Abstract
Cytauxzoonosis, caused by the protozoan parasite, Cytauxzoon felis, is a tick-borne disease of domestic cats causing high mortality. The reservoir is wild felids. In this study, 7 archived cases of the disease were examined through in situ hybridization for localization of the parasite and by immunohistochemistry for various cell markers to characterize infected cells. The riboprobe used was specific for the 16S-like rRNA subunit of Babesia microti, which shares 91% identity with the same gene for C. felis. In situ hybridization highlighted the presence of the organism in several tissues, most prominently lung and spleen, and, in general, there were 2 to 10 times more infected cells seen with in situ hybridization than with HE. Parasite-laden cells were usually found within vessels. These cells were often tightly packed and frequently formed parasitic thrombi. Immunohistochemistry with an antilysozyme antibody confirmed the macrophage origin of the infected cells. Using an antibody specific for calprotectin (Mac387), parasitized cells were markedly devoid of this protein, which may explain the lack of diapedesis and vascular crowding of parasitized cells, providing more circulating parasites for the tick vector. Immunohistochemical labeling for 2 proliferation markers, proliferating cell nuclear antigen (PCNA) and p53, indicated that parasitized cells have a heightened replicative ability, which is probably an additional parasite-driven modification to facilitate survival and transmission.
Introduction
Cytauxzoon felis is a tick-borne protozoan parasite classified in the order Piroplasmida, family Theileridae. First identified and described in North America in 1979, it causes high mortality in domestic cats.15, 23 The parasite is transmitted through the saliva of ticks, predominantly Dermacentor variabilis, and the reservoir are wild felids, especially bobcats (Lynx rufus).3, 11 In domestic cats the disease is acute, with clinical signs developing between 5 and 14 days after experimental inoculation of infected blood or tissue lysate given intravenously.14 Cats become febrile, depressed, dehydrated, anorexic, and reluctant to move around. Jaundice is common. Mortality is close to 100% and dyspnea usually occurs just prior to death.19
The most common gross findings are splenomegaly, generalized lymphadenopathy, pulmonary edema and congestion, an enlarged and mottled liver, and often petechial hemorrhages over serosal surfaces. Histologically the most striking finding is the presence of many large polygonal cells, presumed to be from the monocyte-macrophage lineage, attaching to the inner lining of medium-sized vessels and capillaries. These large cells have cytoplasm distended with basophilic, variably sized (0.75–2 μm), granular parasites (schizonts). The inflammatory response is minimal, and even though vessels may be filled almost to the point of occlusion with organism-containing cells, the vessels themselves show minimal reaction, and the perivascular infiltrate is absent to minimal.6, 12, 23
Cytauxzoon felis shares many common features with the more well known Theileria spp., including ultrastructural shape, replicating cycle, and the presence of both tissue and erythrocytic stages. The cardinal difference between Cytauxzoon and Theileria is that the tissue phase of the latter is in lymphocytes whereas for the former it is believed to be in the monocyte-macrophage.16
Despite its historic relatedness to Theileria, recent phylogenetic analysis reveals many similarities between Cytauxzoon and Babesia spp., especially the small babesias such as B. microti. The 16S-like rRNA subunits of B. microti and Cytauxzoon share 91% identity. Consequently, nucleic acid probes developed for the 16S-like rRNA gene of B. microti should hybridize with the same gene in C. felis. 8, 14, 24
There were two aims of this study. The first was to delineate the presence of Cytauxzoon within infected cells in fixed specimens from clinical cases using in situ hybridization (ISH), perhaps to provide new possibilities for improved diagnostic strategies and to offer a more complete understanding of the spread of the parasite throughout the body. The second aim was to characterize the infected cells further using immunohistochemistry (IHC) for various cell function markers, to gain more insight into the pathogenesis of the interaction between host and parasite. In order to assess immunohistochemically the lineage of the infected cells, Mac387 and antilysozyme antibodies were used. Mac387 is an antibody to calprotectin, a superficial adhesion molecule widely expressed in the cells of monocyte-macrophage lineage.21 Lysozyme is a conserved proteolytic enzyme expressed in many leukocytes of myelomonocytic lineage in mammals.22 Antibodies to proliferating cell nuclear antigen (PCNA) and p53 were used to evaluate the replicative cycle of the infected cells. PCNA is a subunit of DNA polymerase δ and it is mainly expressed during the S phase of the cell cycle.13 The p53 molecule is an oncosuppressor protein, capable of arresting the cell cycle progression and induction of apoptosis in stressed cells. Its immunohistochemical expression is augmented in several tumors, and this has been associated with a lower grade of its biologic activity.1, 2
Materials and Methods
Tissues and histopathology
Formalin-fixed paraffin-embedded tissues (FFPET) from 7 necropsied cats (Nos. 1–7) with a final diagnosis of cytauxzoonosis, were retrieved from the archive of the Department of Pathology and the Athens Diagnostic Laboratory, College of Veterinary Medicine, University of Georgia, Athens, GA. All the cats were submitted to this institution for routine necropsies, and the disease had been diagnosed by gross and microscopic appearances (HE). All tissues were examined histologically for this study. The following tissues were available from the 7 cats: lung, heart (all 7 cats); kidney (6 cats); spleen (4 cats); brain (5 cats); liver (5 cats); and lymph node (3 cats). A quantitative assessment of infected cells was done by counting the mean of parasitized cells per 10 high power field (HPF) at 100×. FFPET from 3 noninfected cats in a laboratory study on diabetes served as negative controls for both immunohistochemistry and in situ hybridization.
In situ hybridization
Preparation of probe
A riboprobe previously used for targeting the presence of B. microti in tissue from experimentally infected hamsters (data not published) was used. Primers for B. microti 16S-like rRNA were as follows: BM16SRW:5′-AACGCTGCGGAAGCGAGATTAATGACAAGGCAG-3′ BM16SFW:5′-CATGTCTTAGTATAAGCTTTTATACAGAA-3′.7
The resulting 600 base pair (bp) polymerase chain reaction (PCR) product was cloned into pGEM-TEasy (Promega Corp., Madison, WI) and sequenced at the Molecular Genetics Instrumentation Facility (University of Georgia) in both forward and reverse directions to obtain the complete sequence of the 5′ and 3′ gene fragments and to verify a match with the Genebank sequence for B. microti 16S-like small-subunit rRNA (BMU09833). The resulting plasmid was linearized with Hind III restriction enzymes (Promega) and transcription was carried out using SP6 RNA polymerase (Promega) and digoxigenin-labeled nucleotides (Boehringer Mannheim, Indianapolis, IN) to create a negative-sense riboprobe of 600 nt in length. Plasmid DNA was removed from the mixture with RQ1 DNase (Promega), and unincorporated nucleotides were removed by ethanol precipitation of the transcription product. The probe was resuspended in water and digoxigenin incorporation verified by dot blot.
