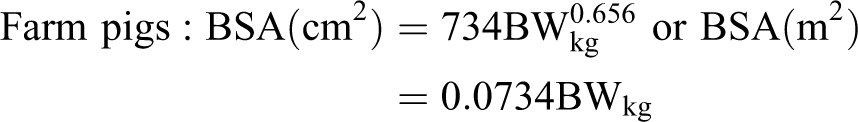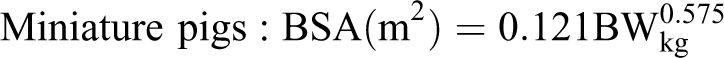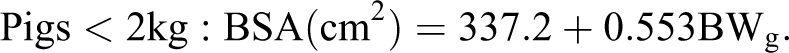Abstract
Swine are considered to be one of the major animal species used in translational research, surgical models, and procedural training and are increasingly being used as an alternative to the dog or monkey as the choice of nonrodent species in preclinical toxicologic testing of pharmaceuticals. There are unique advantages to the use of swine in this setting given that they share with humans similar anatomic and physiologic characteristics involving the cardiovascular, urinary, integumentary, and digestive systems. However, the investigator needs to be familiar with important anatomic, histopathologic, and clinicopathologic features of the laboratory pig and minipig in order to put background lesions or xenobiotically induced toxicologic changes in their proper perspective and also needs to consider specific anatomic differences when using the pig as a surgical model. Ethical considerations, as well as the existence of significant amounts of background data, from a regulatory perspective, provide further support for the use of this species in experimental or pharmaceutical research studies. It is likely that pigs and minipigs will become an increasingly important animal model for research and pharmaceutical development applications.
Swine are considered one of the major animal species used in translational research and are increasingly being used as an alternative to the dog or monkey as the nonrodent species of choice in preclinical toxicologic testing of pharmaceuticals. Over the last 20 years, swine have also replaced dogs as the general surgical model in the international arena for both training and research. Textbooks, article, and proceedings detailing a large number of models and technical procedures for using swine in the laboratory are available. 21,47,50,52,53 There is a large body of literature covering the normal anatomy and physiology of the species, and pathology descriptions related to infectious or degenerative diseases in swine under agricultural conditions are also available. 37,49,51 However, descriptions of macroscopic or microscopic background lesions in research pigs in the laboratory setting or toxicologic pathology descriptions in minipigs have been reported only rarely. 19 This article provides an overview of the common uses of miniature and domestic swine in research and toxicology testing and provides a review of the anatomic and histopathologic characteristics peculiar to this important animal model. The rationale for the use of pigs, and particularly minipigs, as an increasingly common alternative choice of nonrodent species in regulatory toxicology studies is discussed.
Breed Selection
The differences between domestic farm breeds and miniature breeds are related to their growth rate and size at sexual maturity rather than actual anatomic differences in organs and structures. Thus, when different breeds are age matched, the organ sizes will reflect the increased size of domestic breeds compared with miniature breeds; however, the physiologic function should be the same. Conversely, when animals are weight matched, the sizes will be similar for organs and structures; however, the physiologic function will be related to the relative maturity of the animals. As an example, most domestic breeds would be expected to grow from 1–2 kg at birth to more than 100 kg at 4 months of age.
The minipig strains that are used today in research were developed out of a need for a smaller and thus more manageable version of the domestic pig. These minipigs are derived by selective breeding and are in no way to be regarded as transgenic or genetically modified animals. There are various strains available, each with its own characteristics. There is a wide variation in size and weight, which of course has an influence on amounts of drug needed for testing. Furthermore, other characteristic differences (eg, pigmented vs nonpigmented skin varieties) can influence choice of strain. The quality of the animals varies from the conventional to specific pathogen–free types. These animals are bred under controlled conditions and are raised specifically to be laboratory animals. They are around the same price as a dog and essentially can be kept in the same laboratory conditions as dogs, so there is no need for expensive investment to use minipigs. Minipigs are larger and grow faster than dogs. Minipigs also become sexually mature earlier, and from a preclinical toxicology perspective, this implies a greater likelihood of mature animals that will be used in shorter term candidate selection or dose range–finding studies when using minipigs compared with dogs. It makes minipigs easier to use in studies where reproduction is an element of the study (embryo–fetal studies and multigenerational juvenile toxicity studies).
The most common miniature breeds available in the United States are the Hanford, Yucatan, Yucatan micro, Sinclair, and Göttingen (from largest to smallest). These miniature breeds weigh 0.5–1 kg at birth and grow to 17–20 kg for the Hanford and 7–9 kg for the Göttingen in 4 months. Sexual maturity for all breeds occurs between 4 and 6 months of age. Typically, farm pigs are only used in protocols lasting a maximum of 3–6 weeks depending upon the age at the start of the protocol unless growth is part of the study. Problems can arise in using larger pigs. For example, using domestic breeds for implantation of cardiovascular devices for 6-month studies can result in dislodgement of the device caused by the increased length and diameter of the cardiovascular structures. As a general rule, the Hanford miniature pig develops adult human–sized organs and structures between 6 and 8 months of age, whereas the other breeds never develop an equivalent size for most organs and structures.
Anatomic and Histopathologic Considerations
Swine share a number of anatomic and physiologic characteristics with humans that make swine potentially a better model for some procedures and studies compared with other large animal species. Systems that are most commonly cited as being suitable models include the cardiovascular, urinary, integumentary, and digestive systems. These features have led to the increasing use of swine as a major species in preclinical toxicology testing. These characteristics and models have been reviewed in the literature 47,49 –53 and are discussed in further detail below. There are also some specific morphologic and histopathologic characteristics peculiar to the pig of which the pathologist should be aware when reading microscopic experimental studies in swine.
