Abstract
The immunohistochemical features of dystrophic axons in brain tissues of Papillon dogs with neuroaxonal dystrophy (NAD) were examined in comparison with 1 dog with cerebellar cortical abiotrophy (CCA) and a dog without neurologic signs. Histologically, many dystrophic axons were observed throughout the central nervous system of all dogs with NAD. These axonal changes were absent in the dog with CCA and in the control dog. Severe Purkinje cell loss was found in the dog with CCA, whereas the lesions were milder in all dogs with NAD. Immunohistochemically, the many dystrophic axons were positive for neurofilaments, tau, α/β-synuclein, HSP70, ubiquitin, synaptophysin, syntaxin-1, and synaptosomal-associated protein-25 (SNAP-25). A few dystrophic axons were positive for α-synuclein. In addition, these dystrophic axons, especially in the nucleus gracilis, cuneatus, olivaris, and spinal tract of the trigeminal nerve, were intensely immunopositive for the 3 calcium-binding proteins calretinin, calbindin, and parvalbumin. The accumulation of synapse-associated proteins in the dystrophic axons may indicate dysfunction of the synapse at the presynaptic portion. The accumulation of α-synuclein in the dystrophic axon and region-specific appearance of calcium-binding protein-positive spheroids are considered as unique features in NAD of Papillon dogs, providing the key to elucidate the pathogenesis.
Introduction
Canine neuroaxonal dystrophy (NAD) and cerebellar cortical abiotrophy (CCA) are hereditary suspected neurodegenerative diseases with moderate to severe cerebellar atrophy. 10, 47 Because these 2 diseases have similar clinical, neurologic, and magnetic resonance imaging (MRI) features, discrimination between them during life is very difficult. Definitive diagnosis of canine NAD or CCA is made by complete pathologic examination of the central nervous system (CNS). Recently, we have reported the clinicopathologic features of NAD and CCA in Papillon and Papillon-related dogs. 32 Although NAD and CCA showed similar degenerative cerebellar changes, the lesions were more severe in CCA than in NAD. 5, 7, 33, 39, 45 Furthermore, NAD showed characteristic axonal degeneration involving formation of numerous spheroids throughout the CNS. 8, 9, 11, 12, 32, 38, 43
Human NAD is clinicopathologically classified into 4 subtypes: infantile, late infantile, juvenile, and adult types according to the age at onset. Hallervorden-Spatz syndrome, 15, 16, 24 one of the most common infantile NADs, is diagnosed by the presence of spheroids and iron accumulation in the globus pallidus and substantia nigra reticulata. Similar iron accumulation has never been found in canine NADs. Previously, the axonal changes in canine NAD are thought to appear through abnormal accumulation of neurofilament (NF) as a result of distal axonal loss and synaptic abnormalities. 12 Sacre et al. 38 described that the axonal spheroids in canine NAD were located mainly within the sensory systems including the proprioceptive and vestibular nuclei, and that these axonal changes appeared to affect the preterminal or presynaptic portions. Immunohistochemically, accumulation of NF, ubiquitin, Rab-3a, and several synaptic proteins, including synaptophysin, synaptosomal-associated protein 25 (SNAP-25), and synapsin-I, has been observed in the axonal spheroids of Rottweiler dogs with NAD. 43 These findings were considered to reflect severe disruption of axonal transport in these dystrophic axons. In addition, several calcium-binding proteins, including calbindin, calretinin, and parvalbumin, are mainly distributed within the local interneuron containing γ-aminobutyric acid (GABA) as a neurotransmitter. 44 Moreover, these calcium-binding proteins are known to present in morphologically distinct populations of GABAergic interneuron in monkey and human brains. These proteins are used for cyto- and chemo-architectural investigations in several animals. 17 Their function are, although not well defined, suspected to contribute to cytoskeletal organization, cell motility and differentiation, axonal transport, membrane excitability, and the synthesis and release of certain neurotransmitters. 44 Abnormal accumulation of calcium-binding proteins has been demonstrated in several neurodegenerative disorders, including NAD of dogs, cats, and horses. 37, 42, 43 However, the relationship between axonal degeneration and the accumulation of calcium-binding proteins has not been well investigated in canine NAD.
In the present study, immunohistochemical analyses using antibodies against several axonal components, including NF, tau, synuclein, heat shock proteins (HSPs), several synapse-associated proteins, and calcium-binding proteins, were performed to clarify the immunohistochemical features of the spheroids in Papillon dogs with NAD.
Materials and Methods
Tissue samples and histology
Brain tissues including the cerebrum, cerebellum, brain stem, and spinal cords from 5 Papillon dogs, including 3 with NAD, 1 with CCA, and 1 necropsied at 4 months of age without any neurologic signs, were used. The clinical features of these dogs are summarized in Table 1. The details have been reported previously. 32 All tissue samples were fixed in 10% formalin and embedded in paraffin. For microscopic study, 6-μm-thick paraffin sections were stained with HE. Some selected sections also were stained with Berlin blue for the identification of iron deposits. The globus pallidus and substantia nigra reticulate of dog No. 3 also were investigated. The spleen tissue with severe hemosiderin deposits of a necropsied dog was used as a positive control.
Clinical features of cases investigated in this study.

∗ M = male; F = female; NAD = neuroaxonal dystrophy; CCA = cerebellar cortical abiotrophy.
† m = month-old age; y = year-old age.
Immunohistochemistry
Selected sections from the cerebrum, cerebellum, brain stem, and spinal cords were used for immunohistochemistry. After antigen retrieval with an autoclave at 121°C, 5 minutes, endogenous peroxidase was blocked with 3% hydrogen peroxidase in methanol at room temperature for 10 minutes. Primary antibodies against for NFs, tau, α-synuclein, α/β-synuclein, HSP70, ubiquitin, synaptophysin, syntaxin-1, SNAP-25, calbindin, calretinin, and parvalbumin were used. The details of these primary antibodies are shown in Table 2. Each section was incubated with the primary antibody at 37°C, 60 minutes. All sections were incubated with Envision polymer reagent (DAKO-Japan, Kyoto, Japan) at 37°C, 30 minutes, and then these section were visualized with 3,3′-diaminobenzidine (Sigma, St. Louis, MO) in Tris-buffered saline. Sections were counterstained with hematoxylin or light green.
Antibodies used in the present study.