ISH procedure
Briefly, 3-μm sections were deparaffinized, rehydrated, digested with Proteinase K (30 μg/ml) for 15 minutes, and hybridized overnight at 42°C with approximately 2 ng of probe in 100 μl 5× standard sodium citrate (SSC), 50% formamide, 5% blocking reagent (Boehringer Mannheim), 1% N-lauroylsarcosine, and 0.02% sodium dodecyl sulfate (SDS). Following stringent washes (decreasing concentrations of SSC and increasing concentrations of SDS), bound probes were visualized by the addition of sheep antidigoxigenin antibodies (1:300) at 37°C for 2 hours, followed by an amplifying step with a universal biotinylated linker and alkaline phosphatase conjugated streptavidin (Dako Corp., Carpinteria, CA). Fast Red was used as chromogen/substrate, and levamisole was added to avoid nonspecific alkaline phosphatase activity. Substrate development continued for 30–40 min. Sections were counterstained lightly with Mayer's hematoxylin and coverslipped with aqueous mounting media for a permanent record. The assessment of infected cells was perfomed by counting the average of positive cells per 10 HPF (100×). The same procedure was used with the tissues from the noninfected negative control cats. As technique negative controls, tissues from the infected cats were processed with a sense probe.
Immunohistochemistry
Antibodies
The following antibodies were utilized in this study: Mac387, a monoclonal antibody raised in mouse against calprotectin (Dako); antilysozyme (Muramidase Ab-1), rabbit polyclonal antibody (Thermo Fisher Scientific, Fremont, CA); anti-PCNA (clone PC10), monoclonal antibody raised in mouse (Abcam Inc., Cambridge, MA); and anti-p53 (clone D07), monoclonal antibody raised in mouse (BD Laboratories, San Diego, CA).
Procedure
Three-micron sections from FFPET were cut (as serial sections with those used for ISH) onto positively charged slides (Probe On Plus, Fisher Scientific, Springfield, NJ) and deparaffinized. Antigen retrieval was accomplished by microwaving in citrate buffer solution (Antigen Unmasking Solution, Vector Laboratories, Burlingame, CA). Blocking was done with a commercially available blocking solution (Power Block, Biogenex, San Ramon, CA) for all antibodies. For MAC387, PCNA, and p53, endogenous peroxidase activity was quenched with 3% hydrogen peroxide for 10 minutes. The slides were then incubated with the primary antibodies for 2 hours at 37°C or at 4°C overnight. Concentrations were as follows: Mac387, 1:50; anti-PCNA, 1:100; and anti-p53, 1:50. Secondary biotinylated antibodies were anti-mouse (from Vector Laboratories) and were applied to the sections for 1 hour at 37°C, followed by the use of an amplification system with avidin-biotin-conjugated peroxidase (Elite peroxidase, Vector Laboratories). Finally, the antigen-antibody complex was visualized by the reaction with 3′3-diaminobenzidine (DAB). For lysozyme, antibody was applied at concentration 1:200 for 1 hour at room temperature, followed by an alkaline phosphatase-based polymer linker (LabVision Corporation, Fremont, CA) applied for 30 minutes at room temperature. The reaction was visualized by incubation with Vector Red chromogen (Vector Laboratories). Slides were counterstained with Mayer's hematoxylin and coverslipped for a permanent record. For all 4 antibodies, technique negative controls were performed with omission of primary antibodies and substitution with 20% serum from the species in which the antibody was made.
Results
Histopathology
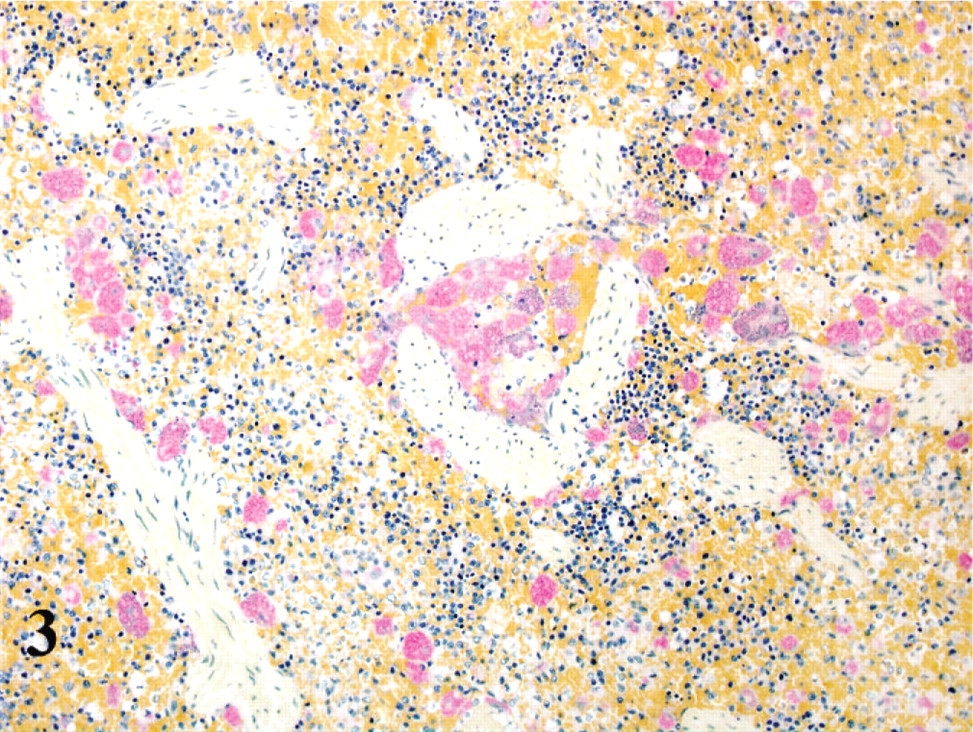
Results of the quantitative assessment of numbers of affected cells are presented in Table 1.