Cardiovascular System
The swine heart is typical of most mammals with a few variations. The distribution of blood supply by the coronary artery system is almost identical to that of humans. 12,47,49 –51 In addition, the size of the heart and blood vessels in sexually mature Hanford minipigs is more analogous to the size in humans than is either the dog or the nonhuman primate. The major anatomic variation from other mammalian species is the presence of the large left azygos (hemiazygos) vein, which enters the coronary sinus. In other mammals, a hemiazygos vein enters the precava. The blood supply to the coronary arteries and conduction system is right-side dominant and is similar to 90% of the human population. Preexisting collateral blood supply is largely absent, and thus occlusion will result in a total infarct. 9,12,49 Other preclinical species, such as the dog, have existing collateral circulation. The peripheral vasculature of swine has some variations from the dog. The external jugular vein is relatively large in the pig, but not superficial as in other common laboratory animals. The vein is located medial to a line drawn from the angle of the mandible to the point of the shoulder, at the same depth as the common carotid artery and internal jugular vein. The cephalic vein is quite prominent and superficial as it crosses the neck at the level of thoracic inlet. Valves are present in the peripheral veins. A review of peripheral vascular injection sites has been previously published. 51 The endocardium and epicardium are activated simultaneously because of the differences in the distribution of the nerve fibers. The conduction system contains large numbers of adrenergic and cholinergic fibers in the atrioventricular node and left and right bundle branches. The pig has a more neuromyogenic conduction system than the human. Despite these differences, the pig has been used successfully in studies of the cardiac conduction system. 12,49,51
Hemodynamically, there are differences between breeds of pigs and between pigs of different ages within the same breed, as is true in all species. For example, heart rates are much higher in young swine than in adults. Swine should be age and body weight (BW) matched for comparison and the values indexed to body surface area (BSA). Formulas for BSA
31,51
need to be matched to the type of pig used in the study:
Histologically, the pig has more prominent Purkinje cells and a more prominent vasa vasorum in the aortic wall than seen in the dog, monkey, or rat. 9,51 Small foci of lymphocytes are occasionally noted within the myocardium of minipigs involved in toxicology studies, similar to those previously described in monkeys 13,55 but much less commonly than is noted in rats with spontaneous cardiomyopathy. 14 These changes are generally considered background lesions of no toxicologic significance but in some cases can be associated with drug treatment when of sufficient severity or associated with a dose response. Valvular lesions have also been produced in pigs, both experimentally and with drug treatment, and they can be assessed by echocardiography as well as histopathology. 8
Spontaneous arteritis has been described in minipigs associated with an idiopathic thrombocytopenia syndrome, particularly in the Göttingen breed (Fig. 1). 2,23 The lesion is similar morphologically to the syndrome of periarteritis nodosa in the rat and histologically is characterized by medial smooth muscle vacuolation and degeneration, intimal hyperplasia with endothelial cell hypertrophy, and perivascular or transmural mixed inflammatory infiltrates in scattered, small to medium-sized arterioles and arteries. Necrosis of the tunica media and thrombosis occur in some vessels, and lesions have included concentric laminar (onion skin) thickening of vascular walls, with partial to complete luminal stenosis. 23 In severe cases of this syndrome, myocardial infarction with myofiber necrosis and mineralization have been described in the heart as a secondary effect of the vasculitis.
Göttingen minipig. Gross necropsy of pig with idiopathic thrombocytopenia syndrome and multiorgan hemorrhage.
The streptozotocin-induced diabetic, hypercholesterolemic pig model has been successfully used to induce coronary atherosclerosis and shares many features of the pathophysiology of hyperlipidemic vasculopathy in humans. 1 There are various modifications to this model involving different regimes of streptozotocin administration and alterations to the diet, but vascular lesions can include atheromatous plaques in larger vessels as well as hypertrophic changes in the walls of smaller vessels.
Lung
The lungs are composed of apical, middle, and diaphragmatic lobes with an accessory lobe on the right lung. Interlobular fissures are incomplete. The larynx is prominent and contains lateral and middle ventricles that cause a narrowing of the passage caudal to the large vestibule. The bronchial tree divisions are typical of other species; however, the right apical lobe has a bronchus that stems from the trachea proximal to the tracheal bifurcation. 30 The pulmonary arteries branch along the dorsal and lateral sides of the bronchioles, whereas the pulmonary veins are mainly on the medial or ventral sides. 30 The mediastinum is flimsy and cannot be considered functionally complete in smaller pigs because it tends to rupture when manipulated. 9,49
Adequate perfusion, fixation, and pulmonary insufflation are important in histologic evaluation of the pig lung, just as they are with other experimental species. In some cases, total body perfusion can be desired or necessary for adequate histologic evaluation in specialized pulmonary procedures, but this is not advocated for routine toxicity studies. Bronchiolar associated lymphoid tissue is often prominent. Foamy alveolar macrophages can be seen in small numbers in minipigs in toxicologic studies as a background change, and these cells can be greatly increased with toxicologic injury and/or can be associated with hemorrhage, inflammation, or alveolar wall necrosis. Even in laboratories with good hygiene and husbandry practices, it is not uncommon to find rare, small granulomas or thickening of the pleura in the lungs of pigs associated with endoparasitism. Agonal interstitial emphysema can be seen as a perimortem event in pigs due to well-developed interlobular septa, but it is much less commonly encountered than in cows or sheep at necropsy.
Gastrointestinal System
Although the physiology of digestion in omnivorous swine is remarkably like that in humans, the anatomy differs significantly from many other laboratory animals and humans. 37,49,51 The stomach is typical of monogastric species except for a prominent muscular outpouching, the torus pyloricus, at the level of pylorus and a more prominent cardia. The small intestine is approximately 10% duodenum, 80% jejunum, and 10% ileum and is approximately 30–40 times the length of the pig’s body. In humans and nonhuman primates, the duodenum is relatively shorter and the ileum tends to be similar in length to the jejunum or even larger. The mesenteric vessels of the small intestine form vascular arcades in the muscularis mucosa of the intestine and not in the friable mesentery, as in other mammals. 37,49,51 The presence of duodenal Brunner’s glands and jejunal Peyer’s patches allows easy histologic differential identification of these structures as it does in other species. 16 Unlike other domestic animals, Peyer’s patches occur in a continuous band along much of the length of the small and large intestine in the pig and form a 2-cm-wide, thickened focus around the ileocecal opening referred to as the cecal tonsil. The large intestine of the pig is quite different anatomically from that of other common laboratory animals. The cecum, the ascending and transverse colon, and the proximal portion of the descending colon are arranged in a series of centrifugal and centripetal coils in the left upper quadrant of the abdomen, collectively forming the spiral colon. The cecum has 3 longitudinal muscular bands (tenia), and the proximal portion of the spiral colon has 2 bands. These result in a series of sacculation (haustra). Xenobiotic-induced toxicologic injury to the stomach or intestine often results in degeneration, ulceration, or hemorrhage, as it does in other species. However, as discussed later in this article, the porcine gastrointestinal tract does not appear to be as sensitive to the ulcer-inducing properties of nonsteroidal anti-inflammatory drugs (NSAIDs) as is the gastrointestinal tract of dogs. 19 Furthermore, ulceration of the nonglandular portion of the stomach is not a common background (incidental) or stress-related finding in pigs, as it occurs in the forestomachs of rodents.