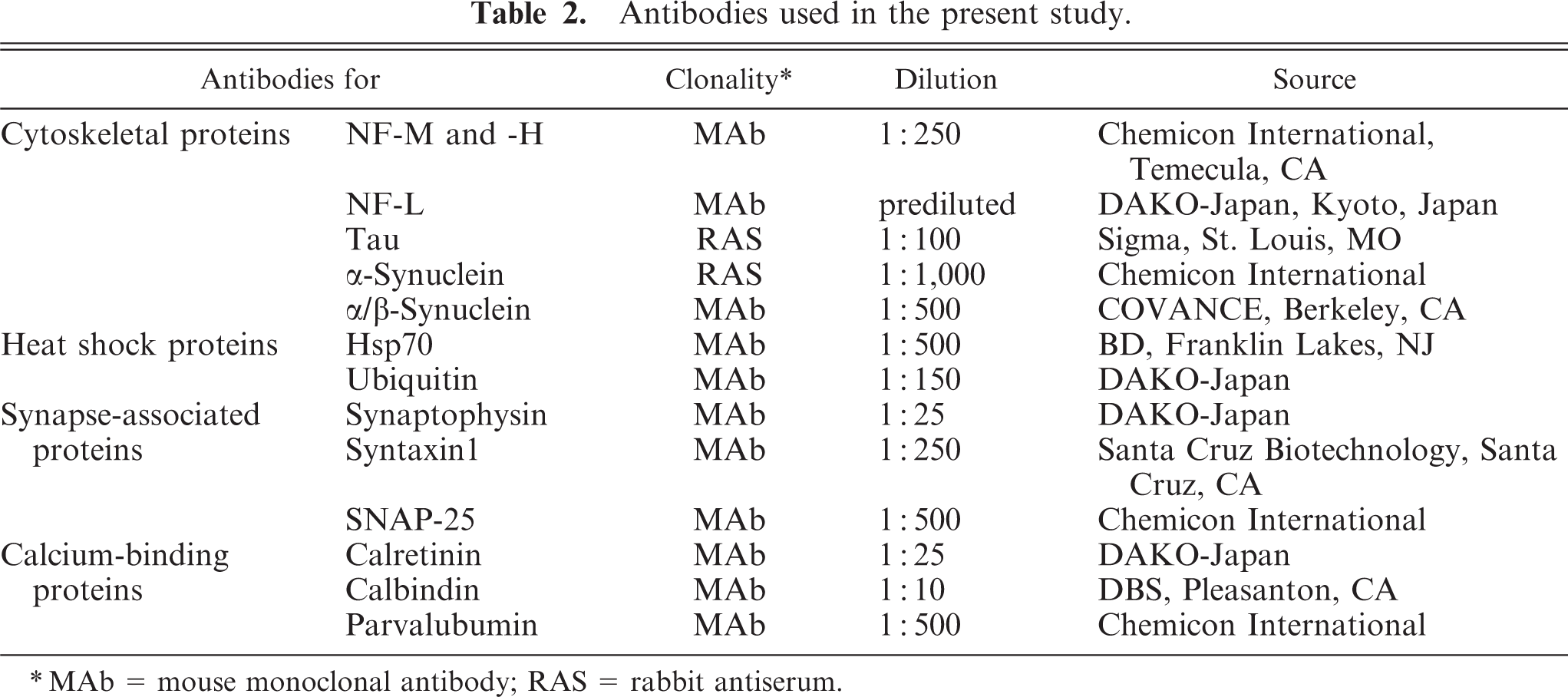
∗ MAb = mouse monoclonal antibody; RAS = rabbit antiserum.
Results
Histologic features
Spheroid formation was a very characteristic feature in all dogs with NAD, while no spheroid was observed in the brain of the dog with CCA and the control dog. On the other hand, cerebellar neuronal cell loss, including Purkinje and granular cells of the dog with CCA, was more severe than that of all dogs with NAD. Spheroids in NAD were scattered throughout the CNS, being especially predominant in the dorsal horn of the spinal cord, nuclei gracilis, cuneatus, and olivaris, and spinal tract of the trigeminal nerve, and its circumference in the brain stem, as described previously. 32 In the cerebellum of all dogs with NAD, many spheroids also were found in the white matter and granule cell layer compared with the cerebrum. They also were densely distributed in the cerebellar nuclei of dog No. 3. The spheroids varied in size from 20 to 50 μm in diameter and were heterogeneous in appearance (Fig. 1). In dog Nos. 2 and 3, severe lesions, including granular and Purkinje cell loss, were located in the vermis. Berlin blue staining revealed no iron deposits in all brain regions of dogs with NAD and the control dog, including the globus pallidus and substantia nigra reticulate of dog No. 3.
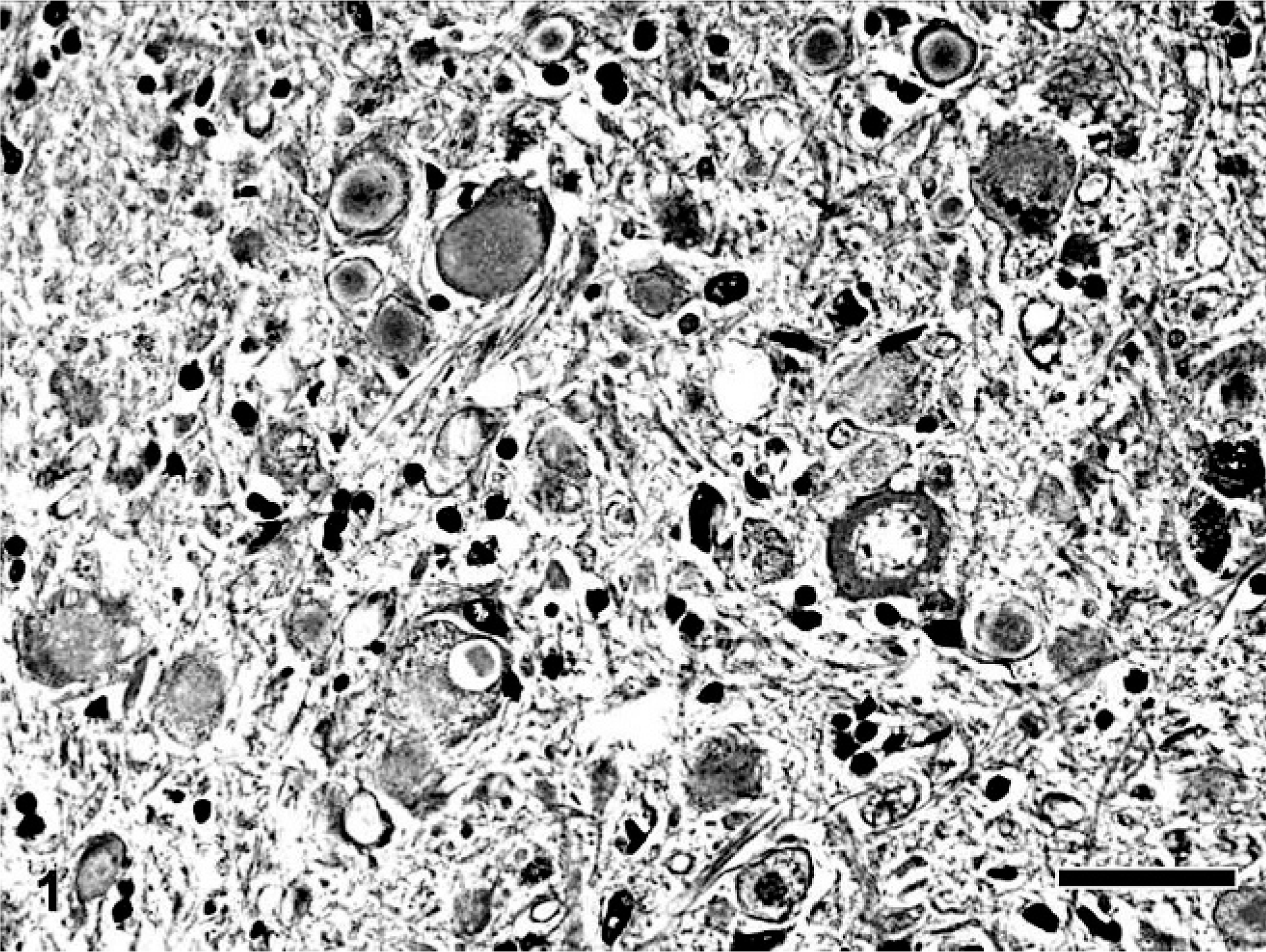
Cerebellar nucleus; NAD, dog No. 3. The spheroids vary in sizes from about 20 to 50 μm and show various morphologies, including homogeneous, granular, and concentric appearance. HE stain. Bar = 50 μm.
