Abstract
During the last 21 years, 7 adult captive Brazilian agoutis (Dasyprocta leporina) from 4 different zoologic gardens were necropsied and histologically examined at the Leibniz Institute for Zoo and Wildlife Research, Berlin, Germany. All animals had polycystic kidney disease as the major pathologic change. Except in 1 case, no clinical signs were recognized prior to death. The animals had macroscopic bilateral alterations of the kidneys ranging from granulated surfaces to severe polycystic changes. Microscopic examination revealed multifocal to generalized, moderate to severe cystic dilatations of Bowman's capsules and renal tubules, moderate mesangial and capsular proliferation of the renal corpuscles, mild interstitial fibrosis, and mild to moderate interstitial lympho-plasmacytic infiltrations. Little information is known about the genetic relationships of these animals, but breeding practice indicates a high possibility of inbred agouti zoo populations in Germany. This is the first report on polycystic kidney disease in Brazilian agoutis with possible genetic background.
Introduction
Agoutis are hare-sized rodents of tropical rainforests in Central and South America belonging to the family Dasyproctidae of the suborder Hystricomorpha. Being frugivorous, they are of great importance for rain forest biodiversity as they spread seeds of their food trees by burrowing excess food into the soil. Although agoutis are often kept in zoologic institutions, where they can reach an age of between 10 and 20 years, little is published about the diseases affecting this species. 2,14
Polycystic kidney disease (PKD) is found in several domestic and laboratory animal species as well as in humans, but little is known about PKD in wildlife and zoo animals. Only a few case reports of PKD in exotic or wildlife species, including young springboks (Antidorcas marsupialis), 16 an adult raccoon (Procyon lotor), 12 adult and infantile rhesus monkeys (Macaca mulatta), 3,17 young and adult slender lorises (Loris lydekkerianus), 27 and a stillborn white-tailed deer (Odocoileus viriginianus), 26 have been published. In general, PKD is described as either 1) a congenital form or 2) an adult form. The congenital form is known to occur in dogs, cats, horses, cattle, sheep, pigs, several laboratory animal species, and humans. In humans, this congenital form is based on an autosomal recessive trait caused by mutation on the PKHD1 gene encoding fibrocystin, which is a receptor protein. 15,31 It is proof that this genetic mutation follows an autosomal recessive trait similar to the childhood form of PKD in humans is also responsible for polycystic kidney disease in Persian kittens, sheep lambs, and West Highland White and Cairn terrier puppies, whereas the inheritance of congenital PKD in other species is unknown. 10
The adult form of PKD is, so far, only described in a few species (see below). In humans, it is caused by a genetic failure on either the PKD1 gene encoding polycystin 1, a membrane receptor, or the PKD2 gene, a Ca2+ channel. 15,31 Both mutations follow an autosomal-dominant Mendelian trait of inheritance. Autosomal-dominant PKD is the most common genetic disease worldwide and an important cause of end-stage renal failure in humans. 9,18,31 PKD in adult animals resembling the autosomal dominant inheritance is described in cats (most of them are Persian or Persian-cross cats) 4,7 and likely in Bull Terriers. 25 Similar to the human form, a mutation on the PKD1 gene causes the majority of adult PKD in cats 13,19 whereas autosomal-dominant PKD in Bull Terriers is likely not associated with a mutation in the canine PKD1 or PKD2 genes. 22,23 Similar alterations are also reported in adult ferrets 6,28 and horses 1,5,29 but the genetic background in these species is unknown.
Both forms of PKD comprise bilateral, moderate to severe, cystic dilatation of renal tubules. The cysts range from a few millimeters up to 3 cm in diameter. They are mainly restricted to the principle parts of the nephrons in the congenital form, whereas in the adult form they are found throughout the whole tubular system. Congenital PKD is associated with abortions or neonatal or infantile death, whereas the adult form develops progressively and is accompanied by mild to moderate interstitial lympho-plasmacytic infiltrations. In both incidences, the resulting renal dysfunction is fatal. In recent years, major efforts have been made to understand the pathogenesis of autosomal dominant PKD. Polycystin 1 is highly expressed during the development of the kidneys and is downregulated after birth. It is part of a multiprotein complex that activates an intracellular signal transduction cascade. Resulting regulation of fetal gene expression is important for an appropriate proliferation and differentiation of tubular epithelial cells. This process is disrupted in PKD, resulting in cyst formation. 32 Alterations of the epithelia include changed polarity of membrane proteins, abnormal secretion, altered cell-matrix interactions, increased rates of proliferation and apoptosis, and proliferative reaction to cAMP. 9,18,31,32 However, the exact process of cyst development is still unclear.
