Abstract
Mycobacterium avium subsp. paratuberculosis (MAP) is the causative agent of chronic enteritis in ruminants (Johne's disease) and a possible etiopathologic agent in human Crohn's disease. The host-pathogen interaction in this chronic disease has largely depended on the randomly collected static lesions studied in subclinically or clinically infected animals. We have established and utilized the neonatal calf ligated ileal loop model to study the early temporal host changes during MAP infection. After inoculation of ligated ileal loop with MAP, samples were analyzed for bacterial invasion, histologic and ultrastructural morphologic changes, and gene expression at several times (0.5–12 hours) postinfection. Our results indicate that MAP invades the intestinal mucosa as early as 0.5 hour postinoculation. Distribution and migration of neutrophils, monocytes/macrophages, and goblet cells were confirmed by histopathology, scanning and transmission electron microscopy. Coincident with the morphologic analysis, we measured by real-time polymerase chain reaction gene expression of various cytokines/chemokines that are involved in the recruitment of mononuclear and polymorphonuclear leukocytes to the site of infection. We also detected expression of several other genes, including intestinal-trefoil factor, profilin, lactoferrin, and enteric β-defensin, which may play significant roles in the early MAP infection. Thus, the calf ligated intestinal loop model may be used as a human disease model to understand the role of MAP in the pathogenesis of Crohn's disease.
Mycobacterium avium subsp. paratuberculosis (MAP) is the causative agent of “Johne's disease” or paratuberculosis, a chronic inflammatory bowel disease in ruminants that results in extreme wasting, loss of productivity, and death of the host. This bacterium has been documented to survive pasteurization, is transmitted in milk, and has been detected in intestinal tissues in a subset of patients who suffer from the human inflammatory bowel syndrome known as Crohn's disease. 5,13,28 Natural progression of MAP infection in cattle is associated with intestinal lesions developing during a prolonged subclinical period (from 2 to 5 years). Progress on understanding the early pathogenesis of MAP infection has been slow due to the extreme difficulty of culturing the organism (3–4 months), and the time required to consistently produce clinical disease after an experimental infection (about 2 years) in ruminants. Diagnosis of MAP is also confused by the close relationship of this organism with M. avium subsp. avium (MAA), which shares many antigenic features but is rarely associated with disease in ruminants. 8 Several laboratory animal models have been developed for MAP infection (extensively reviewed by Harris and Barletta 12 ). The experimental laboratory animal models are somewhat limited in their use for studying MAP infection, because they do not consistently reproduce the disease signs or lesions. For example, C57/B6 and BALB/c mice have been used as an experimental model to study MAP infection. 4,34 However, the disease in this model was predominantly systemic as opposed to intestinal; clearly indicating that it is not a valid model of ruminant MAP infection.
Dysregulation of host immune responses have been proposed to be responsible for the progression of Johne's disease. 3,30 However, early mechanism(s) leading to damage of intestinal tissue as a result of MAP infection are not known. 14 The ligated intestinal loop model was previously utilized to study the pathogenesis of Salmonella typhimurium–induced diarrhea in the neonatal calf. 10,37 Unlike the acute secretory diarrhea and neutrophilic inflammatory response of cattle to Salmonella typhimurium, MAP requires years for the onset of clinical signs associated with the development of chronic granulomatous enteritis. Most clinically important enteric pathogens initiate the disease by invading and traversing through the intestinal epithelium, accompanied by increased expression of pro-inflammatory cytokines and/or chemokines that attract the specific inflammatory cellular populations to the site of infection. This triggering mechanism is generally very rapid and based upon the type of pathogen; the stimulation may be transient or sustained in nature. Thus, to more fully understand the involvement of chemokines/cytokines during the early phases of MAP infection, we analyzed the gene expression profiles of C-C and C-X-C chemokines, pro-inflammatory, and anti-inflammatory cytokines. In addition, we also analyzed the expression of specific host genes involved during the initial invasion by MAP (profilin), genes responsible for the structural integrity of the host cells (trefoil factor), and antimicrobial peptides (enteric β-defensin and lactoferrin). Sequential analysis of the alterations in morphology and gene expression responses during MAP invasion using the neonatal calf ligated intestinal loop model has predicted new insights in the molecular pathogenesis of Johne's disease.
Materials and Methods
Bacterial inoculum
Mycobacterium avium subsp. paratuberculosis strain 19698 was grown in 7H9 broth (Difco Laboratories, Detroit, MI) supplemented with 2.5% (vol/vol) glycerol (Sigma Chemical Co., St. Louis, MO), oleic acid-albumin-dextrose-catalase (Difco Laboratories), 0.05% Tween 80 (Sigma Chemical Co.), and 2 mg of Mycobactin J (Allied Monitor, Inc., Fayette, MO). Single-cell suspensions of MAP and counting of the organisms were performed as described earlier. 17
Animals and surgical procedures
Four male Holstein calves, 3–4 weeks of age, weighing 45–55 kg, were used under an approved animal use protocol in accordance with animal use policy under the supervision of the Texas A&M University Institutional Animal Care and Research Advisory Committee. Calves were fed antibiotic-free milk replacer twice daily and water ad libitum. The calves were clinically healthy before the experiments. Calves were fasted for 24 hours prior to the surgery, anesthetized, and maintained analgesic for the course of the 12-hour experiment. For the nonsurvival surgery to prepare ligated loops of ileum and jejunum, 37 anesthesia was induced with Propofol (Abbot Laboratories, Chicago, IL) followed by placement of an endotracheal tube and maintenance with isoflurane (Abbot Laboratories). The abdominal wall was clipped, and prepared aseptically with chlorhexidine and isopropanol prior to opening. Sterile drapes were used as a barrier, the right abdominal wall was incised, and the entire length of the Peyer's patch of the distal jejunum and ileum exteriorized. Segments of the distal jejunum and ileum (of 6–8 cm length) were ligated with umbilical tape leaving about 1- to 2-cm area as an inter-loop space. At time zero, the length of each loop was measured, and then injected with 3 × 109 colony-forming units (CFUs) of MAP contained in 3 ml of sterile phosphate buffered saline (PBS), using a three-eighths–inch 26-gauge needle. Likewise, 3 ml of sterile PBS was inoculated into the control loops. The loops were replaced into the abdominal cavity, and the incision temporarily closed with Backhaus towel clamps. Dextrose (2.5%) in 0.45% normal physiologic saline at 5 ml/kg/hour was infused intravenously to maintain circulating blood volume. At 0.5, 1, 2, 4, 8, and 12 hours after inoculation, one infected and one control loop were excised. Electrocautery was used to control hemorrhage after excision of loops. Throughout the experimental procedure, the calves were monitored for vital signs (blood pressure, heart rate, status of hydration, degree of anesthesia, and temperature). Tissue samples were collected for quantitation of tissue-associated bacteria, histopathology, ultrastructural studies, and RNA extraction. After the loops were excised, each loop was opened, luminal fluid collected, and the total volume measured in a 50-ml centrifuge tube, and calculated as ml/cm. After the final loops were excised at 12 hours, the calves were euthanized with a rapid overdose (single bolus at 132 mg/kg IV) of pentobarbital sodium.
Quantitation of tissue-associated MAP
A 6-mm biopsy punch was used to collect 2 tissue samples from Peyer's patches for quantitation of tissue-associated bacteria. Intestinal tissue samples were washed 3 times in PBS, weighed, homogenized in PBS, serially diluted, and plated onto Herrold egg yolk Media containing Amphotericin B nalidixic acid and vancomycin (ANV) (Becton Dickinson and Company, Sparks, MD) for incubation at 37°C. The cultures were visually observed weekly for any contamination, and the final counts of CFUs were recorded after 16 weeks of incubation.
Histopathology and acid-fast staining
For histopathologic analysis, formalin-fixed intestinal tissues were processed by paraffin embedding, and 2- to 4-µm thick sections were cut, mounted, stained with HE, and the Ziehl-Neelsen (ZN) method for the identification of acid-fast bacilli. Morphologic evaluation of infected loops was assessed by histopathologic examination and compared with PBS control loops. Two histopathologists were blinded to the treatment group prior to tissue assessment.
Scanning electron microscopy
For scanning electron microscopy (SEM), 1- to 2-mm fragments of mucosa were prepared and were fixed in the solution of freshly made 5% glutaraldehyde and 4% paraformaldehyde in 0.1 M sodium cacodylate buffer at 4°C. The fixed samples were dehydrated in a graded series of ethanol solutions from 10 to 100%, and dried in hexamethyldisilazane (HMDS), a substitute for critical point drying. Dried samples were mounted on aluminum stubs with double-sided sticky carbon tape. The specimens were coated with a 20-nm layer of gold-palladium using an ion-sputter coater (Hummer model from Technics, Inc., Alexandria, VA), examined, photographed, and digitized using a JEOL JSM-6400 scanning electron microscope at an accelerating voltage of 15 kV.
Transmission electron microscopy
For transmission electron microscopy (TEM), small 0.25- to 0.5-mm fragments of intestinal mucosa were fixed in same solution as used for SEM. After overnight fixation, tissues were then washed 3 times with 0.1 M sodium cacodylate buffer, postfixed for 2 hours at 4°C in 1% osmium tetroxide in 0.1 M sodium cacodylate buffer, and then stained overnight at 4°C in saturated uranyl acetate solution. Tissues were dehydrated in a graduated series of ethanol solutions and propylene oxide, and then embedded in Epon Araldite. Sections (0.5 µm) were stained with toluidine blue and examined under a light microscope for selection of the microscopic fields. The blocks were trimmed, and thin sections (60–90 nm) were cut, mounted on copper grids, stained with uranyl acetate and lead citrate, and examined with a Zeiss 10 C transmission electron microscope.