The dental formula of a mature pig is 2(I 3/3, C 1/1, P 4/4, M 3/3) = 44. A full set of permanent teeth is usually present around 18 months of age. 37,49 Macroscopic or microscopic lesions in the oral cavity due to xenobiotic treatments are rare, although broken teeth are frequently encountered in animals on study.
Liver
The liver contains 6 lobes separated by fibrous septae and has a functional gallbladder. Histologically, the liver pattern presents as markedly defined lobulation, and unlike rodents, pigs have well-defined acinar structures with clear portal triads. However, despite this increased amount of interlobular connective tissue, there does not appear to be any increased tendency of the porcine liver to produce cirrhosis after toxicologic injury compared with other nonhuman species. Rather, toxicologic lesions in the porcine liver are similar to those noted in other species including rodents and dogs, and the species is considered predictive for human toxicologic injury. 4 Although macroscopically evident white spots—characteristic of ascaridiasis—are seldom noted in the laboratory setting, rare focal granulomas associated with endoparasitism are still occasionally noted microscopically. Small random foci of lymphocytes and Kupffer cells are frequently noted as a background change of no toxicologic significance. Fatty liver is a common finding in the streptozotocin diabetic pig model, as it is in other porcine models of hypertriglyceridemia. 1
Pancreas
The swine pancreas is related to the proximal duodenum in the usual manner, with a single pancreatic duct entering the duodenal lumen distal to and separate from the common bile duct. The pancreas is more loosely attached to the blood supply of the duodenum than in the dog, facilitating dissection during a pancreatectomy. 51 However, the body of the pancreas separates in the 2 lobes that encompass the portal vein, and a portion of the pancreatic lobe is retroperitoneal, as in humans. Histologically, the pancreas is quite lobulated and fatty. Pig islet cells are functionally similar to humans. 49 –52
Kidney and Lower Urinary System
The left kidney is more cranial than the right. The pig has a true multirenculate, multipapillate kidney with true calices as in humans. 47,49 An important difference and consideration for intrarenal surgery is that the avascular plane of the kidney is transverse in swine, not longitudinal as in dogs and humans. Histologically, both the proximal and distal tubules in the pig appear dilated when the lumen diameter is compared with other experimental species such as the dog or the rodent, but this is a normal finding in pigs and minipigs. Similarly, glomeruli tend to have a greater degree of apparent dilation of the capillary tufts normally, compared with dogs, monkeys, or rodents, when viewed microscopically from paraffin sections. Toxicologic injury is manifested by tubular degenerative changes, glomerular changes, and/or interstitial inflammatory lesions indistinguishable from those in the kidney from other species including humans. Mesangioproliferative glomerulopathy, interstitial fibrosis, and tubular atrophy have been described in minipigs with severe thrombocytopenic purpura syndrome. 23> Pigs develop both vesiculoureteral and intrarenal reflux spontaneously and as a surgically produced model. 51 This can limit their utility in some bladder or ureteral surgery applications. The bladder has a thin flimsy wall but otherwise is like that of other mammals.
Skin
Pigs are well suited for skin studies. Although rat, mouse, and rabbit have been used in experimental and toxicologic dermatologic studies, pig skin has been shown to be the most similar to human skin. 3,49,51 Pig skin is structurally similar to human epidermal thickness and dermal–epidermal thickness ratios. 3 The skin of the pig is relatively hairless and has a fixed subcutaneous layer and dermal hair follicles like humans. The skin tends to be thicker on the dorsum of the neck than in humans and less vascular in all areas. There are prominent basophilic granules along the basal layer. 16 There are few eccrine sweat glands, which are limited to the snout and carpal glands, and this paucity of eccrine glands distinguishes porcine skin from human skin. Swine have apocrine sweat glands and sebaceous glands throughout the skin, but they have minimal function in thermoregulation compared with humans. Interfollicular muscles are present. The cutaneous blood supply and sequence of events in wound healing have made the pig a standard model of wound healing and reconstructive surgical treatments. Pigs have a tendency to develop substantial amounts of subcutaneous fat. 49 Biochemically, pigs contain dermal collagen and elastic content that is more similar to humans than other laboratory animals. Pigs also have similar physical and molecular responses to various growth factors. 24,40 Pig skin wounds heal primarily by reepithelialization rather than contraction, so they are especially useful in studying wound healing and burn lesions. 24
Reproductive System
The boar has a fibromuscular penis with a sigmoid flexure that aids in erection, instead of prominent erectile tissue. The tip of the penis is corkscrew-shaped. These characteristics make it extremely difficult, if not impossible in most individuals, to pass a urinary catheter. Thus, the pig makes a poor choice as a model for penile or ureteral procedures. The preputial diverticulum on the ventral surface of the abdomen is filled with necrotic debris and residual urine and must be avoided or cleaned when performing abdominal surgery. Accessory sex glands of the boar include prominent vesicular glands, a small prostate gland, and bulbourethral glands in an arrangement similar to humans. 49 Focal segmental atrophy or tubular hypoplasia of the seminiferous tubules has been noted as a relatively common background change in minipigs in toxicologic studies, 45 just as it has in rodents and dogs. This can complicate the interpretation of toxicologic testicular injury in drug development studies.
The female reproductive tract has a typical bicornuate uterus like other species that bear litters. The Fallopian tubes are prominent and tortuous, making them readily accessible to surgical procedures. The pig has diffuse epitheliochorial placentation with transplacental transport characteristics similar to those of humans. 49 Because swine are sexually mature at 4–6 months of age (vs 3–4 years for sexual maturity in macaques, used commonly in pharmaceutical safety studies), swine do not have to be housed for as long a time as primates when sexual maturity is critical to study design. Additionally, having litters rather than the single births of primates potentially reduces genetic variability by having age-matched littermates on study.
Endocrine System
The right adrenal gland has a close attachment to the caudal vena cava and is not readily extirpated by surgical means. The thyroid gland is located on the ventral midline of the trachea at the level of thoracic inlet instead of being associated with the larynx as in most species. The single pair of the parathyroid glands is closely related to the thymus rather than to the thyroid gland. The location of the parathyroid glands is variable depending on the age of the pig, but they can generally be found in the first fascial plane medial to the thymus at its cranial end. 51 It is often difficult to locate both parathyroid glands macroscopically, and therefore this tissue may be missing, at least unilaterally, in some toxicology studies, especially if the laboratory has limited experience with porcine tissue lists. With practice and knowledge of the thymic and parathyroid anatomy, this problem can be easily overcome.
