Abstract
Abstract
Purpose
Axial malignant bone tumours are rare in children and adolescents, and their prognosis is still relatively poor due to non-specific symptoms, such as back or groin pain, which may result in late hospital presentation. Therefore, it is very important to raise awareness regarding this pathology.
Methods
We performed a narrative review, including scientific publications published in English. We searched Medline and Google Scholar databases for information on the incidence and prognosis of axial malignant bone tumours in children and adolescents (< 18 years). Outcomes of different surgical management strategies and reconstruction options were assessed.
Results
The incidence of primary malignant bone tumours before the age of 18 years is approximately five per one million population; around 25% of these tumours are located in the axial skeleton. With a five-year survival rate of 50%, tumours in an axial location (chest cage, spine, pelvis) are associated with a poorer prognosis than tumours in more peripheral locations. En bloc excision with clear margins has been shown to improve local control and overall survival, even though obtaining adequate surgical margins is difficult due to the close location of large neurovascular structures and other major organs. Spinal reconstruction options include instrumented fusion with allograft or expandable cage. Pelvic reconstruction is needed in internal hemipelvectomy, and the options include biological, endoprosthetic reconstructions, hip transposition, arthrodesis or creation of pseudoarthrosis and lumbopelvic instrumentation.
Conclusion
Early diagnosis, a timely adequate multidisciplinary management, appropriate en bloc excision, and reconstruction improve survival and quality of life in these patients.
Level of Evidence
V
Introduction
Malignant bony spine tumours are rare in children and adolescents.1–3 In the pediatric age population, malignant osseous spine tumours include osteosarcoma, ewing sarcoma, lymphoma, and metastatic neuroblastoma.1,2 Approximately 50% of bony Ewing sarcomas are in the axial skeleton (spine, pelvis, ribs), and the pelvis is the most common site for Ewing sarcoma (5% to 15%) in young patients. 4 This narrative review focuses on primary malignant bone tumours in the spine and pelvis (Figs 1 to 4).
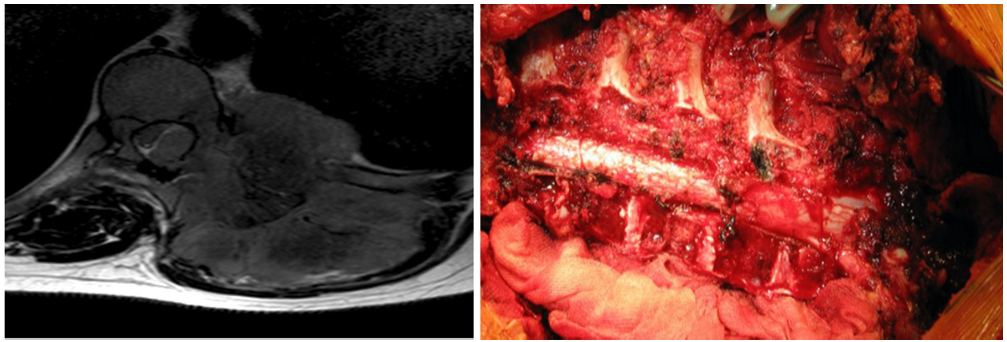
a) Chest wall Ewing sarcoma causing a severe spinal cord compression on a 15-year-old boy. Weak lower legs and severe walking difficulties. T2 axial MRI demonstrates large tumour invading the spinal canal and producing severe deformation of the spinal cord; b) multilevel sagittal excision through the vertebral bodies and discs performed and specimen removed en bloc.