Real-time polymerase chain reaction for analysis of gene expression
Total RNA was immediately extracted from the excised ileal mucosa using Tri-reagent (Molecular Research Center, Cincinnati, OH). The ileal tissue was finely minced with a scalpel blade, and then homogenized with a battery-powered motorized plastic pestle in 1.5-ml Eppendorf tubes resulting in a milky suspension. Thereafter, the protocol for RNA isolation followed the procedure recommended by the manufacturer (Molecular Research Center). In brief, 0.2 ml of chloroform was added to the minced tissue (in 1 ml of Tri-reagent) and mixed very gently. This suspension was kept at room temperature for 15 minutes. The suspension was spun at 12,000 × g for 20 minutes at 4°C. The aqueous layer was removed and equal volume of isopropanol was added. This suspension was stored at −20°C for 2 hours. The suspension was again spun at 12,000 × g for 20 minutes at 4°C. After decanting the suspension, the RNA pellet was washed with 70% ethanol. The final pellet was dried and resuspended in Nuclease-free water. The quality and quantity of extracted total RNA was estimated by UV spectrophotometer and electrophoresis on 1.0% native agarose gels. The RNA was treated with RNase-free DNase to obtain the high quality purified RNA at each of the time points after infection and stored at −80°C until used.
Gene expression was performed by real-time reverse transcription-PCR (Q-RT-PCR) in an Applied Biosystems 5700 DNA Sequence detection system (Applied Biosystems, Foster City, CA). RNA was converted into cDNA by adding 2 µg of total RNA to a 100-µl reaction containing 2.5 µM random hexamer and reagents from a reverse transcription kit from Applied Biosystems (Perkin Elmer Corp., Foster City, CA). The thermal cycle parameters were 10 minutes at 25°C, 30 minutes at 48°C, and 5 minutes at 95°C. The final working solutions diluted to 2 ng in 10 µl. Diluted cDNAs were stored at −80°C until used. Q-RT-PCR was performed with SYBR Green PCR Master Mix (Applied Biosystems). All primers were designed with Primer Express Software (Applied Biosystems) and synthesized by Sigma-Genosys (The Woodlands, TX). The list of primers used in this study is given in Table 1. For each primer pair, a negative control (water) and an RNA sample without reverse transcriptase (to determine genomic DNA contamination) were included as controls during cDNA quantitation.
Sequence of primers used in this study.
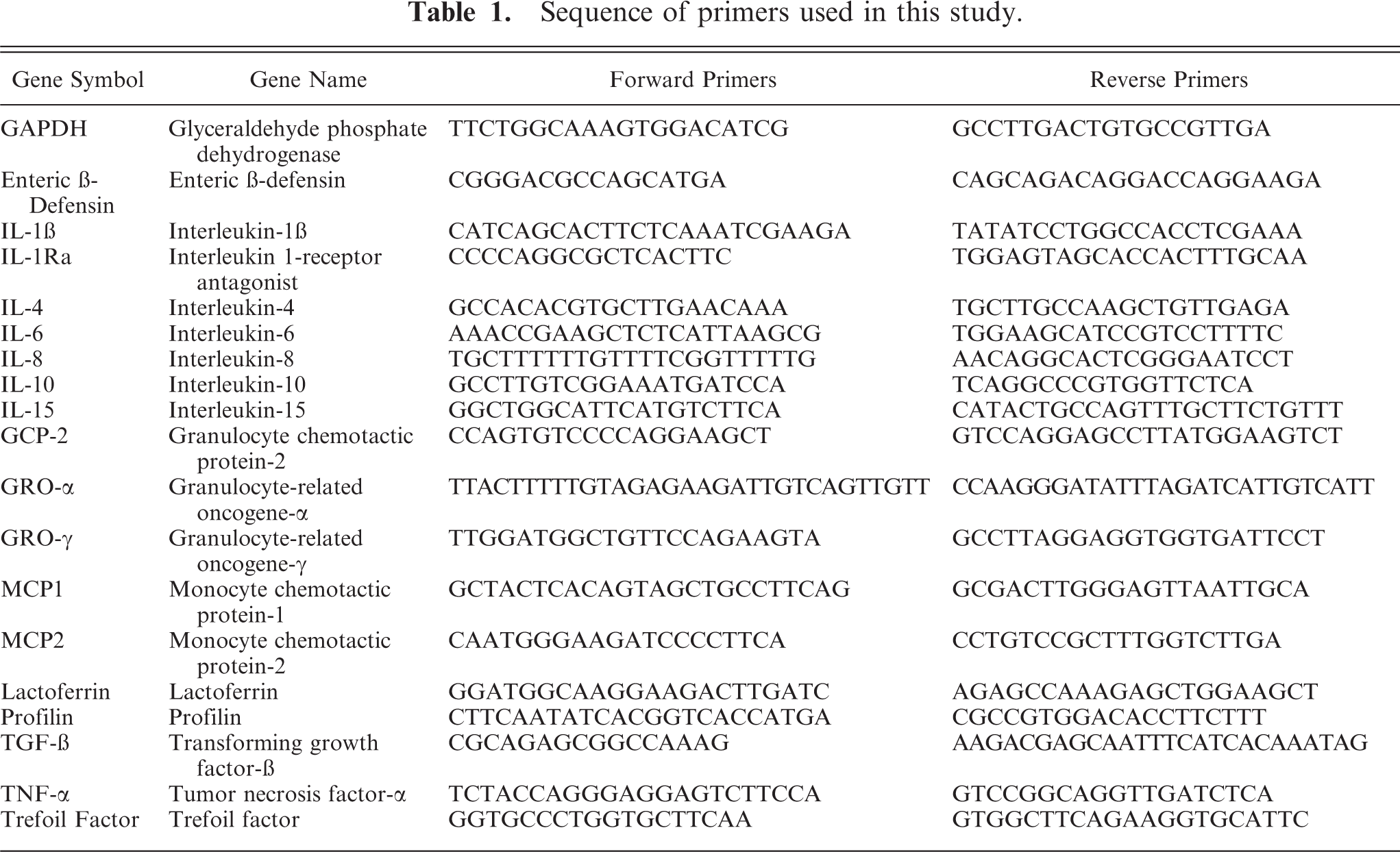
Analysis of real-time-PCR data was performed with the 2−ddCt method as described by Livak and Schmittgen 20 with glyceraldehyde phosphate dehydrogenase (GAPDH) serving as the control housekeeping gene. As per the ddCt real-time-PCR analysis method, cycle number was determined as the cycle at which fluorescence crossed a threshold above background (Ct). The resulting Ct value was recorded for each sample for genes of interest as compared with the control housekeeping gene. The delta-Ct value for each gene was obtained by subtraction of the Ct value from GAPDH. The delta-Ct for the control samples was subtracted from the delta-Ct of infected samples to obtain delta-deltaCt (ddCt). The fold change in the gene expression in infected tissues as compared with control was calculated by converting ddCt into 2−ddCt. The data were expressed as mean ± standard deviation (SD).
Statistical analysis
Colonization of tissue was considered to be positive when MAP was detected by bacterial culture. Tissue burden was defined as the number of CFUs per milligram of tissue. To test if there was a time effect in the expression of genes (in the infected loop) statistical significance was calculated using one-way analysis of variance (ANOVA) (with Newman Keuls test as a posttest) by applying the averaged data mean (fold change), SD, and number of samples in the Graphpad Instat software (GraphPad Software, Inc., La Jolla, CA).
Results
Bacteriology and histopathologic findings
There was no increase in the fluid volume of the ligated ileal loops infected with MAP as compared with control loops at any time after infection (0.5 to 12 hours postinfection). MAP were recovered from the MAP-inoculated ileal tissues at all the time points postinfection (Fig. 1). No bacteria were detected in the PBS inoculated loop, thus confirming that there is no leakage between the loops. No significant changes were found while comparing the invasion of MAP among the infected tissues at various times postinoculation (0.5–12 hours). Acid-fast bacteria were detected on the apical surface of villi (Fig. 2) at 1 hour postinfection and in the lamina propria at 12 hours postinfection (Fig. 3).