Lymphatic and Hematopoietic Systems
The lymph nodes are histologically unique in having an inversion of the cortex and medulla. The germinal centers are more central than in other species, with the outer region being more similar to the medulla. The spleen is more tightly attached to the stomach by the short gastric blood vessels and therefore is not as pedunculate an organ as it is in the dog. 49 Systemic hemorrhage associated with thrombocytopenic purpura syndrome is encountered occasionally in untreated minipigs in both research and toxicology studies and is of uncertain cause. 2,23 Vascular lesions with this syndrome were described earlier under the cardiovascular section. Perivascular hemorrhages are noted in multiple organs and tissues, and in most cases, the vascular changes are associated with thrombocytopenia and regenerative anemia. To date, no causal infectious agents have been identified. Subcutaneous hemorrhage in the region of venipuncture is common and should not be considered to be due to this syndrome or a potential test article effect. Like humans, minipigs are susceptible to bone marrow depletion with cytotoxic drugs and xenobiotics that induce impaired erythropoiesis and granulopoiesis. Extramedullary hematopoiesis is an extremely common finding in the spleen and liver of pigs up to 6 weeks of age and should be considered normal. 45,49
The thymus extends from the thorax into the neck cranially to the level of the larynx in young animals. Thymic lymphoid depletion can occur with stress, as it does in other species in toxicity studies, and this change can be difficult to differentiate from age-related involution. Even as the gland shrinks with age, it does not tend to become completely thoracic in location.
Central Nervous System
The bones of the skull are massive. The brain is relatively large, with structures typical of other mammals. The central nervous system and specifically the brain of the pig have been rapidly evolving as a model system for humans given both the size and anatomic characteristics of the pig brain. 20 Swine have a gyrencephalic brain that is white matter predominant with similar developmental peaks to that of humans. Blood flow characteristics are also similar; however, swine have a rete mirabile. The internal carotid arteries provide most of the blood supply to the brain. The cranial portion of the vertebral artery provides some blood supply to the medulla. The vertebral formula of the pig is C7, T14–15, L6–7, S4, Cy20–23. The cauda equina starts at the level of S2–3. The intervertebral spaces are narrow and the vertebral processes more prominent than most species. 51
Eyes
The eye has 7 extraocular muscles and an open field of vision with a pupil and retina, similar to the human eye. Also present are a nictitating membrane, Bowman’s membrane, and Descemet’s membrane. The tapetum is absent. Retinal rosettes are much less common than in the retina of rats. Corneal abrasions can sometimes occur secondary to rubbing. Meibomian glands are greatly reduced in number compared with the third eyelid of the dog. However, a Harderian gland is present in the third eyelid, and in our experience, this is susceptible to xenobiotic induced atrophy when drugs negatively target or effect sebaceous glands. Swine also have a vitreous humor with characteristics similar to humans. 46,51
Musculoskeletal System
The bones and muscles of the pig are massive, and the pig has only rarely been used as a musculoskeletal model. The epiphyses of the long bones close at different times between farm and miniature breeds, for example, farm pigs 3–4 years, Hanford 2–4 years, Yucatan 2–3 years, and Yucatan micropig 1.5–2 years. There is a predominance of type II B muscle fibers with a lesser number of type II A and II C. Type grouping in the pig consists of islets of slow-twitch type I fibers surrounded by type IIA and more peripherally type IIB fast-twitch fibers. Although type IIB fibers are common in mice, rats, and rabbits and their distribution varies from muscle to muscle, they are much less common in larger mammals such as humans and monkeys, and muscle groupings tend to be in a mosaic pattern rather than the ordered groupings noted in pig muscles.
Intra-articular cartilage and associated ligaments are considered to have close characteristics to the analogous structures in humans. 5,51 Serous atrophy of fat is occasionally noted in the bone marrow of the fibia and tibia of control pigs and can be related to diet, but the syndrome is currently considered of unknown cause. Rarely, small foci of degenerative myofibers are noted in muscle bundles, and this can occur secondarily after prolonged anesthesia and recumbency and especially in the region adjacent to venipuncture. Other histologic lesions in the bone or muscles of laboratory pigs are rarely encountered.
Surgical Models and Procedures
Virtually any surgical procedure that can be performed on other large-animal biomedical models can be performed in swine. It is beyond the scope of this article to describe all the surgically induced models and procedures that can be performed in pigs. Development of these models has been previously described in depth. 50,52 Swine are the default model for nonsurvival surgical training classes. This includes developing expertise in performing general procedures as well as specialized techniques such as endoscopic and laparoscopic procedures. Although there is widespread use of simulator laboratories for training of physicians in some procedures, it is common for swine to be used in training for techniques that cannot be adequately simulated by in silico techniques. The haptics of performing surgery on a mannequin are dissimilar from animals, and the pig is commonly used in training for interventional catheter techniques, complex trauma procedures, and endoscopic procedures. Development of these devices and techniques in laboratories has led to the widespread use of swine as preclinical models for biocompatibility and function of implanted devices. The US Food and Drug Administration has accepted data from swine for both of these functions related to preclinical devices implanted in a variety of organs and systems. The most common survival surgical procedures performed in laboratory animals involve the implantation of catheters and devices, and this is true in swine as well. 44,51 The most common devices implanted include exteriorized catheters for blood collection and administration, vascular access ports (VAPs), and telemetry systems and implantation of devices and materials for preclinical files.
Surgical techniques, prevention of sepsis, and recommendations for specific materials used in experiments with swine models have been previously described and are similar to those of other animals. 48 –50 Using appropriate perioperative care procedures will allow the maintenance of implanted devices for months to years. Cardiovascular models include the following: myocardial infarction, transplantation, pressure overload hypertrophy, volume overload hypertrophy, dilated cardiomyopathy, valvular replacement, stent implantation, aneurysm repair, stent implantation, balloon angioplasty, graft implantation, pacemaker testing, and atherosclerosis-related therapy. The growth of the heart and cardiovascular system of the pig from birth to 4.5 months of age is analogous to the growth of the same system in humans. Consequently, the growing heart model has been used to study the effects of growth on implants and therapies to simulate the effects of growth from birth to the mid-teen years in humans.
The pig has been particularly useful in studies involving the treatment of atherosclerosis and myocardial infarction. The Seldinger technique can be used for interventional catheter-related procedures in either the femoral, carotid, or jugular vessels. Large devices such as telemetry or pacemaker units are best implanted in the jugular furrow. Smaller devices such as VAPs can be implanted subcutaneously and should be placed on the dorsum of the body to prevent the pig from rubbing and irritating the incisions (Fig. 2). 48 As mentioned earlier in this section, the pig’s cardiovascular system has general physiologic, anatomic, and histologic characteristics that make the pig a good model for biomedical research and toxicology testing. The thoracic cavity can be entered via either a lateral thoracotomy or a median sternotomy.
