Abstract
The hepatic progenitor compartment is of vital importance in liver regeneration when hepatocellular replication is impaired, as it occurs in acute fulminant hepatitis or severe liver fibrosis. It consists of resident progenitor cells in the normal liver, and ductular reaction and intermediate hepatobiliary cells in diseased livers. An histologic and immunohistochemical study was conducted to demonstrate putative hepatic progenitor cells in the normal liver (n = 5) and in a range of hepatic diseases (n = 13) in the cat. Formalin-fixed, paraffin-embedded specimens were stained with HE, the van Gieson stain, and the reticulin stain according to Gordon and Sweet, and immunohistochemically stained for cytokeratin-7 (CK7), human hepatocyte marker 1 (Heparl), and multidrug resistance-binding protein-2/ATP binding cassette C2 (MRP2). The normal feline liver contains a liver progenitor cell morphologically similar to humans and dogs, which resides in the canal of Hering. In acute and chronic feline liver diseases a ductular reaction is present, whether in the parenchyma or in a portal or septal location. The putative progenitor cells could easily be demonstrated by staining for CK7, whereas they were generally negative for Heparl and MRP2. In a parenchymal ductular reaction mitotic figures and cells with an intermediate hepatobiliary phenotype could be demonstrated. This is the first account of hepatic progenitor cells in feline liver.
Clinical acute and chronic hepatic disease in both humans and animals may evolve into fatal liver failure from insufficient or impaired hepatic regeneration. Better insight into the underlying mechanisms of hepatic fibrosis versus regeneration could possibly widen the limited therapeutic possibilities available at present. In the liver, regeneration can occur in two ways: 1) replication of mature hepatocytes and 2), in the case of hampered hepatocytic reproduction, by proliferation of the local stem cell/progenitor cell population. 8,9,17,18 A third (theoretical) route by fusion or transdifferentiation of hemopoetic or bone marrow stem cells into hepatocytes is not regarded as clinically important. 3,4,7,11
The progenitor cell population, or progenitor cell compartment, is present in normal and diseased liver. In normal liver, it consists of small, resident progenitor cells that are located in the finest branches of the biliary tree, the canals of Hering. 17 In the diseased liver, the progenitor cell compartment may proliferate and is then expanded by pathologic structures called ductular reactions (DR) and intermediate-sized hepatobiliary cells. The DR encompasses an epithelial component with a distinct trabecular or tubular structure, but also in the complex of stroma, inflammatory cells, and other structures of diverse systems, all of which participate in this reactive lesion. 17,25 This DR may be populated by small cholangiocytes/progenitor cells (<6 µm) and by intermediate-sized hepatobiliary cells larger than 6 µm in diameter but smaller than a mature hepatocyte. A DR should not be confused with ductal plate malformations, such as congenital hepatic fibrosis and polycystic disease, as these are malformations and not reactive lesions. 17
This is the first study to demonstrate the presence of hepatic progenitor cells in the normal feline liver. In a variety of liver diseases in the cat the regenerative and reparative response of the liver, called DR (expansion of the progenitor cell compartment), is characterized by histology and immunohistochemistry.
Materials and Methods
All samples were derived from feline tissues examined at the Faculty of Veterinary Medicine, Utrecht University, The Netherlands. Normal liver tissue (5 cats) was obtained from surplus material from a research project on normal liver function and morphology. Based on histologic evidence of DR with HE staining, liver specimens from 13 cats with acute or chronic liver diseases were selected from the archives of the Department of Pathobiology. Liver samples had been offered as diagnostic biopsy material (n = 1) or were taken from cats submitted for postmortem investigation (n = 12). Patients were referred between 1994 and 2005. No tissue was taken purposely for the reported study. Projects were approved by the responsible ethical committees for the use of experimental animals and for use of client-owned animals according to Dutch legislation.
Wedge biopsies and postmortem samples, taken within 1 hour after submission, were fixed in 10% neutral-buffered formalin and routinely, paraffin embedded. Sections (3 µm) were routinely stained with HE, van Gieson stain, and the reticulin stain, according to Gordon and Sweet, and in 2 cases the rubeanic acid stain for copper (cat Nos. 10 and 17). Histopathologic diagnoses were based on the criteria of the World Small Animal Veterinary Association (WSAVA) Liver Diseases and Pathology Research Group, 2,20–23 and are summarized in Table 1. The copper scoring method was as described previously for the dog. 20
Case summaries.