Immunohistochemical features
Control tissues
By immunohistochemistry for NF-L, M, and H, medium- to large-sized neurons in the cerebral cortex and spinal gray matter, Purkinje cells and cerebellar nucleus, and several nuclei in the brain stem, such as nuclei gracilis, cuneatus, and olivaris, were intensely immunopositive. Tau protein diffusely accumulated in the cytoplasm and their processes of neuronal and glial cells in almost all brain regions examined. Many neuronal and glial cells in the cerebrum, cerebellum, brain stem, and spinal gray matter were feebly positive for HSP70. Some HSP70-positive neuronal and glial cells showed nuclear immunoreactivity. On the other hand, all brain cells were immunonegative for ubiquitin. Immunohistochemistry for synapse-associated proteins, including synaptophysin, sintaxin-1, and SNAP-25, exhibited similar staining pattern. These antibodies diffusely labeled the neuropile of the cerebral cortex, molecular layer, and glomerula cerebellaria in the cerebellum, brain stem, and spinal gray matter. The staining pattern of both α- and α/β-synuclein was similar to that of synapse-associated proteins. By immunohistochemistry for calcium-binding proteins, Purkinje cells were strongly positive for calbindin, and some neurons of the cerebellar nucleus also were weakly positive. On the other hand, Purkinje cells were completely negative for calretinin and parvalbumin. The nerve fibers of the medial lemniscus and trigeminal nerve were immunopositive for calretinin. Parvalbumin immunoreactivity was found in the cerebellar peduncle, lateral and medial lemniscus, longitudinal fibers of the pons, and medial longitudinal fasciculus in the brain stem, and some small neurons in the spinal gray matter. A few neuronal cells in the cerebral cortex were immunopositive for these calcium-binding proteins, while there was no specific distribution pattern of the positive cells.
The immunohistochemical findings of the dystrophic axons of the 3 dogs with NAD are summarized in Table 3. The results for the dog with CCA and the control are not included because of the absence of spheroids. Briefly, the spheroids in almost all CNS regions of all dogs with NAD were intensely immunopositive for NF, α/β-synuclein, HSP70, and ubiquitin. Furthermore, regional restriction was recognized in the immunoreactivities for α-synuclein, syntaxin-1, SNAP-25, calretinin, calbindin, and parvalbumin, as detailed below.
Results of immunostain.
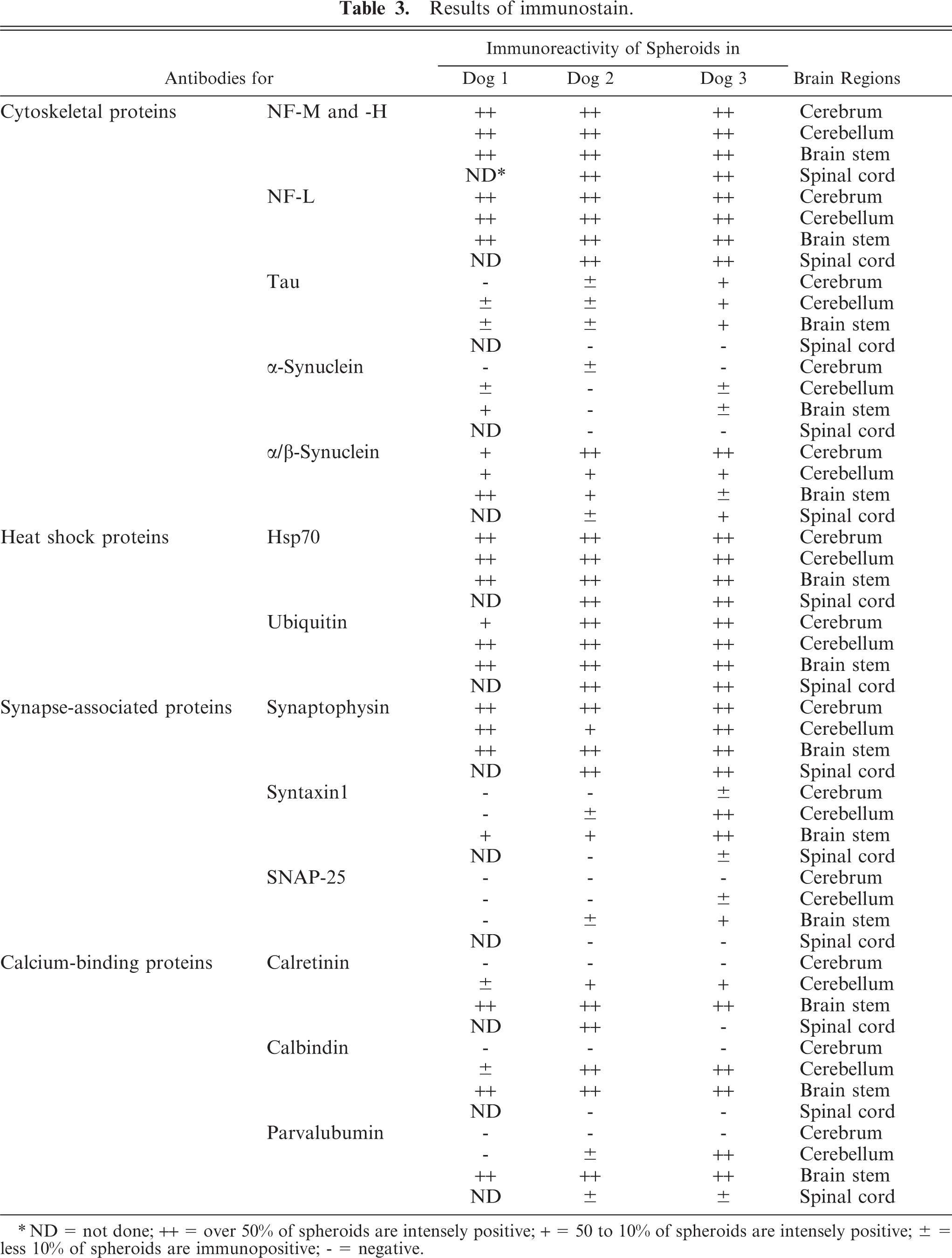
∗ ND = not done; ++ = over 50% of spheroids are intensely positive; + = 50 to 10% of spheroids are intensely positive; ± = less 10% of spheroids are immunopositive; - = negative.