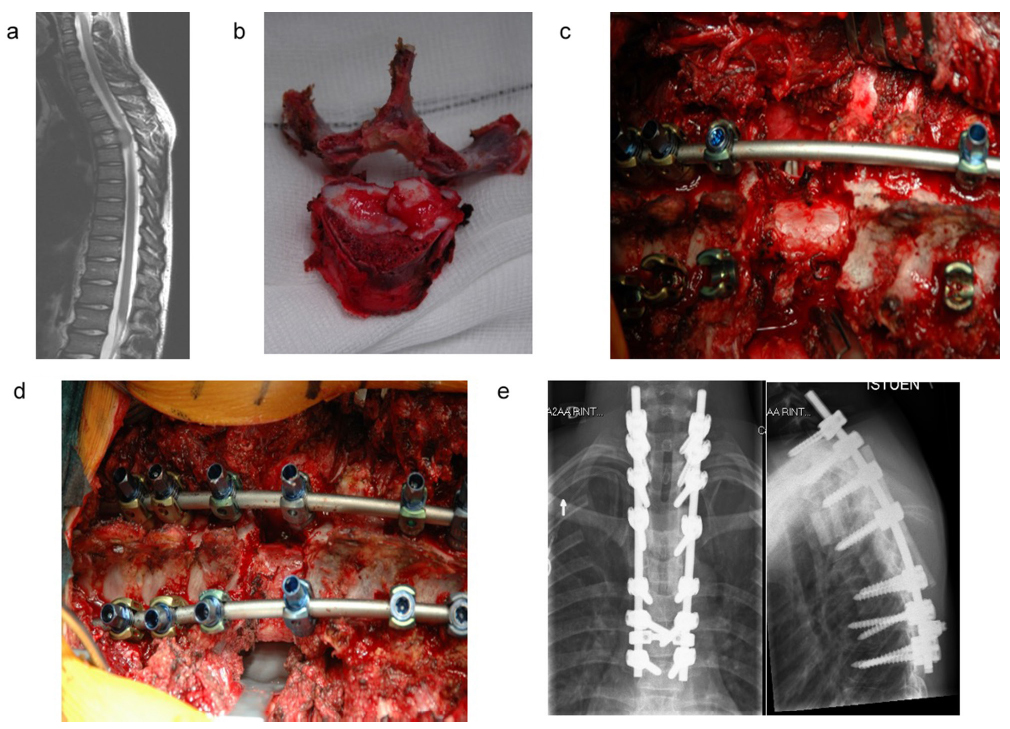
a) Ten-year-old boy with dull back pain between scapulae. Soft-tissue extension behind T4 vertebral body. Biopsy confirmed Ewing sarcoma; b) en bloc excision of T4; c) blunt dissection around the spine (instrument going in front of spine). Nerve roots ligated bilaterally and cut. A space has been created around the spine; d) wide posterior costotransversectomy approach. Humeral allograft strut placed. Pedicle screw instrumentation; e) postoperative radiographs with reconstruction using humeral allograft strut. Extension of instrumentation below an osteoporotic fracture at T6, which occurred during neoadjuvant treatment.
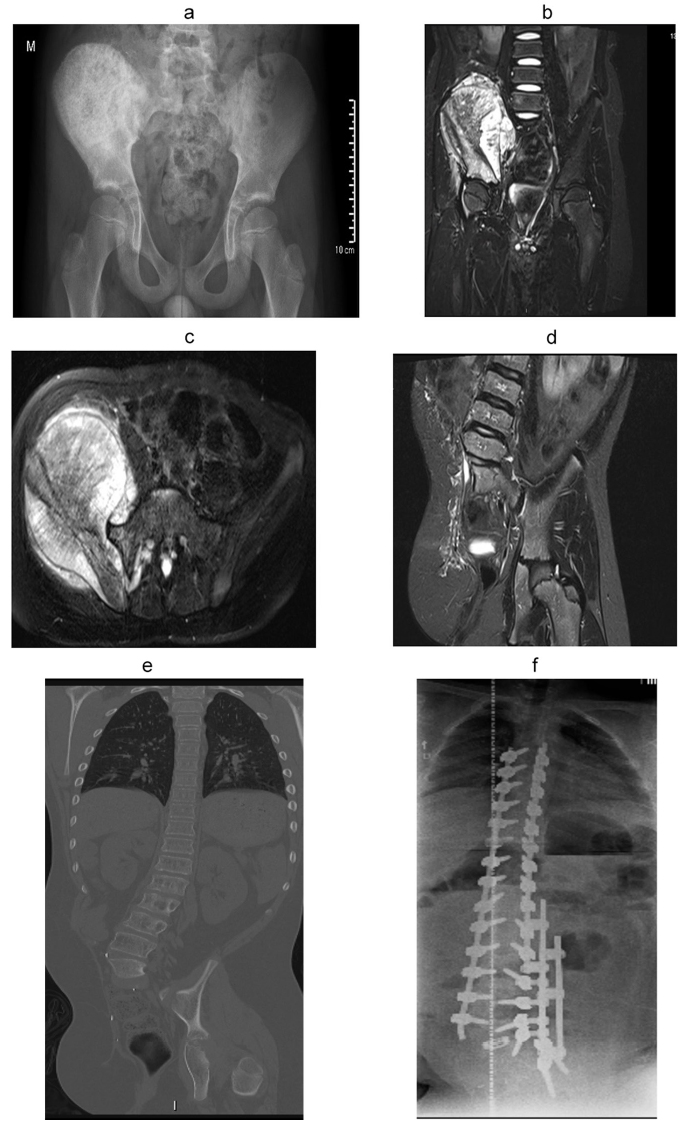
a) Eight-year-old boy with right-sided pelvic osteosarcoma; b) coronal T2 MRI demonstrating acetabular and spinal involvement; c) axial T2 MRI demonstrating L5, S1 nerve root