Abstract
The possibility of modifying the genome in mice has led to an exponential increase in the number of strains that have been developed for biomedical research. This will continue during the next few decades because international programmes plan to develop genetically-modified strains for every known mouse gene. Due to our own experiences and that of colleagues we know that the reproductive performance of many of these modified stains is impeded, despite that the modification is independent from genes that control reproduction. In some cases the spermatogenesis might be disturbed. The reason presumably lies in a defective endocrine function of the testes. This can cause reduced and/or abnormal sperm production. In livestock as well as in humans these disorders can be treated with gonadotropins. One treatment period lasts for the duration of spermatogenesis of the respective species. Up to now, nothing is known about such treatments in laboratory mice to restore or increase reproduction of genetically-modified strains. Spermatogenesis in the mouse lasts approximately 35 days. Therefore, we treated sexually mature male mice of C57BL/6 and BALB/c strains with gonadotropins for this period. The aim of this study was to test the principle suitability of such treatment for the improvement of sperm count, sperm motility, fertilization ability and reproduction.
The methods of transgenesis have led to an enormous increase in the number of genetically-modified mouse strains established for biomedical research. However not rare, besides the expected phenotype other unwanted effects are detected. These effects might be due to position effect variations. 1 Reduced fertility and sterility could be problems associated with transgenic animal models. 2 Special breeding programmes are used for breeding mouse strains with reduced fertility or strains that do not reproduce when the genetic modification is in a homozygous condition. Such strains are maintained as segregating inbred strains by mating heterozygous carrier mice. The offspring has to be genotyped to identify appropriate animals for scientific purposes as well as animals that can be used for further breeding. This is time-consuming and expensive and occupies valuable space in the animal houses. Furthermore, animals that do not carry the desired genotype have to be sacrificed, which does not adhere to the principle of animal welfare.
In our laboratory we noticed genetically-modified mouse strains with poor sperm production. Despite the fact, that superovulation of the females resulted in an increased number of oocytes, only a small portion of the oocytes were fertilized and the females did not become pregnant. The results were the same, when we used wild-type females of the respective genetic background (own, unpublished data). Therefore, we concluded that the problem was on the male's side.
In human medicine,3,4 veterinary medicine 5 and in livestock-breeding programmes gonatotropins like pregnant mare's serum gonadotrophin (PMSG) and human chorionic gonadotropin (hCG) are successfully used to treat male infertility and to enhance spermatogenesis.
PMSG is preferentially used in livestock-breeding programmes. PMSG is a glycoprotein with effects like the follicle stimulating hormone (FSH) of the pituitary gland and the luteinizing hormone (LH). FSH is produced in both genders. In females FSH stimulates the growth of follicles and in males it stimulates spermatogenesis. 6
The choriogonadotropin hCG is present in the urine of pregnant women and is successfully used in human reproductive medicine. Application to men stimulates the production of testosterone. For immature male rats Chemes et al. 7 found that short-term administration of hCG stimulates early meiosis, but causes a reduction of spermatogonia.
Both hormones, PMSG and hCG, are used for superovulation of female mice to increase the number of mature oocytes. The original protocol by Gates 8 is still in use. Superovulation is indispensible for embryocryopreservation programmes and the generation of genetically-modified mice.
In our study we administered PMSG or hCG to mature male C57BL/6 and BALB/c mice over a period of 35 days – the period of mouse spermatogenesis – with the aim to enhance sperm production and quality and thus reproduction. The strains were chosen because they are frequently used to develop genetically-modified strains. At the Central Animal Facility of the Hannover Medical School more than 60% of the genetically-modified and mutant carrying mouse strains are on the genetic background of several C57BL/6 substrains and about 7% are on a BALB/c background. The genetic background of the other strains is distributed over several inbred strains, or is mixed, and often there is no clear information available.
Material and methods
Animals
Maintenance and use of the animals were in accordance with the German Animal Welfare Legislation. 9 All experiments were approved by the local Institutional Animal Care and Research Advisory Committee and permitted by the Lower Saxony State Office for Consumer Protection and Food Safety, Department of Animal Welfare Service (reference number: Az-10/0179).