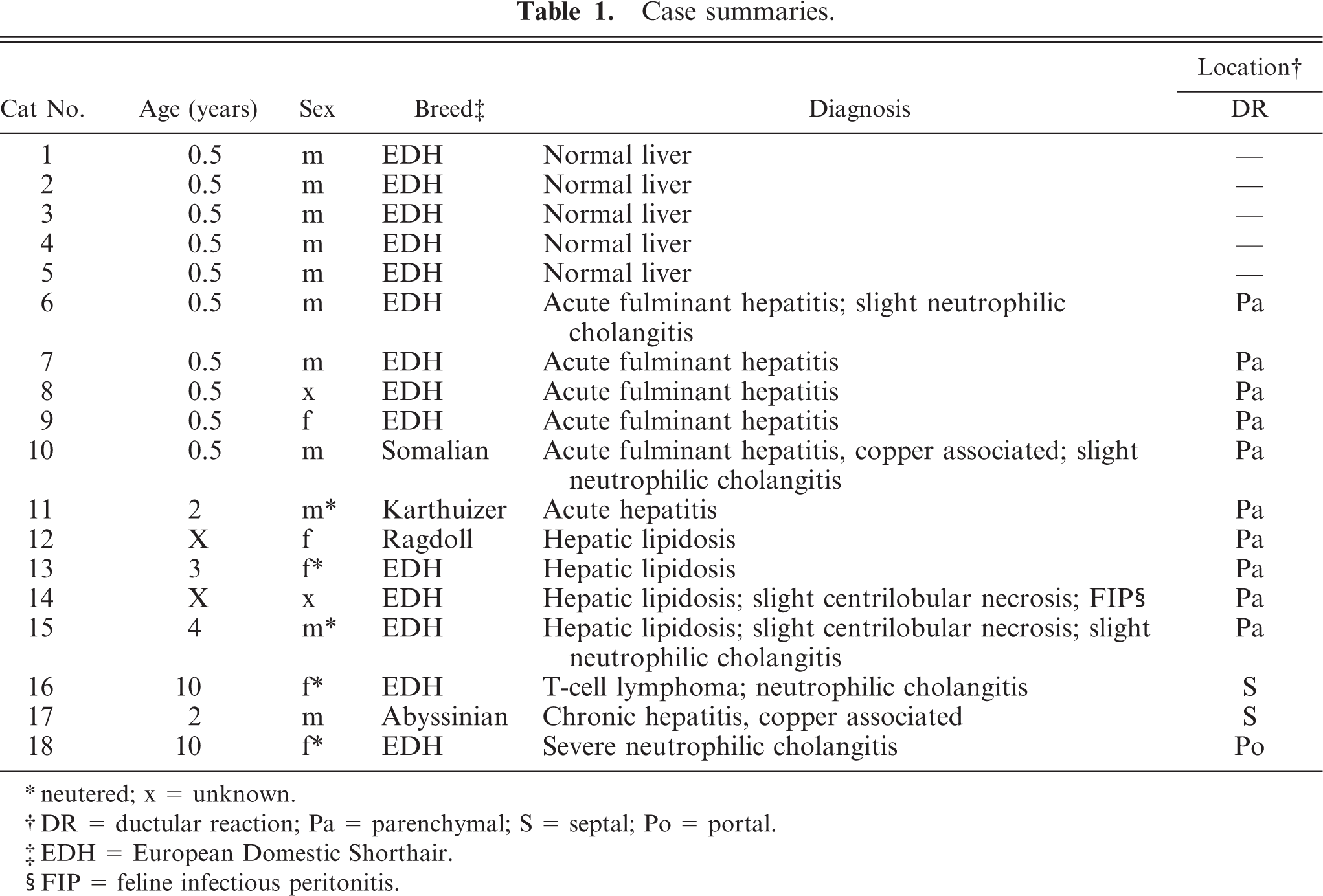
∗neutered; x = unknown.
†DR = ductular reaction; Pa = parenchymal; S = septal; Po = portal.
‡EDH = European Domestic Shorthair.
§FIP = feline infectious peritonitis.
Immunohistochemical staining of normal and diseased livers was performed for cytokeratin-7 (CK7), human hepatocyte marker 1 (Hepar1), and multidrug resistance-binding protein-2/ATP binding cassette C2 (MRP2). Antibody characteristics, manufacturer, dilution, and antigen retrieval methods are summarized in Table 2. Sections (3 µm) were mounted on poly-
Details of immunohistochemical reagents and methods.

RT = room temperature.
† MRP2 = multidrug resistance-binding protein-2.
Results
Histology (HE, van Gieson, reticulin, rubeanic acid)
In the normal liver each portal tract contained one or a few bile ductular cross sections lined by cuboidal to cylindrical cholangiocytes, depending on duct size. The parenchyma did not contain any discernable bile ductular structures. In a variety of liver diseases, a DR pattern was present. A parenchymal DR was prominently present in cats with acute fulminant hepatitis. In this case the DR consisted of small branching ductules, sometimes containing a small lumen, lined by small and intermediate-sized cuboidal epithelial cells in the periportal areas (Fig. 1). Ductules radiated variably deeply into the parenchyma, sometimes appearing as an isolated structure. Although this is often less obvious with HE staining, a DR with small radiating solid branches in the periportal parenchyma was present in severe hepatic lipidosis (Fig. 2). Finally, a marked periportal parenchymal DR was seen in 1 cat with epitheliotropic malignant T-cell lymphoma, associated with moderate hepatocellular necrosis, and slight neutrophilic cholangitis (cat No. 16; Fig. 3). A septal DR was prominently present in a cat with severe copper-associated chronic hepatitis (cat No. 17). In this case regenerative nodules were separated by broad bands containing extensive DR, slight fibrosis, groups of heavily copper-loaded intact hepatocytes, some hepatocellular necrosis, and many copper-containing macrophages. The regenerative nodules only had slight copper accumulation in hepatocytes, particularly at the periphery of the nodules. A moderate DR, restricted to the portal areas, was present in 1 cat with severe neutrophilic cholangitis. In many cats rare mitotic figures were seen in the parenchymal DR (Fig. 1, inset).