Therapeutic interventions targeting heart disease, including augmented mechanical devices and/or biotherapeutic delivery, require safety and efficacy studies, and the pig serves as a major resource for studying these applications. 9,54 The following are some of these applications.
Intravascular stents
From the initial angioplasty procedures in the 1980s, therapeutic procedures rapidly have progressed to intravascular stents (bare metal stents, polymer-coated drug eluting stents, bioresorbable stents). Coronary stents are implanted in thousands of patients every year in the United States (650 000 in 2006). 21 These advances have largely resulted from using pig models, and consensus groups have recommended the pig as the preferred model for studying intravascular stents. 21,38 The pig has also been used as an intravascular model for studying stents for treating pulmonary artery/aorta stenosis and peripheral artery diseases (Fig. 3). 15,38,39
Vascular aneurysms
The pig is a popular model for surgical induction of saccular aneurysms. Experimental surgery techniques have been applied to the carotid artery (unilateral and bilateral) and aorta. 35,38
Prosthetic valves
The pig is used as a model for both noninvasive (percutaneous deployment) and surgically invasive studies for biomechanical replacement of mitral and aortic valves. 7,383
Cardiac transplant
The pig not only has served as a model to understand mechanisms of rejection 27,39 but also has been proposed as a xenograft donor. 32,36
Ischemic heart disease (stem cell therapy/gene transfer)
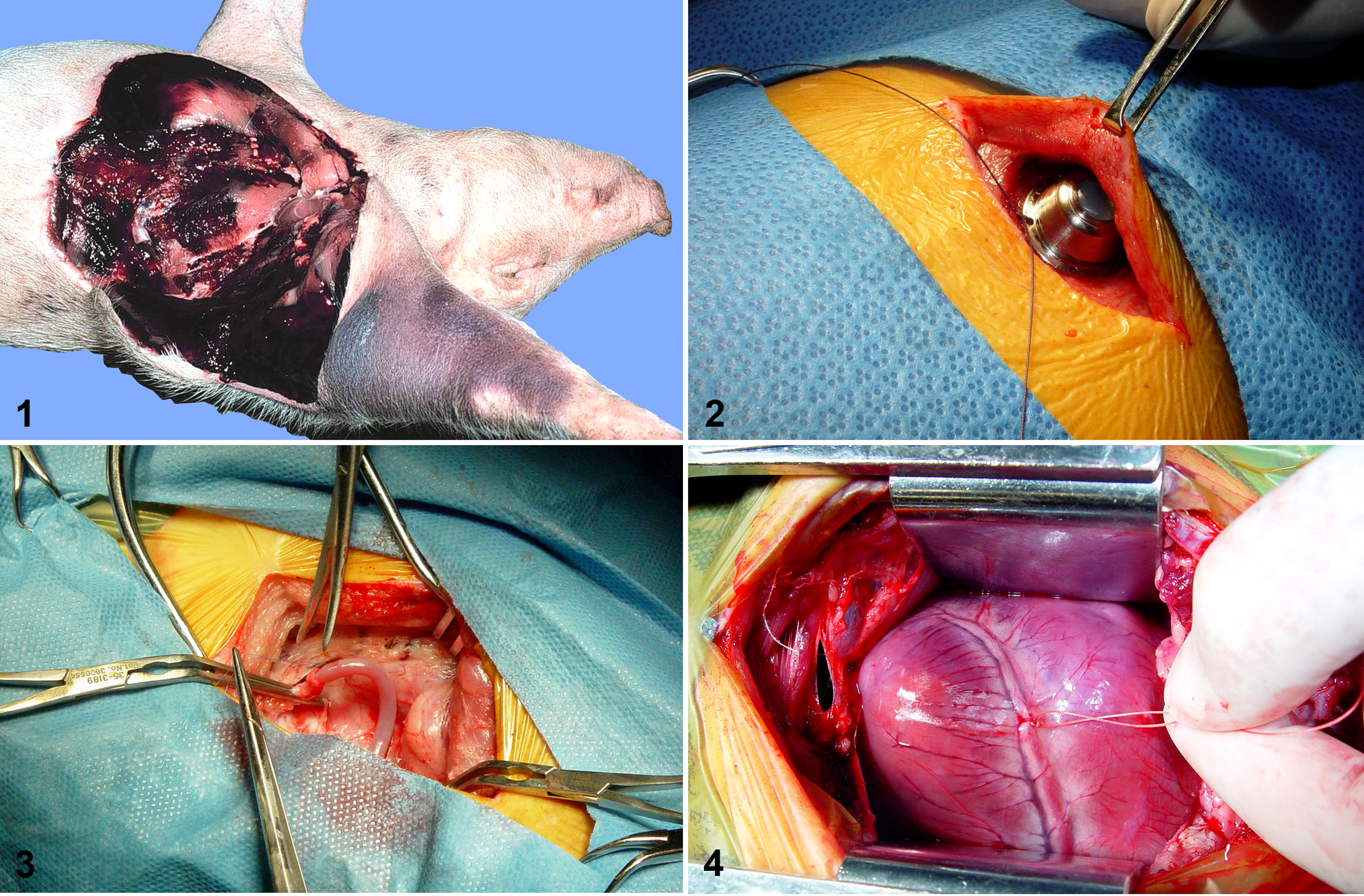
The pig is a model for studying the efficacy of stem cell therapy or gene transfer experiments in acute myocardial infarctions (Fig. 4) and in chronic end-stage heart disease. 27,54
Ventricular assist devices
The pig is a relatively new model for evaluating ventricular assist devices. 10 However, the new generation of pumps are designed for pediatric use in closed systems (transcutaneous energy transmission system), 28 and the pig makes a good model for this size and type of closed system.
Digestive system models
The pig model is used in endoscopic surgery, laparoscopic surgery, natural orifice transluminal endoscopic surgery, liver transplantation, islet cell transplantation, pancreatitis, cholecystectomy, biliary stents, intestinal fistulation, intestinal transplant, endotoxic shock production, and peritoneal dialysis. Abdominal incisions and closure are similar to other species. The intestinal tract requires 2–3 days to empty with fasting, and blood chemistry values are not affected until after 48 hours. Hyperosmotic purgatives can be used to accelerate the cleansing. Swine will readily consume liquid diets such as Ensure or Gatorade. Oral medications and test substances can be hidden and consumed in foods such as catfood meatballs, flavored syrups, fruits, and pastries.
