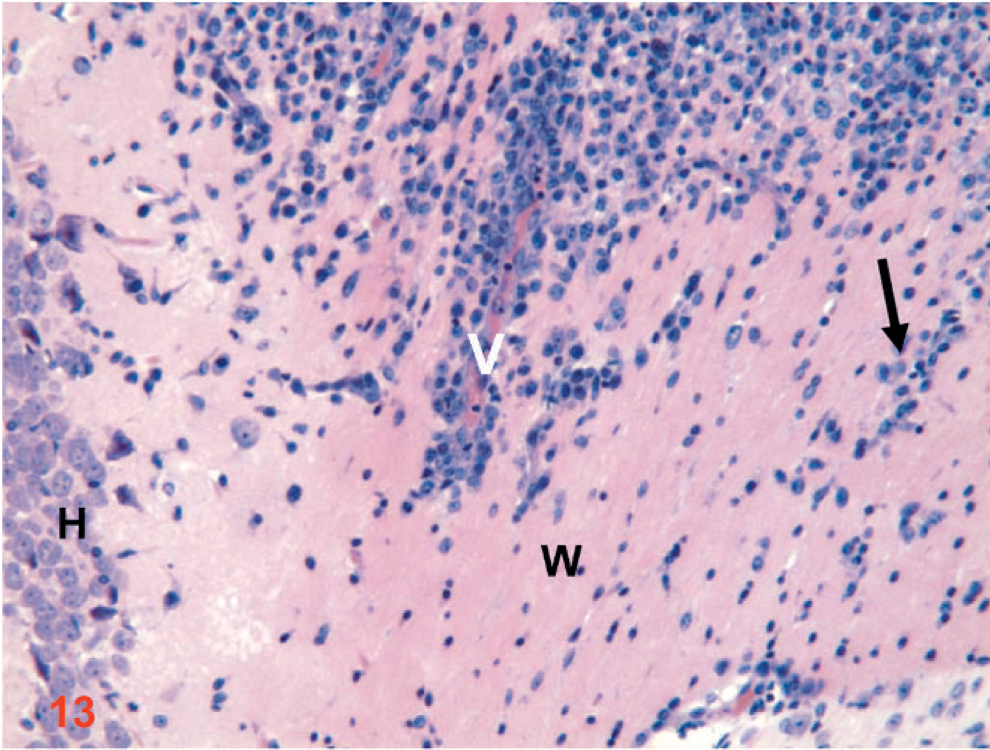
Coronal section: the intracranial orthotopic xenograft of D-GBM cells in a nude mouse (1×106) euthanatized at 10 dpi. Notice the perivascular tumor cells migration within the white matter (V). (W) white matter, (H) hippocampus, and migrating tumor cells into the white matter (black arrow). HE. Original magnification ×20.
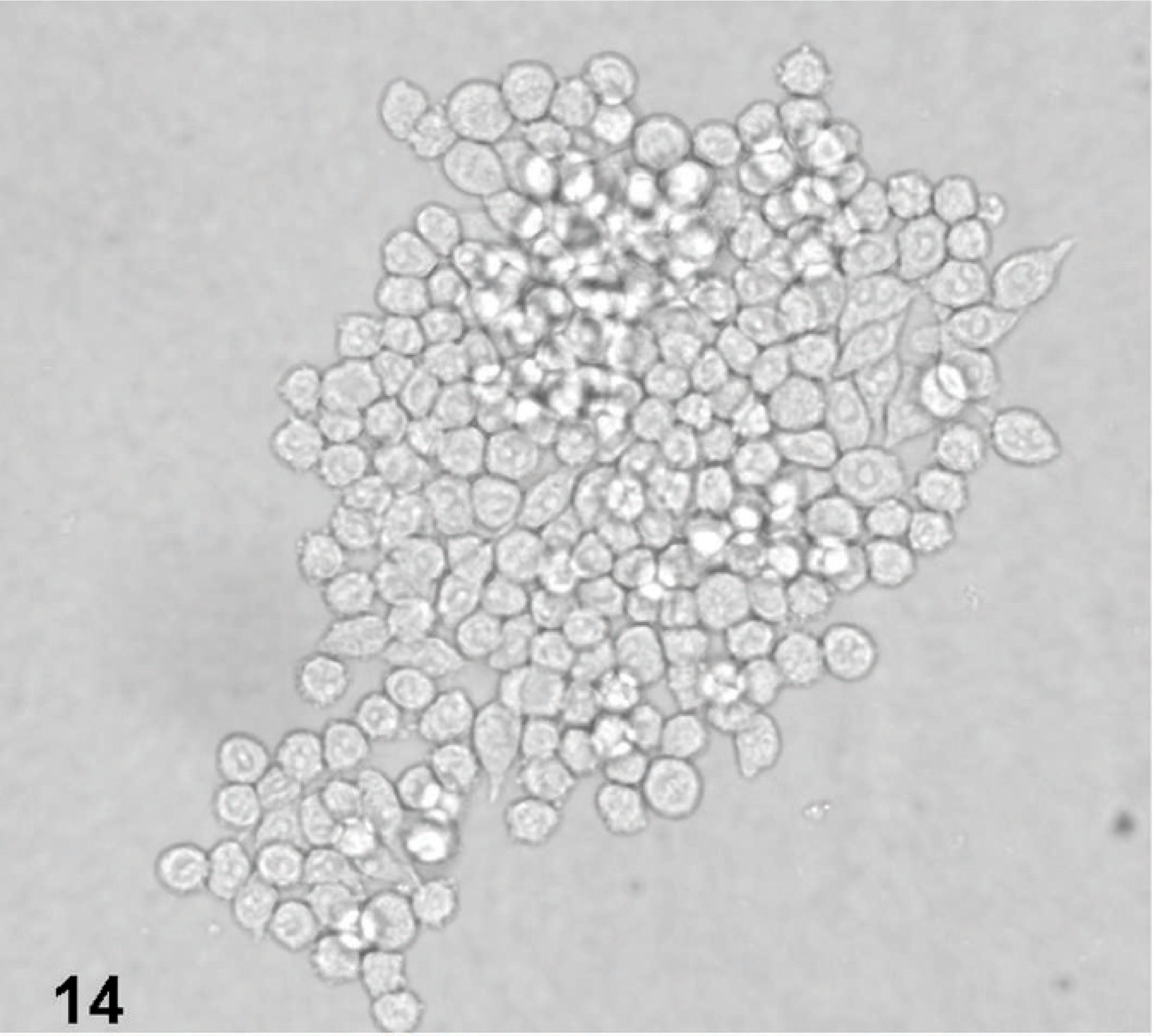
Inverted microscopic appearance of D-GBM xenograft recultured from a nude mouse and maintained in neurobasal medium. D-GBM neurosphere formation in tissue culture.