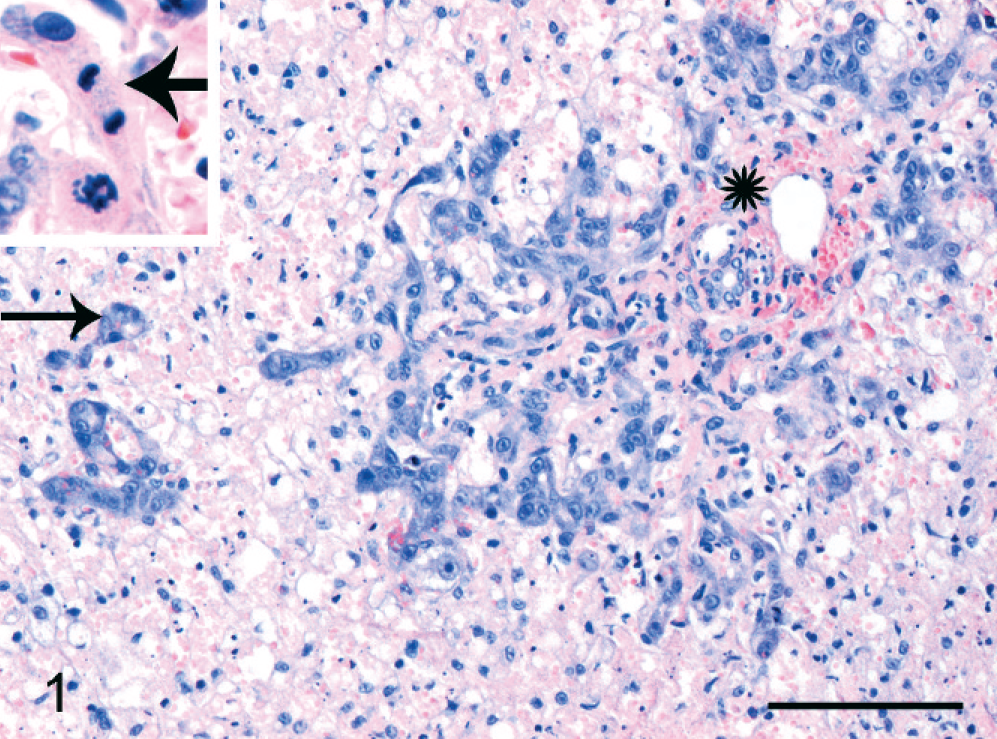
Acute fulminant hepatitis; cat No. 9. Reactive ductules extend from the portal tract (asterisk) into the necrotic parenchyma. Isolated ductules are present beyond the limits of the portal tract (arrow). Inset: Mitotic figure in ductule. HE. Bar = 100 µm.
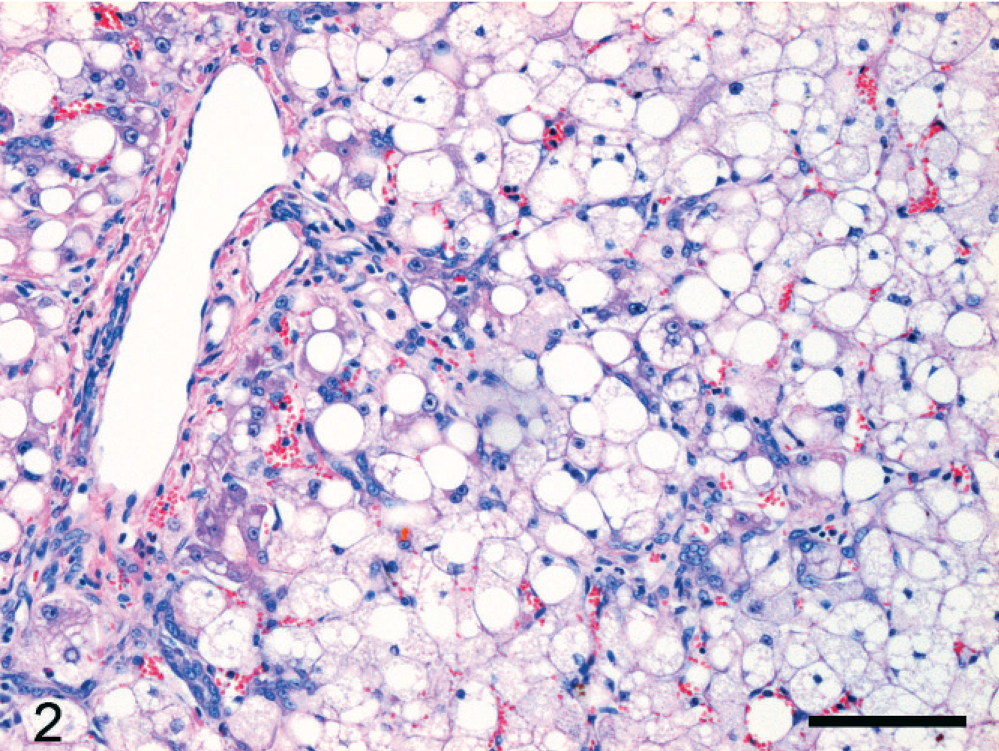
epatic lipidosis; cat No. 13. Extensive micro- and macrovesicular steatosis in all mature hepatocytes. Reactive ductules, mainly without a lumen, extend far into the parenchyma. HE. Bar = 100 µm.
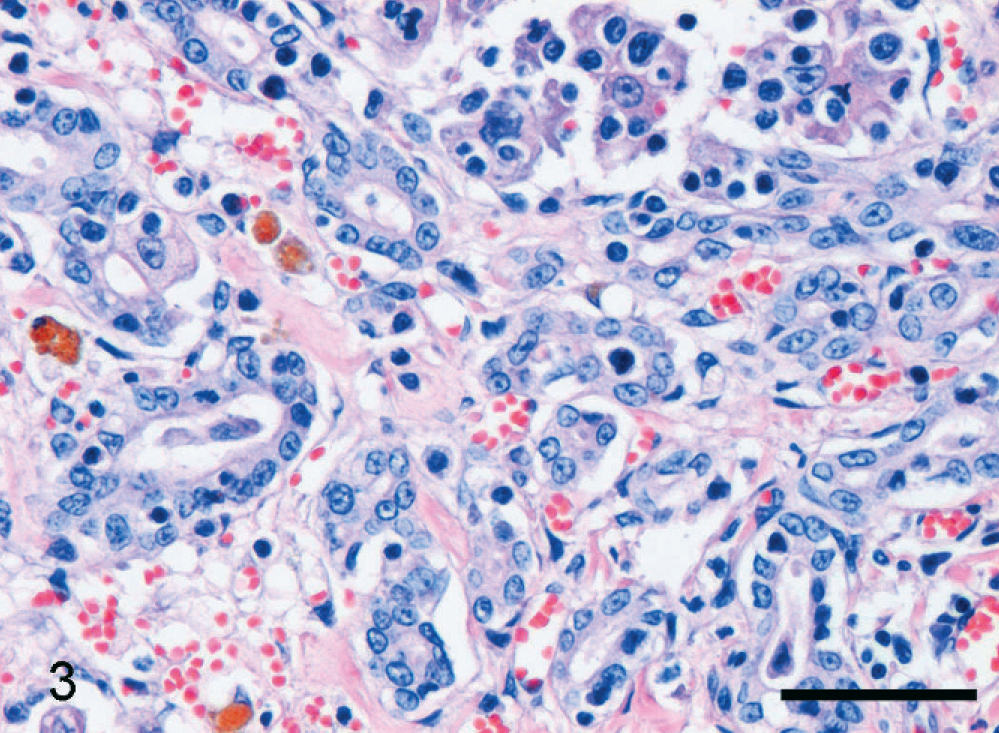
Malignant epitheliotropic T-cell lymphoma; cat No. 16. Branching reactive ductules are present at the porto-parenchymal interface. HE. Bar = 50 µm.