NF and tau
Almost all spheroids in the cerebrum, cerebellum, brain stem, and spinal cord of dogs with NAD showed intense immunoreactivity for NFs. On the other hand, several spheroids were positive for tau. No specific localization pattern of tau or NFs immunopositive spheroids was recognized.
Heat shock proteins
Almost all spheroids in the cerebrum, cerebellum, brain stem, and spinal cord of dogs with NAD were intensely positive for both HSP70 and ubiquitin. Many surviving neuronal and glial cells of dogs with NAD also were positive for HSP70. The immunoreactivity of HSP70 in the cerebellum, brain stem, and spinal cord was stronger than that in the cerebrum. Ubiquitin immunoreactivity of survived neuronal and glial cells of dogs with NAD was very weak compared with that for HSP70.
Synapse-associated proteins
Synaptophysin labeled spheroids in almost all brain regions of dogs with NAD. The immunoreactivity was same equivalent to that of NFs. Only a small number of spheroids in the brain stem and cerebellum were positive for syntaxin-1 and SNAP25 in dogs with NAD, except for dog No. 3 (Fig. 2). In dog No. 3, a large number of spheroids in the cerebellum, brain stem, and sometimes the cerebrum were intensely immunopositive for syntaxin-1.
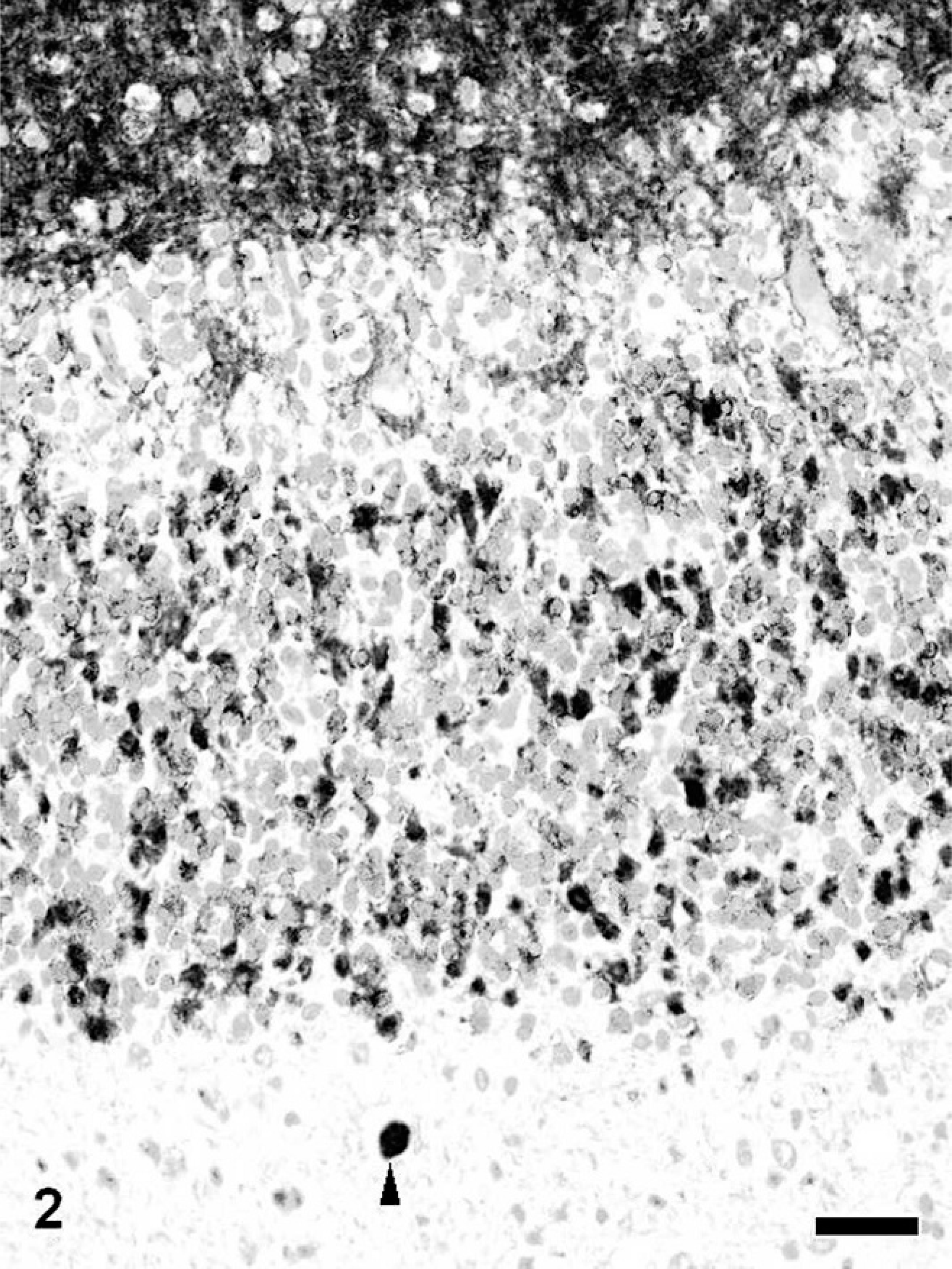
Cerebellum; NAD, dog No. 2. A spheroid in the white matter is immunopositive for syntaxin-1 (arrowhead). The intact molecular layer and glomerula cerebellaria in the granular cell layer also are positive for syntaxin-1. Envision polymer method. Counterstained by Mayer's hematoxylin. Bar = 50 μm.
α- and α/β-synuclein
A large number of spheroids in all dogs with NAD also were intensely immunopositive for α/β-synuclein (Fig. 3). Antibody for α-synuclein also labeled dystrophic axons in dogs with NAD, while the number of immunopositive spheroids was smaller than those for α/β-synuclein.