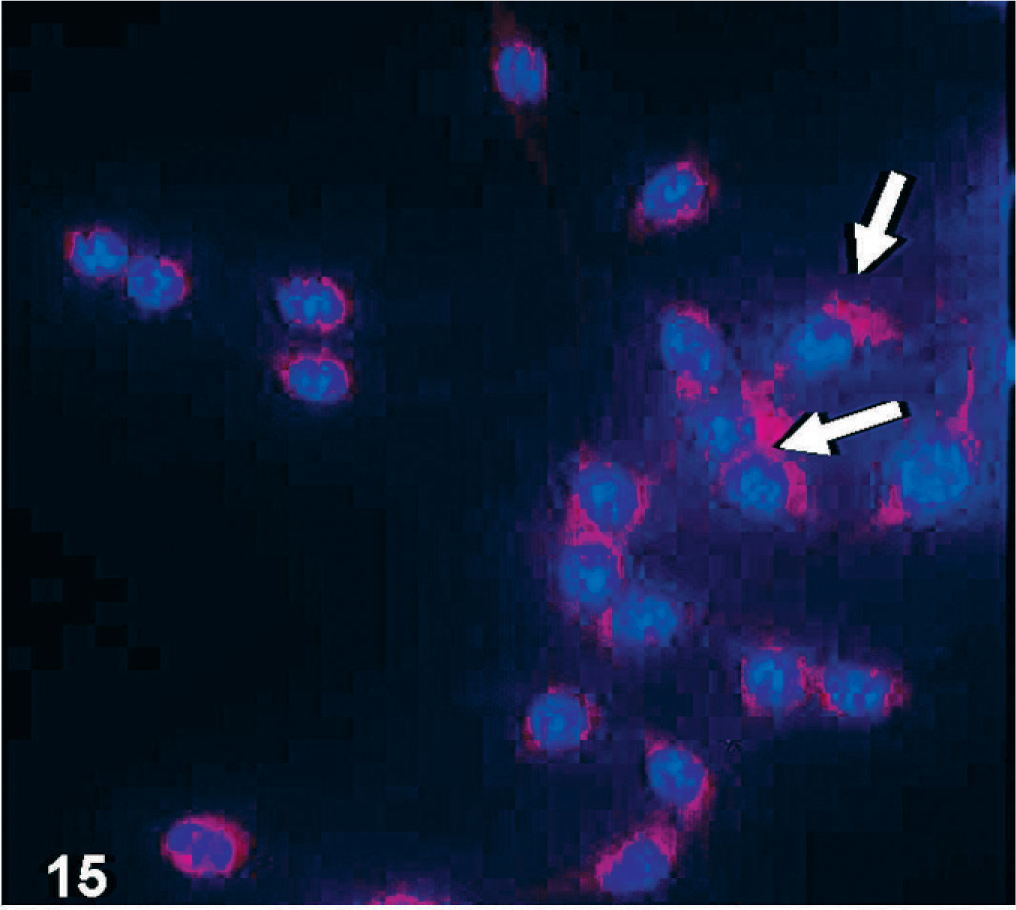
βIII tubulin-immunofluorescence of cultured D-GBM cells in DMEM medium supplemented with 10% FBS. Cell nuclei are indicated by DAPI staining (blue). Notice that the attached cells (arrows) show immunolabeling for neuronal cell differentiation.
Dog glioma cell line has a capacity for differentiation
Glioma stem cells are characterized by the expression of stem cell markers and the capacity for neural cells differentiation. CD133 and nestin are frequently used as markers for an undifferentiated state. When we analyzed CD133 expression by flow cytometry, we detected 8.52% CD133+ cells derived from nonattached neurospheres maintained in neurobasal medium and 3.14 % CD133+ from cells maintained in DMEM medium supplemented with 10% FBS and partly adherent. D-GBM cells maintained in DMEM supplemented with 10% FBS showed neuronal cell differentiation (βIII tubulin labeling) and also astrocytic differentiation (GFAP labeling) (Figs. 15,16). D-GBM cells, which became attached and show neural cells differentiation gradually lost their CSC marker, CD133 (Fig. 17). Some of the differentiated cells (beta III tubulin and GFAP positive) were multinucleated and degenerated in time after few passages. In addition, the immunohistochemistry of xenografts showed strong nestin (a class-VI intermediate filament protein and neural stem cell marker) expression as well as CD133 and CD34 (cell surface markers), beta tubulin class-III (a neuronal filament marker), and GFAP (an astrocytic cell marker). Their immunoreactivity in tumor xenografts demonstrated the existence of cancer stem cells with multipotent neural lineage (Figs. 18–22).
GFAP-immunolabeling of cultured D-GBM cells in DMEM medium with 10% FBS. Note the astrocyte differentiated cells expressing GFAP.
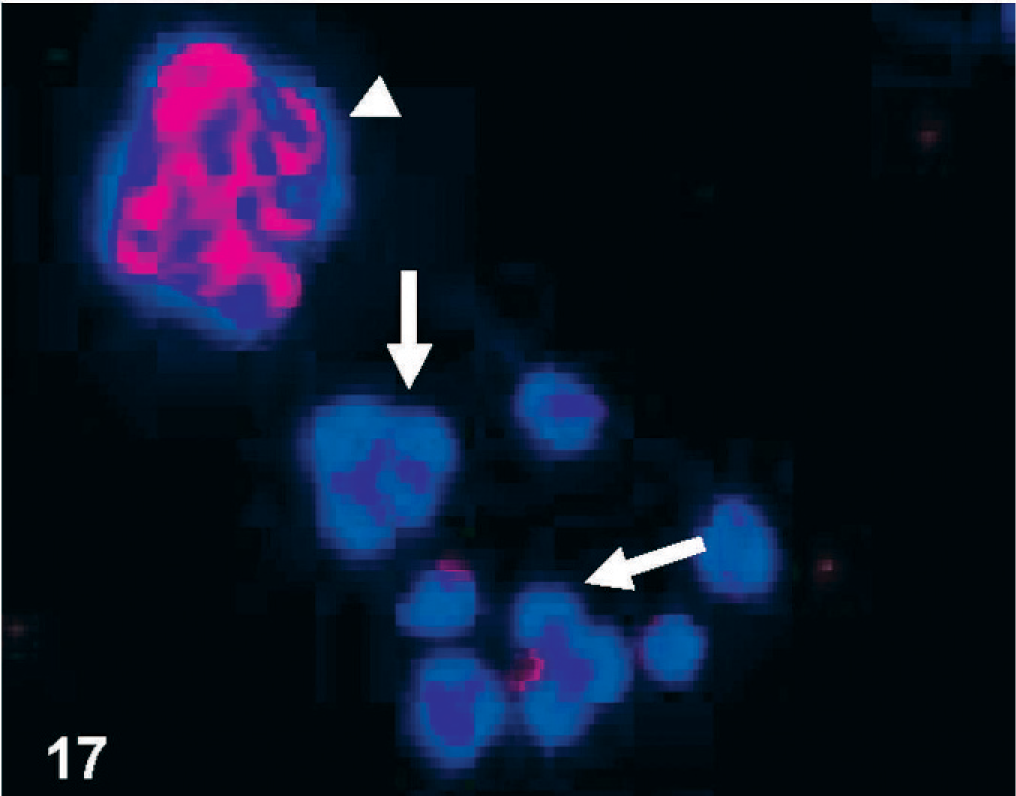
CD133-immunofluorescence of cultured D-GBM cells maintained in DMEM medium with 10% FBS. Cell nuclei are indicated by DAPI staining (blue). Notice the spheroid pattern of growth that characterizes this glioblastoma. Note that the neurosphere (arrowhead) shows intense CD133 immunolabeling and attached differentiated cells show loss of CD133 immunolabeling (arrows).