Materials and Methods
Since 1986, carcasses of 11 agoutis (8 males, 3 females) originating from 4 different German zoos (A–D; Table 1) were submitted to the Leibniz Institute for Zoo and Wildlife Research in Berlin for pathologic examination. All animals belong to the species Dasyprocta leporina (Brazilian, red-rumped or orange-rumped agouti, synonym Dasyprocta aguti). Affected animals included 7 adults and 4 neonates of 2 litters each 1–2 days old. The exact age of the adults was given in 3 cases and ranged from 6 to 8 years. All animals were found dead unexpectedly in their enclosures without showing any previous clinical signs related to renal disease. During necropsy tissue samples were collected from myocardium, aorta, lungs, esophagus, stomach, intestines, liver, pancreas, kidneys, bladder, spleen, lymph nodes, adrenal gland, thyroid, ovaries, uterus, testes, brain, nerves, and skeletal muscles and were fixed in 10% formalin between 1986 and 2000 and in 4% formalin since 2001. They were routinely processed, embedded in paraffin, cut in 2–4-µm sections and stained with HE for light microscopic examination. Histologic sections of the kidneys were also stained with periodic acid Schiff (PAS), Azan (according to Heidenhain), silver staining according to Movat, von Kossa stain for calcium detection, Prussian blue for iron detection, and a prolonged Ziehl-Neelsen method to stain lipofuscin. The urea concentration of the intraocular fluid was measured in agouti No. 6 using urea strips (DiaSys Diagnostic Systems, Holzheim, Germany).
General data of the examined adult agoutis (Nos. 1–7).
Results
Gross findings
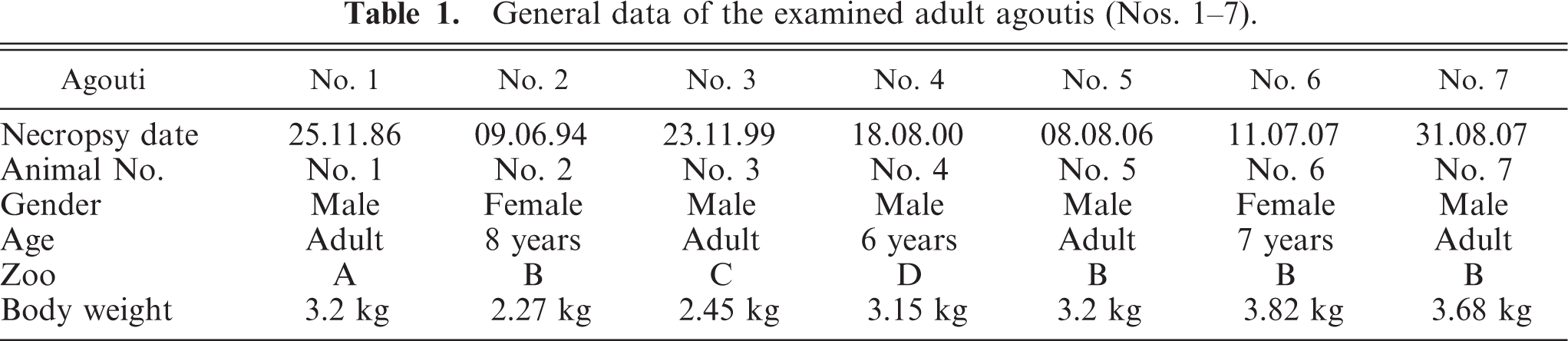
Neonates had no significant changes. All adult agoutis were in poor body condition or cachectic. In 1 agouti (No. 3), the claws of the outer toes of its hind limbs were missing with associated suppurative infection of the stumps. The kidneys of all adult agoutis revealed bilateral moderate to severe changes, ranging from a mildly granulated surface to multiple cysts throughout the whole parenchyma. The cysts ranged from barely visible up to 0.5 cm in diameter. In 1 case, there was unilateral mild atrophy of the left kidney (No. 5) (Fig. 1).
Kidneys; agouti No 5. Polycystic kidney disease. Kidneys with granulated, cystic surface and unilateral atrophy of the left kidney. Bar = 1 cm.