Urinary system models
The pig model is used for renal transplantation, renal hypertension, intrarenal surgery, hydroureteronephrosis, intrarenal reflux, vesicoureteral reflux, artificial bladders, and ureteral stents. All of these models can be induced surgically. Because of the similarity of the anatomy and physiology of the kidneys discussed in the anatomy section of this article, the urodynamics of the pig compare favorably to those of the human. The anatomy is more similar to humans than even that of the nonhuman primates. 36,49,51 The kidneys can be surgically approached using a retroperitoneal flank approach, which avoids having to retract the intestinal mass. Retrograde passage of catheters through the penis is not possible, but that approach is easily performed in the female.
Dermatologic models
The pig model is used in wound healing, plastic surgery techniques, reconstructive techniques, burn models, and artificial skin grafts. Pigs have been used routinely in numerous dermatologic studies, both for experimental purposes and in drug development. The pig has been more commonly used than either primates or dogs for various wound healing and reconstructive surgery given the more similar anatomy of the skin. 47,51 They uses include skin surgery, melanoma research, wound healing, burn studies, microbiology, laser therapy, aseptic necrosis, vitiligo and depigmentation, dry skin, and studies of hypertrophic scarring.
In 1978, Eaglstein and Mertz 3 developed a novel porcine wound-healing model that allowed multiple test sites on the dorsal skin of the same pig. The model had small, identical wounded squares so that various treatment modalities and controls could be confined to a small area on the back of a single pig. This model has been refined and expanded to study various wound types such as partial thickness, full thickness, second-degree burns, ischemic wounds, and infected wounds. 24 Treatment modalities such as ointments, antibiotics, debridement, bandages, and laser therapy have all been evaluated. Wounds can be created by using specially designed dermatomes.
Numerous published reports in the last 40 years have attempted to promote the Sinclair miniature swine as a model of human cutaneous malignant melanoma. 26 Histopathologically, the tumors in pigs and humans are similar, and although these pigs develop a range of pigmented tumors from benign junctional nevi to metastatic malignant melanoma, there are numerous differences between the tumors in pigs and the tumors in humans. The syndrome in pigs is definitely inherited, present at birth but largely gone by puberty. In people, the tumor can be familial but is mostly sporadic and associated with exogenous factors. 52 The tumors seldom appear until after puberty and in the middle decades of life. In regressing tumors of the pigs, there is often depigmentation of the skin. Therefore, although the Sinclair pig has some utility as a model for human melanoma in judging efficacy of experimental therapeutics, caution is warranted in translating results to human tumors because the tumors may not share the aggressive biological behavior of their human counterparts and can even spontaneously regress.
Pigs have been used to study the feasibility of using laser hyperthermia for destroying small breast cancers. 33 Laser hyperthermia is a treatment modality that attempts to achieve tumor destruction by controlled tissue heating. If successful, it results in precise tumor destruction with less tissue trauma and lowered treatment cost. Pigs were chosen for preclinical studies because the subcutaneous tissue from the dorsal back area of the pig is very similar to human breast tissue. It has been demonstrated in the pig model that a fiberoptically delivered laser energy source is capable of precisely destroying small focal areas of tissue very similar to small breast tumors. 33
The red Duroc pig is a useful model for studying hypertrophic scarring, a clinical condition in humans that is often both frustrating and difficult to treat. Hypertrophic scarring can be the sequela to deep dermal injuries such as burns, abrasions, and deep skin graft donor sites. The scars are typically raised, firm, red, and pruritic but do not extend extensively beyond the wound margins. 6,57 They can be so disfiguring as to limit motion. Unfortunately, the cause is unknown and effective treatment protocols are lacking because no well-established animal model exists. Chronic human tissue studies are also limited by logistical difficulties because these hypertrophic scars are slow to develop and do not consistently occur. The female red Duroc pig is a potential useful model for both treatment studies and pathogenesis studies using histopathology and modern molecular techniques. 57 In fact, the female Duroc pig has been used to study hypertrophic scarring in deep donor sites for more than 40 years. 40 More recently, investigators using this model have immunolocalized decorin, versican, tumor growth factor-β1, and insulinlike growth factor-1 to assess the molecular pathogenesis of the lesion. 6
Neurologic models
The pig model is used in neurotrauma, stroke, Parkinson disease, cerebrospinal fluid catheters, cranial reconstruction, epidural catheters, and spinal cord trauma. Pigs have provided an excellent model for stroke associated with saccular aneurysm. 11 A transgenic model of Alzheimer disease is being developed along with behavioral testing for evaluation of models of neurodegeneration. 20 The study of traumatic brain injury is perhaps the most rapidly increasing area of research given the anatomy and size of the brain. Obviously, nonhuman primates have inherent behavior that is more similar to humans, but the small size of the brain in commonly used breeds compared with pigs is a disadvantage to their use. In addition, conservation associated with nonhuman primates is an issue with the development of significant studies in the field of trauma. In the literature, the use of pigs in neurosciences is the most rapidly increasing area of research for this species. 20,51 The main issue with neurosurgery in the pig is the thickness of the skull and the massive bone structure of the vertebrae. These approaches require power tools and skillful surgical approaches. Another specialized model involves fetal surgical techniques for implantation of catheters and fetal wound-healing studies. These techniques are highly specialized and require intense efforts to maintain fetal normothermia and homeostasis.
Obviously, perioperative care techniques such as caging, husbandry, and handling are required to successfully produce chronic surgical models, and these will not be addressed in this article. Likewise, many failures of chronic models involve the inappropriate design of the anesthetic and analgesic protocols. A series of DVD training modules have been developed to cover these aspects of the porcine surgical models, and videos of some surgical techniques are available. 18
Minipigs as Toxicologic Models in Safety Assessment Studies
From the foregoing, it is clear that the pig and minipig represent good models for humans in many ways. This includes models used in safety studies in the evaluation of pharmaceuticals, biopharmaceuticals, and other chemicals. In fact, the popularity of the minipig in pharmacology, pharmacokinetic, and toxicologic safety evaluation studies has increased rapidly over recent years. 4 In this context, the conventional pig and the minipig are regarded to be equivalent; however, given the minipig’s size and the fact that it is purpose-bred for research, the minipig is the standard porcine model used in modern regulatory safety testing. An increasing appreciation of the physiological similarities between human and pig, plus developing awareness of metabolic similarities, contributes to use of the minipig model. Added to this, the growing concerns over the use of other nonrodent species such as nonhuman primates and dogs in research have contributed to the rising interest in alternative species, including minipigs. There is an increasing amount of literature on use of minipigs in research, including overviews of their use in toxicology.20,31,45 The existence of significant amounts of background data, from a regulatory perspective and the perspective of good laboratory practices, provides strong support for the use of this species in mainstream research.