Numerical presence of Cytauxzoon in formalin-fixed paraffin-embedded tissues as determined by HE and in situ hybridization. ∗
ISH = in situ hybridization; NA = not applicable.
Mean of positive cells per 10 HPF. The counting was performed in the parenchymatous part of the organs, avoiding the large vessels, which contain extensive aggregates of parasites, and in which the comparison between ISH and HE would not be effective.
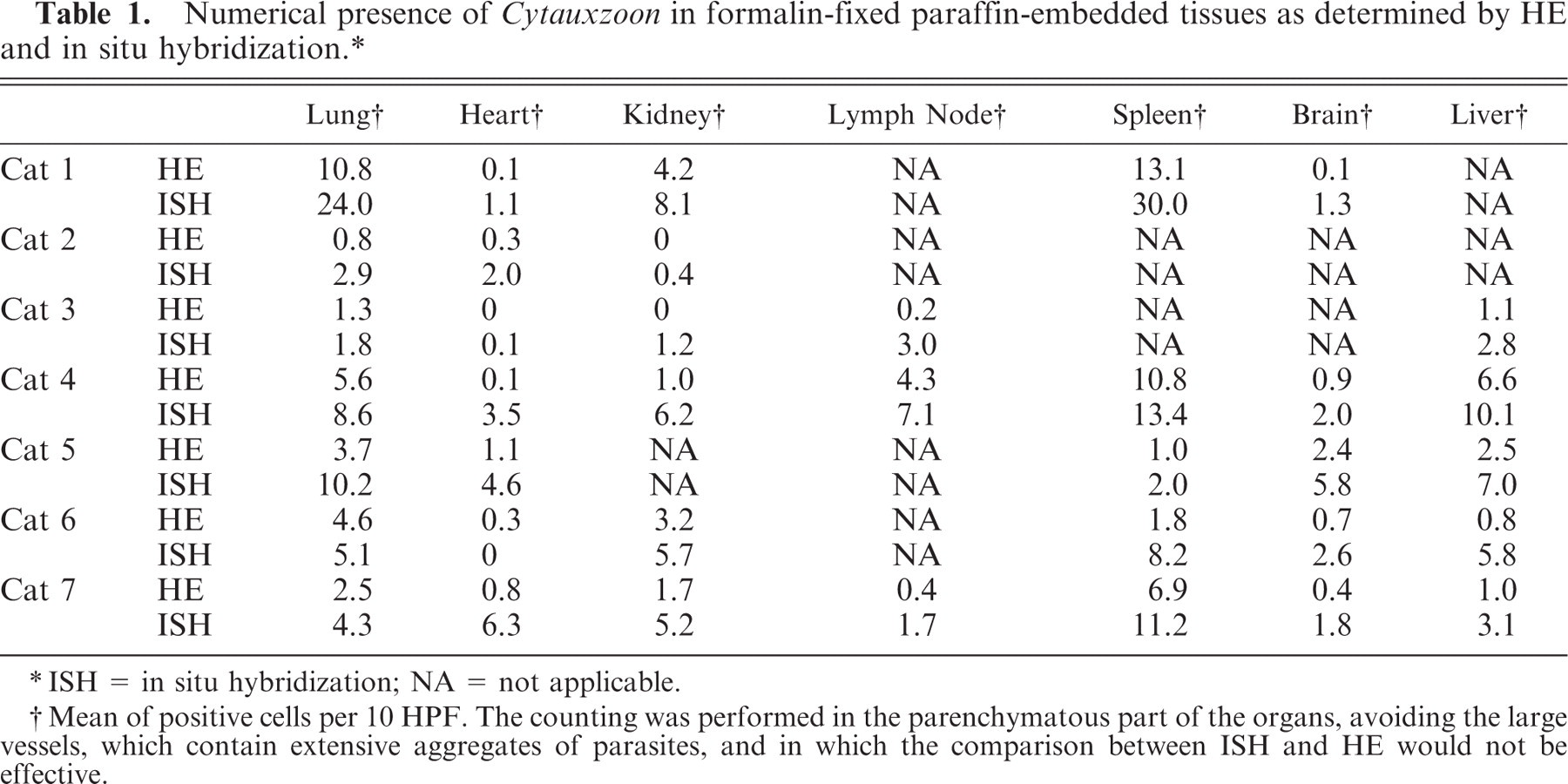
Overall the most striking finding was the presence of emboli of parasitized cells in the medium-sized vessels of most organs in all the infected animals (Fig. 1). The cells with visible parasites had extensive amounts of cytoplasm and were distended by a widespread basophilic granularity. The nuclei were large, indented, and vesicular, with a single prominent nucleolus, consistent with monocytes or macrophages. The infected cells were most easily detected in those organs that had a loose cellular architecture (lung, brain, lymph nodes), while in other more densely cellular organs, it was often challenging to discern parasitized cells.
Lung, artery; cat No. 6. Cytauxzoon felis within the cytoplasm of infected cells. HE.
The most heavily parasitized organ was spleen, where the medium-sized vessels and the red pulp were frequently filled with large polygonal cells whose cytoplasm was distended with granular organisms. The lung was also heavily parasitized, with similar cells crowding the medium-sized vessels and the septal capillaries. In scattered septal walls, small necrotic foci were present, with neutrophilic extravasation and infiltration (cats Nos. 1 and 2). Cat No. 6 showed multifocal, moderate, medial arterial hypertrophy. In all cats, the lung showed moderate to severe diffuse edema, and a septal hypercellularity. The kidney of cat No. 4 showed membranoproliferative glomerulonephritis with a mild mononuclear periglomerular infiltrate (lymphocytes, macrophages, and rare plasma cells) and numerous tubular protein casts, and in the cortex of the kidney of cat No. 7, multifocal interstitial lymphoplasmacytic infiltrates were observed. In the heart of cat No. 3, the perivascular spaces were distended, and parasites were present in some of the extravasated mononuclear phagocytic cells. Also, in this same cat, there was multifocal and locally extensive myocardial degeneration characterized by increased eosinophilic cytoplasmic staining, cellular swelling, and loss of intercellular bridges.
In situ hybridization
Results are summarized in Table 1.