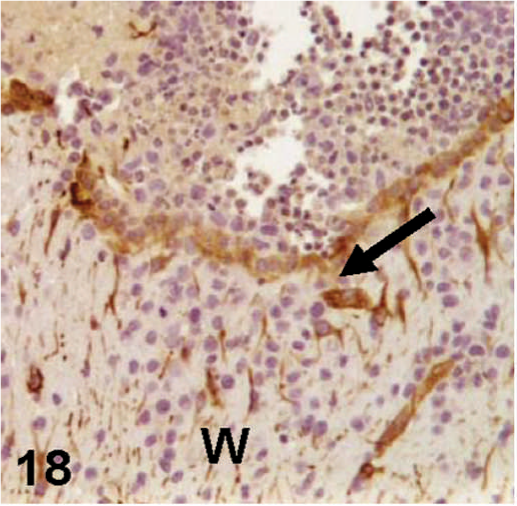
Nestin-immunohistochemical labeling of the nude mouse brain xenograft, inoculated intracranial with D-GBM cells. Nestin-labeled cells (arrow) fingering into the corpus callosum, and white matter (W).
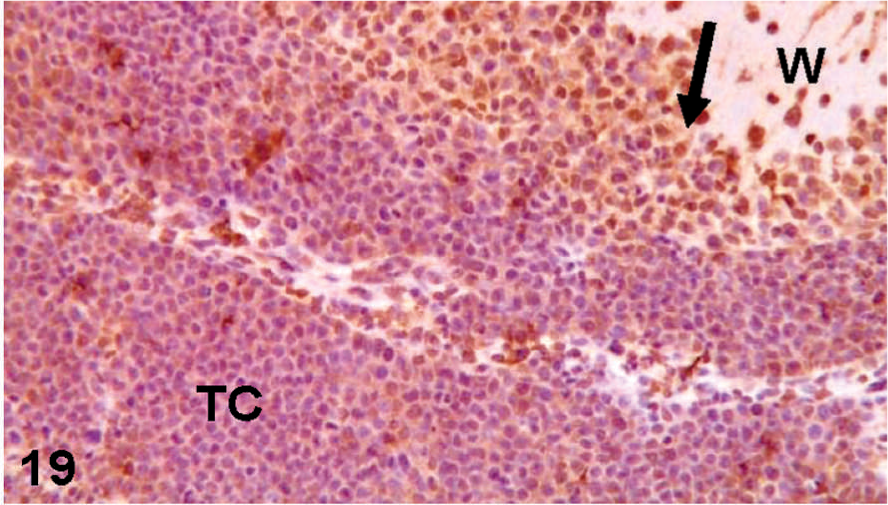
CD133-immunohistochemical labeling of the nude mouse brain xenograft, inoculated intracranial with D-GBM cells. TC illustrates the unlabeled tumor core cells; arrow illustrates the CD133 labeled cells, mostly at the invasion front into the brain white matter (W).
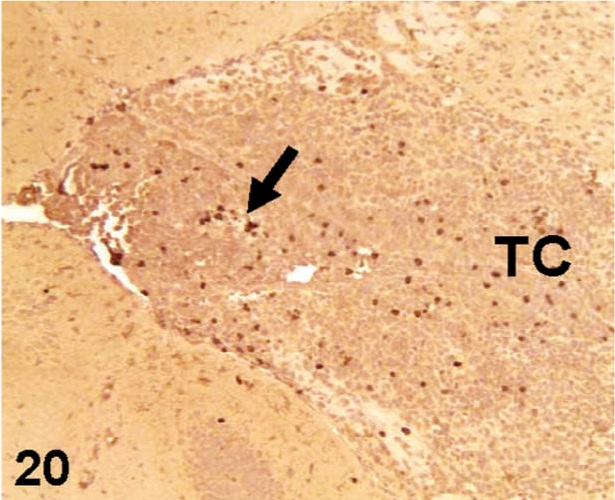
CD34-immunohistochemical labeling of the nude mouse brain xenograft, inoculated intracranial with D-GBM cells (arrow). Notice that most of the cells in the tumor core (TC) are unlabeled for CD34.
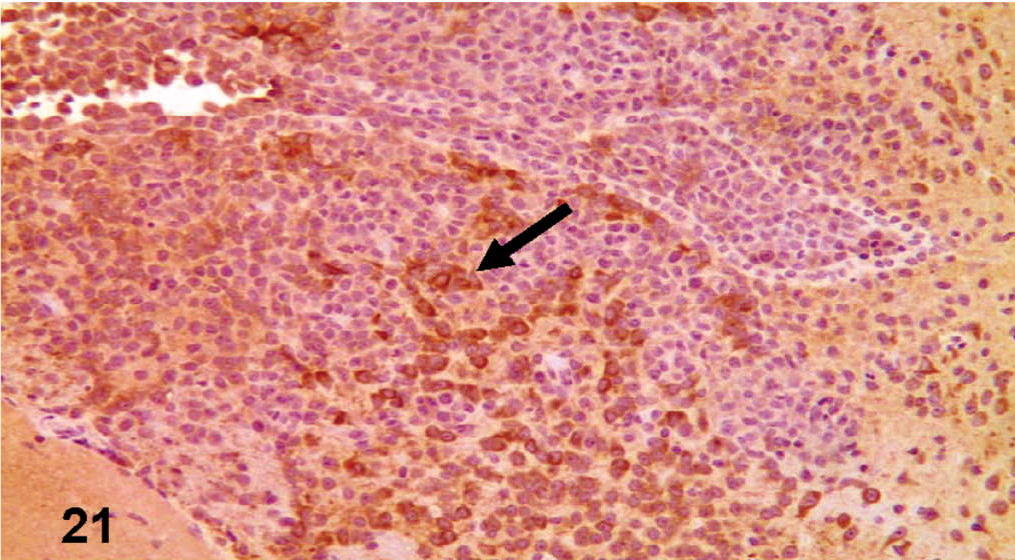
βIII-tubulin immunohistochemical labeling of the nude mouse brain xenograft, inoculated intracranial with D-GBM cells. Notice strong expression of βIII tubulin (arrow).