The van Gieson stain, as well as the reticulin stain, revealed enhanced positivity in fibrous septa.
In both tested cats (Nos. 10 and 17) copper scores in the rubeanic acid staining were 4+.
Immunohistochemistry
Cytokeratin-7 (CK7)
The normal liver bile ducts showed diffuse staining of the cholangiocytic cytoplasm. Also, few immediately periportally located small cells, which formed small groups or appeared as isolated cells, reacted positively (Fig. 4). A parenchymal DR displayed strong cytoplasmic positivity of small and intermediate-sized cells (Figs. 5, 6). Furthermore, the intermediate cells featured a stronger membranous staining, fading away in the largest intermediate-sized positive cells (Fig. 5). A septal and portal DR was present in a cat with chronic hepatitis (cat No. 17) and severe neutrophilic cholangitis (cat No. 18), which also reacted positively. Here, the DR was restricted to the septa, to the portal areas, and no intermediate cells were seen.
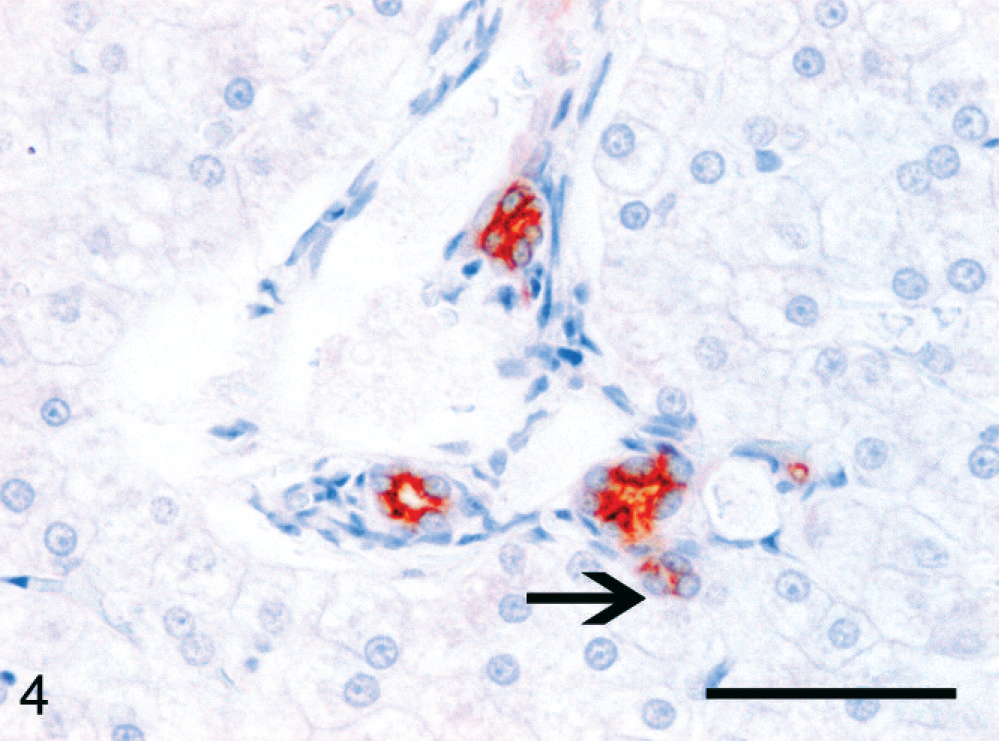
Normal feline liver; cat No. 4. Strong cytoplasmic staining (red) in mature cholangiocytes in the portal area. Few positive cells form a small ductular structure just beyond the limits of the portal area (arrow). This suggests the presence of progenitor cells in the canal of Hering. CK7 immunolabeling, Mayer's hematoxylin counterstain. Bar = 50 µm.