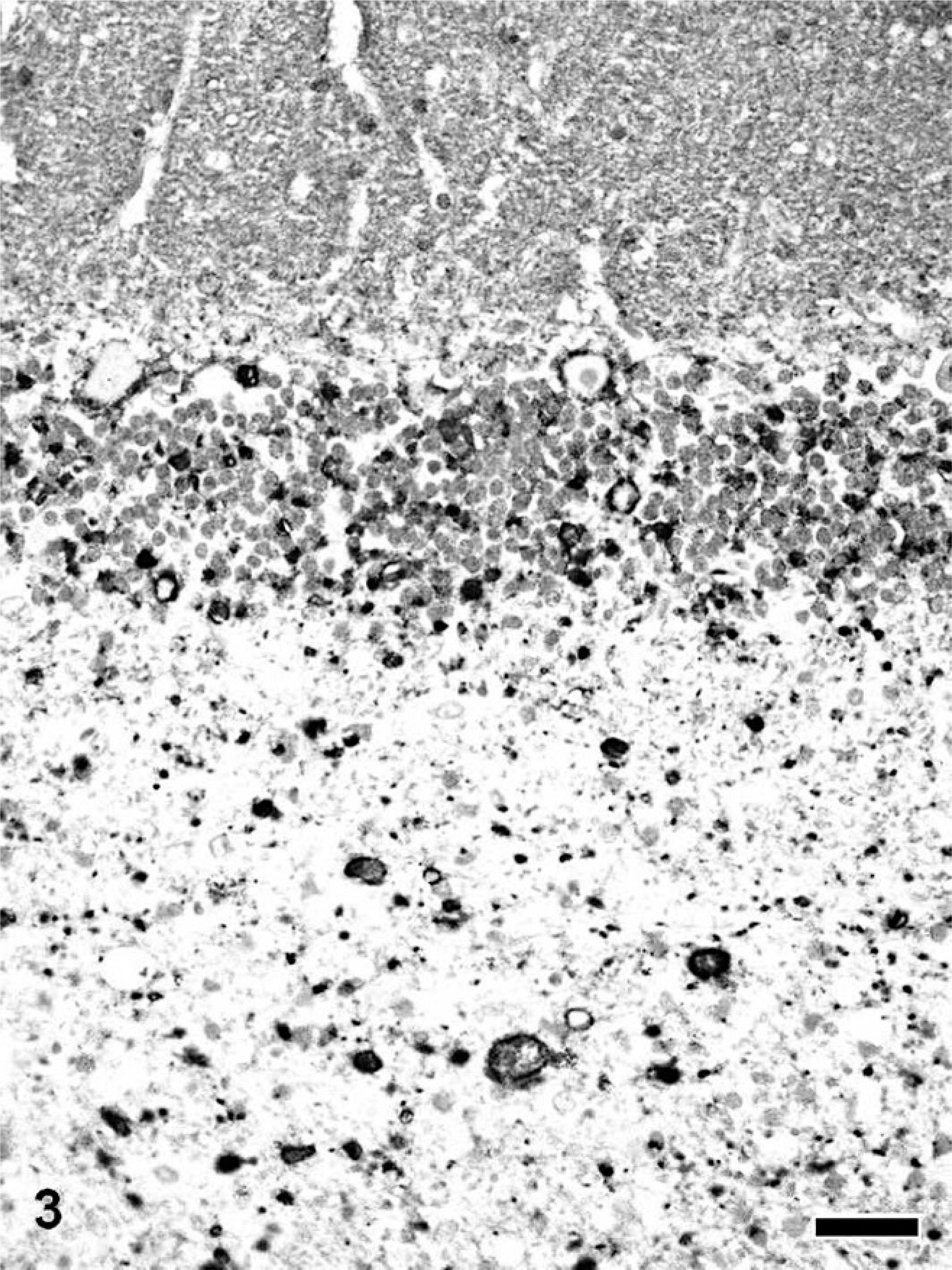
Cerebellum; NAD, dog No. 1. Many spheroids immunopositive for α/β-synuclein are evident in the granular cell layer and white matter. Envision polymer method. Counterstained by Mayer's hematoxylin. Bar = 50 μm.
Calcium-binding proteins
Calbindin-, calretinin-, and parvalbumin-immunopositive spheroids were distributed in the limited region of the CNS. In the cerebrum of dogs with NAD, the spheroids were completely negative for these antibodies. On the other hand, in the cerebellum, many dystrophic axons in the granule cell layer and white matter were intensely immunopositive for calbindin (Fig. 4), calretinin, and parvalbumin. Furthermore, in dog No. 3, the spheroids in the cerebellar nucleus also were intensely immunopositive for the proteins. Antibody to calbindin also labeled morphologically intact Purkinje cells and their processes of dogs with NAD (Fig. 4). In the brain stem, many spheroids in certain nuclei, including the nucleus gracilis, cuneatus, olivaris, and the spinal tract of the trigeminal nerve, were immunopositive for these 3 antibodies (Fig. 5), while the intensity was varied in each nucleus. The spheroids in the nucleus gracilis of dog No. 3 showed very intense immunoreactivity for calretinin, whereas those in the nucleus cuneatus exhibited weakly or no immunoreaction (Fig. 6). The immunoreactivity of spheroids for parvalbumin was more intense in the nucleus olivaris and spinal tract of the trigeminal nerve than those in nucleus gracilis and cuneatus. In the spinal tract of the trigeminal nerve of dog No. 2, some parvalbumin-positive spheroids were adjacent to the neuronal cell body (Fig 7). Although spheroid formation was prominent in the spinal dorsal horn, only a few spheroids were feebly positive, and most were negative for calbindin, calretinin, and parvalbumin in all dogs with NAD. Exceptionally, the spheroids in the dorsal horn of dog No. 2 exhibited intense immunoreaction for calretinin.