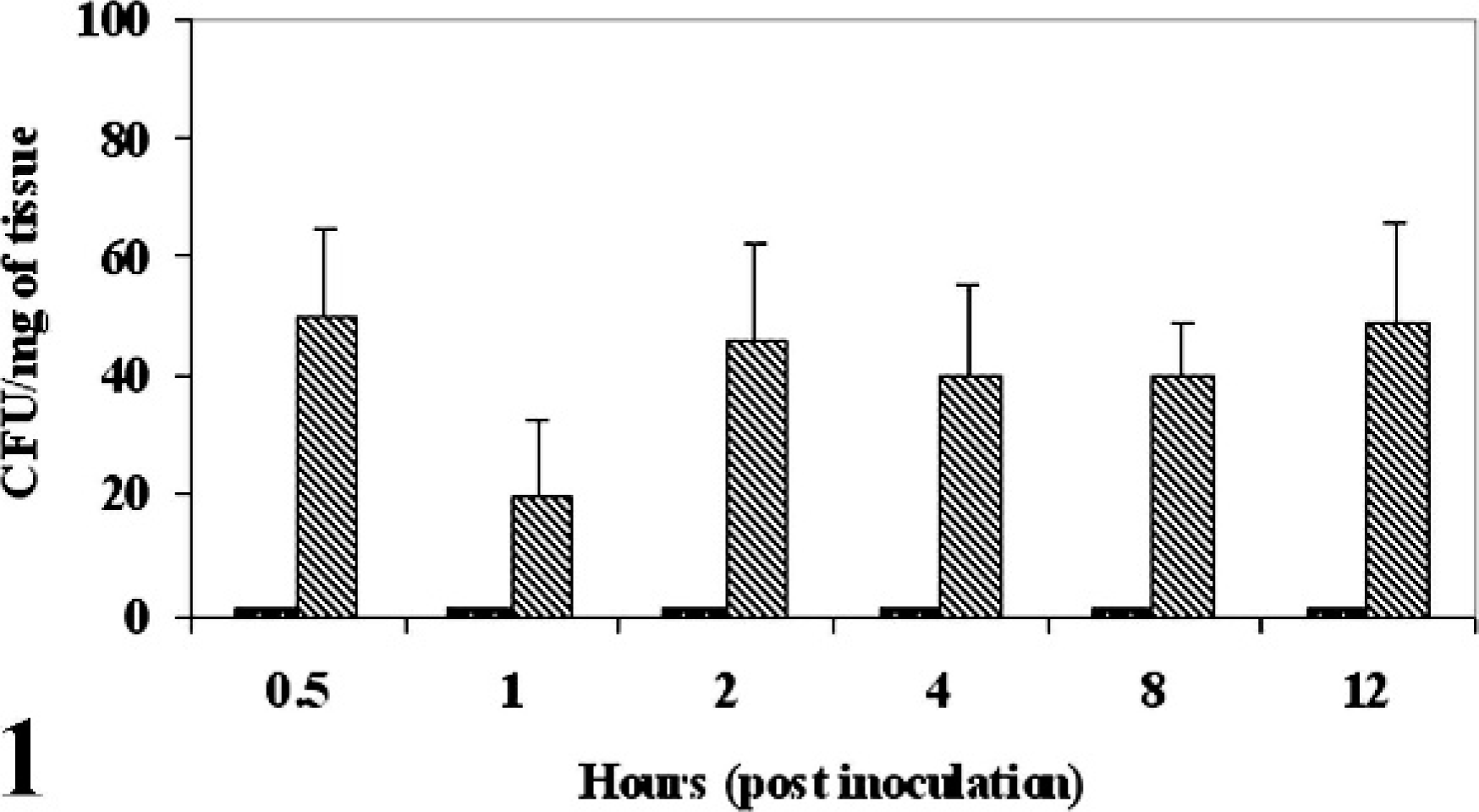
Sequential burden of MAP in infected bovine ileum. The bar graph (solid bar = control, cross-hatched bar = infected loops) indicates the mean ± standard deviation of bacterial colonies from 4 biological replicates counted after 16 weeks of incubation.
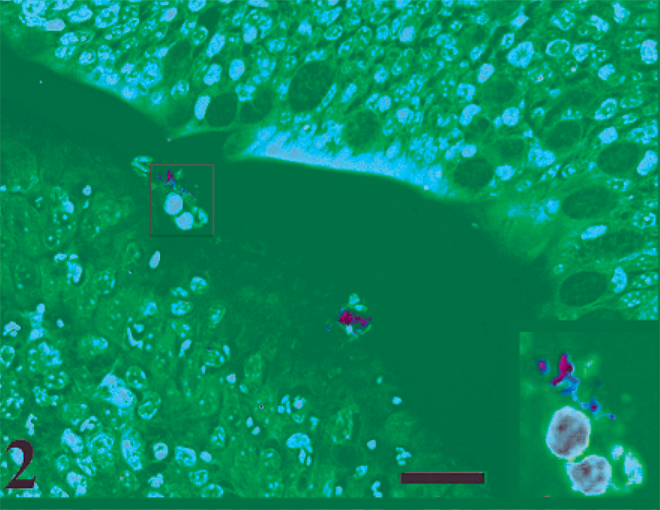
ZN-stained acid-fast bacilli in bovine ileum at 1 hour postinfection on the apical surface of villi. ZN stain. Bar = 20 µm.
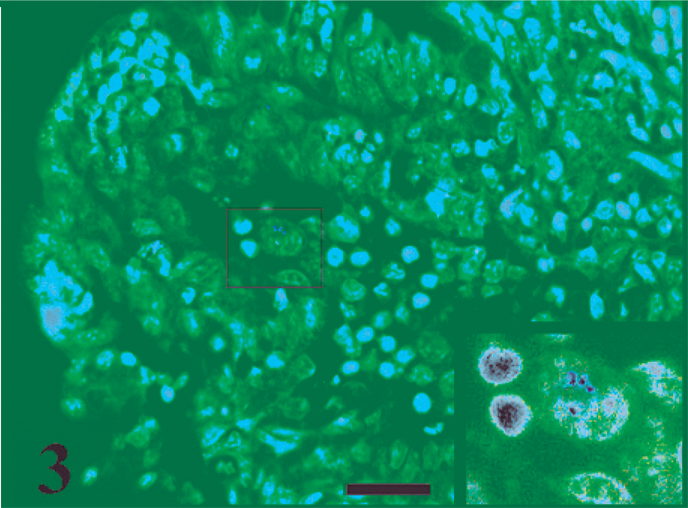
ZN-positive bacilli at 12 hours postinfection within a macrophage located in the lamina propria. ZN stain. Bar = 20 µm.
Macroscopic lesions were not observed at any time point. HE-stained ileal sections revealed sequential development of histopathologic changes that varied with the time of host–pathogen interaction (Table 2), while control loops had no significant histologic changes at any time point. The first histologic lesions detected in MAP-inoculated loops were characterized by migration of mononuclear cells into the lumen at 0.5 hour postinfection (data not shown). At 1 hour postinfection, ZN stain revealed acid-fast bacteria in the lumen surrounded by mononuclear cells (Fig. 4). At 2 hours postinfection, crypts containing viable and degenerate polymorphonuclear neutrophils (PMNs) admixed with karyorrhectic debris (crypt abscesses) were detectable which was mild initially, but became more pronounced at the later time points (8–12 hours postinfection) (Fig. 5). At 4 hours postinfection, a mild increase in the number of PMNs and mononuclear cells was observed in the lamina propria (Fig. 6). These mild changes became progressively more pronounced at the later time points (8–12 hours) with transepithelial and perivascular migration of PMNs and moderate-to-marked accumulation of PMNs and fibrin in the crypts and intestinal lumen. In some cases, the host-MAP interaction resulted in the formation of a fibrinous pseudomembrane in the intestinal lumen (Fig. 7). Goblet cell hyperplasia was observed at 12 hours postinfection, and blunting of the absorptive villi was detected early in some infected loops and then occurred in all infected loops at later time points.
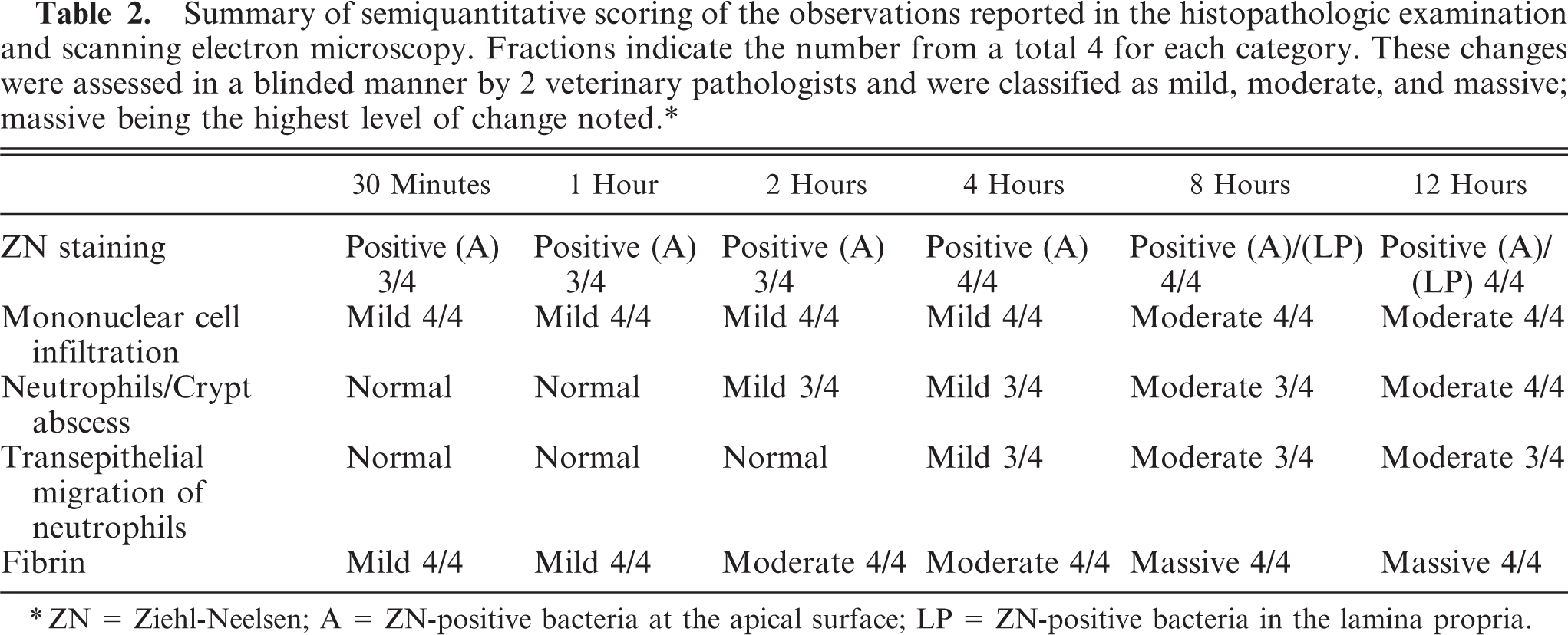
Summary of semiquantitative scoring of the observations reported in the histopathologic examination and scanning electron microscopy. Fractions indicate the number from a total 4 for each category. These changes were assessed in a blinded manner by 2 veterinary pathologists and were classified as mild, moderate, and massive; massive being the highest level of change noted.∗
∗ ZN = Ziehl-Neelsen; A = ZN-positive bacteria at the apical surface; LP = ZN-positive bacteria in the lamina propria.