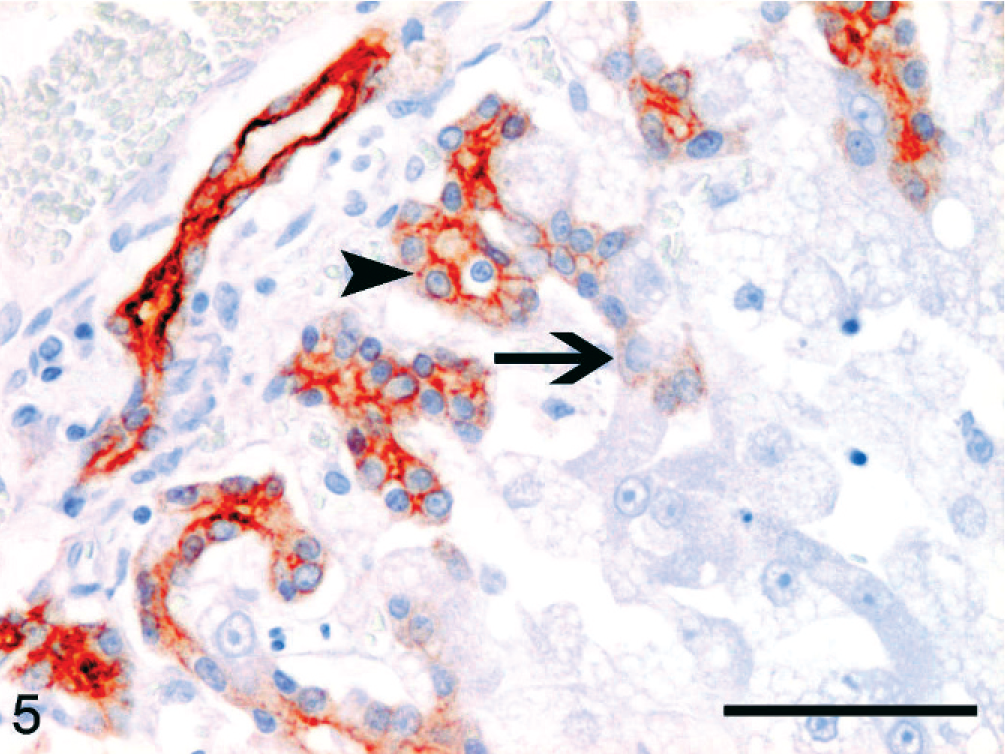
Hepatic lipidosis; cat No. 14. Small and intermediate-sized positive cells (red) compose the ductular reaction perpendicular to the preexisting bile duct (left top), which is compiled of small, diffusely positive cholangiocytes. The ductular reaction is composed of intermediate-sized cells with a membranous staining pattern (arrowhead). On the edge of the hepatic cords, also larger intermediate-sized cells appear that display less intense cytoplasmic reactivity (arrow). CK7 immunolabeling, Mayer's hematoxylin counterstain. Bar = 50 µm.
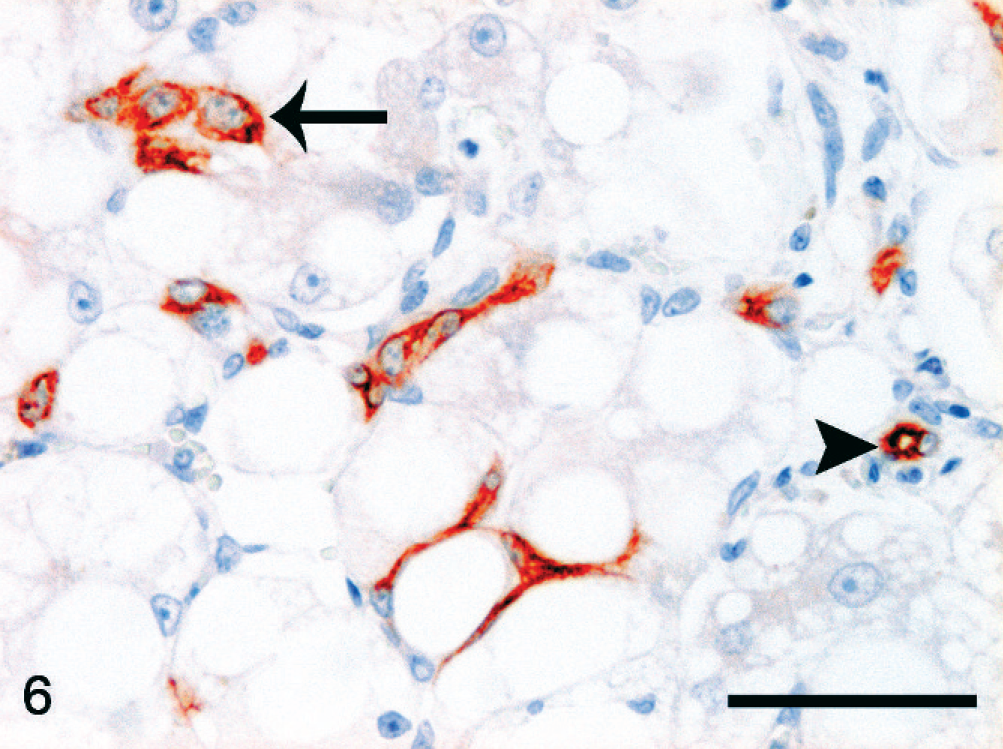
Hepatic lipidosis; cat No. 13. Branching interface reactive ductules in the hepatic parenchyma. Cytoplasmic CK7 reactivity (red) is present in small cells of a preexisting bile duct (arrowhead) and in intermediate cells of varying size (arrow at largest positive cell), which are dispersed between hepatocytes with extensive fatty change. CK7 immunolabeling, Mayer's hematoxylin counterstain. Bar = 50 µm.
Hepatocyte paraffin 1 (Hepar1)
In the normal feline liver Hepar1 provoked strong granular cytoplasmic reactivity, exclusively in mature hepatocytes (Fig. 7). A few, either periportally or centrolobularly located, individual hepatocytes displayed even more intense staining. In diseased livers, viable hepatocytes had granular, strongly positively staining cytoplasm (Fig. 8). Most necrotic hepatocytes showed a less intense and more smoothly dispersed signal. Ductular reaction, whether parenchymal, septal, or portal, showed no positive staining in general (Fig. 8). However, in acute fulminant hepatitis, positively reacting small or intermediate-sized cells in or directly adjacent to a ductular structure were incidentally seen (Fig. 9). In addition, some staining of individual cholangiocytes in preexistent bile ducts was rarely observed in 2 cats (Nos. 6 and 11).