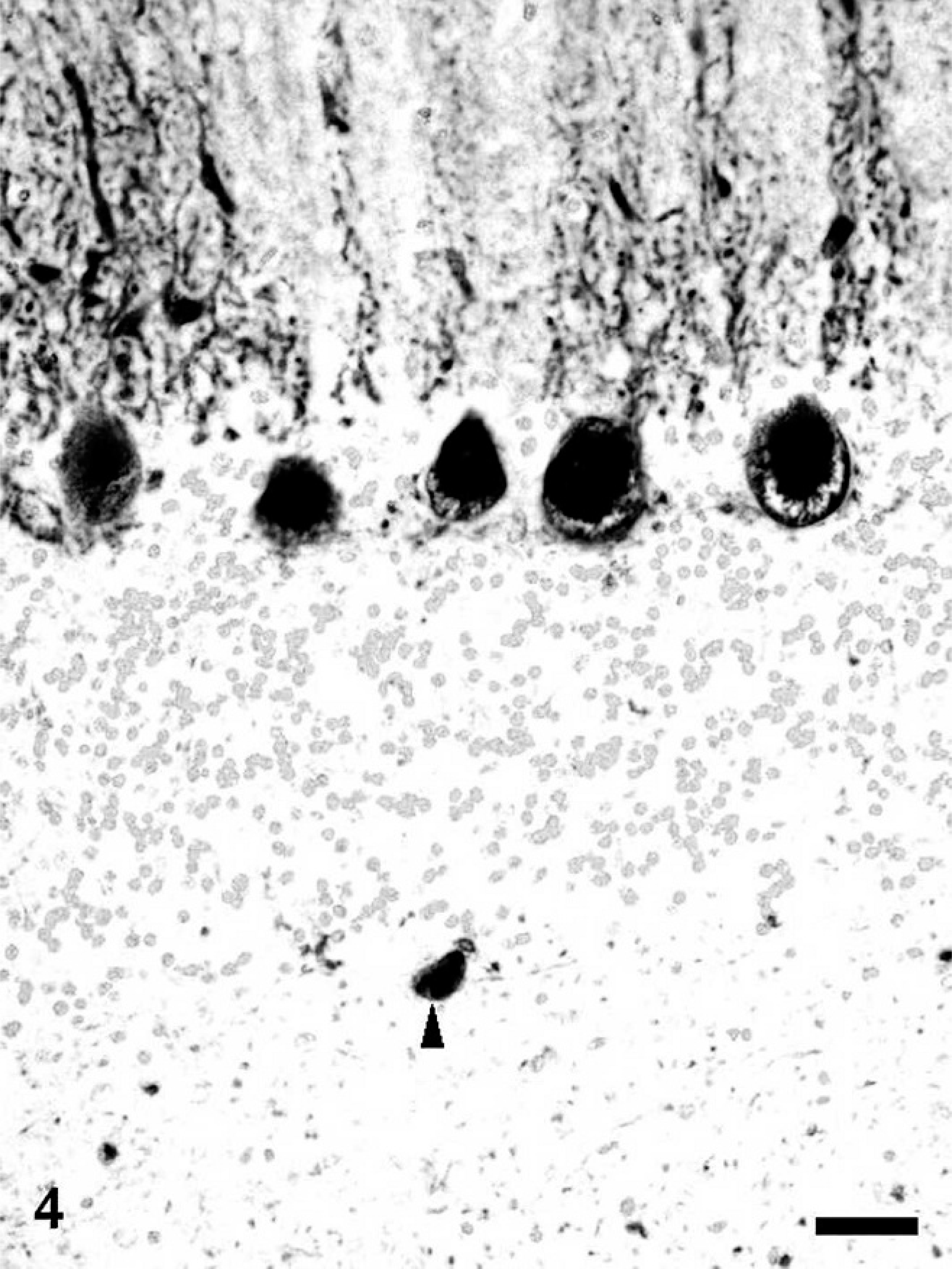
Cerebellum; NAD, dog No. 2. A spheroid in the white matter is immunopositive for calbindin (arrowhead). Morphologically intact Purkinje cells and their processes also are positive for calbindin. Envision polymer method. Counterstained by Mayer's hematoxylin. Bar = 50 μm.
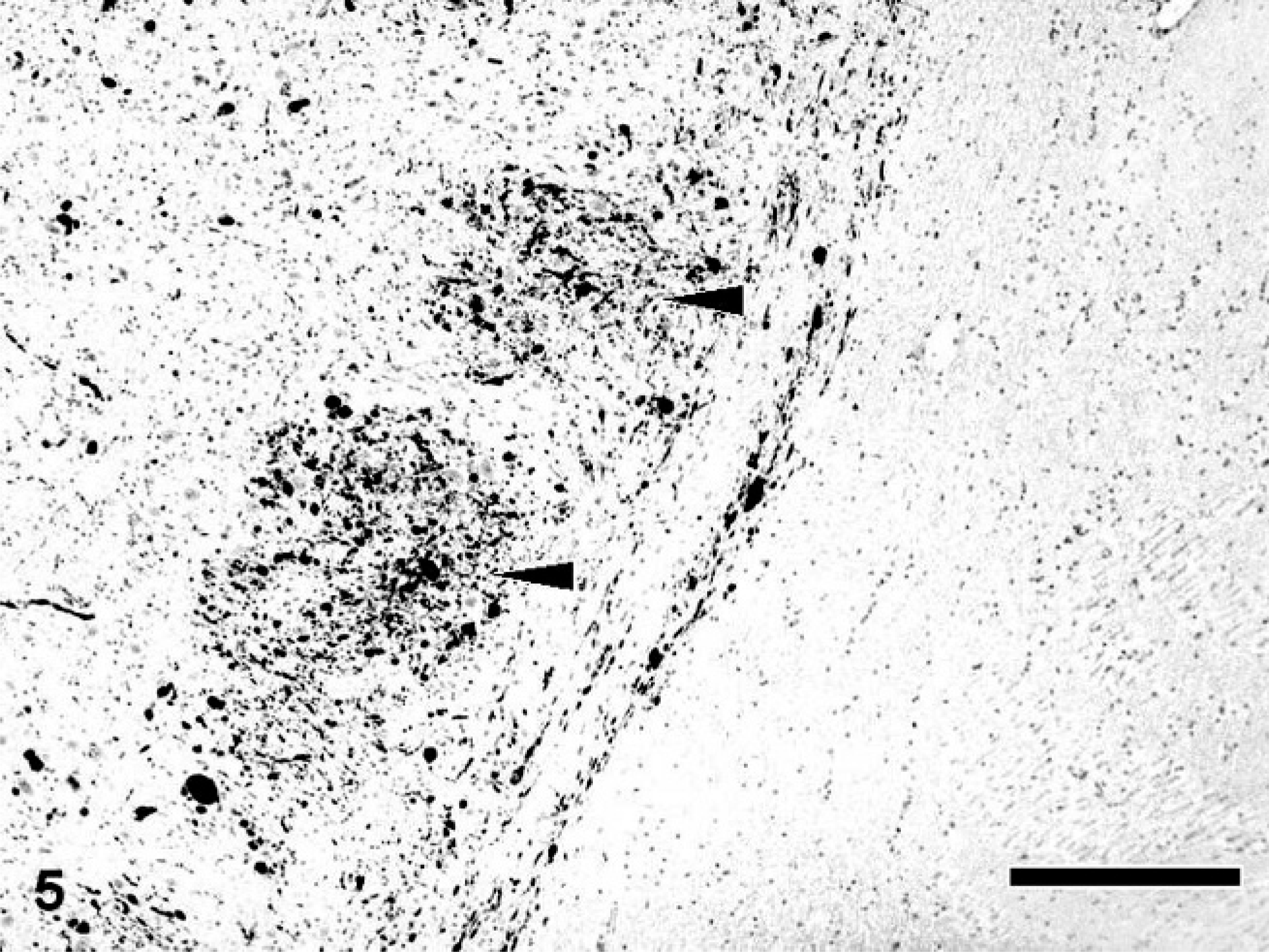
Medulla oblongata; NAD, dog No. 2. Many spheroids immunopositive for parvalbumin are evident in the nucleus of the spinal tract of the trigeminal nerve (arrowhead). Envision polymer method. Counterstained by Mayer's hematoxylin. Bar = 50