General microscopic findings
In 1 agouti (No. 2), suppurative bronchopneumonia was diagnosed in the cranial lobes of lung, with multifocal moderate to severe broncho-alveolar infiltration with neutrophilic granulocytes, macrophages, and a few lymphocytes and plasma cells. No pathogenic bacterial organisms were isolated. Four other agoutis had diffuse moderate to severe alveolar edema (Nos. 1, 3, 4, and 7). In addition, 3 agoutis revealed mild pulmonary interstitial lymphocytic infiltration (Nos. 1, 2, and 4). In agouti No. 7, a mineralized cartilage of a single bronchus showed focal destruction combined with proliferation of connective tissue. The myocardium from the same animal had small multifocal fibrous foci. Occasionally, mild lymphocytic infiltrations were observed within the stomach (Nos. 2, 4, and 6) or small intestine (Nos. 1 and 6). The livers of agoutis Nos. 3, 6, and 7 revealed mild fatty degeneration of hepatocytes. In the spleen, moderate follicular hyperplasia was detected in 2 animals (Nos. 1 and 2), and a moderate to severe siderocytosis was detected in 5 cases (Nos. 1, 2, 4, 5, and 7). All other organs (pancreas, bladder, lymph nodes, adrenal gland, thyroid glands, uterus, ovaries, testes, brain, nerves, skeleton muscle, bone, and skin) had no morphologic alterations except the kidneys (see below and Table 2).
Pathologic changes of the kidneys found in each individual agouti.∗

∗ex = cachexia; + = mild; ++ = moderate; +++ = severe.
Detailed microscopic examination of the kidneys and related changes
Almost every kidney had multiple atrophic renal corpuscles, and in 2 cases (Nos. 1 and 2) the total number of corpuscles seemed to be decreased compared with the kidneys of the other animals. Renal corpuscles of all kidneys revealed multifocal mild to severe dilatation of Bowman's capsules filled with a slightly eosinophilic, proteinaceous fluid (Figs. 2, 3). Occasionally Bowman's space contained mild to moderate amounts of fibrin deposits (Nos. 4 and 5) with additional sparse erythrocytic accumulations (Nos. 2, 4, and 6). Many Bowman's capsules were thickened by fibrotic tissue (Fig. 4) and comprised mild epithelial proliferations. In 3 cases, a few Bowman's capsules had distinct cuboidal parietal epithelia. There were multifocal moderate and in some cases severe mesangial proliferations and a few adhesions between the glomerular loops and the capsules in all kidneys. Two agoutis (Nos. 5 and 6) had calcified foci within their glomeruli as well as within the parietal capsular epithelium of some renal corpuscles.
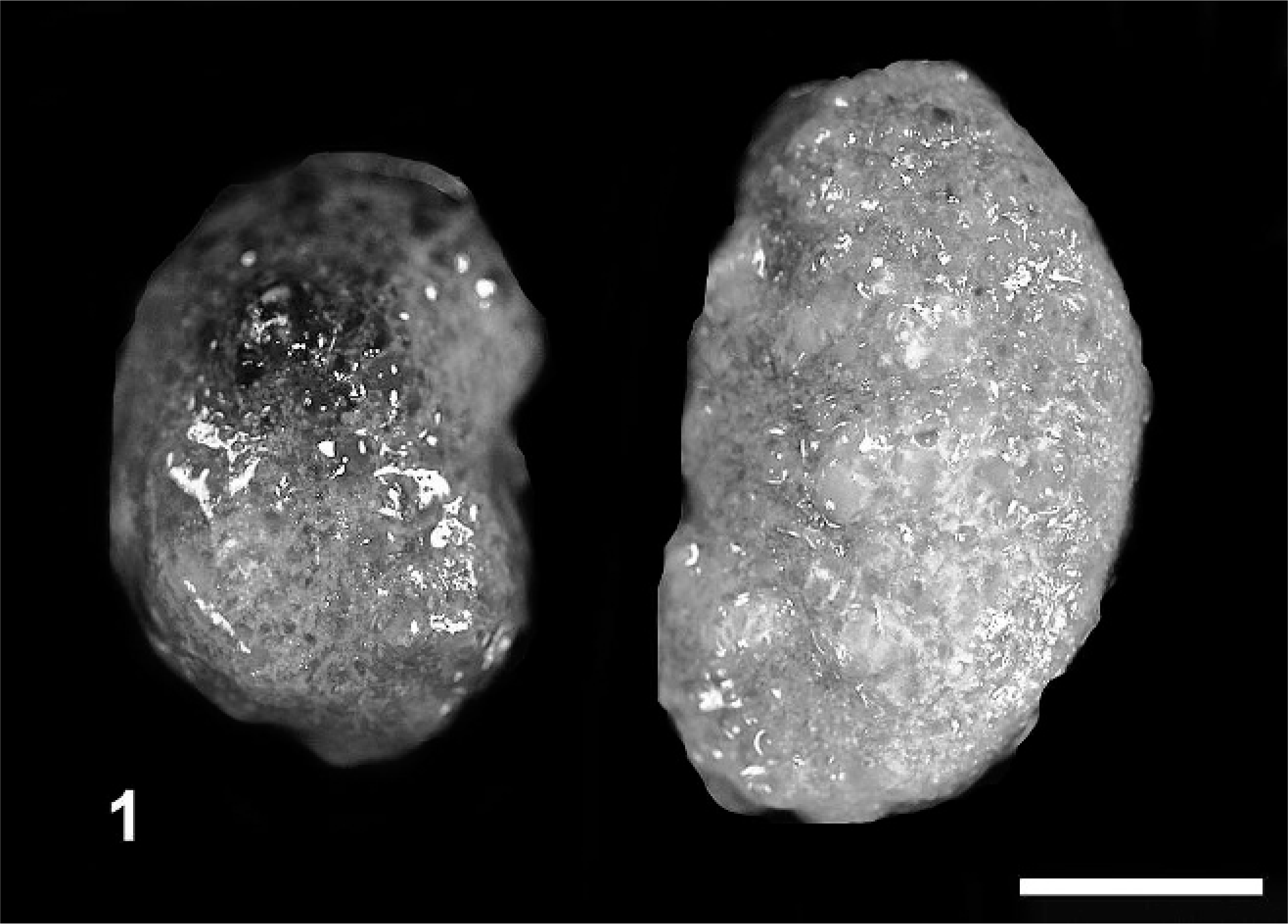
Kidney; agouti No 4. Polycystic kidney disease. Micrograph, overview. Generalized dilatation of tubules and glomerular corpuscules. PAS stain.