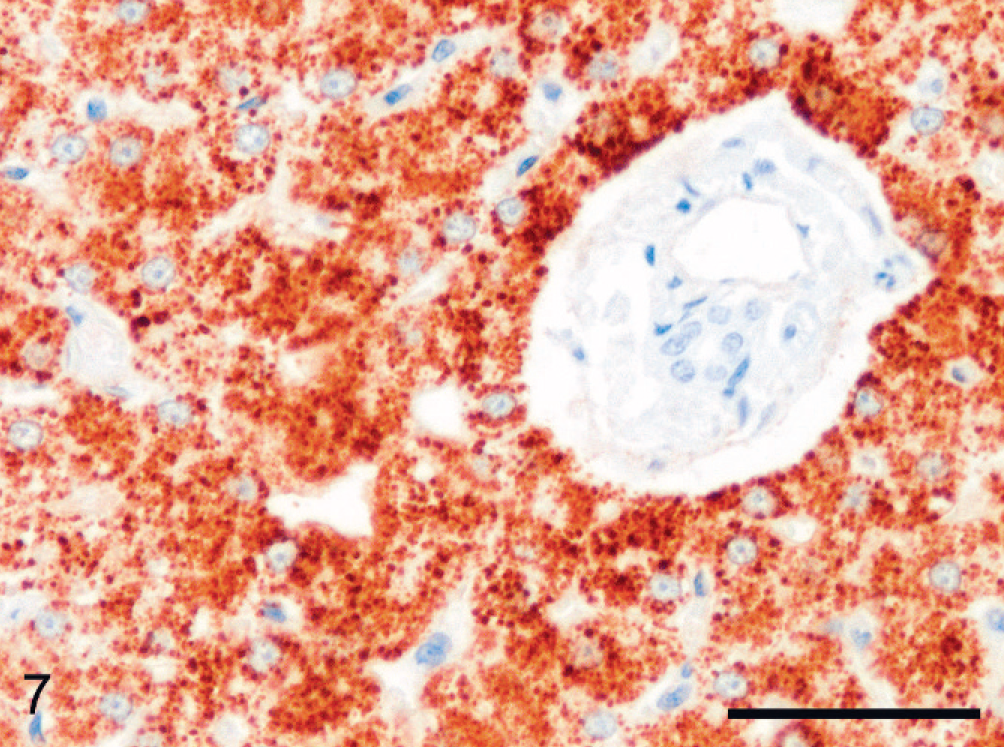
Normal feline liver; cat No. 2. In all depicted periportally located hepatocytes a strong positive signal (brown) is present. All other cells are negative. Hepar1 immunolabeling, Mayer's hematoxylin counterstain. Bar = 50 µm.
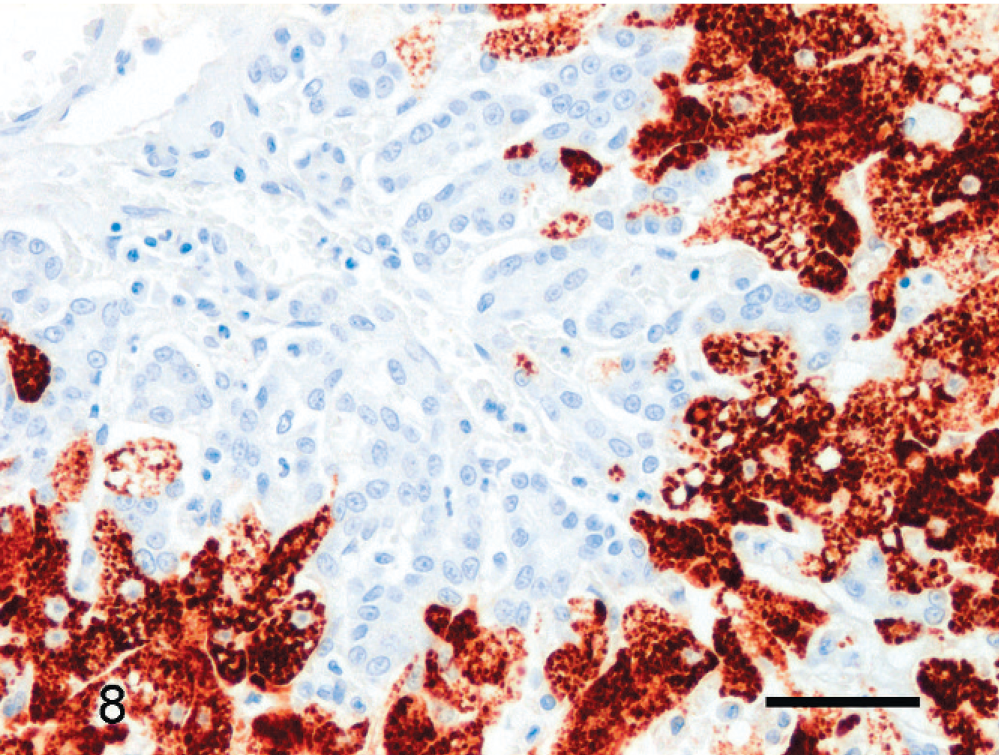
Hepatic lipidosis; cat No. 14. Epithelial cells in ductular reaction are negative (left top and center), and larger, mature hepatocytes show intense granular cytoplasmic positivity. Hepar1 immunolabeling, Mayer's hematoxylin counterstain. Bar = 50 µm.