C57BL/6NCrl and B6C3F1Crl (C57BL/6N × C3H/HeN) mice were purchased from Charles River, Sulzfeld, Germany, and BALB/cJRj were purchased from Janvier, Le Genest-Saint-Isle, France at the age of 4–6 weeks. B6C3F1Crl female mice were used as mating partners for the treated males. The mice were allowed to adapt to the new conditions for two weeks. At the Central Animal Facility of the Hannover Medical School they were housed in an individually ventilated caging system in type 25 cages (BioZone, Ramsgate, Kent, UK) under standard conditions (mean temperature of 22 ± 2°C, 50 ± 10% relative humidity, 14:10 h light–dark cycle) on sterilized aspenwood granulate bedding (AsBe-wood GmbH, Ahrensfelde, Germany). They received a commercial pelletized diet (Altromin 1314 TPF, Altromin GmbH, Lage, Germany) and water ad libitum from an automated watering system. The mice were kept as sibling groups or as trios (one male with two females).
The animals had been tested according to the FELASA recommendations 10 and were free of the listed microorganisms.
Hormones
PMSG (Intergonan®, Intervet GmbH, Unterschleissheim, Germany), hCG (CG5; Sigma-Aldrich Biochemie GmbH, Hamburg, Germany) and luteinizing hormone-releasing hormone (LHRH, Sigma L4513) were diluted according to the recommendations of the manufacturer to a concentration of 1000 units in 1 mL. Aliquots of 1 mL were stored at –20°C until use.
Hormone treatment
In total, 18 male mice per group were treated with gonadotropins over a period of five weeks. This means that 18 males of C57BL6/NCrl and 18 males of BALB/cJRj were treated with PMSG, 18 males of each strain received hCG and 18 males of each strain served as controls which received only the diluting agent. Hormones and diluting agent were applied by an intraperitoneal injection (10 IU; 200 μL).
On the day of the last injection six males of every treatment group were mated to two superovulated females each of the respective strain, or to B6C3F1 females. Superovulation was induced by intraperitoneal injection of 10 IU PMSG followed by an intraperitoneal injection of 5 IU hCG 48 h later. B6C3F1 females were included, because their response to superovulation is constantly high, whereas the response differs between C57BL/6N and BALB/cJ mice (own, unpublished data).
On the following morning females with a vaginal plug (VP) (day 0.5 of pregnancy) were counted and sacrificed for embryo collection.
To evaluate the reproductive performance, another group of females received one intraperitoneal injection of 5 μg LHRH to synchronize the reproductive cycle. Four days later oestrus was assured by a vaginal smear and they were mated to males (n = 6) of each treatment group. These females were allowed to deliver. The number of litters and pups per litter were recorded.
Embryo collection and in vitro culture
For this the oviducts of plug positive females were excised on day 0.5 of pregnancy and placed in a Petri dish (ø 30 mm; Nunc GmbH + Co KG, Wiesbaden, Germany) containing 500 μL phosphate-buffered medium (PB1). 11 The cumulus–oocyte complexes were prepared from the ampulla as described by Nagy et al. 12
The oocytes were counted and were kept for further in vitro development in M16 medium (Sigma M7292). They were incubated at 37°C, 5% CO2 and 50 ± 5% humidity in four-well Petri dishes (Nunc). The number of fertilized oocytes was evaluated by counting the two-cell embryos on the next day.
Evaluation of the sperm
The hormone-treated and control males (n = 6) that had not been mated were used one day after the last hormone injection. Upon sacrifice, the epididymidis was prepared and transferred to a 1 mL reaction tube (Eppendorf) containing 500 μL HTF medium (Chemicon MR-070-D; Hofheim, Germany). The epididymidis was cut into pieces with fine scissors. The sperm was allowed to swim out from the tissue at room temperature for 15–20 min. The supernatant was used for sperm counting. The testes were removed and fixed for histological analyses.
Histopathology
The testes of three unmated males of each treatment group were prepared for histopathology. They were fixed in 10% formaldehyde and embedded in paraffin. Serial sections (3 μm thickness) were stained with haematoxylin and eosin, and examined using a light microscope.