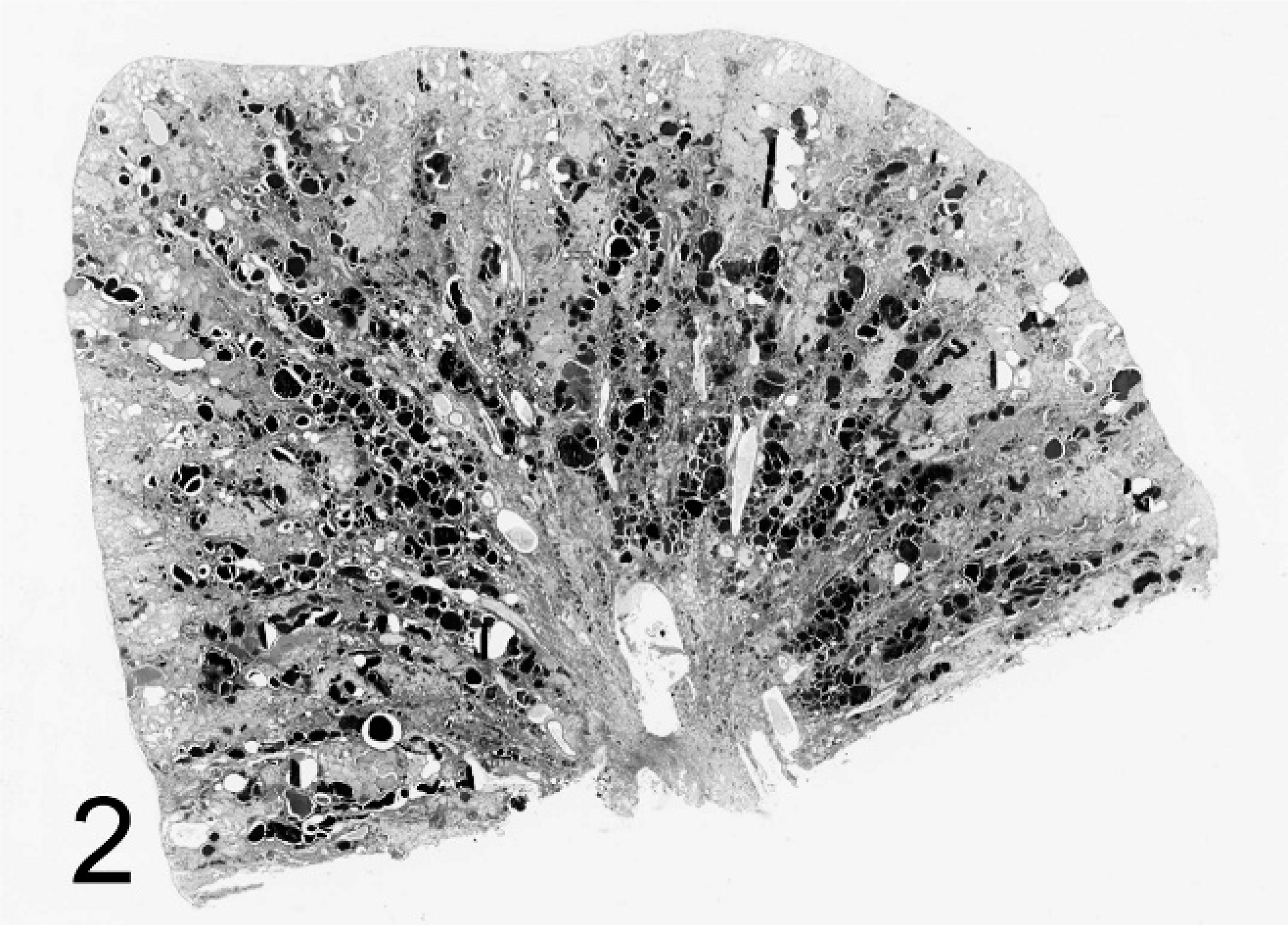
Kidney; agouti No 3. Polycystic kidney disease. Marked dilatation of tubules and Bowman's capsule. PAS stain.