Although especially suited for dermal routes of drug administration, pigs are a good alternative for all routes of administration and durations of study when traditional species are found to be unsuitable. An increasing amount of literature indicates similarities between pigs and humans in the way they metabolize xenobiotics both in vitro and in vivo. 41 –43,56 The P450 system has been studied and partially characterized, and porcine metabolic pathways have been found to be relatively similar to humans, with significant overlap in substrate specificity. 29,56 Total P450 in conventional pigs is similar to that of humans, whereas in Göttingen minipigs, total P450 is 2- to 3-fold higher. 41 –43 When compared with total P450 for dogs, monkeys, and humans, there are some differences, with higher flavin monooxygenase (FMO)-dependent oxygenation activities in minipig liver microsomes and lower CYP-2A activity. Castration of male minipigs is reported to result in a 10-fold increase in CYP-2A activity, indicating that activity is regulated sexually. 42,43 However, CYP-1A, 2B, 2C, 2E, and 3A–dependent drug oxidation activity has been shown to be present and relatively comparable in minipigs. 29 As in rodents, hepatic metabolism in the minipig is susceptible to induction (eg, induction of CYP-1A using omeprazole), resulting in potentially increased metabolism of some drugs and compounds, but this has not posed a major problem in comparative pharmacokinetic modeling or interpretation. It is recommended that in the drug discovery/early development process, the in vitro metabolism studies should include minipig or pig microsomes in the screening battery, alongside those of humans and other standard laboratory species for comparison. Histologic evidence of induction-associated liver hypertrophy or thyroid hyperplasia, as noted in rodents, has not been published, and if these conditions occur at all in swine, they are extremely uncommon. Metabolism of lipoproteins occurs in a similar fashion to humans and makes swine an excellent model of atherosclerosis, as noted above. 49 –51
The body of evidence thus demonstrates that the minipig is at least as good as other species as a model for humans in safety studies. From a practical point of view, all of the standard measurements (ophthalmoscopy, electrocardiography, etc) are easily performed and the background is well-characterized in minipigs. Furthermore, in addition to routine toxicology studies, the minipig is used as an alternative species in performing embryo–fetal reproductive toxicology studies where other species are not suitable for particular reasons. Use of minipigs as models in juvenile studies in development of pediatric pharmaceutical products is increasing. A number of efficacy models have been developed in the minipig to specifically address Parkinson disease, 20,25 atherosclerosis, 11 and diabetes 18,34 (type 1 and type 2) research.
Choice of Species
The choice of which nonrodent species to use for safety assessment of new pharmaceuticals is extremely important and should always be based on good scientific principles. In the past, there has been a tendency to use the dog as a default, and if the dog was not suitable, the nonhuman primate was the next choice. Setting aside the testing of certain biological molecules, for which the nonhuman primate would normally be the best or even the only choice, ethical concerns over use of primates and dogs has led to the search for alternatives. The increased interest in the minipig is helped, as discussed earlier, by the physiological similarities between pigs and humans in several major organ systems and the growth in availability of background data and regulatory acceptance.
Applying sound scientific principles, investigators should always choose the nonrodent species that is the most relevant. There can be many cases where one species does not have a great advantage over others; however, there are some obvious examples where certain species should not be used. For example, it is widely accepted that the dog is an unsuitable model for testing NSAIDs and other compounds such as mitogen-activated protein kinase inhibitors. Low tolerance to NSAIDs in dogs, seen as gastrointestinal lesions, prevents or restricts their ability to demonstrate other toxicities because it is not possible to set dosages high enough. 19 Minipigs can tolerate higher doses of NSAIDs, they are more tolerant of gastric lesions, ulcers heal better, and dosing of up to 1 year is possible. 19 In this regard, the minipig responds to NSAIDs more like humans than does the dog. Cardiotoxic compounds such as doxorubicin produce widespread myocardial inflammation and necrosis in swine, in a pattern similar to the patterns noted in other species. However, the dog has been demonstrated to be more sensitive to some specific cardiomyotoxins than is the pig, such as in the sympathomimetic class of drugs. 20 In dogs, cardiac toxicity is seen following treatment with sympathomimetic drugs at doses close to the therapeutic level, and therefore the pharmacodynamic effects prevent possible demonstration of other relevant toxicity. Using a benzodiazepine that caused comparable increased heart rate in dogs and minipigs, investigators showed that although dogs developed myocardial necrosis, minipigs were unaffected, and that in this way minipigs respond in the same way as humans. 19 It has been suggested that minipigs can be a suitable species of choice where a drug demonstrates a significant degree of first-pass metabolism, because these particular hepatic metabolic pathways more closely resemble those in humans than do those in dogs. 17,41 –43
Minipigs in Safety Studies
As previously discussed, from all practical considerations, minipigs can be regarded as interchangeable with dogs as experimental animals, and their use has increased tremendously in certain areas of drug development such as in preclinical acute, subacute, and chronic toxicity studies. In contrast, the minipig’s use as a model in early drug discovery or efficacy studies has remained much more limited. The reason is attributable to limited availability of drug material in the early stages of drug discovery. As compound is normally in very short supply during this stage of pharmaceutical development, most experimentation is either performed in vitro, ex vivo, or in rodents. Larger animal models such as swine, dogs, or monkeys normally are used when lead candidates move from discovery into preclinical development and drug supply is scaled up. There are exceptions in which swine efficacy models are preferred in early drug discovery, as was noted previously with atherosclerosis or other cardiovascular models, or occasionally in dermal applications. Early discovery efforts involved with biomedical devices are another exception. When assessing biomechanical devices such as stents, drug substance isn’t an issue, and the similar size of the animals makes them an ideal model in these experiments. In fact, pigs have increasingly been used throughout all development stages to assess the preclinical safety of mechanical implements such as stents or mechanical heart valves. 7 The current ISO guidelines indicate that the pig is a suitable, if not preferred, model for testing local effects after implantation. 7 In the field of food safety, pigs are used throughout the discovery and regulatory process given the similar omnivorous eating patterns of pigs and humans and the previously mentioned similarities in the digestive tract. 7 A large historical database is available for pigs in the food safety industry.