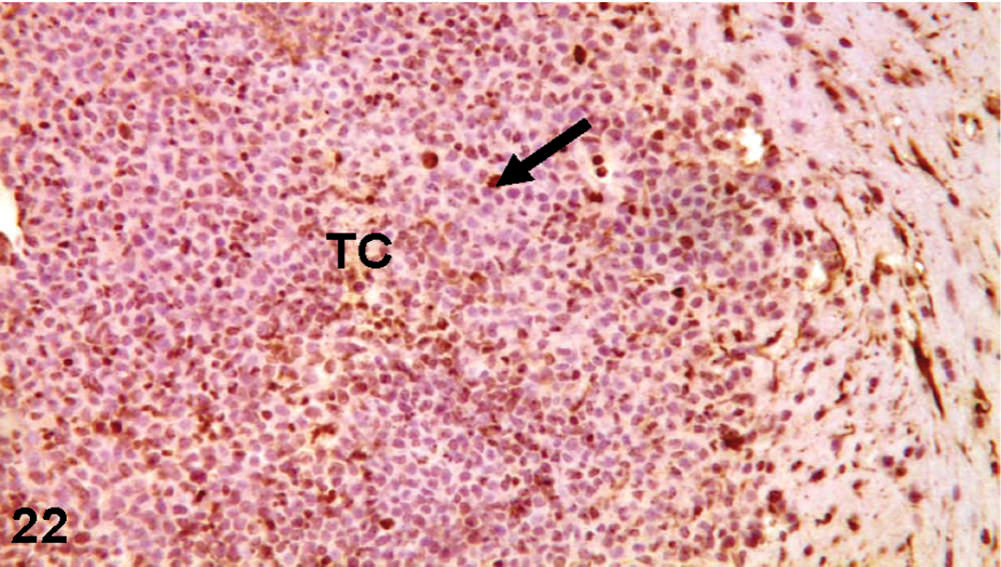
GFAP-immunohistochemical labeling of the nude mouse brain xenograft, inoculated intracranial with D-GBM cells. Immunolabeling of GFAP: Notice, GFAP-labeled cells (arrow) dispersed throughout tumor core (TC).
RT-PCR shows mRNA expression of pluripotency markers
RT-PCR analysis of Boxer D-GBM grade IV, revealed the expression of CD133, nestin, GFAP, and βIII tubulin (Fig. 23). CD133 and nestin were absent in grade-I pilocytic astrocytoma cells. Normal dog brain tissue was used as a control. These results confirmed the expression of CD133, nestin, βIII-tubulin, and GFAP in Boxer D-GBM grade-IV by flow cytometry and immunohistochemistry demonstrating the differentiation potential of D-GBM CSCs.
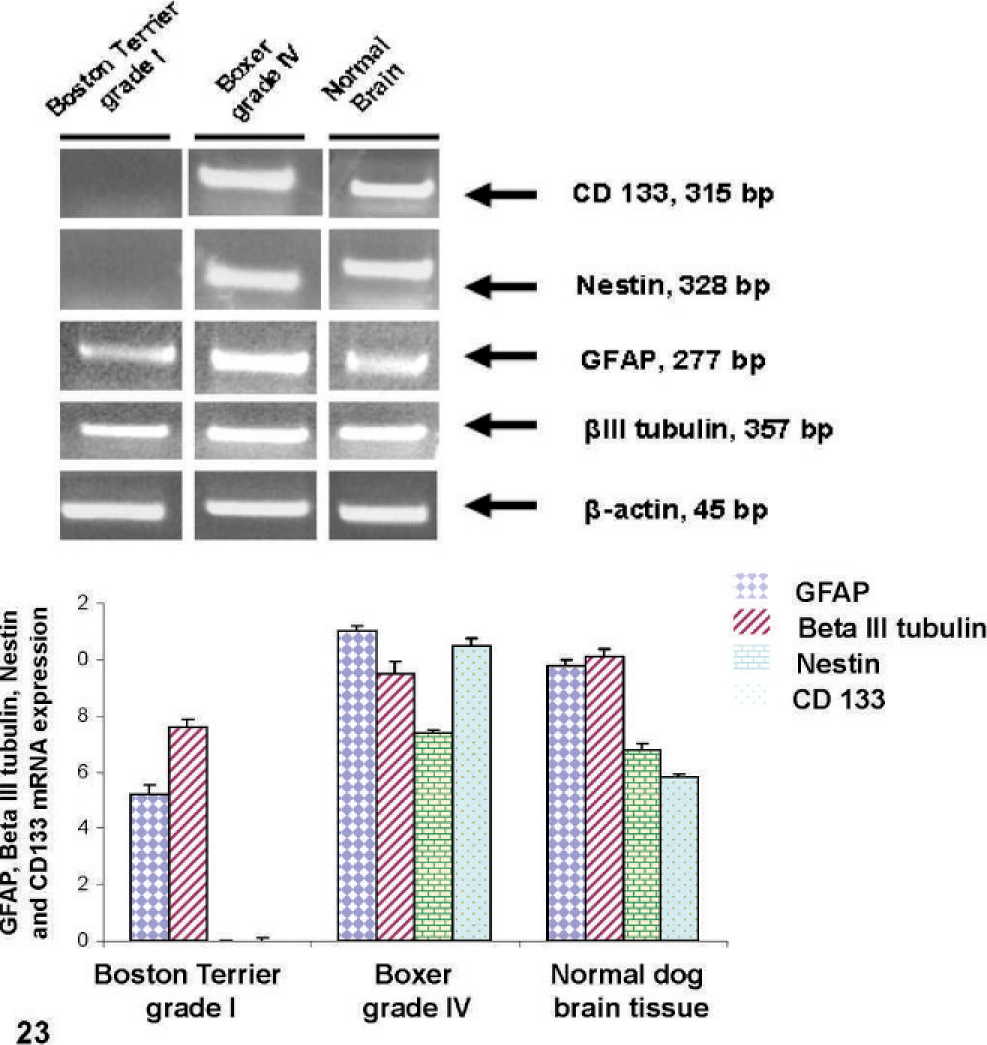
Gene expression profile of dog brain tumor, pilocytic astrocytoma (PCA), grade I and glioblastoma multiforme, grade IV as analyzed by RT-PCR. Normal dog brain was used as a control. Total RNA was extracted from primary cell culture of a Boston Terrier brain grade-I PCA and Boxer grade-IV D-GBM cloned line, and from normal dog brain tissue. Higher levels of CD133, nestin, βIII tubulin, and GFAP were found in Boxer grade-IV D-GBM compared with Boston Terrier grade-I PCA. CD133 and nestin mRNA were not detected in Boston Terrier grade-I brain tumor. β-actin served as a loading control. Quantitative analysis was performed by densitometry. RT-PCR was performed 3 times on each sample, and the results represent the mean ± SD.
D-GBM and xenografts express VEGF and VEGFR2
Among VEGF receptors, VEGFR-2 is responsible for the mitogenic signaling and the proliferative response of cells. As such, it has been thought to be the principal receptor for VEGF signaling. 50 VEGFR2 (Flk-1) showed immunolabeling of the endothelial cells and neoplastic cells primarily around the new forming blood vessels (Figs. 24–27). Double immunostaining for Factor VIII and VEGF showed that VEGF was expressed in nude mice D-GBM tumor xenograft (Fig. 28). RT-PCR demonstrated the expression of VEGF and VEGFR2 on D-GBM cells (Fig. 29).