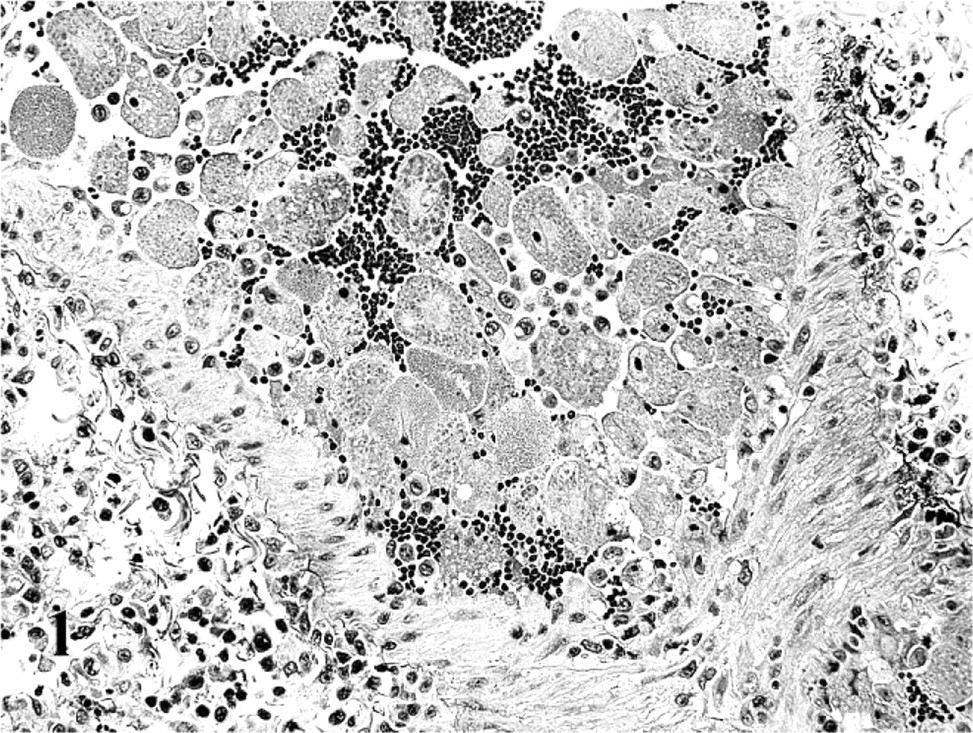
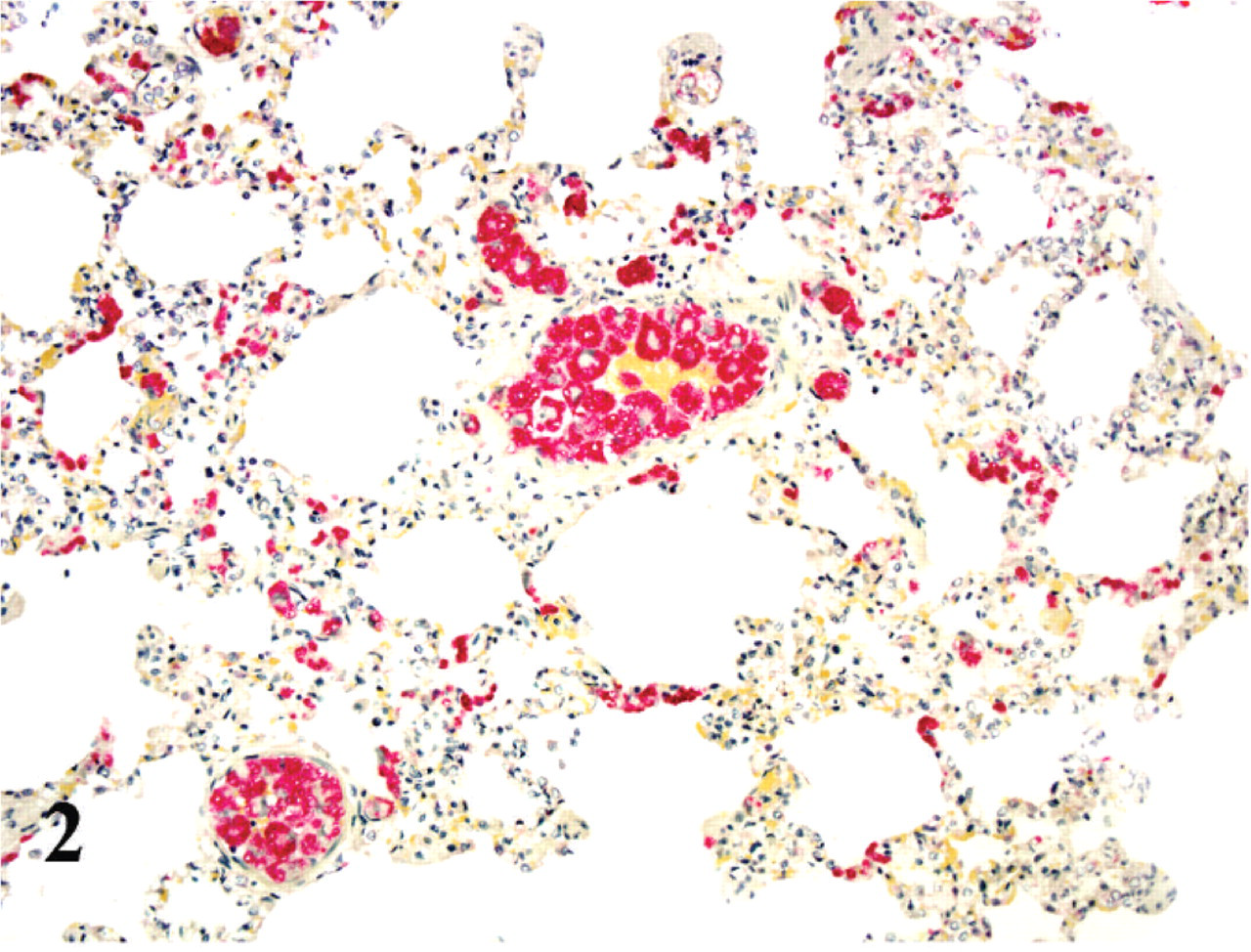
As with HE, the highest numbers of organisms were seen in spleen and lung; however, there were often 2–10 times more organisms visualized with ISH (Figs. 2 and 3). As with HE, all positively labeled cells were morphologically consistent with mononuclear phagocytic cells (monocytes and macrophages). In the lungs, ISH-positive cells filled many vascular spaces, and on rare occasions, pulmonary intravascular macrophages and alveolar macrophages appeared positive as well. In the kidneys, numerous glomeruli had several positively labeled cells entrapped in the capillary loops, and there were scattered positive cells in the interstitium of cortex and medulla. In the heart, rare capillaries harbored positively labeled monocytes, which seemed to elongate to fit better in the narrow luminal spaces. In the brain there were ISH-positive cells in almost all the sections examined, and overall the structures with the greatest number of positive cells were the choroid plexus and the choroid villi. In these structures many positively labeled monocytes were attached to the endothelial lining. Scattered rare positive cells were attached to the ependyma, and also free in the ventricular cavities.