involvement; d) postoperative T2 coronal MRI after extended external hemipelvectomy; e) patient developed painful scoliosis one year after external hemipelvectomy; f) reconstruction using long posterior pedicle screw instrumentation with Transforaminal Lumbar Interbody Fusion (TLIF) cage in the remaining L5/S1 disc and multiple rod construct to prevent rod fractures.
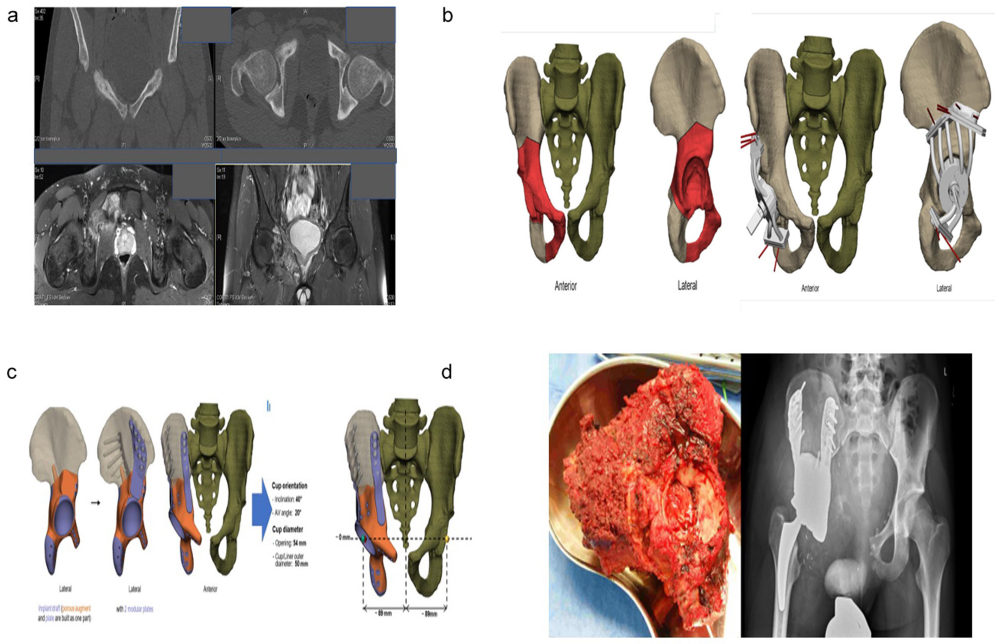
a) 17-year-old boy. Right-sided Ewing sarcoma causing a pathological pubis stress fracture and MRI showed extension in the pubic and acetabular bone; b) defining the resection planes and planning the patientspecific cutting guides; c) planning of the implant (Scaffold with femoral prosthesis) with reconstruction of the original femoral head centre; d) postoperative radiograph showing a resection Type II+III (Enneking) with clear margins and reconstruction using a scaffold wih femoral prosthesis.