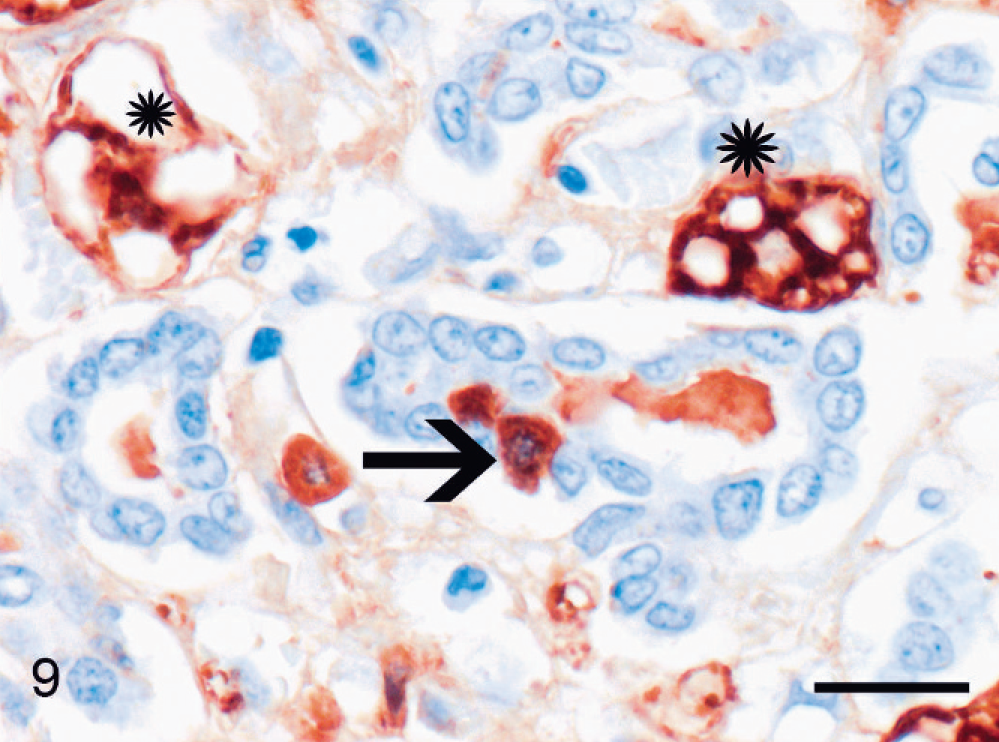
Acute hepatitis; cat No. 11. Reactive ductules composed of small epithelial cells are dispersed between degenerated hepatocytes (asterisks). Few small, strongly positive (brown) epithelial cells are present (arrow) in the ductules. Hepar1 immunolabeling, Mayer's hematoxylin counterstain. Bar = 20 µm.
Multidrug resistance-binding protein-2/ATP binding cassette C2 (MRP2)
In the normal liver linear staining at the canalicular hepatocellular membranes was present, consistent with the anatomic location of bile canaliculi (Fig. 10). The staining was more pronounced centrolobularly than periportally. In addition, slight background staining appeared in the cytoplasm of the hepatocytes. In all sampled diseased livers necrotic hepatocytes were negative, as well as all reactive ductules, whether these were located parenchymal, septal, or portally. Infrequently, a positive reaction was observed at the junction of DR and mature hepatocytes.
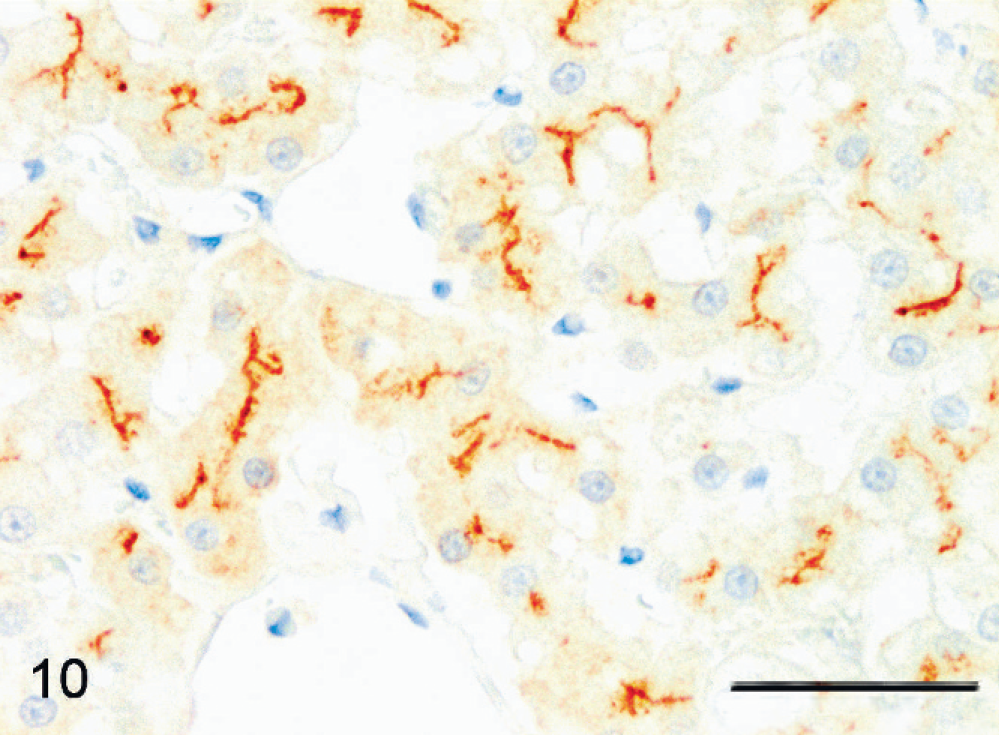
Normal liver; cat No. 1 (P05 4718-1). Hepatocellular bile canalicular membranes react positively (brown). Unapparent cytoplasmic background staining appears in hepatocytes; all other cells are negative. MRP2 immunolabeling, Mayer's hematoxylin counterstain. Bar = 50 µm.