Lung; cat No. 3. ISH to identify Cytauxzoon. Numerous positively labeled cells are present within vessels and scattered in alveolar septa. Alkaline phosphatase method, hematoxylin counterstain.
Spleen; cat No. 1. ISH to identify Cytauxzoon. Positive cells are numerous, but labeling is fainter than in other organs. Alkaline phosphatase method, hematoxylin counterstain.
The pattern of the labeling varied, between different cells in the same affected organ, in the same HPF, and even within the same vessel. Though some cells presented a very intensely labeled fine granular dust, often clustered into larger aggregates, many cells showed fainter labeling, with the parasite shape becoming defined with the counterstaining. Particularly in the spleen, although there was an abundance of positively labeled cells, the overall strength of the signal from each cell was weaker compared with other affected organs.
Immunohistochemistry
All negative control IHC (protocols done with omission of primary antibody) were devoid of any positive labeling.
Anti-lysozyme
In the noninfected control cats and in the infected cats, the antibody reacted with the mononuclear phagocytic cells in a variety of tissues, in the expected distribution. In addition, in the infected cats, all parasitized cells were labelled, and this labelling was smooth, evenly distributed, and diffuse throughout the cytoplasm (Fig. 4).
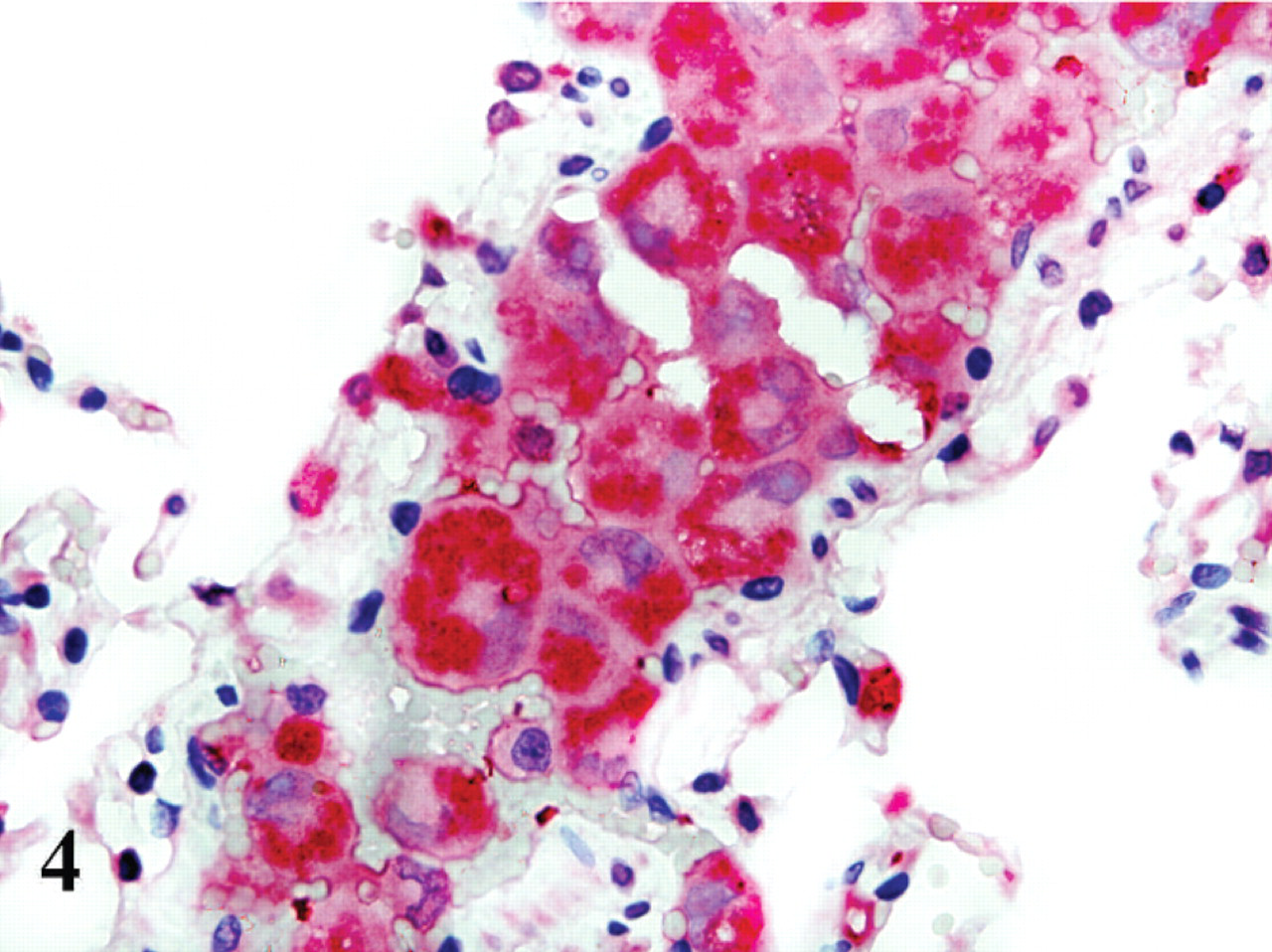
Lung; cat No. 4. Antilysozyme antibody highlights numerous parasitized cells within a vessel. Polymer alkaline phosphatase method, hematoxylin counterstain.
Mac387
In tissue sections of noninfected cat, Mac387 highlighted the presence of cells histologically consistent with monocytes and resident macrophages in all organs. The reaction was cytoplasmic, slightly granular, with a more prominent peripheral distribution, subjacent to the cell membrane. In the sections from Cytauxzoon-infected cats, none of the parasitized cells showed positive labeling, while noninfected macrophages were positively labeled as in the noninfected cat tissues (Fig. 5). The discrimination between infected and noninfected cells was performed by the use of serial contiguous sections of the paraffin block with ISH, then compared with the slides immunolabeled for Mac387. Also, parasitization of many of the large cells was visible, even in the IHC sections, because they were counterstained with hematoxylin.