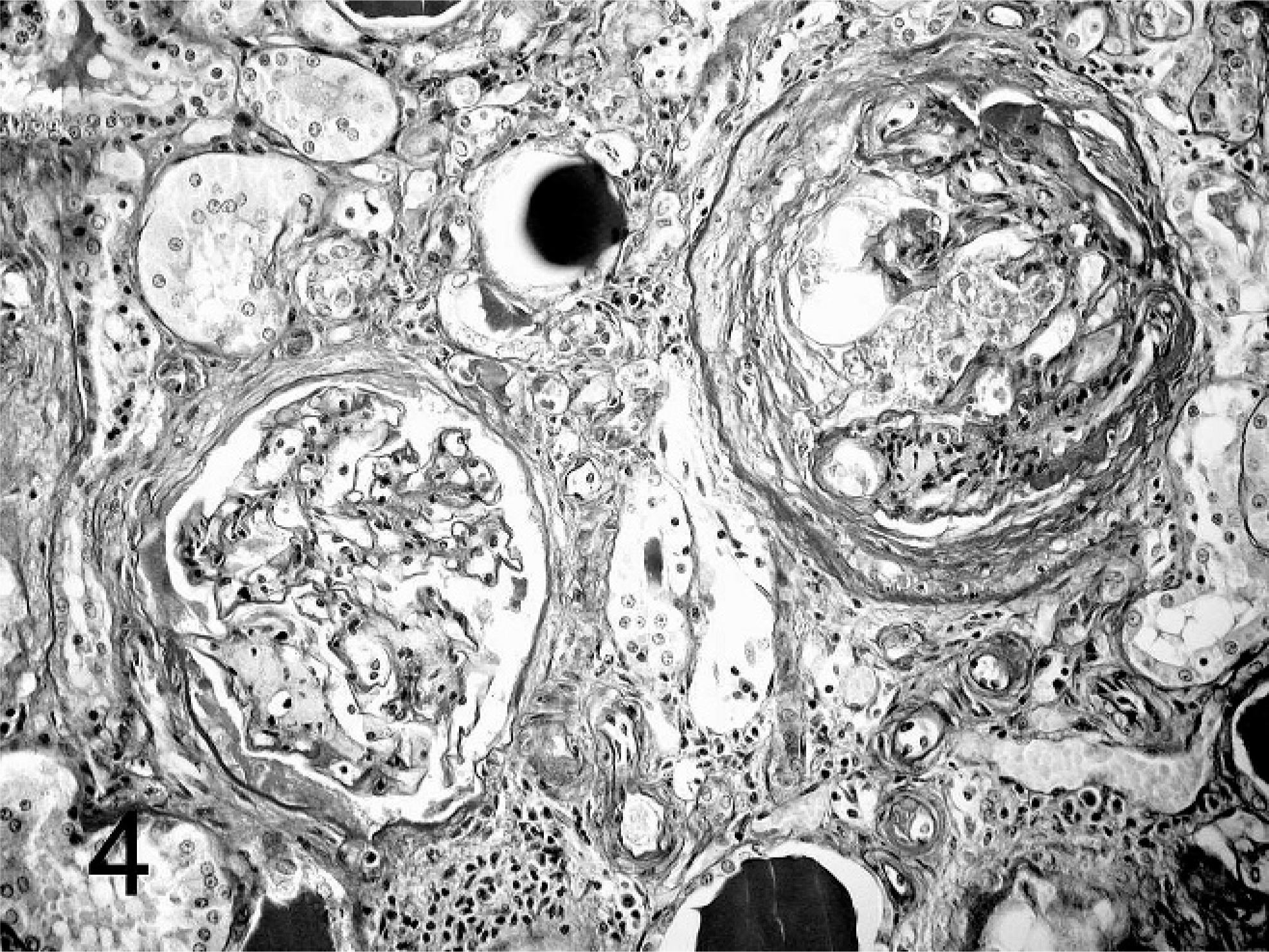
Kidney; agouti No 4. Polycystic kidney disease. Proteinaceous effusion and fibrosis of Bowman's capsule. PAS stain.