In a national study, the overall incidence of bone sarcomas among children and adolescents (< 18 years of age) was 5.1 per one million population: 3.6 for osteosarcoma, 1.2 for Ewing sarcoma and 0.3 for chondrosarcoma. 5 Of these tumours, 24% (21/88) were located in the axial skeleton, i.e. chest cage, spine or pelvis. 5 Even if bone sarcomas are rare among children, it is very important to raise awareness regarding this pathology. Our aim is to provide a review of the existing knowledge on the presenting symptoms of axial malignant bone tumours, diagnostic work-up, treatment principles, and prognosis.
Materials and methods
We performed a narrative review, including scientific publications published in English. We searched Medline and Google Scholar databases for the years between 2000 and 2020, for information on the incidence and prognosis of axial malignant bone tumours in children and adolescents (< 18 years). Outcomes of different surgical management strategies and reconstruction options were assessed.
Presenting symptoms
The symptoms of axial bone tumours include back or inguinal pain, which is typically relentless or dull in nature and may present during the nighttime may or may not be exercise-related.1,2,6–9 Spinal deformity and neurological deficits are less common. Even if neurological symptoms are less prevalent in children than in adults, large soft-tissue mass involvement that causes spinal cord or nerve root compression may produce neurological symptoms depending on the location (Fig. 1a). 2 A typical neurological symptom is walking difficulties. Some patients can also present with constitutional symptoms, e.g. fever, weight loss and night sweating (Ewing sarcoma).
The time interval between the onset of symptoms (intermediate pain vs. a night pain) and the diagnosis in a pelvic location is usually significantly delayed (a mean of seven months (1 to 48)) because of intrapelvine growth and a variaty of differential diagnoses that need to be considered and excluded.7,8 In one study, 8 the pelvic sarcoma was not recognized initially in roughly half of the patients, and an inaccurate diagnosis was made. Besides inguinal hernia or urinary-tract infections, misdiagnoses often include particular spine-related diseases such as herniated lumbar disc, spinal stenosis and spondylolisthesis. Therefore, the tumour volumes have often increased to > 100 ml at the time of diagnosis. Common symptoms and findings on physical examination include buttock pain, low-back pain, pain in the hip and in the groin, a palpable mass, bladder dysfunction and sciatic nerve symptoms.7,8
Diagnostic work-up
A full clinical investigation of the child is indicated when a clinical suspicion of axial bone tumour is raised.1,2 Palpation of the tumour area may evoke the pain symptoms but is barely possible in cases of intrathoracal and/or pelvic growth. On neurological examination, the Babinski reflex can be positive, and the patella and achilles refelexes may be brisk (cord compression) or attenuated (nerve root compression) depending on the location of the tumour. The straight leg raising test becomes positive (< 60°) early with nerve root or spinal cord compression (Fig. 1). Heel and toe walking are sensitive screening tests to examine the neurological function of the lower extremities: general strength and ataxia (spinal cord, myelopathy), L3-4 (knee extension), L5 (hip abduction and ankle dorsiflexion) and S1 nerve root (plantar flexion). Rectal tone should also be evaluated (cauda equinae). 2 In pelvic sarcomas, a direct compression of sciatic or femoral nerve-related symptoms/neurological deficiencies should be feasible to detect in clinical exmination. Tenderness and groin pain is more common than night pain. 8
Standing radiographs of the spine and/or pelvis are indicated when the patient is presenting with the above-mentioned symptoms or with obvious palpable pelvic/spinal mass or deformity.1–3,6–9 Radiological diagnosis of pelvic/spinal sarcomas with simple radiographs is difficult because of the organs and soft-tissue mass that lies in front of these bones. Therefore, most symptomatic patients have to be evaluated by MRI with intravenous contrast. Primary malignant bone tumours are rare before school age, whereas infections and other paediatric cancers (leukemia, neuroblastoma) are more common. 1 Typical imaging findings in malignant bone tumours are soft-tissue mass and bony destruction (Fig. 2a and Fig. 4b).