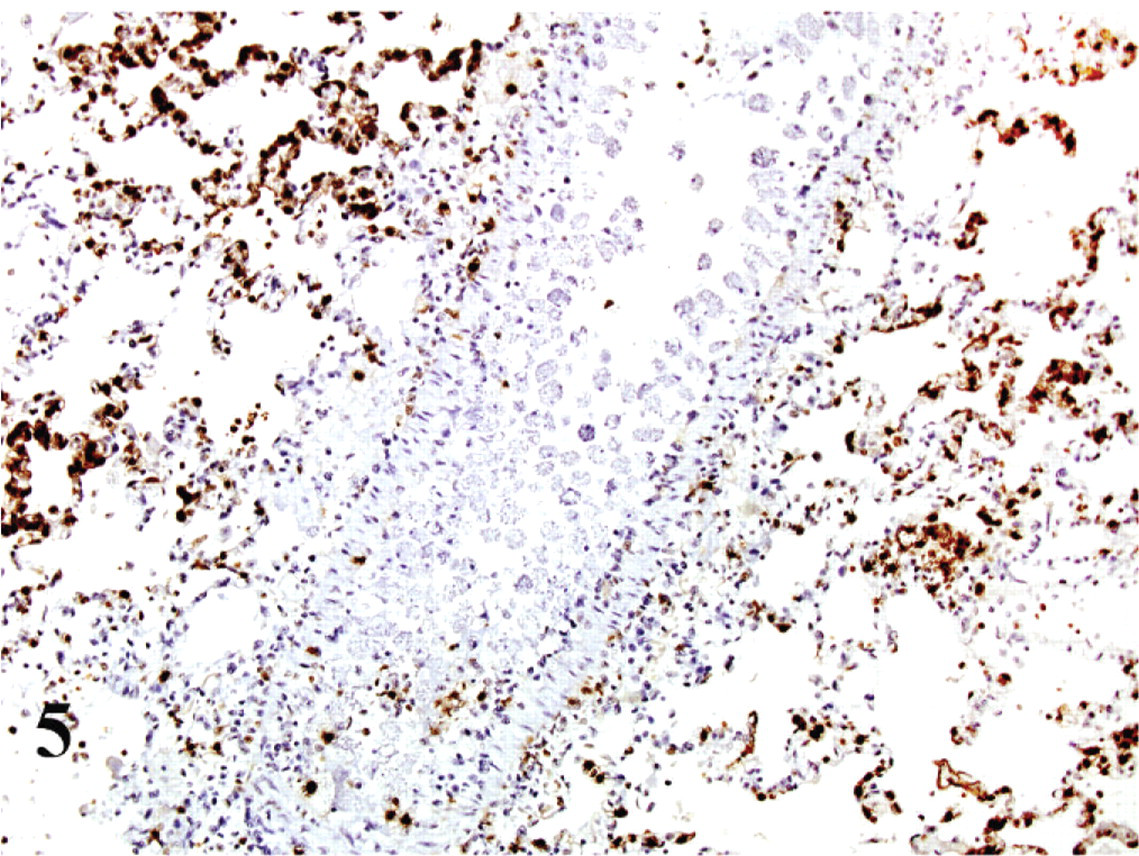
Lung; cat No. 2. Mac387 antibody labels macrophages within vessels and alveoli in lung, but parasitized cells within the large blood vessel are nonreactive. ABC diaminobenzidine method, hematoxylin counterstain.
Anti-PCNA
In both the infected and noninfected cats, with the exception of parasitized cells, positive labeling was very similar and was found in the nuclei of epithelial cells lining the bronchi and bronchioles, and in the transitional epithelium of the urinary bladder. In the Cytauxzoon-infected cats, IHC with anti-PCNA antibody highlighted the nuclei of many parasitized cells (approximately 80%), especially in the lungs, where many infected cells had an intense reaction (Fig. 6).
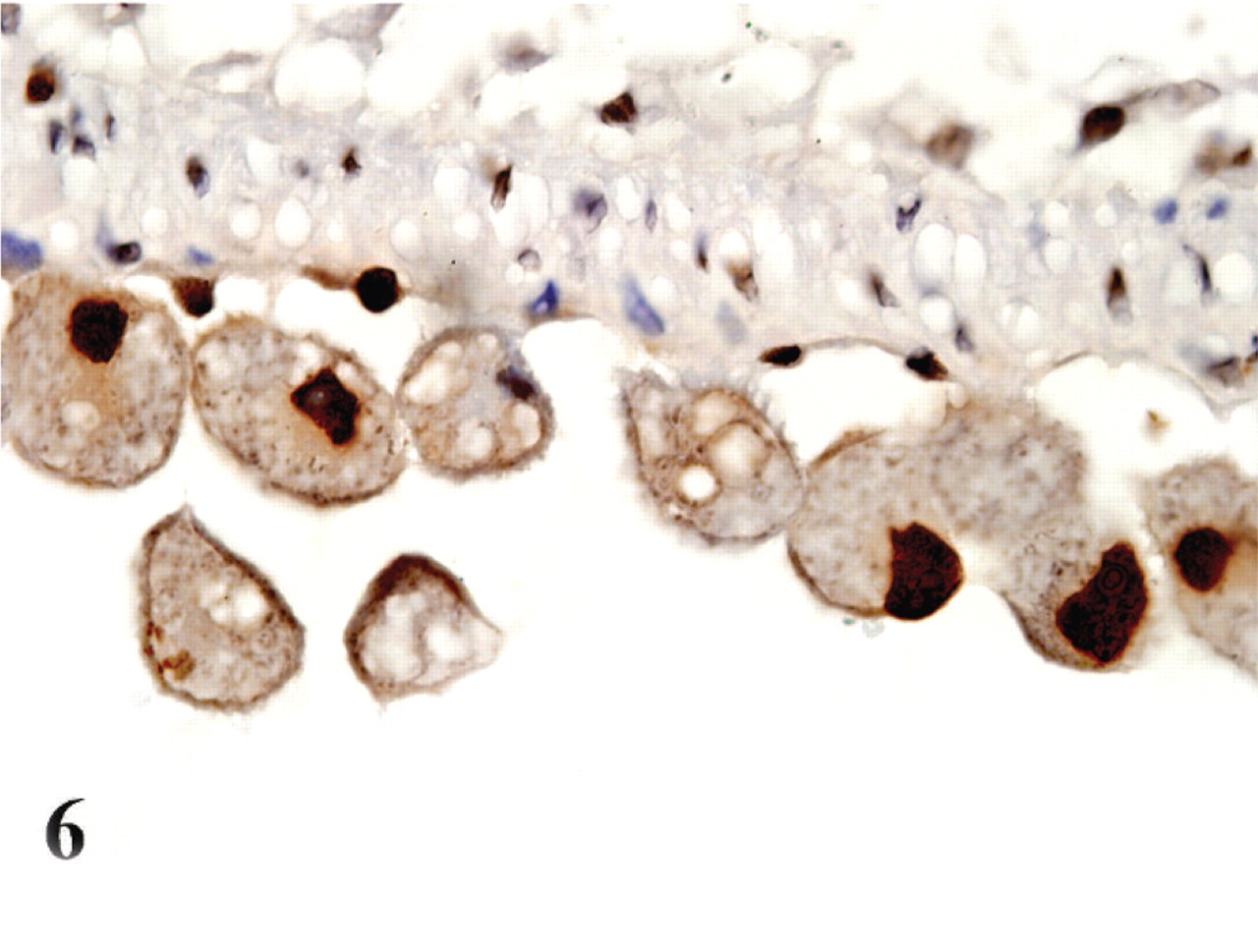
Brain; cat No. 4. Infected monocytes in the blood vessels of the meninges have positive labeling of their nuclei for PCNA. ABC diaminobenzidine method, hematoxylin counterstain.