The renal tubules in the cortex of all kidneys comprised multifocal or, in 5 cases, generalized (Nos. 1, 2, 3, 5, and 6) moderate to severe dilatation with markedly flattened epithelia. Changes in the medulla were less severe, but in 2 agoutis tubules of both cortex and medulla were affected (Nos. 1 and 5). Occasionally, in all kidneys, tubular epithelial cells comprised moderate intracytoplasmic brown granules that mostly stained positive with Prussian blue, indicating cellular iron storage. Rarely, some of these intracytoplasmic granules were positive for lipofuscin with a prolonged Ziehl-Neelson stain. The intraluminal fluids were highly proteinaceous, and, in some agoutis, protein droplets had accumulated in the lumen of the tubules. The kidneys of 5 animals (Nos. 2, 3, 5, 6, and 7) revealed focal to multifocal calcification of intraluminal tubular contents (Fig. 5) and, in three cases, the tubular basal membranes (Nos. 2, 5, and 6).
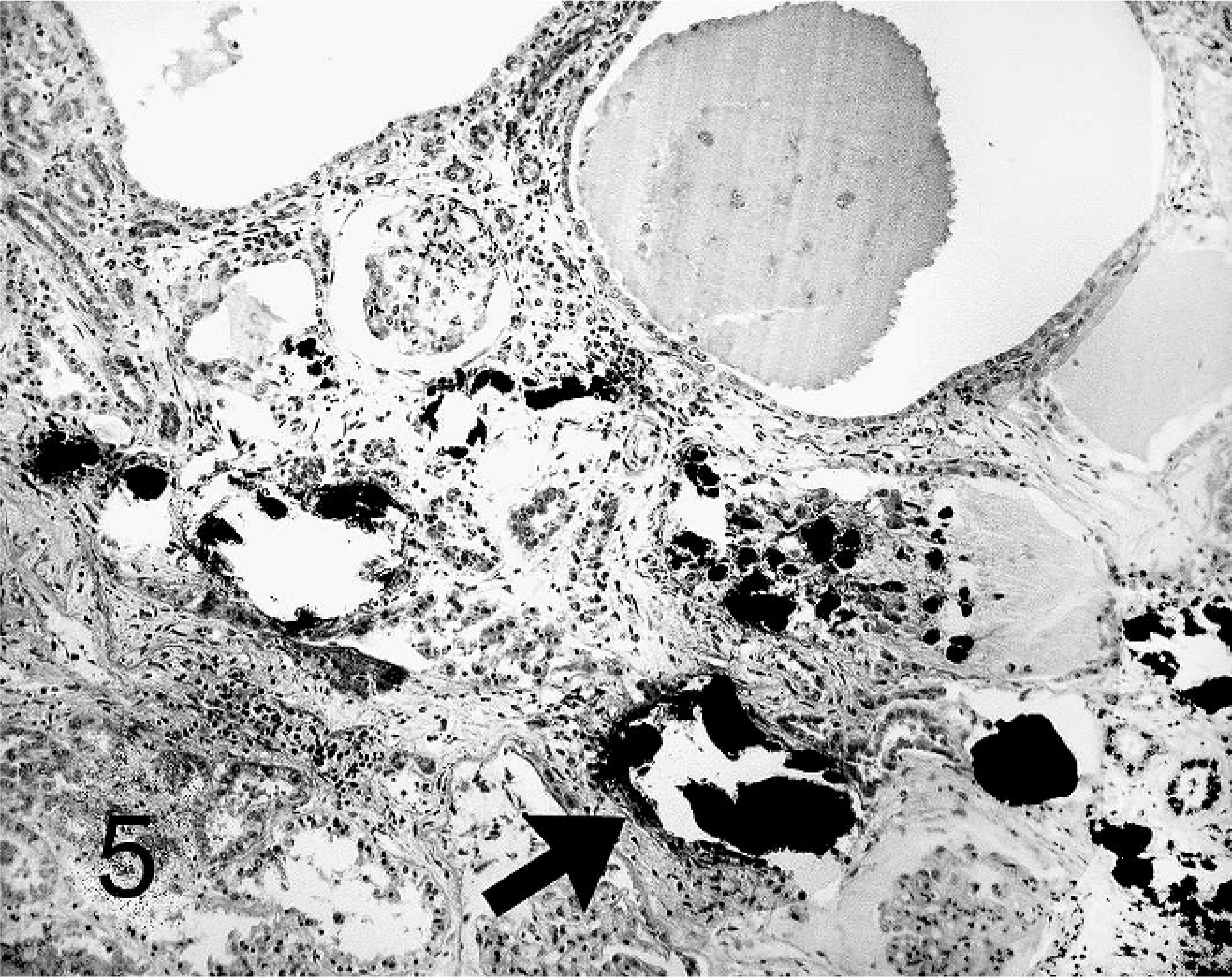
Kidney; agouti No 5. Polycystic kidney disease. Tubular intraluminal mineralization (arrow). Von Kossa stain.