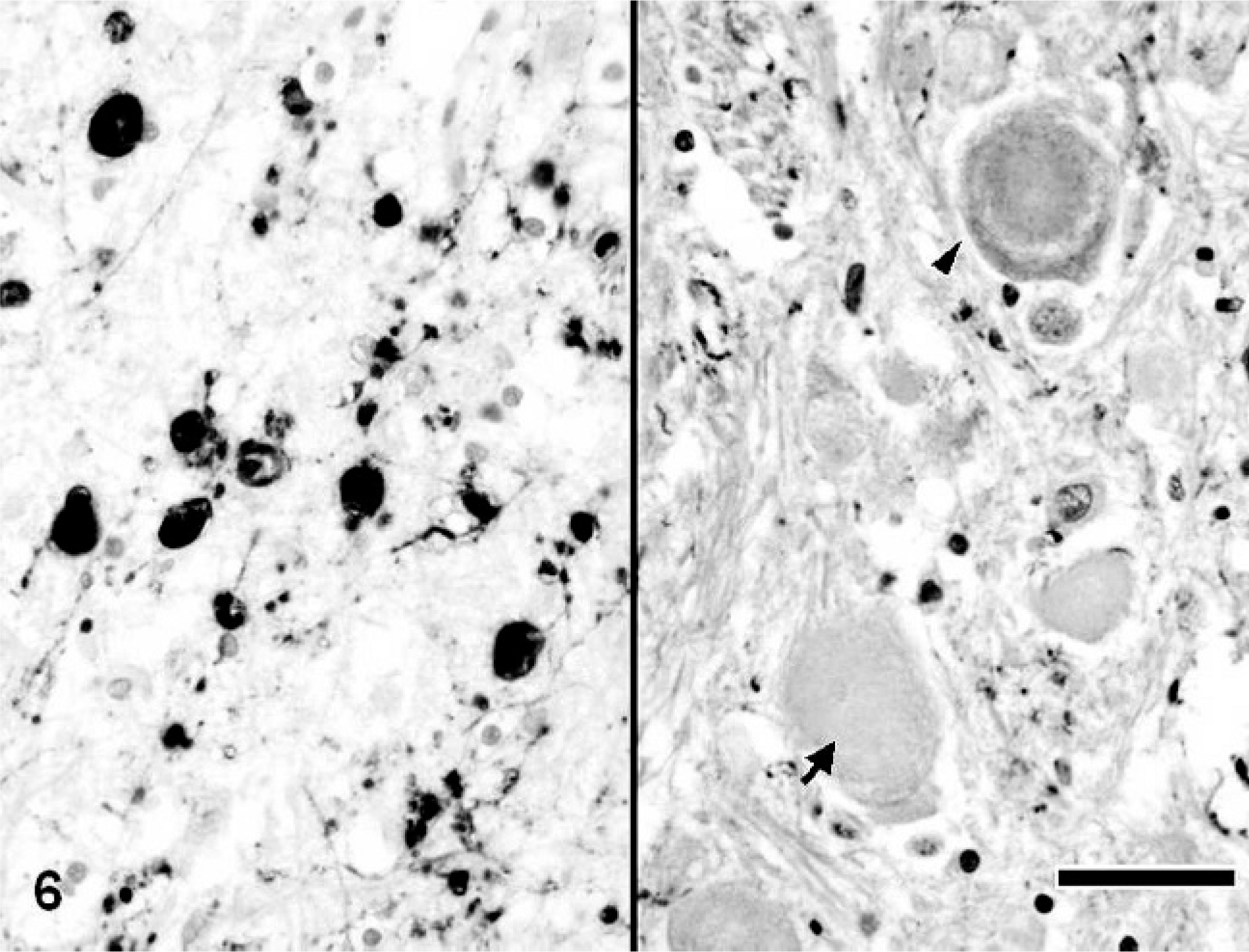
Medulla oblongata; NAD, dog No. 3. Many spheroids in the nucleus gracilis are intensely positive for calretinin (left), while those in the nucleus cuneatus exhibit weak (arrowhead) or no (arrow) immunoreaction (right). Envision polymer method. Counterstained by Mayer's hematoxylin. Bar = 50 μm.
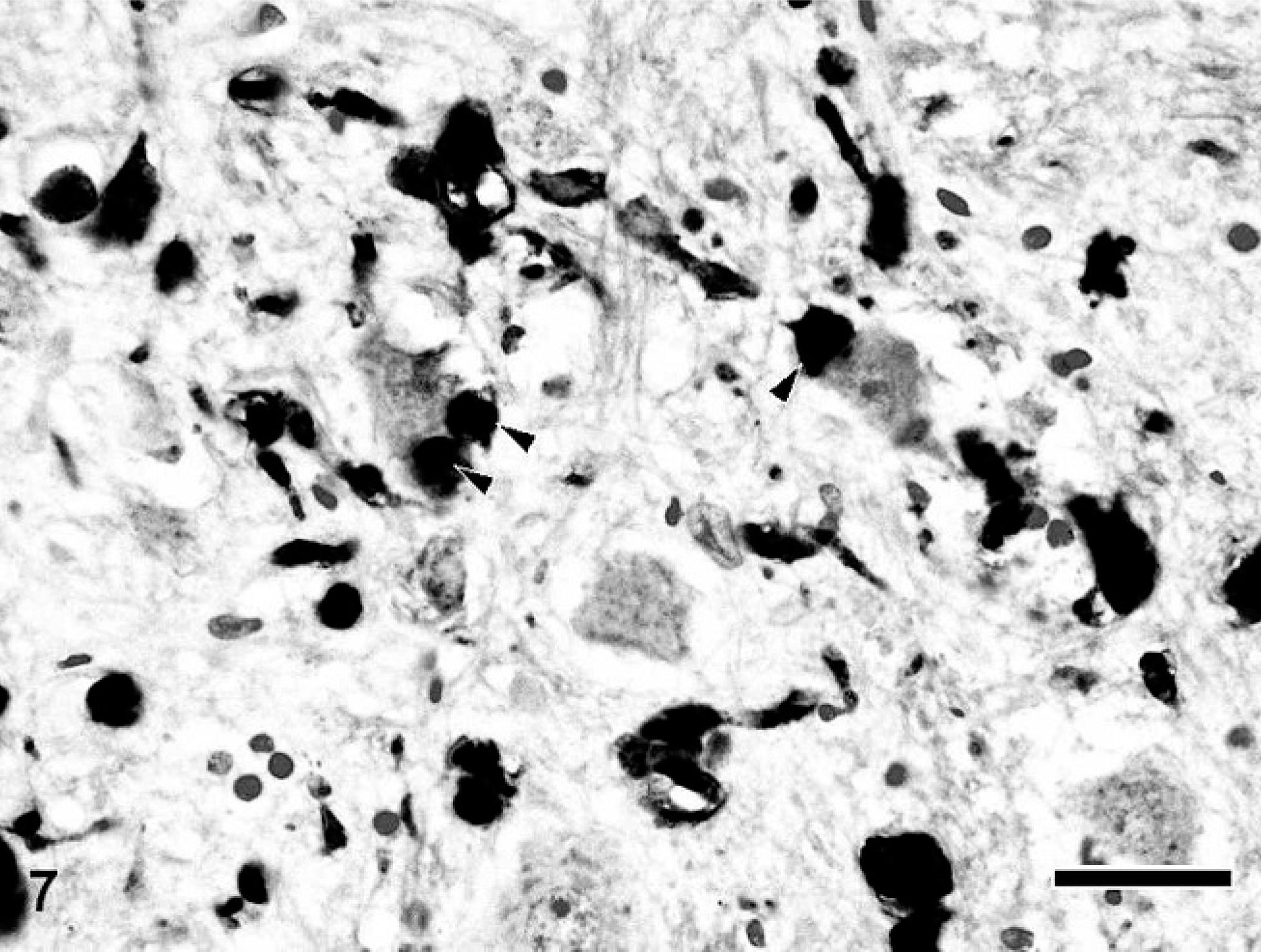
Medulla oblongata; NAD, dog No. 2. Parvalbumin-positive spheroids are sometimes adjacent to the neuronal cell body (arrowhead). Envision polymer method. Counterstained by Mayer's hematoxylin. Bar = 50 μm.