p53
Immunohistochemistry for p53 showed no immunolabeling in the noninfected cats, while there was positive labeling in most if not all parasitized cells (Fig. 7). There was a diffuse cytoplasmic granular reaction, stronger around the parasitic bodies. There was no evidence of labeling in the nucleus or nucleolus of the cells.
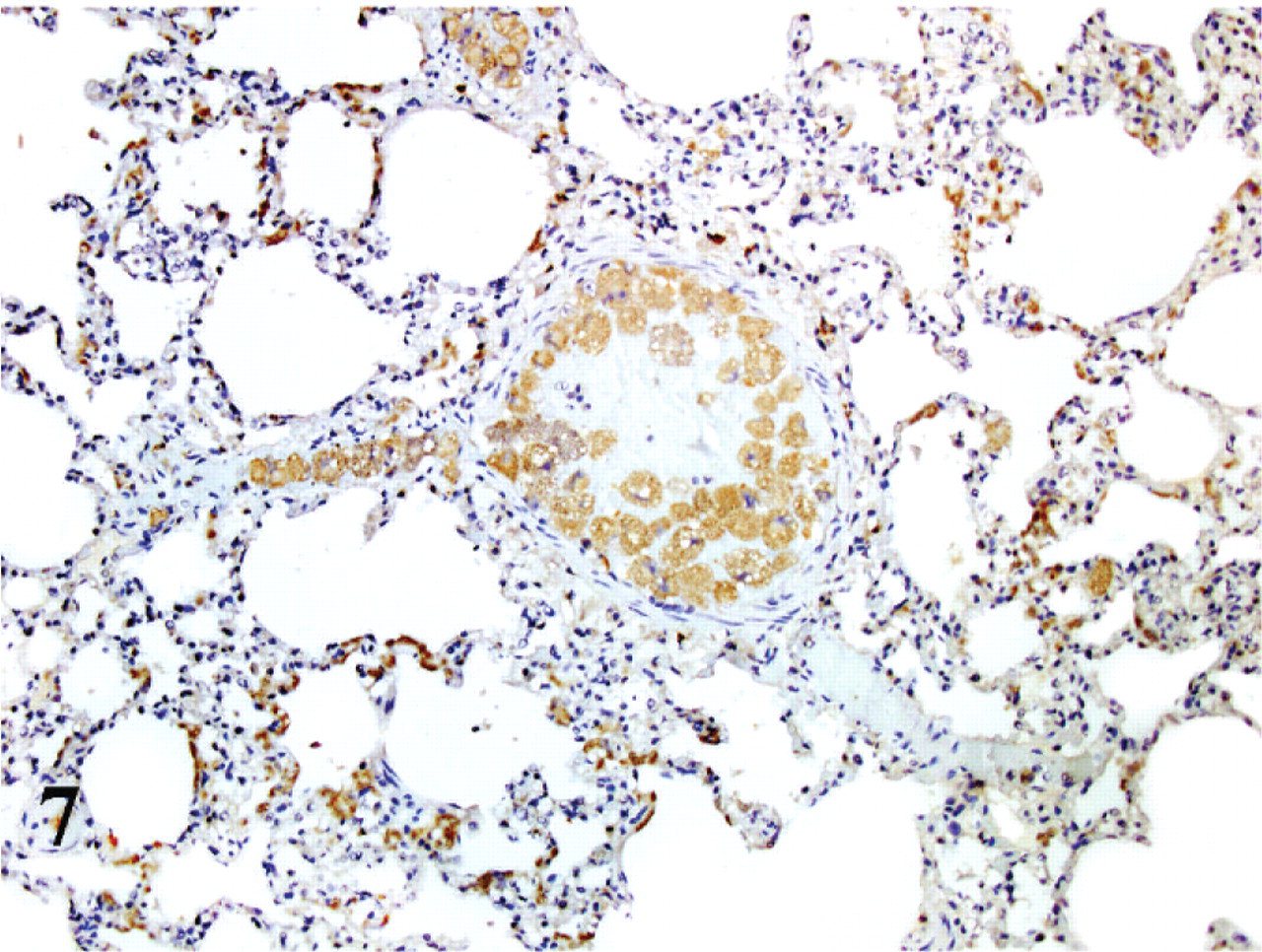
Lung; cat No. 1. Infected cells within blood vessels have positive cytoplasmatic labeling for p53. ABC diaminobenzidine method, hematoxylin counterstain.