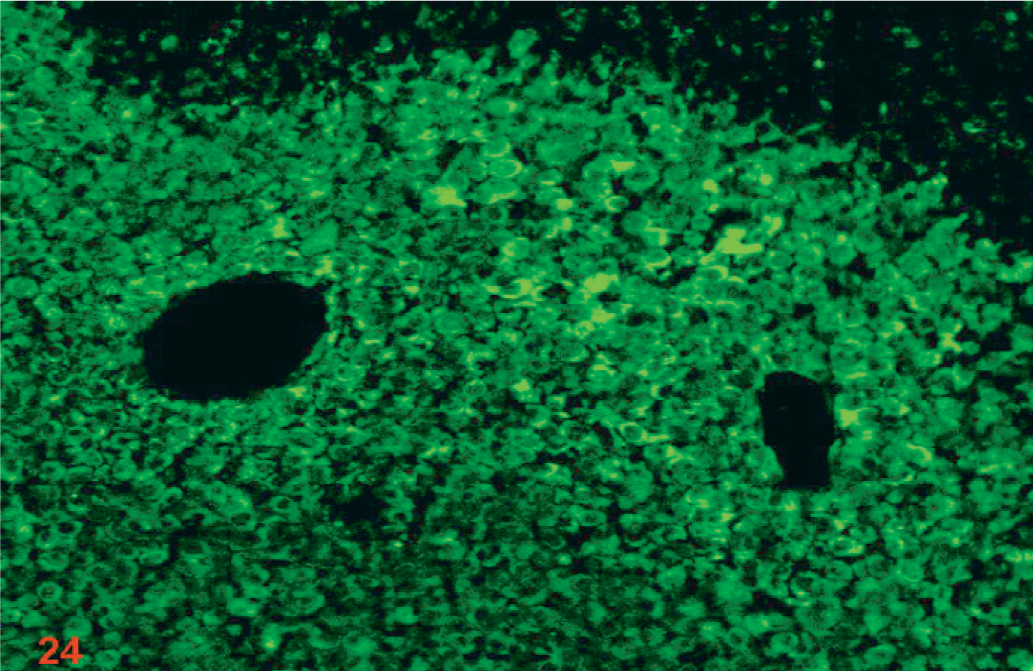
Nude mouse brain xenograft inoculated intracranially with D-GBM cells showing diffuse VEGFR2 immunofluorescent labeling of cells. Original magnification ×10.
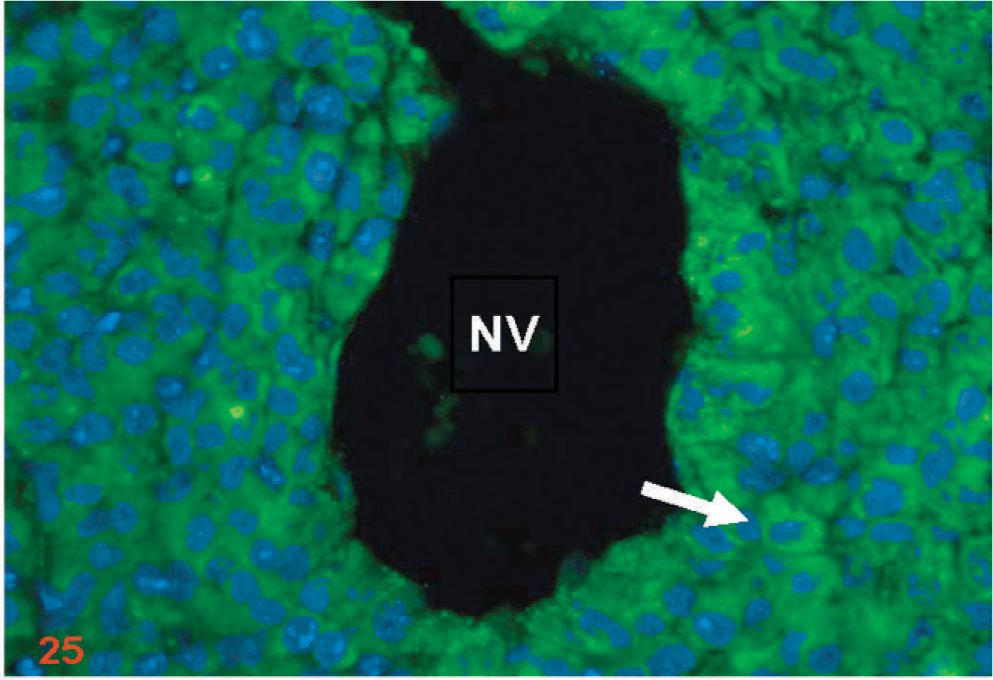
Nude mouse brain xenograft inoculated intracranially with D-GBM cells showing VEGFR2 immunofluorescent labeling of cells (arrow) around the neovessels (NV). Cell nuclei are indicated by DAPI staining (blue). The photograph is a high-power view of Fig. 24 (original magnification ×20).

Nude mouse brain xenograft inoculated intracranially with D-GBM cells showing cell nuclei as indicated by DAPI staining (blue). This represents the negative control slide where primary antibody was omitted and only secondary antibody and DAPI were used.

Nude mouse brain xenograft inoculated intracranially with D-GBM cells showing some background level of the secondary antibody staining (green). This represents the negative control slide where primary antibody was omitted and only secondary antibody and DAPI were used.
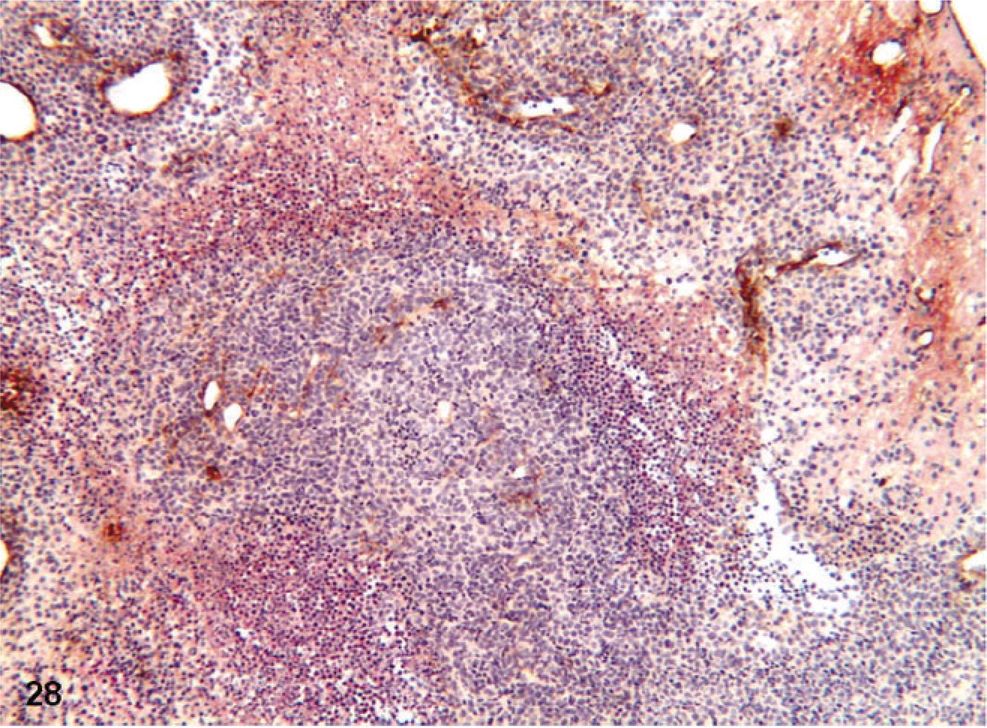
Double labeling immunohistochemistry for VEGF and Factor VIII of D-GBM. D-GBM nude mouse xenograft showing VEGF immunolabeling (red) and Factor VIII (brown).