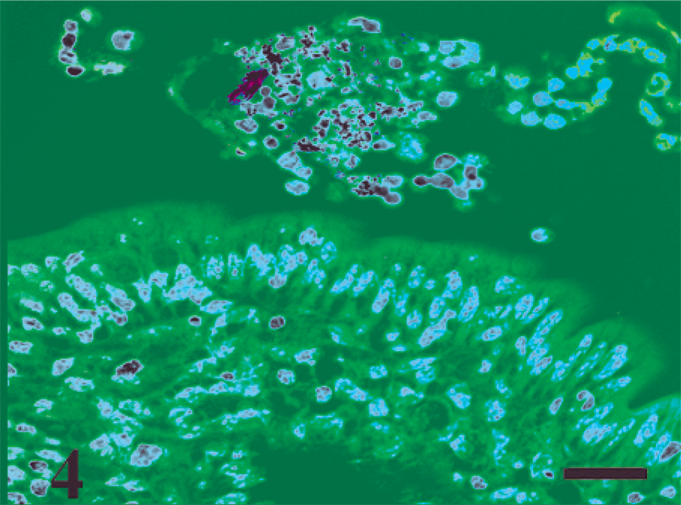
Mononuclear cells and ZN-positive bacteria in the lumen at 1 hour postinfection. ZN stain. Bar = 20 µm.
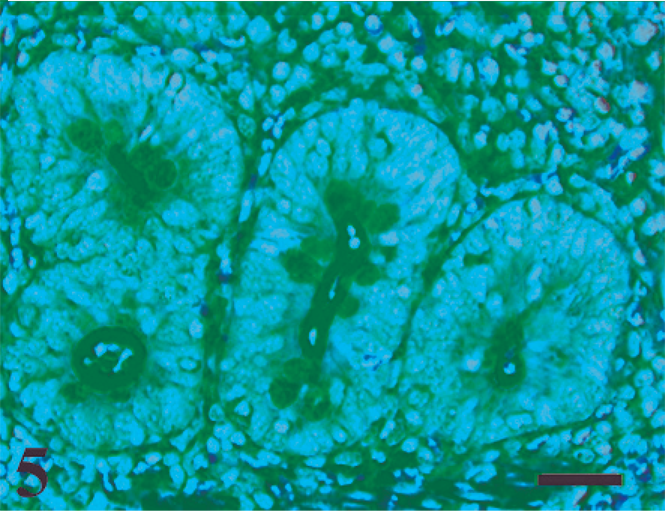
Crypt abscessation at 8 hours postinfection with a significant increase in the infiltration of polymorphonuclear leukocytes in the lamina propria. HE. Bar = 50 µm.
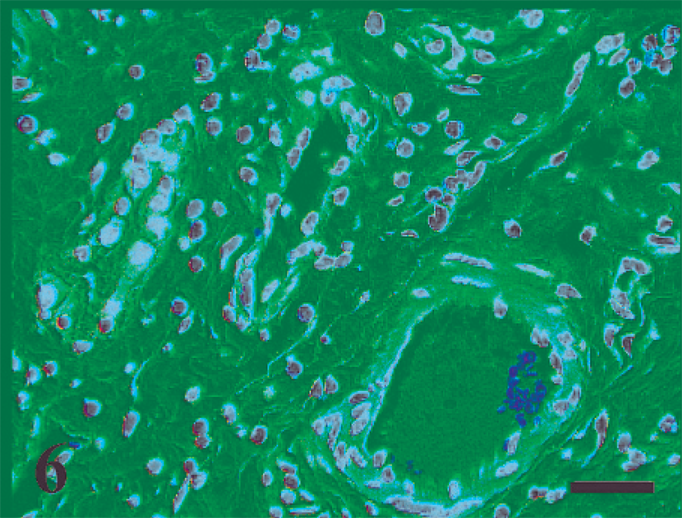
Perivascular migration of polymorphonuclear leukocytes (4 hours postinfection). HE. Bar = 40 µm.
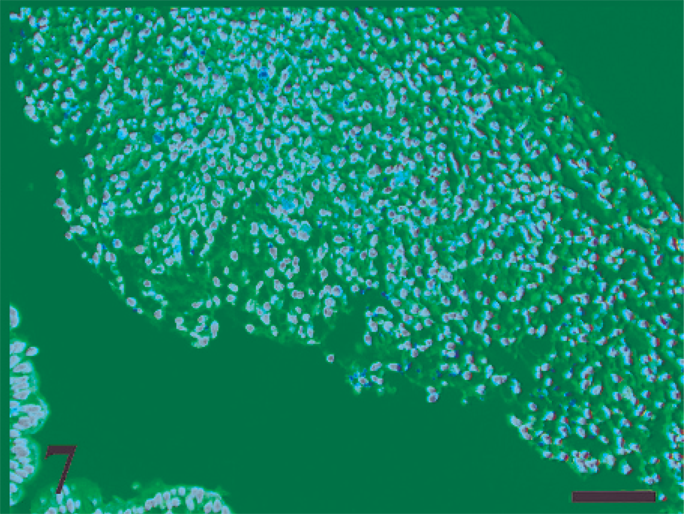
Moderate to massive accumulation of polymorphonuclear leukocytes, mononuclear cells, and fibrin in the intestinal lumen at 12 hours postinfection. HE. Bar = 200 µm.
Scanning electron microscopy
Uninfected ileal loops had normal Peyer's patch that is covered with domed epithelium and is surrounded by absorptive villi (Fig. 8). As early as 0.5 hour postinfection, MAP-infected loops had fibrin overlaying the ileal mucosa (Fig. 9). At 1 hour postinfection, bacteria were observed overlaying the M cells (Fig. 10). As the time postinfection progressed, fibrinous and mucoid exudates covered a larger area of ileal mucosa, and blunting of absorptive villi were observed (Fig. 11). Increasing numbers of inflammatory cells were observed overlaying the ileal mucosa in MAP-infected loops (Fig. 12). At 12 hours postinfection, mononuclear cells and activated platelets (Fig. 13) were observed on the ileal mucosal surface near MAP.

SEM of the uninfected control ileal surface at 0.5 hour. Bar = 66 µm.
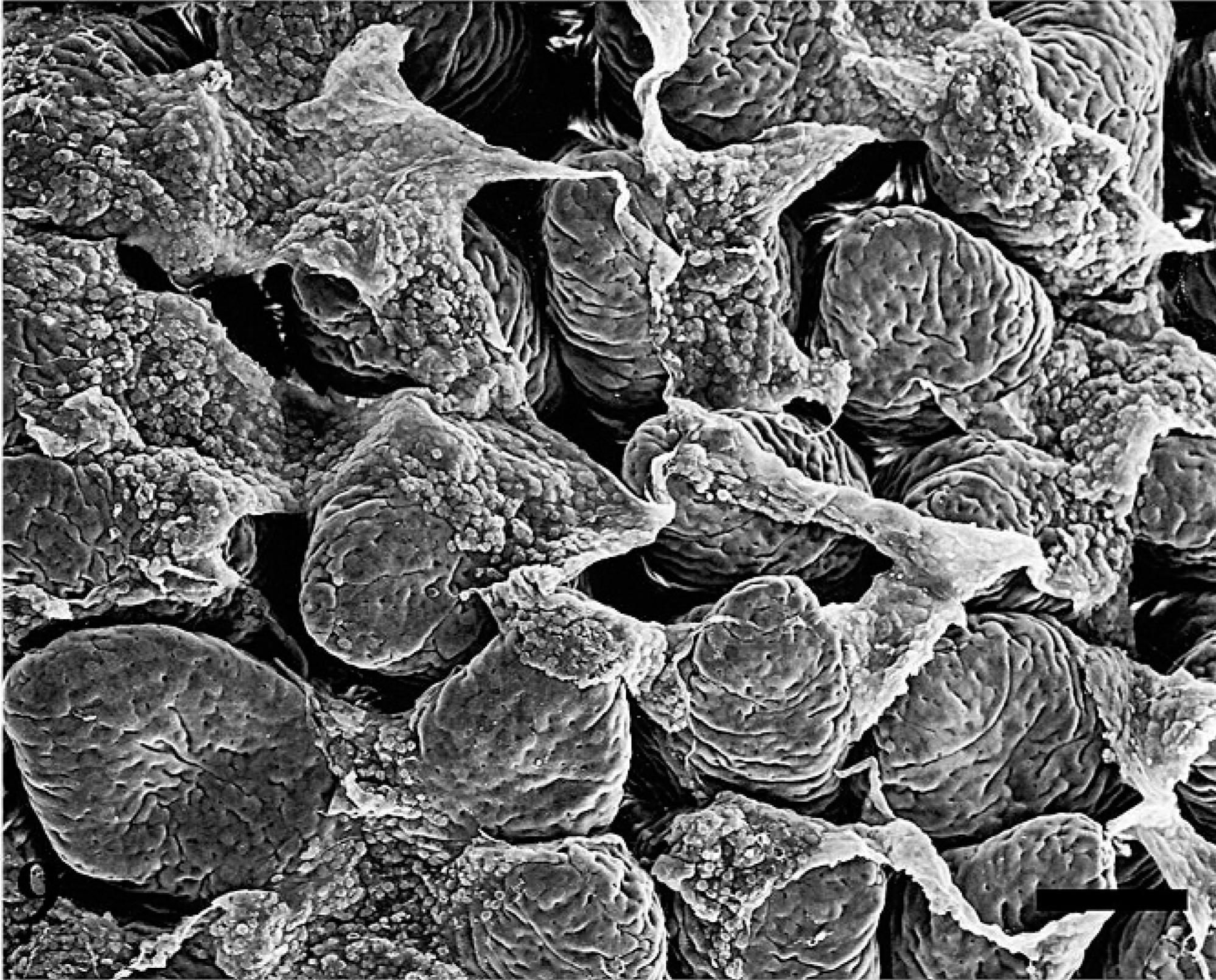
SEM of the infected ileal mucosa at 0.5 hour postinfection. Mucus is present on the mucosa as early as 0.5 hour postinfection. SEM. Bar = 66 µm.