The interstitium of all kidneys comprised multifocal mild to moderate and in 2 cases severe (Nos. 2 and 5) infiltration with lymphocytes, plasma cells, and a few neutrophilic granulocytes (Fig. 6). In all but 1 agouti (No. 1) there was mild to moderate interstitial fibrosis. In 3 cases (Nos. 1, 2, and 6), sporadic interstitial calcification was detected.
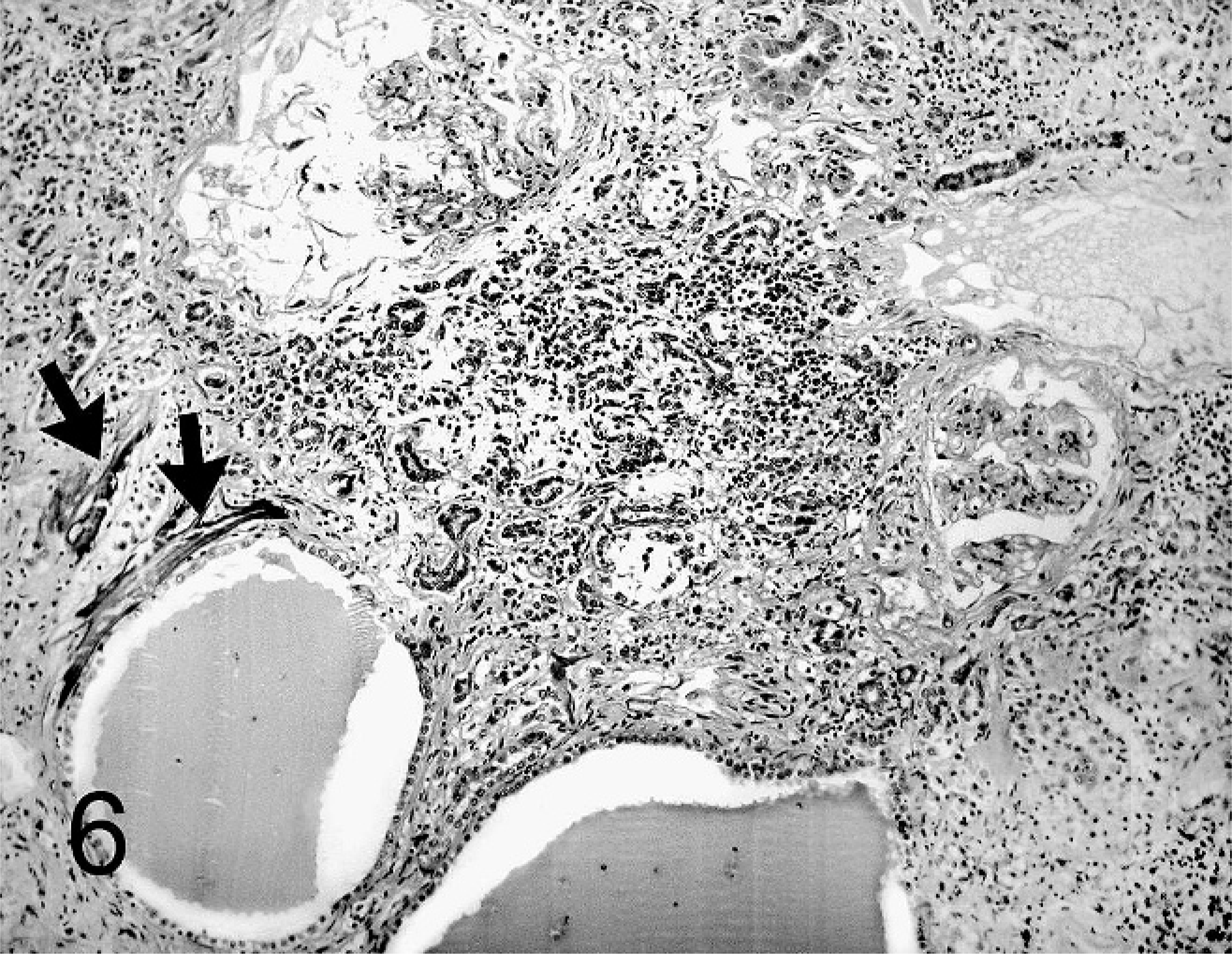
Kidney; agouti No 5. Polycystic kidney disease. Dilatation of tubules and glomerular corpuscules with fibrosis and calcification (arrows) of Bowman's capsule and mild interstitial fibrosis with mononuclear infiltration. HE stain.