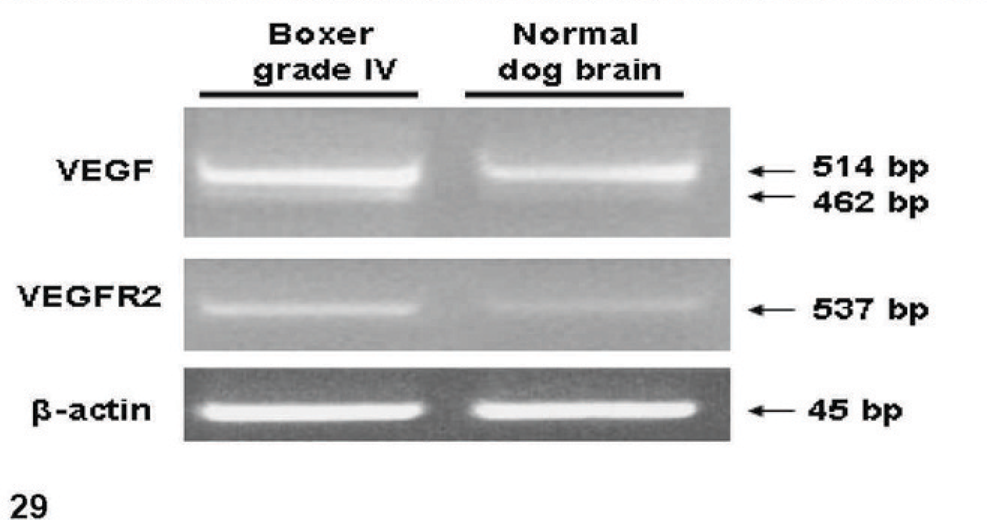
Semiquantitative RT-PCR was used to detect VEGF, and VEGFR2 in total RNAs extracted from Boxer grade-IV D-GBM cells and normal dog brain tissue. β-actin was used as an internal control. For VEGF, 2 alternative splicing variants were identified. A 462 bp product, corresponding to the VEGF164 isoform, and a second 514 bp product corresponding to the VEGF188 isoform were detected. For VEGFR2, a 537 band was detected. Each PCR sample was run in triplicate.
Expression of integrins in dog glioblastoma
The RT-PCR analysis of integrins was performed in D-GBM (grade IV) cells isolated from Boxer, and pilocytic astrocytoma (PCA, grade I, WHO classification) primary culture isolated from a Boston Terrier. Our results demonstrate that grade-IV D-GBM expresses higher levels of α-5 integrin compared with grade-I PCA tumor (see Fig. 30). Alpha-1 integrin was not detected in grade-IV glioblastoma. Beta-3 integrin expression was higher in grade-IV glioblastoma compared with grade-I PCA.
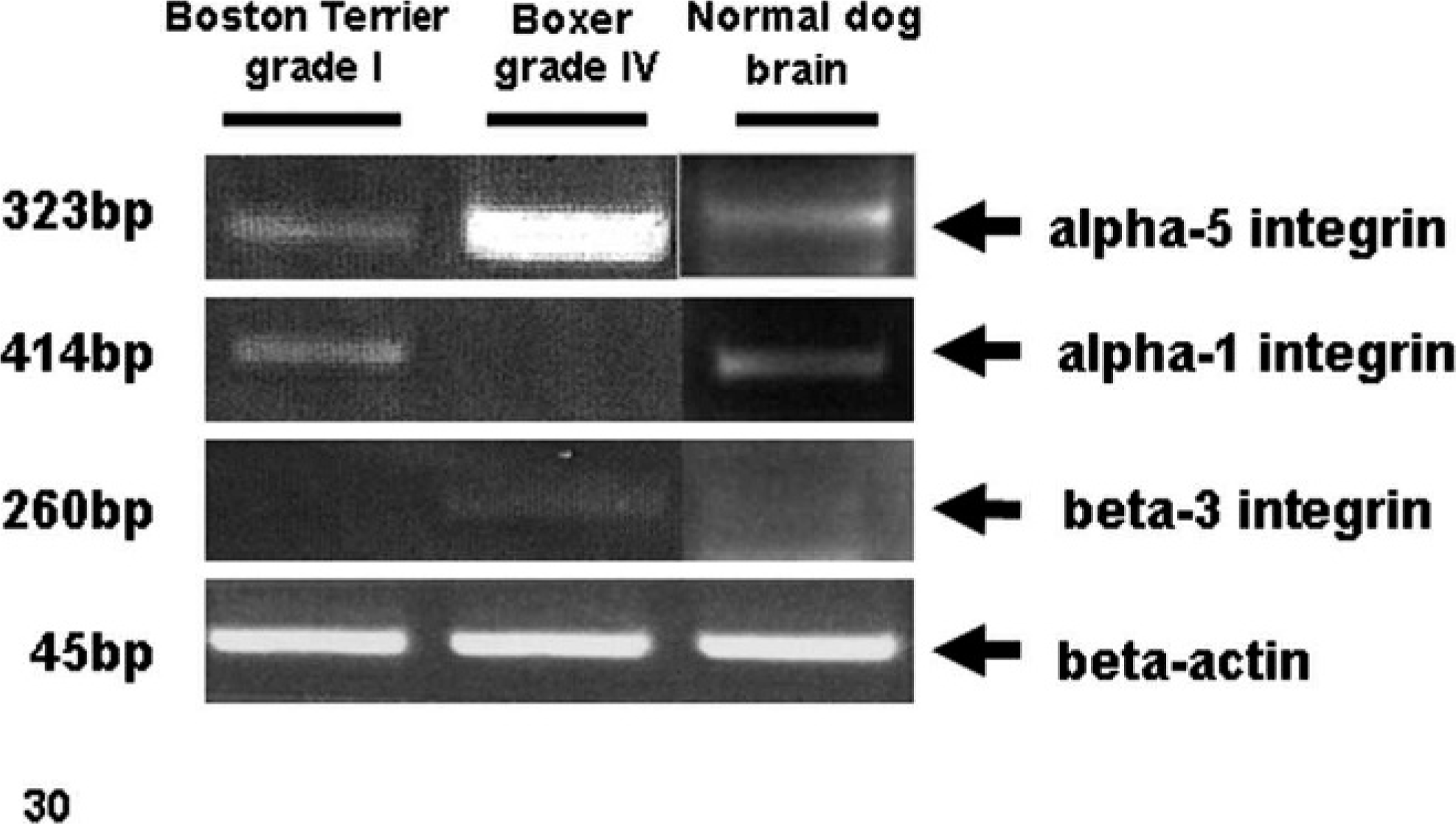
Differential integrin expression in dog brain gliomas grade I and IV as analyzed by RT-PCR. Normal dog brain was used as a control. Total RNA was extracted from primary cell culture of a Boston Terrier grade I, PCA and Boxer D-GBM, grade IV, and from normal dog brain tissue. Boxer D-GBM, grade IV expresses higher levels of alpha-5 integrin compared with Boston Terrier PCA, grade I and normal brain tissue. Alpha-1 integrin was not detected in grade IV D-GBM. Beta-3 integrin level was higher in Boxer grade IV D-GBM and was not detected in grade I PCA and normal brain. β-actin served as a loading control.