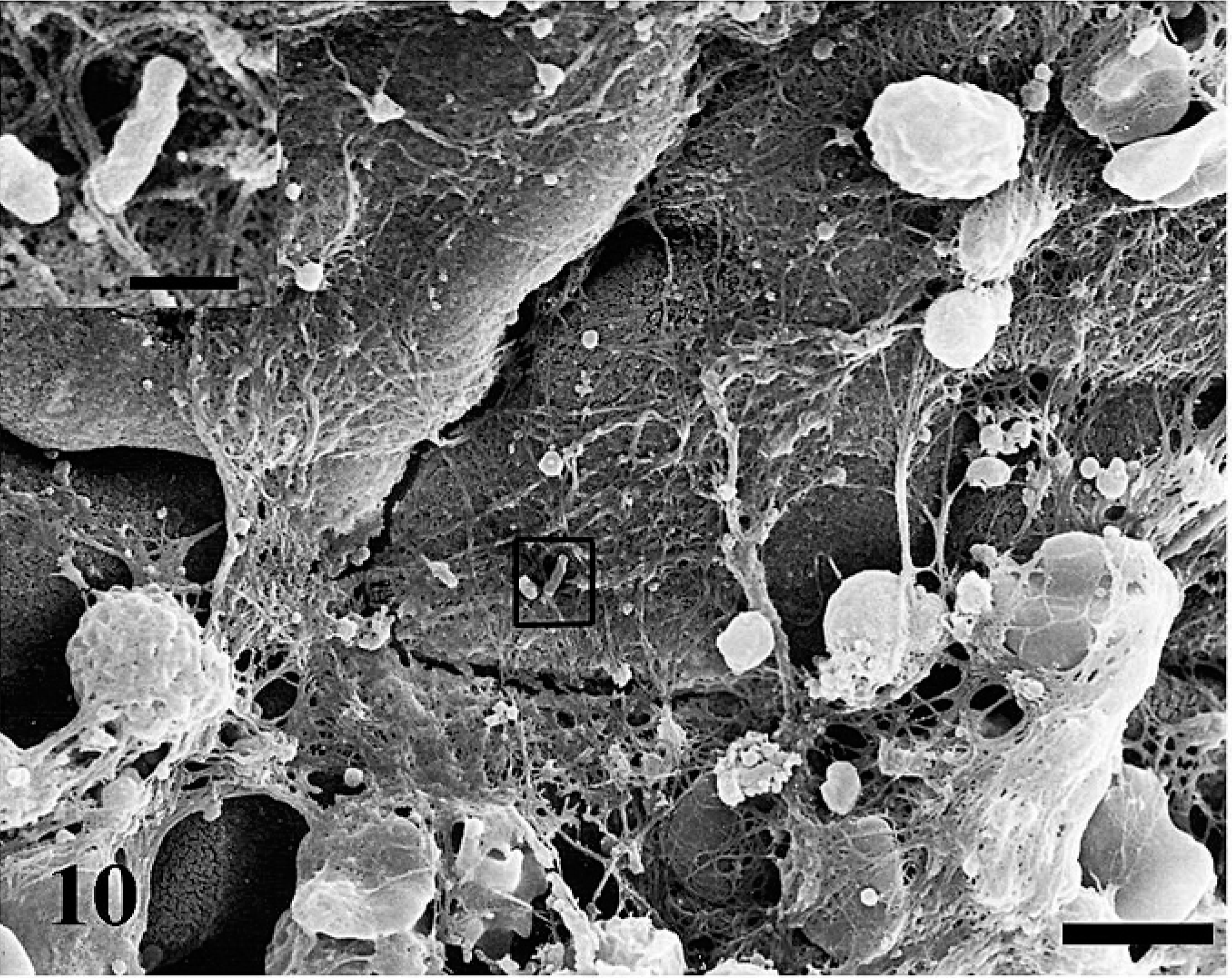
Fibrin accumulation at 1 hour postinfection on the mucosal surface of the ileum Bar = 5 µm. Box demonstrates bacteria overlaying M cells accompanied by fibrin and mucus. SEM. Bar = 1 µm.
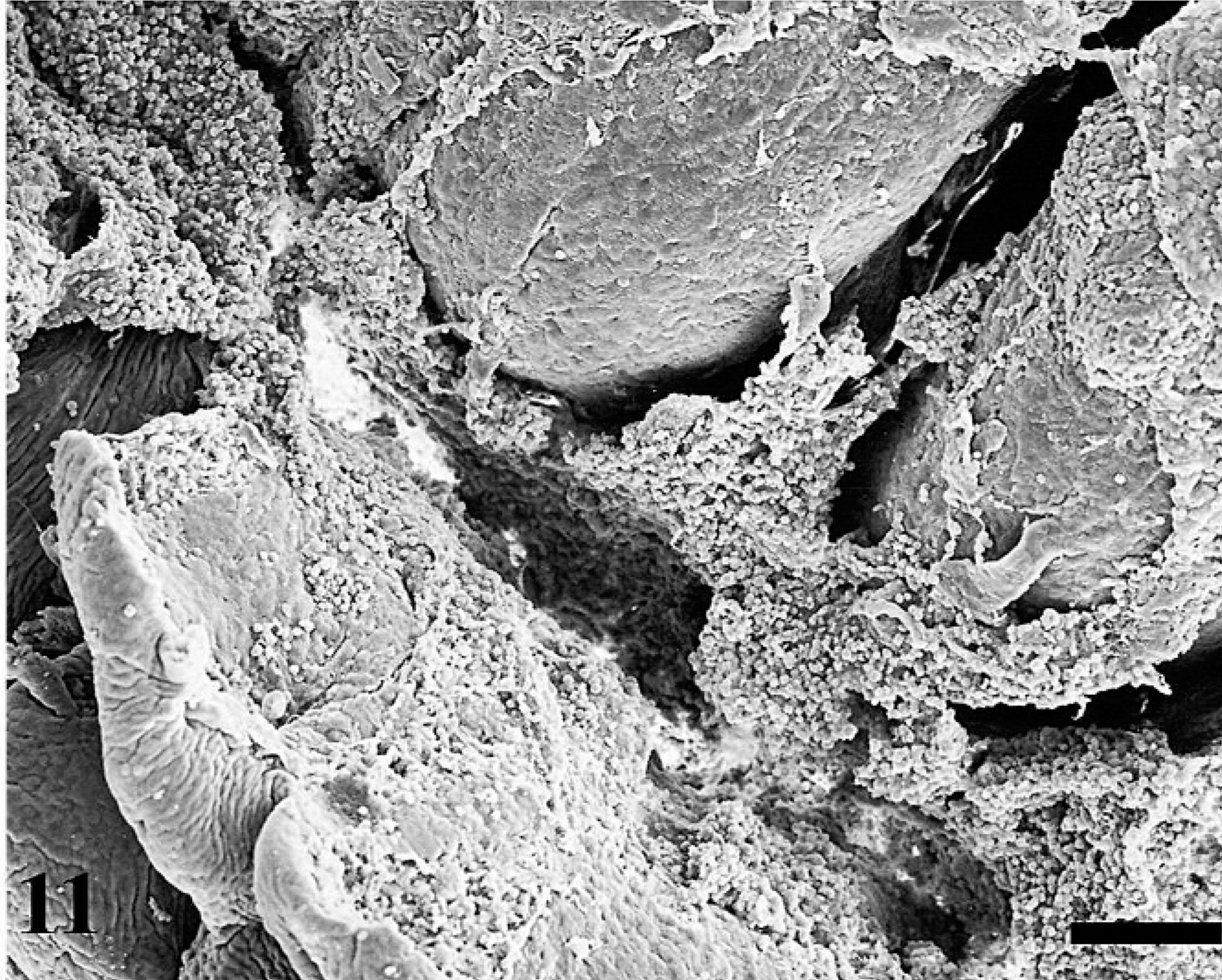
Increased accumulation of mucus on the mucosal surface at 4 hours postinfection. Bar = 66 µm.