Biopsy
The diagnosis of malignant tumours is confirmed by a well-planned biopsy.1,2 The biopsy should preferentially be performed percutaneously since the associated 0.37% risk of tumour cell seeding is significantly lower than the 32% risk associated with the open incisional biopsy.10,11 Advanced imaging helps guiding the percutaneous biopsy, which in many institutions is performed by an interventional radiologist under CT guidance after interdisciplinary discussion with the responsible surgeon and the pathologist. At the spine, a percutaneous biopsy via the pedicle allows relatively easy access to the vertebral body. Transthoracic biopsies should be avoided as they can contaminate the whole chest cavity. The diagnosis of a malignant bone tumour can often be obtained from the soft-tissue component of the tumour.
Treatment management of axial tumours
The management of the most common malignant axial tumours (Ewing sarcoma, high grade osteosarcoma) in children includes a neoadjuvant and postoperative chemotherapy, even in children with neurological compromise. A prompt start of the neoadjuvant chemotherapy has been shown to improve neurological deficits and it should, therefore, be the primary management if the tumour is sensitive to the chemotherapy. 12 The typical surgical window opens after ten to 12 weeks of oncological management depending on the primary sarcoma.
The advances in chemotherapy and surgical techniques have changed the way primary osseus malignancies are treated nowadays. The complex anatomy of the spine and pelvis and its close relation to adjacent neural, vascular and visceral structures, however, make these techniques and principles inapplicable, according to Enneking. 13 A wide resection at the cost of significant spinal cord/lower limb function would probably not be acceptable for most patients, but surgery is still considered beneficial, at least when marginal resections within anatomical barriers like a nerve sheat, fascia etc are achievable.6,9,14–18 However, in radiation sensitive bone tumours like the Ewing sarcoma or in high grade osteosarcomas, (proton) radiation therapy could be an alternative or additional adjuvant treatment option.19,20
Additional preoperative steps such as embolization of large tumours, aortic ballon occlusion or application of a tempororary stoma in case of sacrum tumours can be helpful for reducing intra- and postoperative complications like massive blood loss or infection.19,20
Spinal tumours
The anatomy of spine and neural elements does not allow radical tumour resection, therefore, the resections of tumours in the spine are typically marginal or wide. 15 It should be noted that there is typically only one good chance to perform a curative en bloc excision for spinal malignancy, and this should be performed only by the most experienced orthopaedic spine surgeons. A difficult discussion with family arises if the child is already presenting with significant neurological deficits (Fig. 1a). Surgical decompression will alleviate the neurological deficit but this will also result in local spill of malignant cells and contamination of the surrounding tissues, which makes curative en bloc exision impossible. A prompt start of the neoadjuvant management using chemotherapy and radiotherapy has been shown to improve neurological deficits as well and should, therefore, be the primary management. 12
The three main procedures used to achieve en bloc resection in spine tumours include: 1) spondylectomy (removal of full vertebral body en bloc with or without posterior elements) (Fig. 2b); 2) sagittal resection through the spine (Fig. 1b); and 3) posterior arch resection. 15 To help decide which operation is required, surgical staging systems have been developed. The two most commonly used include the Weinstein, Boriani, Biagnini 15 (WBB, Fig. 5), and the Tomita staging systems. 21 Dr. Stener, from Gothenburg, was the first to report the en bloc excision of the vertebral body, i.e. spondylectomy for a primary malignant spinal bone tumour, in 1971. 