Statistics
Sperm count, the percentage of fertilized oocytes and live born pups of the different treatment groups were compared by one-way analysis of variance.
Results
Sperm count and motility
Unmated, hormone-treated and control males (n/group = 6) were sacrificed and spermatozoa were counted as described. PMSG as well as hCG treated C57BL/6N males showed an increased sperm count. However, the increase was not significant. PMSG treatment of BALB/cJ males had no effect; hCG treatment decreased sperm count. The difference, however, was not significant. These results are summarized in Table 1.
Sperm count after hormone treatment

n/group = 6, sperm number (× 103)
PMSG: pregnant mare's serum gonadotropin; hCG: human chorionic gonadotropin
The number and the portion of motile spermatozoa increased in C57BL/6N mice after the treatment with PMSG and hCG significantly. The hormone treatment in BALB/cJ mice led to a decrease in motile spermatozoa (Table 2).
Sperm motility after hormone treatment
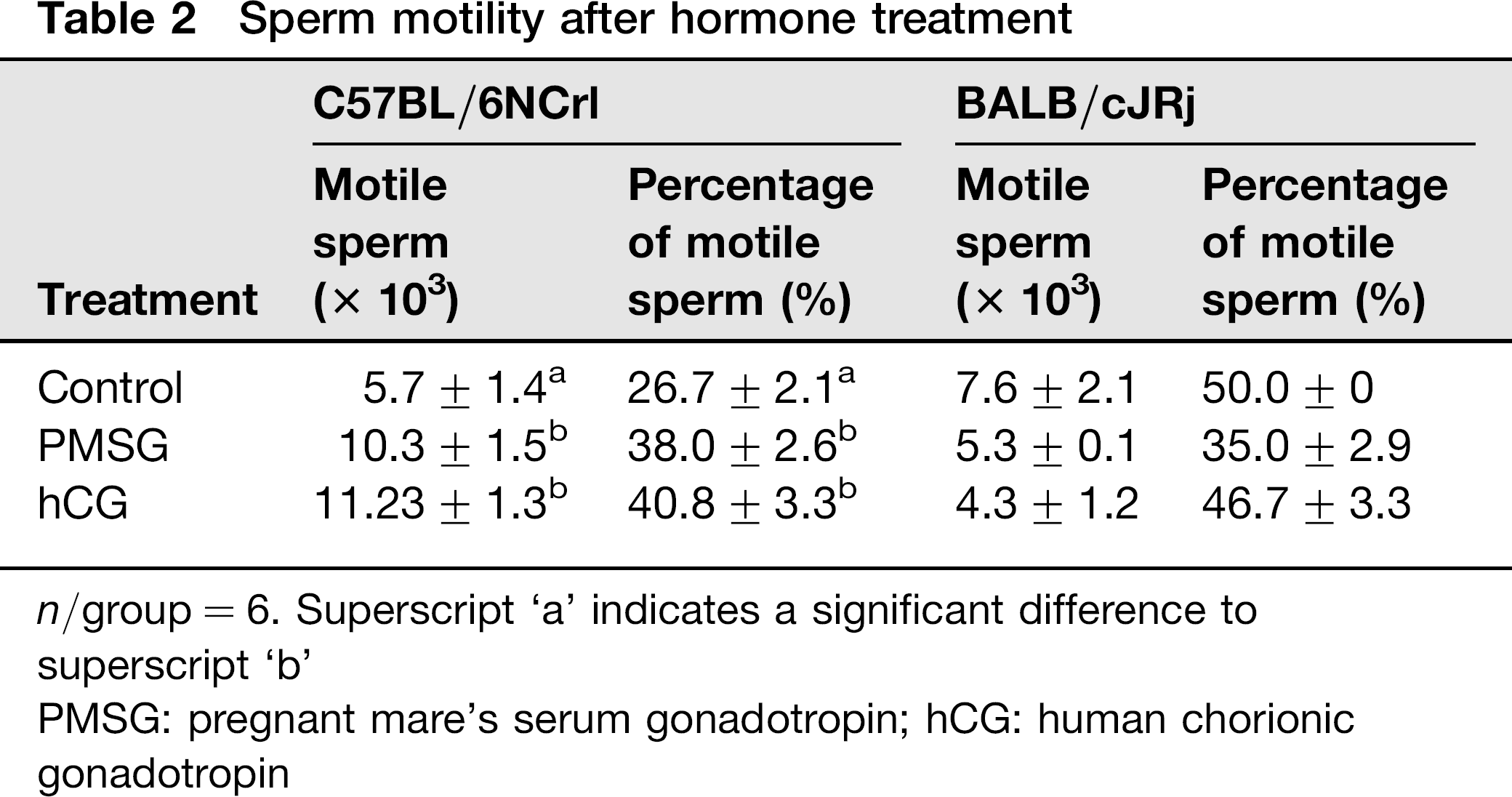
n/group = 6. Superscript ‘a’ indicates a significant difference to superscript ‘b’
PMSG: pregnant mare's serum gonadotropin; hCG: human chorionic gonadotropin
Fertilization rate, reproductive behaviour and reproduction