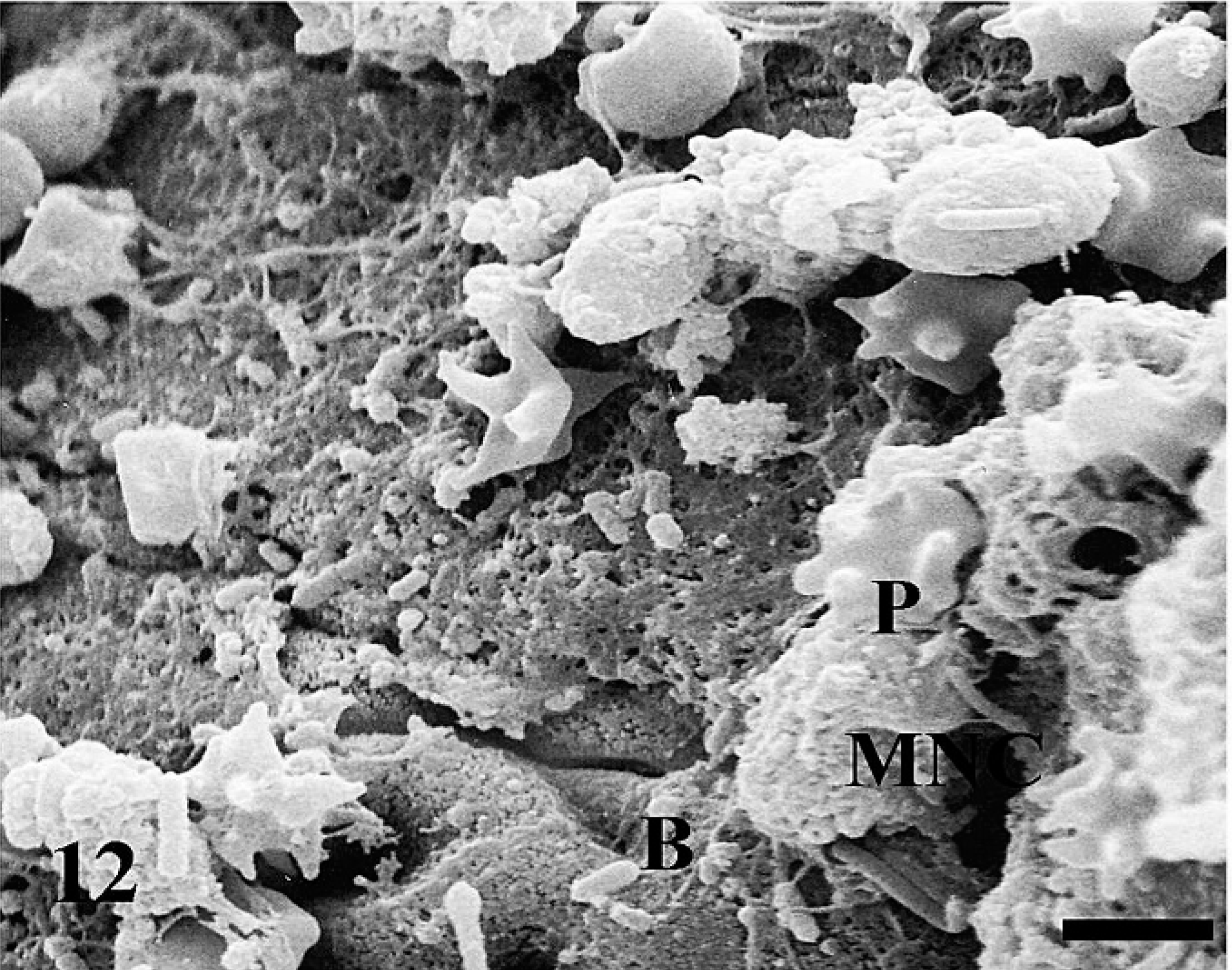
Bacteria (B) are present on the mucosal surface at 4 hours postinfection. Mucosal surface is covered with mucus and various cells, which include mononuclear cells (MNC), and platelets (P). Bar = 3.12 µm.
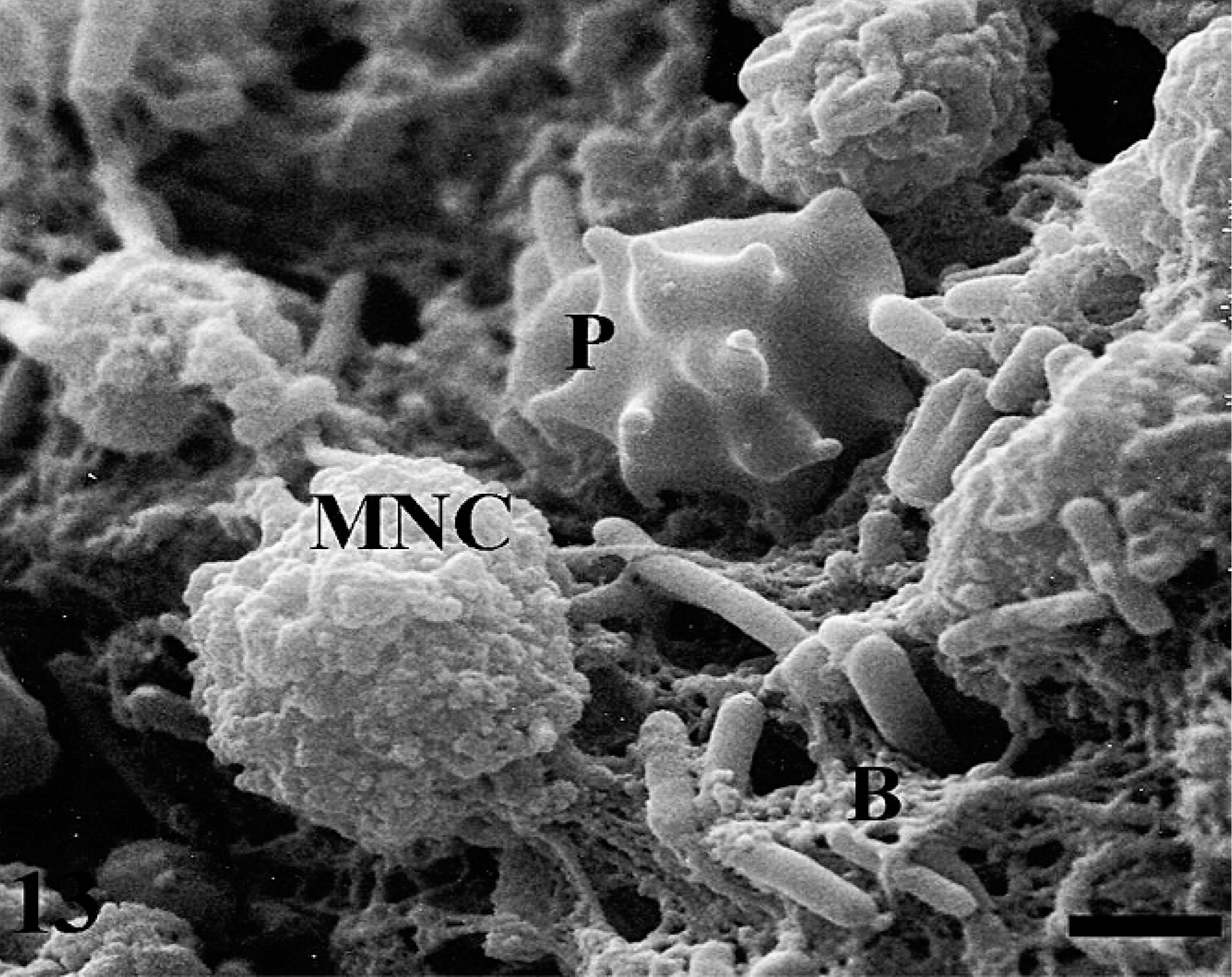
At 12 hours postinfection, mononuclear cells (MNC), activated platelets (P), and bacteria (B) are present on the ileal mucosal surface. Bar = 1.4 µm.
Transmission electron microscopy
No morphologic changes were observed in the ultrastructural analysis of uninfected control ileal loops (Fig. 14); however, invasion by MAP through epithelial surfaces occurred within 1 hour postinfection (Fig. 15). Examination of MAP-inoculated ileal loops revealed goblet cells on absorptive villi showing ruffling-like formation associated with bacterial invasion and exudation of mucus into the lumen. Preference for infection of specific intestinal epithelial cell types was not observed as all intestinal epithelial cell types were found to contain MAP.
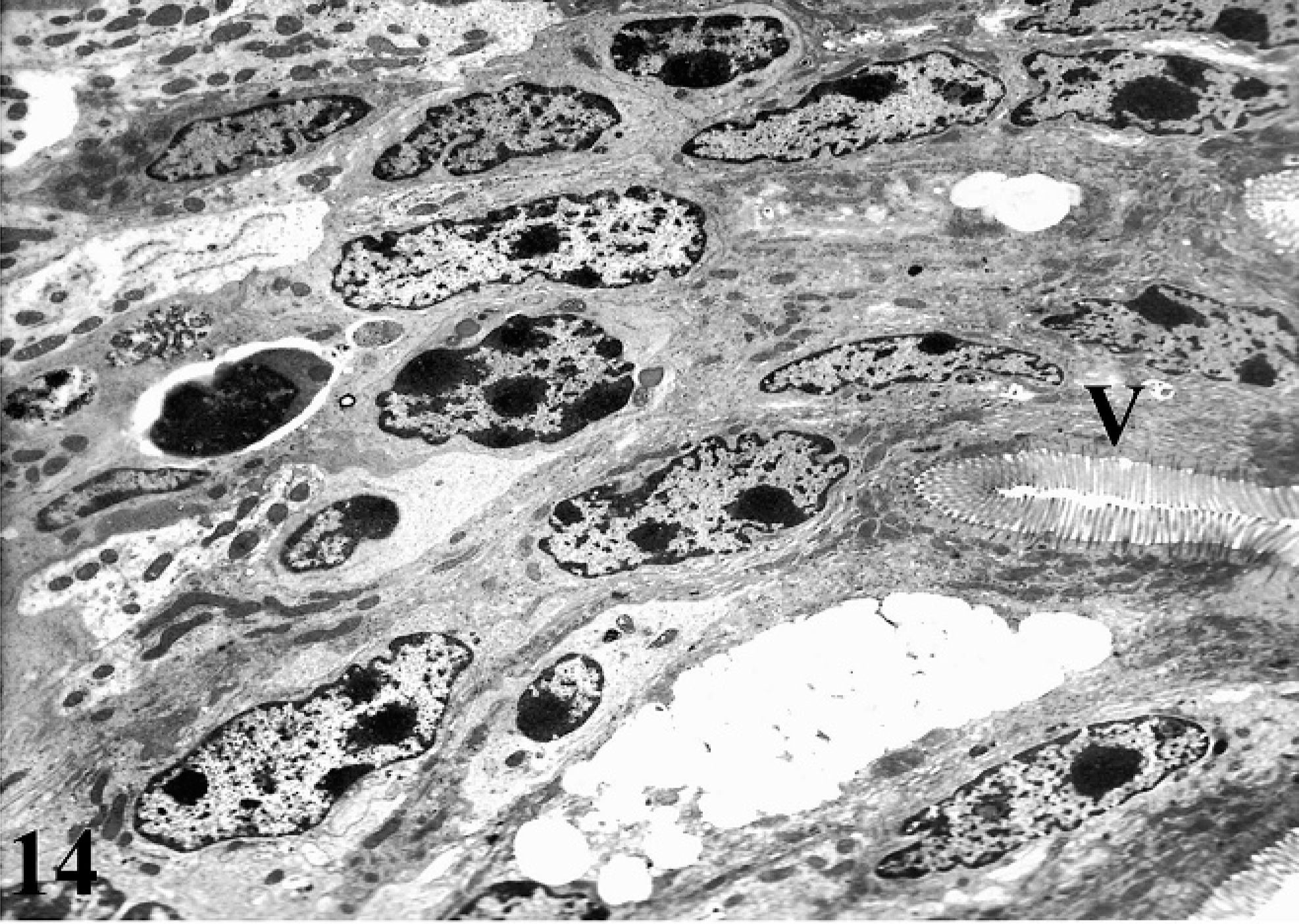
TEM of uninfected ileal mucosa with normal villi.