22 En bloc spondylectomy can be performed either via a posterior-only approach or a combined anterior and posterior approach, depending on the affected part of the spinal column and the surgical staging of the tumour.23,24 Basically, for the tumour to be removed en bloc, one pedicle has to be free of tumour, otherwise the operation will inevitably be at least minimally intralesional.15,21,23 In the case of a large tumour extension outside the vertebral body, a combined approach is usually needed to allow full excision of the tumour with adequate margins. The need for en bloc spinal excisions is very rare in children and, therefore, a surgical team that brings together expertise from both paediatric and adult spine surgery is very useful. 25
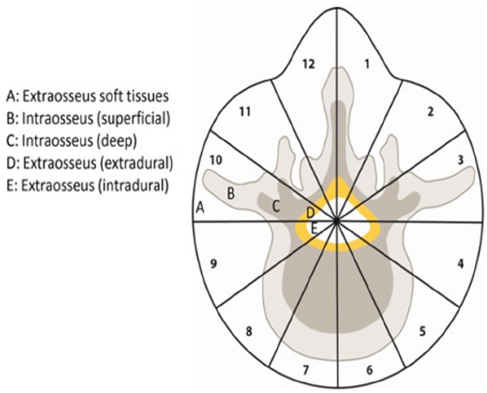
The Weinstein-Boriani-Biagnini (WBB) surgical staging system divides the vertebra into 12 radiculating zones and into five layers. Based on Boriani et al. 15
The Tomita procedure (all posterior en bloc spondylectomy) encompasses a wide bilateral posterolateral approach. 21 To obtain this, two or three pairs of ribs around the malignant tumour are excised from the vertebral body to 5 cm lateral. Pleura and large vessels are dissected away from the spine using blunt dissection (Fig. 2c). Pedicle screws (the author prefers three pairs above and below) with a safety rod are applied. Pedicles, if free from tumour, are cut at the base of the vertebral body and the posterior element is lifted en bloc. A space around the spine is created and the anterior longitudinal ligament, discs, as well as the posterior longitudinal ligament above and below the vertebral body are cut to allow removal of the body en bloc.
Successful en bloc resection has been shown to improve survival in chondrosarcoma 26 and osteosarcoma. 27 Total en bloc spondylectomy may also improve prognosis of spinal Ewing sarcoma, at least when combined with radiotherapy. 28 Neoadjuvant chemotherapy is recommended in spinal Ewing sarcoma even if the patient presents with neurological deficits, since the soft-tissue mass will typically disappear which makes the excision more effective. 12 As major complications are common, the decision of whether or not to perform total en bloc spondylectomy should be made by experienced, multidisciplinary teams. 29
Management of pelvic tumours
Hemipelvectomy involves removal of the innominate bone and can be performed internally (preserving the ipsilateral extremity; Figs 4b to 4e) or externally (hindquarter amputation; Figs 3b to 3e). External hemipelvectomy is indicated when iliac vessels (artery or vena iliaca communis or branches) and/or when nerves like the lumbosacral plexus or the sciatic nerve are involved (Fig. 3). Closure of the external hemipelvectomy is typically performed with a posterior flap using gluteal muscles and skin. If posterior structures are involved, an anterior flap along with superficial femoral artery can be applied.
Based on their location, pelvic tumour resections are typically divided into iliosacral (type 1), acetabular (type 2), ischiopubic (type 3) and including sacral (type 4), but combinations of different types are possible. 30 When the pelvic ring is disrupted, a reconstruction becomes necessary. Resection at the pelvis is anatomically complex and, therefore, navigation or patient-specific cutting guides/3D models may improve the accuracy of pelvic tumour excisions and, therefore, the prognosis of the patient (Figs 4a to 4c).31,32
Zhang et al 33 proposed a new classification of Enneking type 4 resections of the sacrum. They divided the excisions and reconstructive procedures into four subtypes (a to d) according to the extent of the tumour invasion in the sacrum. Type a includes tumour invading the ipsilateral sacral wing; type b the ipsilateral sacral foramina; type c the contralateral foramina; and type d the whole sacrum. A type a excision can be carried out with an ilioinguinal approach, while subtypes b to d require a posterior sacral approach first to sever the sacral nerve roots and to obtain an adequate surgical margin.