Hypoxia is a factor in D-GBM cell migration
Matrigel migration assay was performed to evaluate the migration potential of D-GBM cells. Neurosphere cells were used for this procedure. Our data demonstrate a significant increase in tumor cell migration when kept under hypoxic conditions compared with cells kept under normoxic conditions (see Fig. 31).
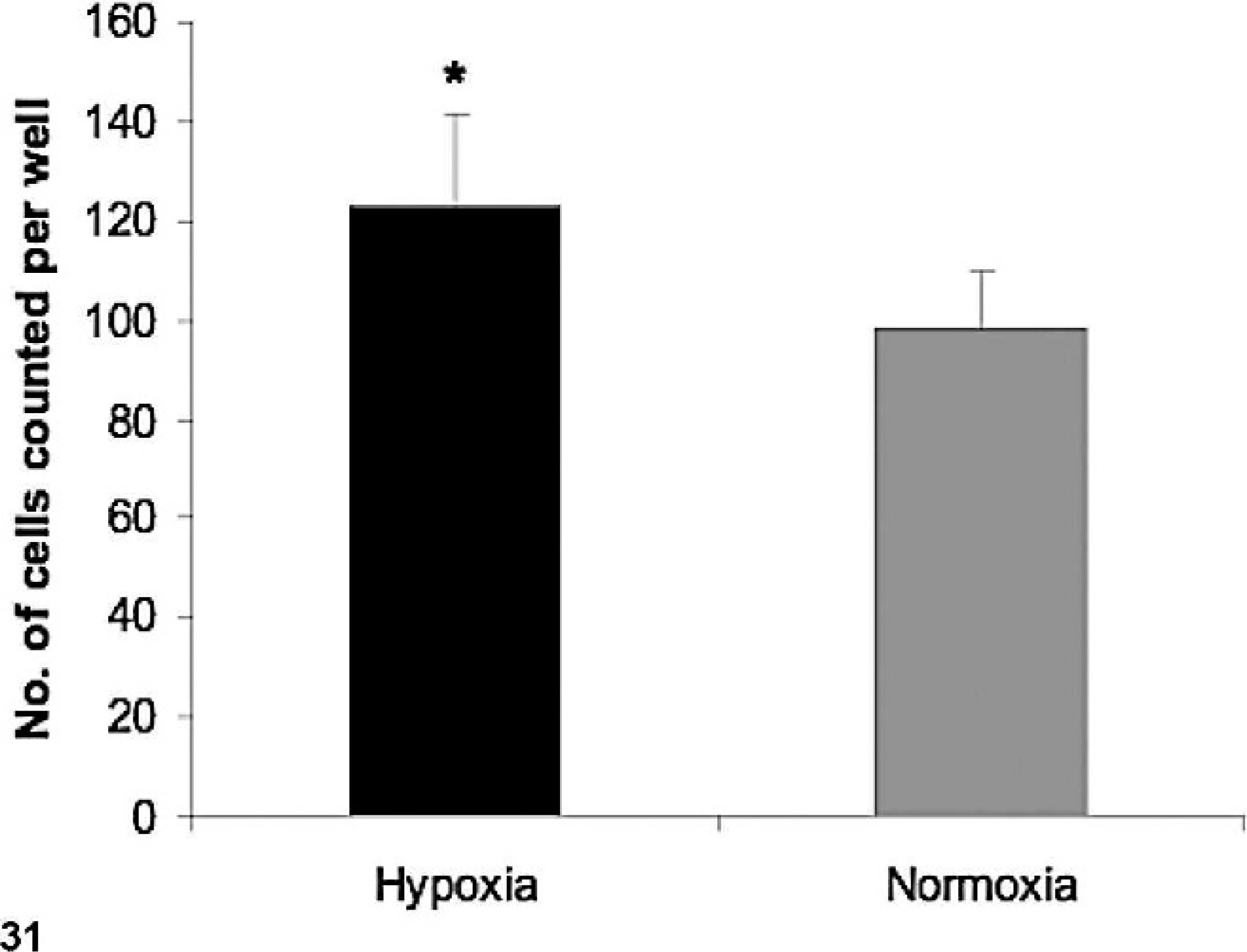
Migration potential of hypoxic dog GBM grade IV. An in vitro invasive chamber assay was performed as described in Materials and Methods section. The results shown in the histogram are the mean ± SD from 3 independent experiments; (∗) indicates a statistically significant difference (P < .05).
Discussion
GBM is the most common and lethal primary malignant brain tumor in human and dog. GBM is a nonmetastatic tumor, but highly locally invasive, diffusely disseminating into the brain parenchyma and placing cancerous cells outside the margin of therapeutic intervention. 21, 42
The GBM infiltrative path into the normal brain is not random; it often follows white matter tracts and extends along perivascular spaces, the glial limitans externa, and the subependyma. 28 Their diffuse infiltration suggests the activation of genetic and cellular programs that distinguish them from cells in the tumor core. Additionally, migrating tumor cells may activate the host brain microenvironment to facilitate tumor dispersal.
The expression of CSCs in D-GBM cloned cell culture was demonstrated by flow cytometry analysis and immunohistochemistry. The neurospheres expressed positive immunolabeling for nestin and CD133, which are considered stem cell markers. D-GBM maintained in the DMEM medium with 10% FBS demonstrated neuronal and astrocytic cell differentiation capacity (Figs. 15–17). The differentiated attached tumor cells lost their CSC markers, nestin, and CD133.
The D-GBM nude mouse xenografts clearly demonstrated the pattern of brain invasion by the CSCs. The inoculated tumor cell migration pathways followed the white matter of the corpus callosum, glial limitans of lateral ventricles, and the pial surface, and also perivascular venue (Fig. 13) as described in human gliomas. 28 The CD133+ and nestin-expressing tumor cells were at the migration front in the nude mouse brain white matter inoculated with D-GBM cells (Figs. 18,19). The nude mouse xenografts of D-GBM also demonstrated the differentiation capacity of the CSCs. Besides CD133+ and nestin, tumor cell xenografts showed various degrees of immunoreactivity for CD34, (hematopoietic progenitor and stem cell marker), βIII tubulin (neuronal marker), and GFAP (an astrocyte lineage marker), which imply a multi-lineage differentiation capacity of D-GBM in vivo (Figs. 20–22). The differentiation capacity of D-GBM was also supported by RT-PCR results.