Hormone-treated males and control males were mated to superovulated females of the respective strain and to B6C3F1 females. Each male was mated to two females. The following morning the females were checked for the presence of a VP. The number of females with a visible VP was increased in the PMSG treatment group for both strains, irrespective of whether the females were from the same strain or B6C3F1.
In the hCG treatment group the number of VP-positive females increased, if male and female were from the same strain. Fewer B6C3F1 females had a VP when they were mated to hCG-treated C57BL6N males, whereas more B6C3F1 females had a VP when they were mated to hCG-treated BALB/cJ males. In no case the differences were significant compared with the controls (Table 3).
Percentage of females with a visible copulatory plug on the morning after mating
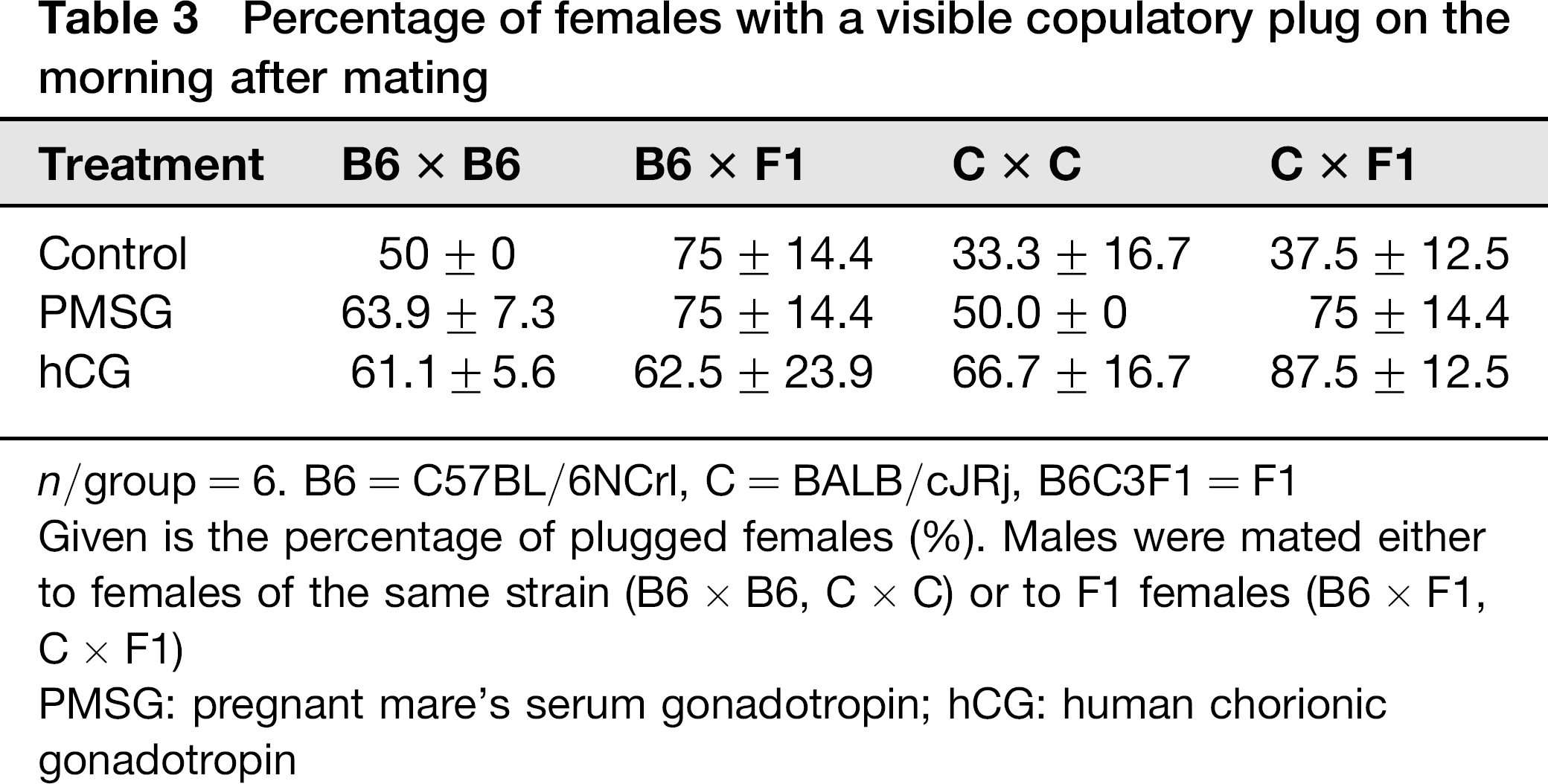
n/group = 6. B6 = C57BL/6NCrl, C = BALB/cJRj, B6C3F1 = F1
Given is the percentage of plugged females (%). Males were mated either to females of the same strain (B6 × B6, C × C) or to F1 females (B6 × F1, C × F1)
PMSG: pregnant mare's serum gonadotropin; hCG: human chorionic gonadotropin