Reconstruction options in spinal tumours
Reconstruction after en bloc spondylectomy involves instrumented spinal fusion using pedicle screws. The gap between adjacent vertebral bodies is typically reconstructed using an allograft (Figs. 2d and 2e) or an expandable titanium cage. Reconstruction should be strong and stable enough to allow early mobilization, postoperative radiotherapy and continuation of the neoadjuvant therapy. Carbon implants are especially suited for postoperative radiotherapy. Posterior element excision (laminectomy) on a growing child requires instrumentation and spinal fusion to prevent development of postlaminectomy kyphosis. 34
Reconstruction options in pelvic tumours
External hemipelvectomy for a growing child does not require any reconstruction but will typically result in a compensatory scoliosis, which may cause pain (Fig. 3e). Treatment of compensatory scoliosis after hemipelvectomy involves a long pedicle screw instrumentation, interbody fusion with a cage and multiple rods to prevent a rod fracture. All sacral excisions will include lumbopelvic fixation with pedicle screw instrumentation. 33 When the excision also includes the acetabulum, the lumbopelvic instrumentation can be connected into a modular hemipelvic prosthesis.6,9,16
Hip joint reconstruction after internal hemipelvectomy can be performed using a megaprosthesis, a hip transposition to the sacral ala according to Winkelmann, an iliofemoral fusion or the joint may be left unreconstructed as a ‘flail hip’.6,9,16 This option also has to be discussed in palliative cases, because pelvic reconstruction after tumour resection has a high risk of major complications (30%) and long time stay in the hospital could be possible. 35
When the acetabulum does not need to be excised but the integrity of the pelvic ring is lost, a reconstruction is needed to provide stability of the spine and pelvis. Reconstruction options include lumbopelvic instrumentation with iliac or fibular autografting or massive allograft. Both autograft options can be performed as vascularized or non-vascularized.36,37 Using double fibular grafting with lumbopelvic instrumentation, Ogura et al 36 obtained bone union in five of out of eight patients but two patients developed a painful scoliosis. Extracorporeal irradiation is an option in Ewing sarcoma. 38 This includes internal hemipelvectomy and reimplantation of the extracorporeally irradiated autograft. Growth disturbance of the autograft and deep surgical site infection are limitations of this technique.
Prognosis of malignant axial bone tumours
There is very limited data on the prognosis of primary malignant bony spine tumours in children. The Surveillance, Epidemiology, and End Results database collects national cancer data from the United States. According to this database, the five-year survival rate is approximately 18% in all patient age groups with primary spine osteosarcoma and 41% in those with spine Ewing sarcoma.14,39 In a Finnish nationwide retrospective study, children with axial Ewing sarcoma had a significantly lower ten-year survival (56%) than children with a peripheral tumour location (100%). 5 Laitinen et al 4 evaluated the outcome of 113 pelvic bone sarcomas in children < 16 years of age. Ewing sarcoma accounted for 88 and osteosarcoma for 25 of these. One-third showed metastasis at presentation. The five-year survival was 37% in Ewing sarcoma and 32% in osteosarcoma. Chemotherapy response was a predictor of local recurrence in Ewing sarcoma, with the lowest recurrence rate for those with good chemotherapy response treated with a combination of radiotherapy and surgery. In children with osteosarcoma, both chemotherapy response and surgical margin influenced the local control. Patients with pelvic chondrosarcomas showed a direct correlation between survival and grading. The risk of disease-related death was 3% for grade-I tumours, 33% for grade-II tumours and 54% for grade-III tumours. Identified risk factors for impaired disease-specific survival were the resection margins and maximal tumour size. However, only a few patients in this data set were adolescents (youngest patient 15 years old). 40
It is important to monitor and compensate for limb-length discrepancy after pelvic surgery. Consistent tracking and documentation of the patients’ bilateral extremity growth is advised at regular six-month intervals. 41 Growth milestones for the acetabulum have not been well studied or documented in children, but the acetabular physis is thought to naturally reach maturity and ‘close’ at 12 to 14 years of age.