Given the morphological and physiological similarities between human and porcine skin compared with other laboratory animal species, the minipig has become a preferred model to evaluate the safety profile of dermally applied xenobiotics. The minipig shares all of the advantages of comparative dermatology with its larger porcine relatives, but the smaller size precludes the difficulties associated with handling larger pigs in long-term toxicity studies. Hence, the Göttingen minipig, in particular, is increasingly replacing the rabbit and guinea pig for dermatotoxicity testing in drug development. 19,22 However, interpretation of dermal toxicity studies in minipigs is still hindered somewhat by individual variability in dermal tolerability and a potentially wide spectrum of treatment-induced (and not necessarily drug-induced) skin alterations, just as it is in other species used for this purpose. 22 Viscous vehicle preparations in particular, such as gels, ointments, or cream applications, can result in inflammatory reactions, epidermal hyperplasia, and folliculitis of the skin in minipig dermal studies as they do in guinea pig or rabbit skin. 22
Although dermal dosing is a routine procedure, the amounts of the drug product that can be delivered to the animal are limited. The amount of the surface that can be exposed is in the region of 10% of the total body area, although larger areas can be used. Once this target starts to approach areas in the region of 20% of body surface, the dose applied to skin may not be uniform, because the nature of pig skin, especially thickness, varies over the body of a minipig.
All dose routes are possible in the minipig, from the more standard (eg, oral, subcutaneous, intravenous, and intramuscular) to the less standard (eg, infusion, intravaginal, and intravesicular). The size of the animals permits collection of sufficient volumes of blood for most purposes. Standard parameters such as ophthalmoscopy and electrocardiography are routinely measured. Certain practical issues need to be considered with respect to use of any species, and minipigs are no exception. Although dogs can tolerate quite large doses of administered substances packed in capsules, the tolerance of minipigs is somewhat less. Therefore, both the size of the capsules that can be administered and the number that can be given at any one time are smaller in the case of minipigs compared with dogs. 4 The lack of any sizeable peripheral veins in the minipig means that repeated intravenous injections can only be achieved via previously implanted catheters. Blood samples, however, are readily collected from the jugular vein or bijugular trunk without the need for prior surgical preparation. Detailed descriptions of vascular access, oral dosing, and catheter implantation have been published. Pigs can be readily trained to accept oral or dermal test substances using standard methods with food treats. Pigs have also been trained to weigh themselves using the same methods. 20,51
Handling of minipigs is straightforward and requires good technique, patience, and experience. In the hands of a skilled technician, the animals can be as easy to handle as dogs, despite their size. Certainly, the size of the animals should not be a deterrent from using them purely for practical reasons, although the requirement for test drug can be prohibitive. Operant conditioning techniques such as clicker-training are now being widely used to get the animals to comply during the performance of dosing and sampling procedures, reducing stress for both animals and handlers while at the same time reducing the number of staff needed to perform these basic techniques.
Safety Pharmacology Studies
The use of minipigs in safety pharmacology studies, specifically in cardiovascular studies, is increasing. 4 It has always been the trend that the nonrodent species selected for the cardiovascular safety pharmacology study is the same one used for general toxicology studies—based on the appropriateness of the species in relation to the product under test. It therefore makes sense that in cases where the minipig is used in the main toxicology studies, it should also be used for the safety pharmacology. As we have noted earlier, the cardiovascular system of the pig, including the vasculature of the heart, is anatomically very similar to that of human. Several groups are now using minipigs in these safety pharmacology studies, with an increasing amount of background data generated. This includes data demonstrating that the minipig is a suitable model to identify compounds that have an effect on QT interval. 4 The amount of literature on the subject is limited, although more is being presented in the form of posters at scientific meetings, and the field is expected to grow.
Reproductive Studies
Traditional species used in assessment of potential effects of drugs on reproduction and development have been rats (fertility and prenatal/postnatal studies) and rats and rabbits (embryo–fetal studies). Where rat or rabbit has not been suitable for some reason, the most commonly used species have been mouse and, for embryo–fetal studies, the nonhuman primate. Other species, such as the dog and minipig, have certain advantages and disadvantages as alternatives, particularly compared with the rabbit or nonhuman primate. There is a slowly increasing use of the minipig as an alternative nonrodent species for embryo–fetal studies, with a growing database of background and spontaneously occurring abnormalities and anomalies.
Developmental and Juvenile Toxicity Studies
In the field of developmental toxicology, there is an increasing focus on effects of drugs in juvenile animals according to the guidelines that have been published by regulatory authorities in relation to administration of drugs to human pediatric populations. From a purely practical point of view, the minipig is a very useful species for use in juvenile toxicity studies where a nonrodent is required. Minipigs are easy to handle, their reproduction is such that sufficient numbers of young animals can be obtained as required to perform studies in the shortest time and with a sensible study design, and they are of a size that permits performance of basic procedures, such as blood sampling, from an early age.
Although much work has already been done to develop this species within the field of safety evaluation of human pharmaceuticals, there are still gaps in the knowledge and background database that need to be filled and areas identified where minipigs may not be a suitable choice. For example, in developmental studies the minipig is less versatile than the rat in regard to tests of learning and memory. Fortunately, test methods are being developed that can add valuable information in relation to the potential effects of drugs on the nervous system of growing pigs. Other potential areas within drug development where the use of minipigs could be further exploited, such as within the fields of immunotoxicology and inhalation toxicology, will continue to be developed as more background data are assimilated.
Conclusion
The usefulness of pigs in experimental research and particularly the use of minipigs in routine safety testing studies are clear. A brief review can only scratch the surface of the potential for swine research models, but it illustrates the versatility of the pig and the wide variety of studies in which pigs can be used. Our combined experience suggests that there is an increasing interest in, and demand for, these animals in both regulatory and experimental research. As the demand has increased so has the development of background data to the extent that minipigs have become regarded as mainstream models in safety evaluation. From this, we can conclude that pigs and minipigs will continue to be useful tools for the investigator, toxicologist, and research pathologist.
Footnotes
Acknowledgements
This work was originally presented as a series of lectures at the “Pigs and Minipigs as Research Models” minisymposium at the 60th annual meeting of the American College of Veterinary Pathologists in Monterey, California, on December 6, 2010. We appreciate the assistance of Dr Justin Vidal for suggestions and review of this manuscript; Dr Aaron Sargeant, Dr John Van Fleet, and Stephen Davis for potential photographic contributions; and Beverly Maleeff for technical assistance.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.