Multifocal moderate to severe calcification was also observed in the media of the aorta (No. 5) and in a number of arteries in different organ systems (intestine, myocardium, muscle, adrenal gland; Nos. 2, 6, and 7); in intramuscular fascial connective tissue (No. 6); in the gastric mucosa and muscular tunic (No. 2); and in some bronchial cartilage (Nos. 1, 2, 4, 5, and 7). The urea concentration of the intraocular fluid in agouti No. 6 was markedly elevated to levels of 120 mmol/liter (reference value: 1.45–2.9 mmol/liter).
Discussion
To our knowledge this is the first description of PKD in Brazilian agoutis. At necropsy all 7 adult agoutis from different zoologic gardens revealed severe bilateral polycystic changes of their kidneys. The severity of these lesions and the lack of other significant pathologic findings make renal failure, with resulting uremia, the most likely cause of death. This finding is supported by the high intraocular urea concentration in agouti No. 6.
Macroscopic and histologic changes were characteristic for PKD. Although all cases had mild to severe multifocal mononuclear infiltration accompanied by mild interstitial fibrosis and, in 1 agouti (No. 5), unilateral renal atrophy similar to chronic interstitial nephritis, the main features typical for acquired PKD led to its diagnosis. Severe generalized multifocal cystic dilatations of the entire renal tubular system are considered to be the main change in PKD. 9,10 In addition, dilatation of Bowman's capsules is described in adult human PKD as well as in Bull Terriers. 20,24 These changes are not described in chronic interstitial nephritis, whereas secondary mononuclear interstitial infiltration is also a common pathologic finding in PKD. 4,7,11,18,24 Interstitial fibrosis and thickened Bowman's capsules are also described in autosomal dominant PKD in adult Persian cats, Bull Terriers, and humans. 7,18,21,24,30 In contrast to other reports on PKD in adult animal species and humans, none of the agoutis revealed cysts in any other organ system, including, for example, intrahepatic bile ducts or the ducts of the exocrine pancreas. 4,7,9,18 Accumulations of erythrocytes in Bowman's capsules and cysts are also described in PKD of humans and cats. 7,20 Mild to moderate epithelial iron storage in all agoutis might be a result of hemoglobin reabsorption, which is also a component of human PKD. 20 Damaged renal tubules also have an impaired ability to reabsorb calcium sufficiently, thus causing multifocal intraluminal accumulation of calcium salts. Accompanying secondary hyperparathyroidism may have caused multifocal calcification mainly found in the tunica media of arteries in at least 4 agoutis (Nos. 2, 5, 6, and 7). The observed focal destruction of mineralized bronchial cartilage in agouti No. 7 might be the result of enhanced parathyroid hormone production. Mineralization of bronchial cartilage (Nos. 1, 2, 4, 5, and 7) could be a similar side effect of hyperparathyroidism or related to an aging process. Mineralization of gastric mucosa (No. 2) as a result of ulcerative processes is often associated with uremia.
The genetic background of PKD in adult agoutis remains unknown. Preliminary investigations into the origin of the animals indicate that Brazilian agoutis kept in German zoos were bred from only a few founder individuals, and the PKD could therefore be a result of inbreeding. This hypothesis of a genetic disorder is supported by the fact that all 7 adult agoutis comprised the same renal lesions and that they originated from 4 different German zoos. This fact makes nutritional- or environmental-related factors less likely as a cause of PKD. Further investigations are currently being undertaken into the genetics of agoutis to clarify the role of inbreeding in this disease. In human medicine, clinical trials are currently being conducted, but no satisfactory therapy is known for both autosomal-recessive and autosomal-dominant PKD. 8,31,32 Several animal models have already been introduced for the congenital and adult form of PKD. Nevertheless, none of these rodent models seem to sufficiently resemble the human adult form of PKD in all aspects. 31 If PKD in agoutis is determined to be based on a genetic mutation, this species may provide a new animal model for autosomal-dominant PKD. Successful husbandry of agoutis as a laboratory animal species is already described by Baas, Potkay, and Bacher. 2
In conclusion, in this retrospective study all adult agoutis submitted for necropsy revealed severe bilateral renal alterations similar to the human form of PKD. As the population of Brazilian agoutis in Germany is the result of breeding from a small number of founder animals, a genetic background for the alterations seen in the kidney seems highly likely.
Footnotes
Acknowledgements
We thank Z. Mezoe, D. Krumnow, and M. Biering from the Leibniz Institute for Zoo and Wildlife Research, Berlin, for excellent technical assistance and Dr. G. Strauss and K. Rudloff from the Tierpark Berlin-Friedrichsfelde for information on the origin of agoutis kept in German zoos. We especially thank Dr. Alan Curry for copy-editing the final version of the manuscript.