The immunolabeling pattern for VEGF shows a widespread distribution on the tumor cells xenograft. The VEGFR2 labeled cells in the mouse xenograft are mostly located around sites of new blood vessel formation (Figs. 24,25). RT-PCR assay also demonstrated that D-GBM produces VEGF. This preliminary finding is interesting and suggests that dog glioblastoma cancer stem cells might reside in a vascular niche as has been hypothesized by others in human glioblastoma 8 and mouse neural stem cells. 36 Our data demonstrate that D-GBM xenografts from nude mice showed an excessive vascularization pattern. These neovessels were incompletely formed, hyperdilated, and had irregular size and shape. Tumor cells appear to cluster and remain in close contact with the endothelial cells of the new forming vessels (Figs. 10–13). It is suggested that tumor cells can be incorporated into the growing vascular bed and contribute to tumor angiogenesis. In the brain, the formation of multiple vascular CNS niches, each with the capacity to generate numerous tumor cells, might significantly facilitate brain tumor growth and invasion. If the niche hypothesis is correct, then targeting these microenvironments could prove a highly effective, novel therapeutic approach to glioblastomas.
GBM is one of the most lethal primary brain tumors in human and animal species. This tumor is composed of a phenotypically heterogeneous cell population. 42, 23 The mechanisms responsible for the GBM heterogeneity and the ability of tumor cells to migrate into the brain parenchyma are incompletely understood. Localized hypoxia appears to upregulate migration-associated genes, leading to migration of tumor cells away from a central hypoxic center. Necrosis is a central feature of the highest-grade malignant gliomas. No histologic feature is more powerful in predicting poor prognosis. 23 There is a vicious cycle involving necrosis-induced hypoxia and various angiogenic and growth factors that combine to foster the highly malignant state of glioblastoma. In the setting of rapidly dividing cells with high metabolic demands, small regions of necrosis may develop in areas where metabolic demands exceed supply. Another recently suggested possibility is that in response to hypoxia, small clones of tumor cells acquire molecular characteristics that encourage more active migration, thus clearing a central region more susceptible to necrosis. This hypothesis is based on work showing that glioblastoma tumor cells surrounding necrotic centers (pseudopalisades) are less proliferative and more apoptotic than adjacent cells. 4, 5 These perinecrotic cells also express hypoxia-inducible genes, such as hypoxia-inducible factor 1 alpha (HIF-1α), 4 and in vitro studies have demonstrated that hypoxia increases cellular migration and gelatinase activity. 30 When selected molecules that are preferentially expressed in palisading cells are used as prognostic markers in glioblastomas, they correlate with shorter patient survival. 11 This finding supports the notion that these perinecrotic regions are zones in which more malignant clones are actively selected. In addition, the interactions between hypoxia, necrosis, upregulation of growth factors, and selection of malignant clones might provide an explanation for the marked histologic heterogeneity noted in glioblastomas and a possible explanation for the marked resistance to conventional cytotoxic therapies seen in clinical patients with glioblastoma. 28
Tumor progression leading to metastasis or invasion appears to involve equipping cancer cells with the appropriate adhesive (integrin) phenotype for interaction with the ECM. The integrins constitute a family of transmembrane receptor proteins composed of heterodimeric complexes of noncovalently linked alpha and beta chains. Integrins function in cell-to-cell and cell-to-ECM adhesive interactions and transduce signals from the ECM to the cell interior and vice versa. 32 Previous studies reported the expression of various integrins in brain tumors. 34– 38 There have been few investigations of the expression of αvβ5 and αvβ3 in gliomas. Few data are available regarding the relationship between the expression of these integrins and angiogenesis and invasion in this type of tumor. Two αv integrins, αvβ3 and αvβ5, are necessary for cytokine- or tumor-induced angiogenesis. They mediate 2 distinct pathways of angiogenesis—expression of αvβ3 is required for fibroblast growth factor 2 (FGF-2) and tumor necrosis factor α-induced angiogenesis. The integrin αvβ5 is required for VEGF- and transforming growth factor β-induced angiogenesis. 12 Hypoxia has been reported to stimulate expression of integrins αvβ3 and αvβ5 in retinal microvascular endothelial cells and to inhibit expression of integrin β1. The hypoxic induction of the αv integrin is partially mediated through VEGF induction in an autocrine-paracrine manner. 44 The angiogenic integrins have been reported to not only mediate vascular cell migration but also to regulate metalloproteinase activity and cell proliferation. These specific increases in the tumor cellular integrin expression may contribute to neovascularization and invasion observed in dog glioblastomas. Therapies directed at influencing integrin expression and function are presently being explored for inhibition of tumor growth, invasion, and angiogenesis. However, further studies are needed to elucidate the mechanisms by which hypoxia and VEGF regulate integrin functions in glioblastoma. It appears that invasive glioblastoma cells re-activate developmental migration mechanisms to affect motility and that the interaction between CSCs and the microenvironment plays an important role in glioma invasion.
Germinal regions, such as the subventricular zone (SVZ), have long been proposed as sources of gliomas. Many gliomas are either periventricular or contiguous with the subventricular zone, and they frequently express the progenitor-cell markers nestin and CD133. 2, 25 Recent studies suggest that human brain tumors are organized as a hierarchy and are maintained by a small number of tumor cells that have stem cell properties. 10 Based on the anatomic location of the D-GBM isolated from a Boxer dog, intuitively we believe that the cells of origin for this glioblastoma multiforme most likely reside with the neural stem cells from the subependymal lateral ventricle region. Histologic observation derived from this tumor and others that were diagnosed in our laboratory demonstrated a similar intraventricular or periventricular location, and fragments of ependymal lining cells were described within the tumor. In addition, the migration pattern in nude mouse xenografts is highly suggestive that D-GBM has a homing property directing migration from the site of implantation to the ventricular region. However, this observation is just correlative. If the cell of origin for this type of tumors can be demonstrated to be derived from SVZ neural stem cells it will have a great impact on the future classification of brain tumors and novel therapeutic modalities.
In conclusion, we have demonstrated for the first time the presence of CSCs in dog brain tumors. Understanding the basic mechanisms underlying glioma cell invasion into the brain parenchyma requires improved and diverse experimental models of glioma invasion. The spontaneous dog glioma appears as an excellent animal model for investigating cancer stem cell invasion. A dog glioma model has a higher incidence and less restrictive rules than in humans. The invasive nature of glioma cells is the obvious obstacle to effective therapy, making this issue of great significance.
Footnotes
Acknowledgements
This study was supported in part by Canine Health Foundation (Grant no: 0800587). We would like to thank Cynthia J. Meininger for critical reading of the manuscript. The authors would also like to thank Andy Ambrus and Sarah Jones for assistance with immunohistochemistry analysis and histology. Also, thanks to Kent Claypool for flow cytometry analysis and Xuie Xie for technical assistance on nude mice inoculation and necropsy.