Discussion
In situ hybridization with a probe designed to target B. microti proved to be a reliable system for the detection of the Cytauxzoon parasite in FFPET. Although the 2 parasites (B. microti and Cytauxzoon) belong to different families (Babesiidae and Theileridae), the 91% identity in the 16S-like rRNA region allowed the B. microti probe to hybridize effectively with Cytauxzoon.
The ISH procedure proved superior to HE in detecting the presence of the parasite. Although the original diagnosis was rendered via HE alone, with ISH it was apparent that the parasite was present in many more locations than what was visible using only routine histopathology. There are other diagnostic tests available for Cytauxzoon (e.g., PCR from fresh tissues, which are more sensitive). However, ISH has the advantage of use on archived tissues and also demonstrates the presence of the parasite in the context of the morphologic tissue changes visible in the histologic section.
There was variability in ISH reactivity in different organs, but also in cells within the same vessels. The spleen, even if extremely parasitized, appeared to show an overall weaker ISH signal than that in other organs, even though basophilic bodies representing schizonts or meronts were equally discernible in HE-stained sections of the various organs. This difference could be explained by the different replicating status of the parasite in the cells. When the parasite is undergoing very active replication and protein synthesis (early schizont), more ribosomal RNA is produced, thus offering more target for the ISH probe. When the parasite replication slows (late schizont-meront), the ribosomal RNA tends to be less abundant in the cells, thus offering a weaker or even negative signal. The infected cells were more strongly positive in the lung than in the spleen, which may indicate that the spleen could be the site where older stages of the parasite are sequestered.
In an effort to delineate the cell types visibly infected by Cytauxzoon, we used 2 macrophage-specific markers antilysozyme and Mac387. Lysozyme is an enzyme present in macrophages.17, 22 Using antilysozyme IHC, the parasitized cells were clearly positive, demonstrating that the parasitized cells belong to the monocyte-macrophage lineage. This finding is consistent with previous studies using electron microscopy to determine infected cell type.8
The Mac387 antibody is considered to be specific for inflammatory cells (mostly monocytes and macrophages). It has been widely used to identify macrophages in tissue sections. The antibody reacts against the superficial protein calprotectin, which is extremely important in the process of diapedesis. Calprotectin allows the inflammatory cells to attach to the endothelial lining, then to attach to the extracellular matrix and reach the inflammatory focus.21 In this study, although mononuclear phagocytic cells in both infected and noninfected cats were identified by the Mac387 antibody, the parasitized cells in the infected cats were not labeled. If this lack of reaction corresponds to reduced expression of calprotectin protein in the infected cells, this might mean that these cells are less able or unable to undergo diapedesis. This downregulation of such a critical protein in the inflammatory cells could, in part, explain the crowding of the infected cells in the vessels, and the formation of the parasitic thrombi. The putative inability of these cells to undergo diapedesis may also help to explain the lack of vasculitis. The Cytauxzoon parasite is not encapsulated in a parasitophorous vacuole, but rather is free in the cytoplasm and thus may more easily be able to modify the translational and transcriptional machinery of the host cell. Teleologically, the possibility of more infected cells circulating in the bloodstream would facilitate the parasite's survival by increasing the number of parasites available to the tick vector.
PCNA is a 29KDa essential protein of the DNA replication machinery, functioning as the accessory component for DNA polymerase δ, required for processive chromosomal DNA synthesis, and DNA polymerase ε.13 PCNA is normally used to detect the proliferating compartment in a cell population. It has been mainly used in cancer studies to determine proliferation index, and thus serves as a prognostic marker. In this study, anti-PCNA reacted with the nuclei of the majority (80%) of the infected cells. This PCNA expression indicates that the cell cycle is deregulated in the parasitized cells, and that these cells are being stimulated to undergo replication. This could be a great advantage for the parasite, whose schizogonous phase would spread from a previously infected cell to a newly formed one. Alteration of cell cycle has been well documented in another protozoan infection, Theileria parva, where it has been shown that the organism triggers the NF-kB signaling pathway, leading to marked proliferation.10 Also, the PCNA antibody targets the cells that are in an active anabolic state (during the S phase not only DNA is duplicated but also all the organelles and the proteins). This may be useful for the parasite, as an active anabolic state would facilitate parasite replication. The approximately 20% of the parasitized cells that did not react with the PCNA antibody may be a reflection that the parasite does not always trigger the cell cycle.
Protein 53 is a well-known proapoptotic protein, and its activity is initiated by many noxious stimuli. The p53 needs to be translocated to the nucleus, where it acts as a transcription factor, enhancing the transcription of several proapoptotic proteins. The overexpression of this protein has been detected in several human and animal cancers, and its mutations have been associated with their molecular pathogenesis and progression of the tumor. Under normal, nonneoplastic conditions, wild type p53 is not detectable by IHC, as it is ubiquitinated and eliminated by the proteasome very quickly. Consequently, when p53 is detectable through IHC, it may be a consequence of a longer half-life caused by a mutation or alteration of its subcellular location.1, 2 There could be several explanations for the positive p53 signal in the cytoplasm of the parasitized cells in this study. Upregulation of p53 in Cytauxzoon-infected cells could be a cellular response to the parasitic infection, as has been shown in murine models of Chagas disease and trichinellosis.4, 5 In addition, p53, when detected (as in tumors), is more commonly found in the nucleus than the cytoplasm. It is known that p53 is moved at subcellular level by the cytoskeleton.18, 20 In this study, the absence of p53 labeling in the nucleus could be a reflection of the parasite's interference with the cytoskeleton, inhibiting the transport of p53 to its active location. Another protozoan, Encephalitozoon, has been described to prevent p53 nuclear translocation in cell cultures, which decreases transcription of pro-apoptotic genes, prolonging the lifespan of the cells.9
In summary, ISH highlighted the presence of Cytauxzoon felis in the examined tissues from 7 archived cases and was proven to have a higher sensitivity than HE staining for detection of infected cells. The infected cells were shown to be of the monocyte-macrophage lineage, as determined by antilysozyme immunohistochemistry. However, infected cells, which were predominantly intravascular, did not express Mac387, suggesting an evolutionary adaptation of the parasite that interferes with diapedesis of infected cells. In addition, infected cells showed heightened anabolic state and increased replicative ability, as demonstrated by increased PCNA and p53 positivity, which may also be an adaptation that favors replication of the parasite. In this way, the highest percentage of the parasite will be in blood monocytes, where it can transfer to the erythrocyte and finally be taken up by the tick again to complete its life cycle.