Females with a VP were sacrificed to collect oocytes. The oocytes were taken into culture overnight to evaluate their developmental capacity. In C57BL/6N the treatment with either hormone increased the number of fertilized oocytes, independent of the genetic background of the female mating partner.
PMSG treatment of BALB/cJ males resulted in an increase in the fertilization rate, if they were mated to B6C3F1 females, but in a decrease if they were mated to BALB/cJ females. The hCG treatment of BALB/cJ males resulted in an increase of the fertilization rate, irrespective of the strain of the females. However, the differences were not significant. Table 4 summarizes the results.
Fertilization ability
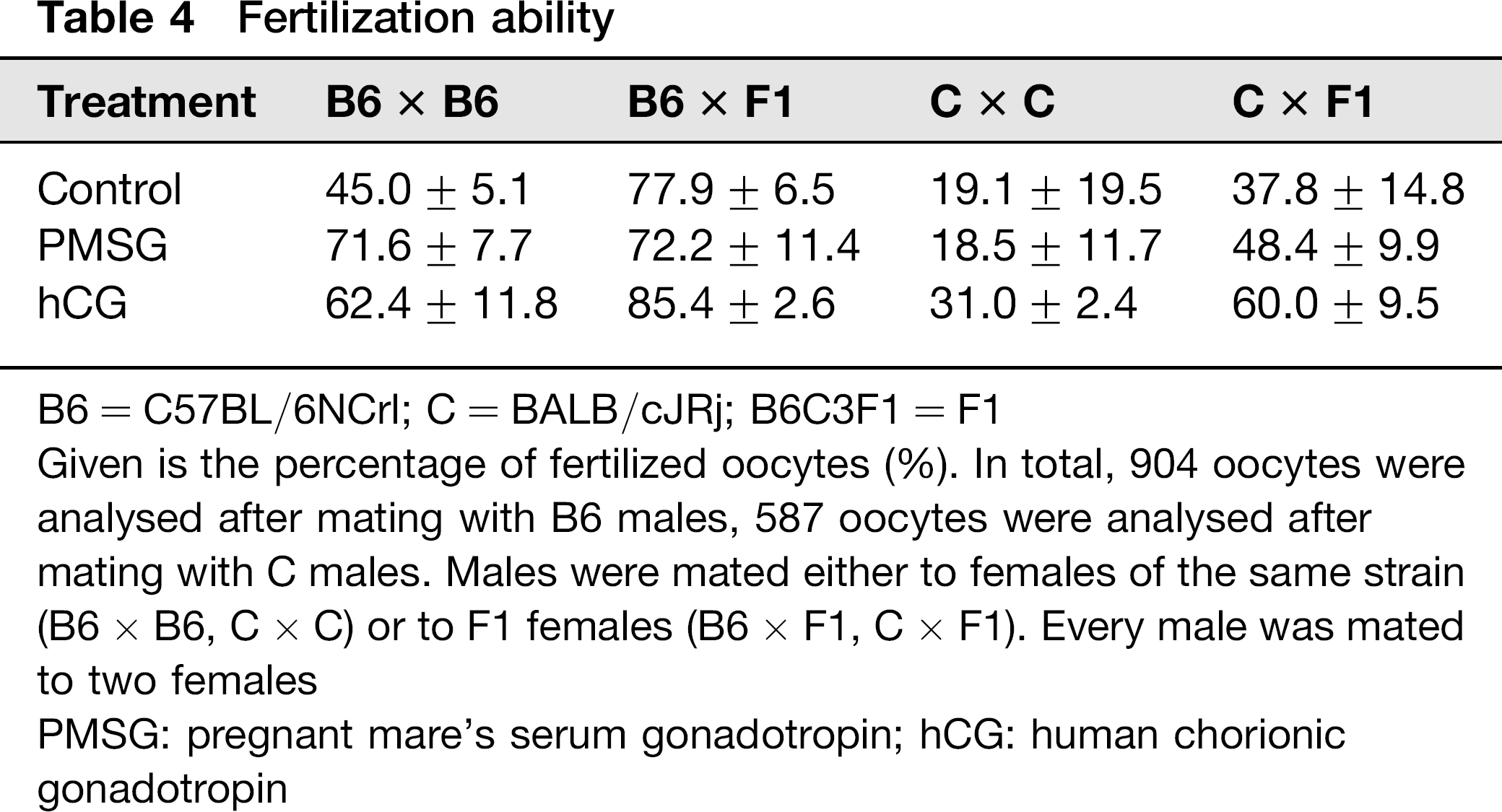
B6 = C57BL/6NCrl; C = BALB/cJRj; B6C3F1 = F1
Given is the percentage of fertilized oocytes (%). In total, 904 oocytes were analysed after mating with B6 males, 587 oocytes were analysed after mating with C males. Males were mated either to females of the same strain (B6 × B6, C × C) or to F1 females (B6 × F1, C × F1). Every male was mated to two females
PMSG: pregnant mare's serum gonadotropin; hCG: human chorionic gonadotropin
Pregnancies, weaned offspring and pups per litter
To synchronize the reproductive cycle, B6C3F1 females received an injection of LHRH. Four days later they were in oestrus and mated either to hormone-treated or control males for one week. Each male (n = 4/treatment group) was mated to two females. In C57BL/6N males hormone treatment did not change the number of pregnant females; however, the litter size increased for both hormones. The increase was not significant. Five out of eight females got pregnant and weaned their litter in the PMSG as well as in the hCG and control group.
In BALB/cJ the number of pregnant females as well as the litter size decreased in the hormone-treated group. Again, the decrease was not significant. In the control group all females got pregnant, in the PMSG group five females and in the hCG group seven of eight females, respectively, got pregnant. All litters were weaned. Table 5 summarizes these results.
Litter size

Males: n/group = 4, each male was mated to two B6C3F1 females for one week. Given is the average number of pubs born per litter
PMSG: pregnant mare's serum gonadotropin; hCG: human chorionic gonadotropin
Histology
In Figure 1 the testis of hormone-treated animals is compared with the testis of an untreated male. All stages of spermatogenesis are present in the control male as well as in both treatment groups. Also the numbers of spermatogonial stages are similar to the control as well as to the treatment groups. No differences could be found.