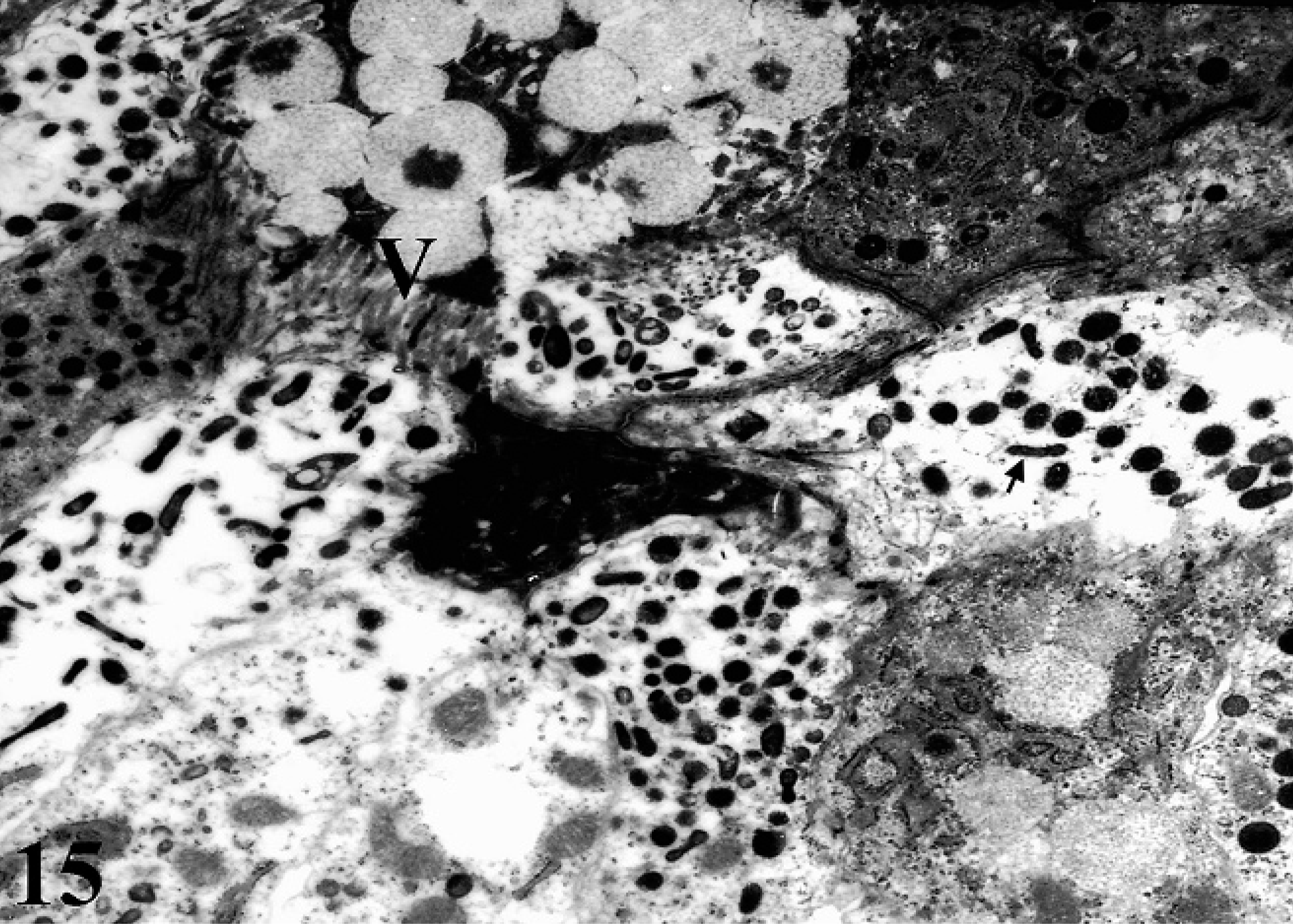
TEM of infected ileal mucosa depicting the presence of MAP (arrow) and goblet cells releasing the mucus near the absorptive villi (V).
Real-time PCR for intestinal gene expression
To further define the role of the ileal mucosa in initiating and modulating the host response to infection with MAP, we analyzed mRNA expression levels of some genes involved in the innate response of the host. Profilin, an invasion-related gene, was down-regulated until 1 hour postinfection and thereafter had increased expression (Fig. 16a). Intestinal mucosal epithelium serves as the first line of defense against any external stimuli. Intestinal trefoil factor, which promotes epithelial-cell restitution within the gastrointestinal tract, was up-regulated immediately after infection and was persistent; however, it later had a transient decrease at 2 hours (Fig. 16a). The gene encoding enteric ß-defensin had a gradual increase in the mRNA expression level until 4 hours and decreased at later times (Fig. 16b), whereas the expression level of lactoferrin peaked at 4 hours postinfection and remained elevated. However, the differences in the expression of these genes were only trends, which did not show statistical significance.
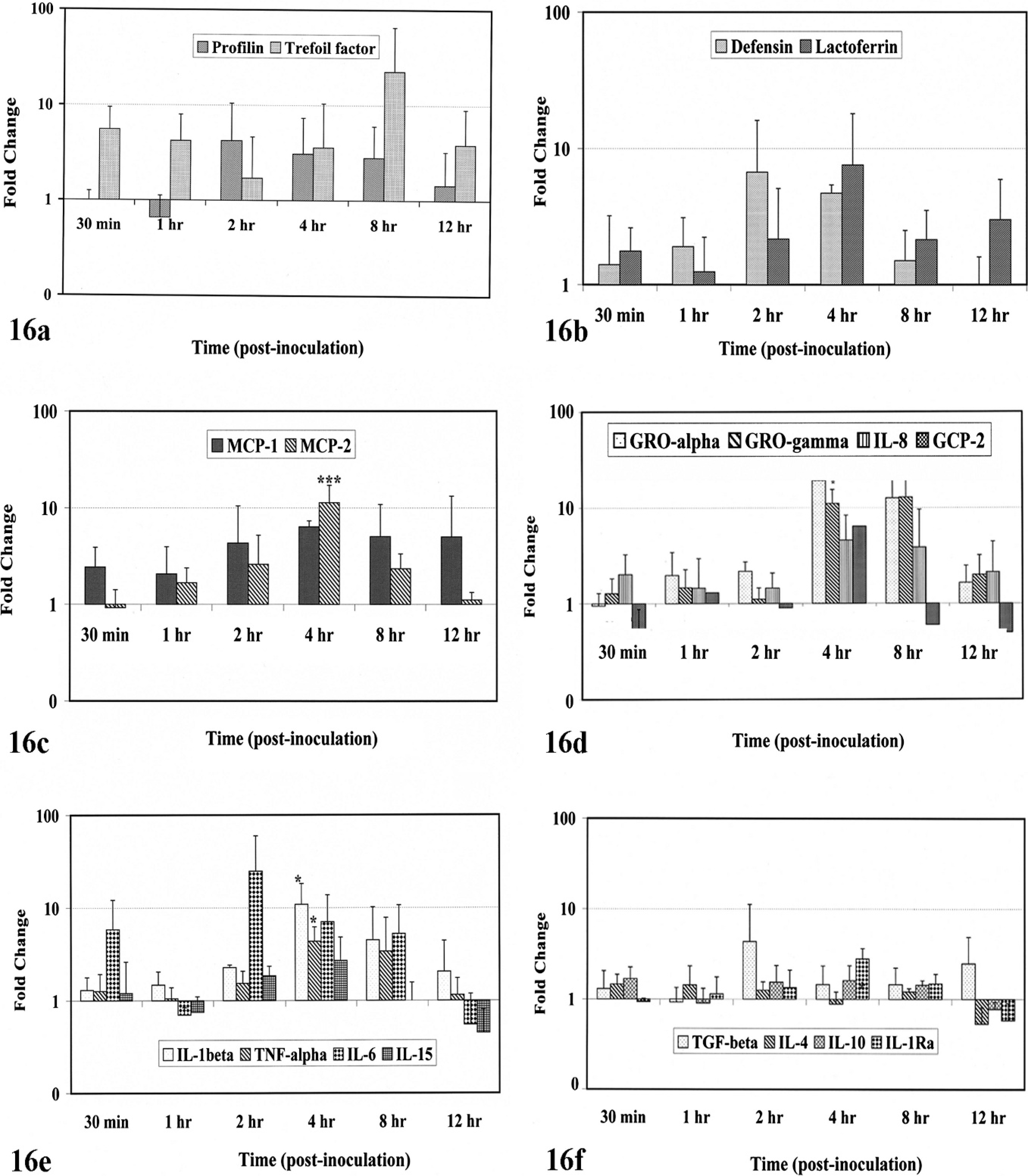
Real-time PCR analysis of mRNA expression for genes involved in innate immune response at the mucosal surface. Y-axis shows the fold change in the gene expression of infected loops as compared with the uninfected loops. In the plots shown, the bars represent mean ± SD of 4 different experiments. Asterisks indicate times with statistically significant differences in gene expression between treatment groups Real-time PCR analysis of mRNA expression for trefoil factor and profilin (Fig. 16a), enteric β-defensin and lactoferrin (Fig. 16b), C-C chemokines (Fig. 16c), C-X-C chemokines (Fig. 16d), pro-inflammatory cytokines (Fig. 16e), and anti-inflammatory cytokines (Fig. 16f).