For patients with upper acetabular involvement (type IIA), reconstruction has been suggested, as this area could bear the majority of the load in the hip joint movement. 42 Implanted allografts do not grow like the other components of the acetabulum. To match the host's femoral head, the surgeons usually implanted a slightly larger osteoarticular allograft, which provided more space for the growth of the femoral head. Patients undergoing type I excision also required to avoid pelvic instability and leg length discrepancy, whereas those undergoing type III excisions did not.
The management of leg-length discrepancies can be simplified for small discrepancies that are < 2 cm through the use of a shoe lift and nonoperative management. At the time of pelvic resection, a reconstruction can be carried out in a way that effectively lengthens the limb by placing a pelvic implant or allograft that is slightly (1 cm to 2 cm) larger than preoperative length for the correction of future leg length discrepancies. This method is limited to corrections of 10 mm to 15 mm and requires precise pelvic reconstructive metrics that include the proximal femur, acetabulum and ilium.
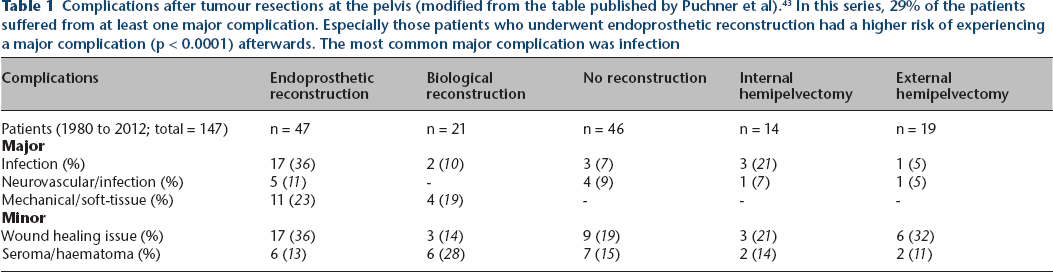
Complications are frequent after spinal or pelvic en bloc excisions.29,43 Complications reported after spinal tumour excisions include massive intraoperative blood loss (aorta, vena cava), neurological deficit, untintentional dural lesion, deep surgical site infection and nonunion with a reported range between 13% and 56%. 29 The most common complications after pelvic tumour excision include wound healing issues, deep surgical site infection and mechanical complications after endoprosthesis reconstruction (Table 1). 43
Complications after tumour resections at the pelvis (modified from the table published by Puchner et al). 43 In this series, 29% of the patients suffered from at least one major complication. Especially those patients who underwent endoprosthetic reconstruction had a higher risk of experiencing a major complication (p < 0.0001) afterwards. The most common major complication was infection
Conclusion
Night-time or non-exercise-related dull back, inguinal or buttock pain should raise the suspicion of an axial bone tumour. Neurological deficits are rare, but even with neurological deficits, neoadjuvant treatment should be started before surgery. A prompt and effective work-up is needed to confirm the diagnosis and to plan multidisciplinary oncologic and orthopaedic management. A well-planned and performed percutaneous biopsy in musculoskeletal tumour units confirms the malignancy diagnosis. En bloc excision with clear surgical margins improves the survival of children with axial bone tumours but there is only one good chance for curative treatment and, therefore, these should be left only to the most experienced orthopaedic spine and oncological surgeons. Using navigation or 3D patient-specific guides may provide more accuracy/safety for the resection in anatomically difficult locations and, therefore, can improve the prognosis for this patient. Therefore, cautious decision-making is necessary in the indication for axial reconstruction by the interdisciplinary sarcoma team. Nevertheless, a restrictive approach to reconstruction is not necessarily sensible, especially if the other option is major amputation.
Footnotes
Acknowledgements
The authors would like to thank the members of the EPOS Musculoskeletal Tumors, Infections & Arthritis Group for valuable comments during the preparation of this manuscript.
AK: Literature review, Critical revision of the manuscript.