Discussion
The most important hepatobiliary diseases in cats include cholangitis, hepatic lipidosis, and, less frequently, acute fulminant hepatitis; chronic hepatitis/cirrhosis is uncommon. 22 This is reflected in the selected cases, where compared with canine patients, a preponderance of acute cases (n = 11, of 13 patients; see Table 1) is present.
In the normal feline liver CK7 immunohistochemistry revealed positive bile ducts and highlighted small periportally located cells (Fig. 4). Morphology and location of these CK7-positive cells were identical to resident hepatic progenitor cells located in the canal of Hering, as described in humans and dogs. 6,17 To the best of our knowledge such putative hepatic progenitor cells have never been reported in the cat.
The term DR refers to a reaction of ductular phenotype, possibly, but not necessarily of, (bile) ductular origin, 17 as it can arise from preexisting cholangiocytes, progenitor cells, or hepatocytes. Determination of the origin of the epithelial cells in ductules of the present cases was beyond the scope of this study, as it requires longitudinal studies and multiple markers. Therefore, we preferred the more neutral terminology of DR, instead of the older term reactive biliary hyperplasia.
In humans, dogs, and rodents, the occurrence of DR in the parenchyma indicates impaired hepatocellular proliferation at times of demand of hepatic regeneration. 8,9,13,16,18,24 Our findings in the cat are in accordance with those in humans, dogs, and rodents. 5,6,16,19
The classic rodent “oval cell” proliferation is very easily provoked and is often dispersed throughout the lobules. 5 Each cat in the present study showed DR to some extent, because case selection was based on the presence of DR in HE staining. Compared with rodent liver, the parenchymal DR in the cat was less extensively present. It varied markedly in extent and was located in the periportal parenchyma (Figs. 1–3). This pattern more closely resembled DR in humans and dogs, where it appears less common (although still often) and less extensive than the rodent oval cell proliferation.
The existence of intermediate-sized cells positive for CK7 could indicate differentiation of progenitor cells into mature hepatocytes (Figs. 5, 6). However, as all studied cases were “snapshots” in time, dedifferentiation of hepatocytes into proliferating progenitor cells cannot be excluded. 10,16,24 Parenchymal DR in the cat was not only associated with massive hepatic necrosis (acute fulminant hepatitis) and lipidosis, but was also seen in a chronic case, epitheliotropic malignant T-cell lymphoma (cat No. 16). Some hepatocellular necrosis was also present. Compared with other species, cats are very sensitive to hepatic lipidosis, which can be a sequel to relatively short anorectic periods, with and without any other underlying disease. 2 The extensive DR found in cats with severe hepatic lipidosis indicates serious hepatocellular injury. In humans and mice, hepatocellular replicative senescence in fatty liver disease has been attributed to oxidative stress. 15,24
The CK7 staining of the bile ducts and the small cells of portal DR (cat No. 18), as well as the septal DR in chronic hepatitis (cat No. 17), is as described in other species (Figs. 5, 6). 6,15,19 The absence of any intermediate cells in the portal and septal DR in these cases might indicate that progenitor cell maturation is restricted by this setting. 12,13
In the normal feline liver, staining for mature Hepar1 was restricted to mature hepatocytes (Fig. 7). Ductular reaction in general appeared negative for Hepar1 (Fig. 8). However, in some cats with acute fulminant hepatitis solitary small epithelial cells in the wall of a reactive ductule was positive (Fig. 9). These cells always were in close contact with the hepatic parenchyma and were located at the interface of ductules and mature hepatocytes. In our opinion, this indicates hepatocellular commitment of the progenitor cells composing these reactive ductules.
Excretion pump MRP2 stained bile canalicular membranes in viable mature hepatocytes in both the normal feline liver and in diseased livers (Fig. 10). Because necrotic hepatocytes appeared negative, this marker could serve as a viability marker for hepatocytes. Hepar1 is unsuitable because necrotic hepatocytes react to the antibody (in immunohistochemistry). MRP2 has an important function in the biliary excretion of endogenous metabolites, as well as many exogenous compounds. 1 Because these detoxification processes are usually concentrated in the centrolobular area, this could explain the immunohistochemical accentuation of MRP2 expression in this location.
In this study, the putative progenitor cell compartment in the cat is identified by CK7 immunohistochemistry, but not by mature hepatocyte markers Hepar1 and MRP2. In humans and rodents, the progenitor cell compartment comprises several cell types, with transient expression of many different markers during proliferation and differentiation toward the hepatocytic or cholangiocytic cell lineage. 14,18,25 To the best of our knowledge, MRP2 never tested positive in progenitor cells of any species. In one recent study, 25 Hepar1 tested positive in variably sized epithelial cells, admittedly at the end of the hepatocellular maturation, which is in line with the present findings in the cat. Double positivity with CK19 or NCAM, bile ductular and progenitor cell markers, respectively, was never encountered. 25 Therefore, to confirm the presence of the feline progenitor cell compartment, additional immunohistochemical characterization could include CK19 and NCAM. 14,18
In conclusion, this study shows that the feline liver exhibits a putative progenitor cell compartment that is morphologically very similar to that in the human liver. In the normal feline liver resting progenitor cells reside in the canal of Hering. Parenchymal DR is induced in feline liver diseases with extensive parenchymal damage. These ductules show mitotic figures of small progenitor cells as well as differentiation into intermediate cell types. Portal DR remained restricted to the portal area, and did not contain intermediate cells. CK7 is a good progenitor cell marker in formalin-fixed feline liver. MRP2 may serve as a marker for viable mature hepatocytes.
Footnotes
Acknowledgements
We express our gratitude to Prof. Dr. J. E. van Dijk for critically reading of the manuscript.