Gene expression of the C-C chemokine monocyte chemotactic peptide (MCP-1) was rapidly expressed with a twofold increase as quickly as 0.5 hour postinfection and increased until 4 hours postinfection and then declined at later time points. MCP-2 was only transiently up-regulated at 4 hours postinfection (Fig. 16c). The expression of MCP-2 was significantly higher at 4 hours (P < .001) when compared with other time points. Expression of the PMN chemoattractants granulocyte chemotactic protein-2 (GCP-2), growth-related oncogene (GRO)-α, GRO-γ, and interleukin (IL)-8 were transiently up-regulated with delayed kinetics at 4 hours (Fig. 16d). The expression of GRO-α and GRO-γ were statistically significantly different (P values ≤ .05) between treatment groups when compared at 30 minutes postinfection. Expression of pro-inflammatory genes (IL-1β, TNF-α, IL-6, and IL-15) and anti-inflammatory genes (IL-1Ra, IL-4, IL-10, and TGF-β) were differentially regulated. In general, most of the pro-inflammatory genes had peak expression at 4 hours postinfection (P values ≤ 0.05) and then tapered at 12 hours (Fig. 16e). However, the anti-inflammatory genes were down-regulated until 1 hour, after which these genes were transiently up-regulated at 2 hours postinfection (Fig. 16f). Only TGF-β was up-regulated at later points.
Discussion
Cattle are natural hosts for Mycobacterium avium subsp. paratuberculosis. Neonatal calves are more susceptible to infection than adults. Infected adult cattle transmit MAP to neonates via feces or contaminated milk. Transplacental transmission of MAP from infected pregnant dam to fetus has been also described. However, regardless of the mode of infection, signs of illness do not appear until the cattle are young adults. Interaction of MAP with host eventually produces a cascade of events that ultimately results in profound chronic granulomatous enteritis. 12 MAP is detected in fecal samples of cows within a few hours of oral ingestion and persists in ileal mucosa for several weeks. 33
The neonatal calf ligated ileal loop model has been used to examine the acute infection, but its utility has not been tested beyond the site of invasion of MAP in the ileum. 24 Wu et al. performed surgical inoculation of animals with MAP in order to examine intestinal invasion and colonization until 9 months following infection. 36 To our knowledge, this is the first study to utilize the neonatal calf ileal loop model for studying the earliest lesions and transcriptional events in the ileum. This model has identified pathogen-specific responses of the ileal tissue. For example, infection with Salmonella enterica Typhiurium induced the accumulation of fluid in the bovine ileal loops, 26 yet MAP failed to cause any luminal fluid accumulation during the 12-hour experiment. The bovine ligated ileal loop model provides a platform to study changes in morphology during early infection, innate immune responses against the pathogen, and changes in host and/or pathogen gene expression important in the pathogenesis of Johne's disease. Adult cattle chronically infected with MAP develop very similar clinical signs and lesions to those of human Crohn's disease; thus the calf ileal loop model may be useful to investigate the early phases of this inflammatory bowel disease.
MAP successfully entered the ileal mucosa within 0.5 hour of inoculation as evidenced by bacterial culture and TEM. The invasion of MAP into the tissues did not increase with time, perhaps due to the clumping nature of MAP in the lumen, sensitivity of MAP to antimicrobial peptides, and the slow rate of replication of MAP. Another possibility is the secretion of mucin into the lumen after MAP exposure (as seen by SEM) that might have formed a protective layer over the intestinal mucosa, 9 which blocks subsequent bacterial invasion. The large inoculum in a confined area (ligated loop) may be a physiologic limitation of the ligated loop method; nonetheless, the model has provided new data that have not been reported previously and are unique to the MAP–host interaction.
The ileum has been considered the primary site of infection in Johne's disease. In the neonatal calf, the continuous linear Peyer's patch of the terminal jejunum and ileum has a massive follicle-associated epithelium composed of enterocytes and M cells that cover the domed villi. Domed villi are surrounded by absorptive villi composed of enterocytes and goblet cells. Various pathogens gain access to the intestinal wall either via specialized M cells or enterocytes. An in vitro study reported that there are no differences in attachment of MAP among different regions of the intestinal tract. 27 Another study has shown that MAP moves from ileum to liver and local lymph nodes via blood or/and lymphatic circulation. 36 However, little is known about the manner in which MAP interacts with the intestinal mucosa during the earliest phases of the infection. In the present study, the earliest interaction of MAP was with M cells and goblet cells. M cells are unique among cells of the intestinal epithelium as they display a high density of ß1 integrins on their luminal surface, 31 which have affinity for the fibronectin attachment protein of MAP 29 and, therefore, may facilitate the entry of MAP into the ileal mucosa. In contrast, goblet cells may impede the entry of MAP into the mucosa by secreting mucin glycoproteins and trefoil peptides, which form a continuous gel on the mucosal surface. 18 In this study, mucin secretion was increased in the MAP-infected ileal tissue as evident from SEM. Moreover, the gene expression of trefoil factor showed a trend of up-regulation during early infection. Trefoil factor not only controls gastrointestinal repair but also regulates mononuclear cell inflammatory responses. 19 The functional effects of mucin and trefoil factor in the early phase of MAP infection remain uncertain, as the differential regulation of these genes was only a trend and will require more detailed studies to understand the role of these unique innate mediators.
Transmigration of the mononuclear cells was evident within 4 hours of the inoculation. Profilin may play a role during monocyte migration and endothelial transmigration during infection of MAP, as suggested by Matias-Roman et al. during extravasation of circulatory cells in the process of inflammation. 21 Profilin has also been found to increase the surface expression of fibronectin receptors 22 and thus may contribute to the formation of focal contacts between MAP and endothelial cells. 22
Antimicrobial proteins may also play a role in innate immunity against the invading pathogen. The presence of both enteric β-defensin and lactoferrin has been reported in the chronic stage of Johne's 23,32 and Crohn's 11,35 diseases. In our study, because there was no significant increase in the number of MAP after 1 hour, we reasoned that MAP may be sensitive to even a minimal expression of antimicrobial proteins, as the expression of these genes was only a trend and was not statistically significant.
The immune system of calves responds to the MAP invasion by recruiting mononuclear cells (macrophages and lymphocytes) to the site of infection. These mononuclear cells release a variety of chemokines and cytokines to increase the bactericidal power of the macrophages and recruit more cells to combat the infection. Monocyte chemoattractant protein-1 and -2 (MCP-1 and MCP-2) are C-C chemokines that are chemotactic for monocytes but not PMNs. CCR2, the ligand for MCP-1, has been shown to be up-regulated in T lymphocytes isolated from the intestine of Crohn's patients. 6 Chemoattractants for PMNs include the C-X-C chemokines GRO-α, GRO-γ, and IL-8. A synchronized up-regulation of IL-8 and GRO-α has been proposed to be involved in the PMN-mediated tissue injury in patients with inflammatory bowel disease. 15 The C-X-C and C-C chemokines are generally not stored within cells; rather their production is induced at the transcriptional level upon appropriate stimulation. 1,2 In our study, both C-X-C and C-C chemokines were transiently induced during the early host–pathogen interaction. The gene expression of regulatory cytokines (Th1 and Th2 type) was also transiently induced. Cattle that had progressed to the clinical stage of Johne's disease had higher TGF-β and IL-10 mRNA levels compared with subclinically infected or healthy cows. 16 However, IL-10 gene expression was consistently enhanced by MAP stimulation of peripheral blood mononuclear cells (PBMCs) from subclinically infected cattle. 7 In vitro studies have demonstrated that MAP stimulation of PBMCs derived from infected and uninfected cows caused a decrease in the gene expression of Th1 type of cytokines (IFN-γ, IL-1α, and IL-6). Our study clearly documented an increased expression of these Th1 cytokine genes in MAP-infected ileal tissue. However, expression of IL-15 was transient. IL-15 has a dual role in the intestine; it works as a Th1/pro-inflammatory cytokine and reduces mucosal damage by preventing intestinal epithelial cell apoptosis. 25 The interaction between the Th1 and Th2 type of cytokines is considered critical to protective immunity against pathogenic mycobacteria and may contribute to the ability of the MAP to establish infection in the susceptible host.
In summary, we have provided a proof of concept that early responses of MAP can be effectively evaluated in the neonatal calf ligated intestinal loop model by simultaneously measuring the changes in gene expression and morphology. The expression of genes profiled in this study coincided and provide a conceptual framework for the temporal changes in the histopathology, TEM, and SEM. Study of early responses in chronic enteric infections like Johne's disease (MAP, as the causative agent) and Crohn's disease (MAP, as one of the potential etiopathologic agents) has been a challenge. Our studies provide a comprehensive and integrated view of early host–pathogen interactions achieved by an inclusive morphologic and multigenic discovery approach to understanding some of the mechanistic pathways influencing the pathogenesis of Johne's disease. Studies are underway in our laboratory investigating the reciprocal host-pathogen interactions by evaluating in vivo MAP gene expression and how bacterial expression is modulated by the bovine host during early infection.
Footnotes
Acknowledgements
We thank posthumously Dr. Helga Sittertz-Bhatkar (Image analysis laboratory, Texas A&M University) for her assistance in SEM and TEM experiments, and Mr. Alan Patranella for the assistance with animal care. The study was supported by funds from the Texas Johne's Working Group and U.S. Department of Agriculture Cooperative State Research, Education and Extension Service-National Research Initiative-Coordinated Agricultural Project-Johne's Disease Integrated Program (2004-35605-14243 subaward Q6286224133).