Discussion
Axonal degeneration characterized by abundant spheroids is a pathologic hallmark of canine NAD. Although abundant iron accumulation also is a characteristic feature of human NAD, 24 it has never been described in canine NAD in either Papillon or Rottweiler dogs. 8, 11, 12, 32, 43 Even in this study, iron accumulation was not found in all dogs with NAD. Recently, at least 2 causative genes, PANK2 and PLA2G6, have been identified in human infantile NAD, 6, 23, 30 although alteration of these genes in canine NAD may not be responsible for the pathogenesis because of the difference in the pathologic features. Because the spheroids in canine NAD are commonly formed adjacent to the neuronal cell bodies, axonal degeneration has been suggested to occur in the presynaptic area. 38 Previously, Sisó et al. 43 reported that the spheroids in NAD of Rottweiler dogs were immunopositive for several synapse-associated proteins, including synaptophysin, synapsin-1, SNAP-25, and Rab3a, together with other molecules such as phosphorylated NF, ubiquitin, and α-synuclein. In this study, similar observations were obtained in Papillon dogs with NAD.
Spheroids arise in a variety of circumstances or diseases, and their contents vary in each case. 48 Ultrastructually, they are mixture of NFs, tubulovesicular structures, mitochondria, lysosomes, and membranous bodies. 48 This study revealed that NFs were one of the major components of the spheroids in canine NAD. The accumulation of NFs is considered a rather common property of axonal spheroids in several CNS disorders 47 and is not specific to canine NAD. Tau is a microtubule-binding protein in normal neurons and glial cells, which associates with transportation of cytoplasmic proteins and cell organelles. Thus, the local accumulation of tau might indicate the abnormality of axonal transport. Hsp70, which is one of the major heat shock proteins and which is expressed under several stress conditions, protects cells from various stresses and assists damage repairs. 4, 22 Ubiquitin exists in cells universally, participating in protein modification and being involved in various physiologic processes, such as protein degradation, DNA repair, translational control, and signal transfer. 28, 51 The accumulation of these heat shock proteins within the axonal spheroids in NAD might be a response directed against accumulated proteins within the presynaptic area. The phenomena also might indicate that the level and/or nature of these deposits resulted from exceeding the ability of degradation by the proteasome system and the lysosomal system. Synaptophysin is an integral protein of the synaptic vesicle membrane, interacting with other synaptic proteins and participating in exocytosis. 20, 31 The accumulation of synaptophysin in the dystrophic axons of Papillon dogs with NAD may indicate the functional obstruction of the synapse at the presynaptic portion. As compared with those positive for synaptophysin, only a small number of spheroids were immunopositive for syntaxin-1 and SNAP25. Syntaxin-1 is an anchor protein involved in exocytosis, like SNAP-25, which is a plasma membrane protein located at presynaptic nerve terminals. 46, 50 These differences in location and biologic function might reflect the spheroid immunoreactivity seen in canine NAD. Synuclein family has 3 types of proteins, including α-, β-, and γ-synuclein. This has a highly conserved alpha helical lipid-binding motif with similarity to the class-A2 lipid-binding domains of the exchangeable apolipoproteins. The α- and β-synuclein are cytoplasmic proteins found predominantly and abundantly in the presynaptic nerve terminals in the brain, primarily near the vesicles. Although the functions of α- and β-synuclein are not well elucidated, their participation in synaptic plasticity has been proposed. Abnormal axonal transport or metabolism of presynaptic portion is thought to be one of the causes, but the mechanism remains unclear.
In humans, accumulation of phosphorylated tau 2, 14, 36, 53 and synuclein 19 proteins is one of the important factors of several neurodegenerative diseases. The terms “tauopathies” and “synucleinopathies” are general terms of diseases that are caused by the accumulation of phosphorylated tau or synuclein proteins. Alzheimer's disease is one example of a tauopathy. 40 A mechanism of neurotoxicity is suggested that hyperphosphorylated and aggregated tau impairs axonal transport. 27 Likewise, abnormal accumulation of α-synuclein is a pathologic feature of “synucleinopathies.” 19, 41 Human multiple systems atrophy (MSA) 26, 35 is one of the spinocerebellar degenerations (SCDs), which is one form of “synucleinopathy” and is commonly characterized by intracytoplasmic accumulation of α-synuclein. 19 Although intracytoplasmic accumulation of α-synuclein was not found in the dog with CCA, the molecules accumulated in the spheroids in all dogs with NAD. Similar observations have been reported previously in Rottweiler dogs with NAD. 43 Because the accumulation of α-synuclein in dystrophic axons has not been reported in human NAD, the features might be specific for canine NAD.
With regard to calcium-binding proteins, a large number of immunopositive spheroids were found in the cerebellum, medulla oblongata, but there were few spheroids in the cerebrum of dogs with NAD. Calcium-binding proteins are widely distributed throughout the CNS, and cell-specific expression has been reported in cats, 1 monkeys, 3 and rats, 29 although in dogs, there is a report about the abnormal expression on calbindin and parvalbumin in brain tissue under several pathologic conditions. 44 Hof et al. reported the distribution of calcium-binding proteins, including parvalbumin, calbindin, and calretinin, in the neocortex and hippocampus of dogs. 17, 18 Physiologic distribution of these proteins in other parts of canine CNS was not been well elucidated. In our study, almost all regions of the cerebrum showed weak and vague immunoreactivity for all calcium-binding proteins examined. However, in the cerebellum and brain stem, many immunopositive spheroids were predominantly distributed. Because the phenomena may reflect simply the normal localization of the proteins, further fundamental studies on these molecules in the CNS of dogs will be needed to elucidate the meaning or roles in canine NAD.
In humans, dysfunction of the calcium channel in CNS is quite important to many neurologic diseases. 21, 25 Human SCD is a hereditary disease characterized by atrophy of the spinal cord and cerebellum and is caused by expansion of a CAG repeat. 34 Among the subtypes of human SCDs, SCA6 is caused by an elongated CAG repeat of the CACNA1A gene encoding the P/Q-type voltage-dependent calcium channel. 13 Several murine models with mutations at the α1 subunit of the gene such as groggy rat 49, 52 are thought to have some clinical and pathologic similarity with canine NAD. To discuss the relationship between the calcium channel and canine NAD, further molecular investigations, including gene analysis, are necessary.