Histopathological evaluation of spermiogenesis in hormone-treated and control male mice. Shown here are cross-sections though the testis of C7BL/6N mice. (a) Untreated male, (b) hCG-treated male and (c) PMSGtreated male. No differences in spermatogonial stages and numbers could be detected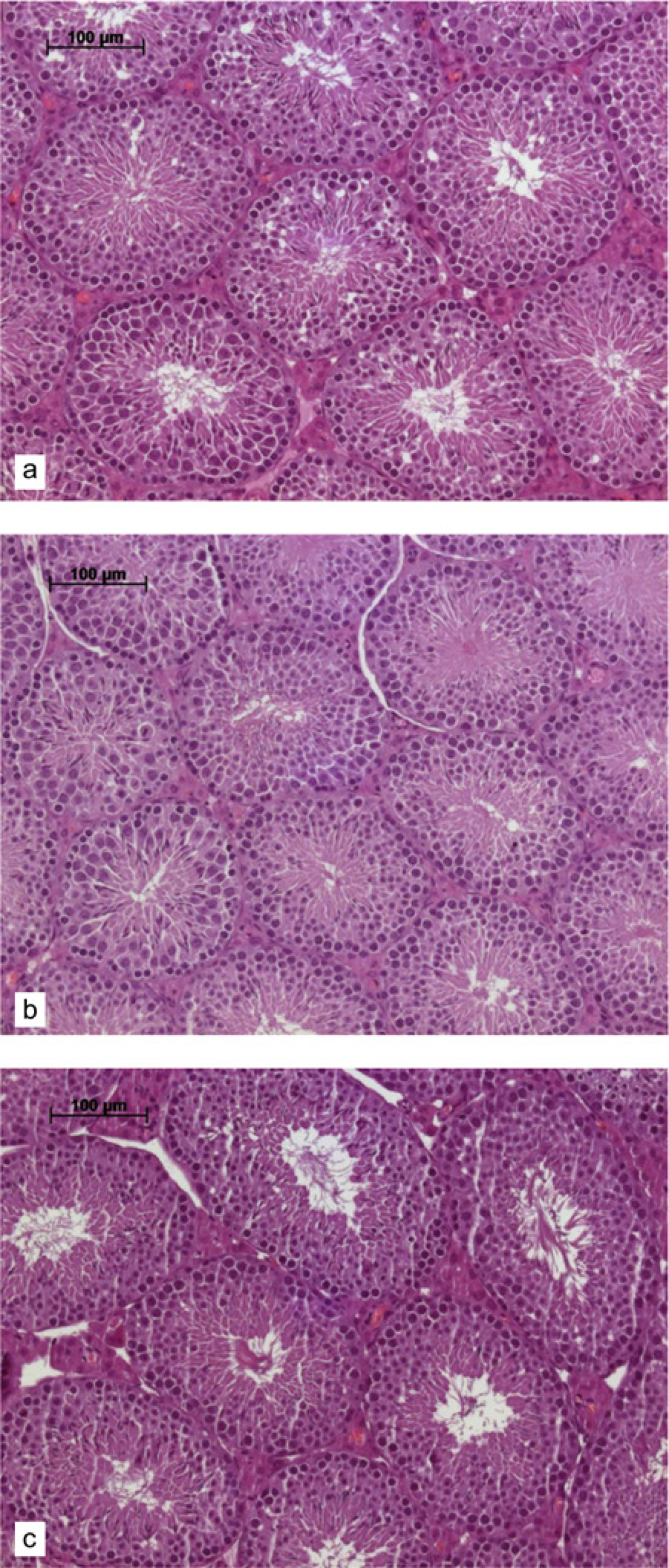
Comparative evaluation of the results
Figure 2 summarizes the results of the measured parameters. For all parameters that were analysed, the hormone treatment of the males showed an effect, although not significant for most of them. The effects of the hormones on tested parameters differed between the two strains included in this study. For C57BL/6N both hormones showed a slightly positive effect, except for the pregnancy rate, which was not influenced by the treatment. In BALB/cJ the hormones had a slightly negative effect on four of the six analysed parameters (Figure 2).
Comparative evaluation of the results: C57BL/6N = B6; BALB/cJ = C; VP = vaginal plug; ↗ increase; ↘ decrease; → no effect. *Effect depends on whether the males were mated to females of the same strain or to B6C3F1 females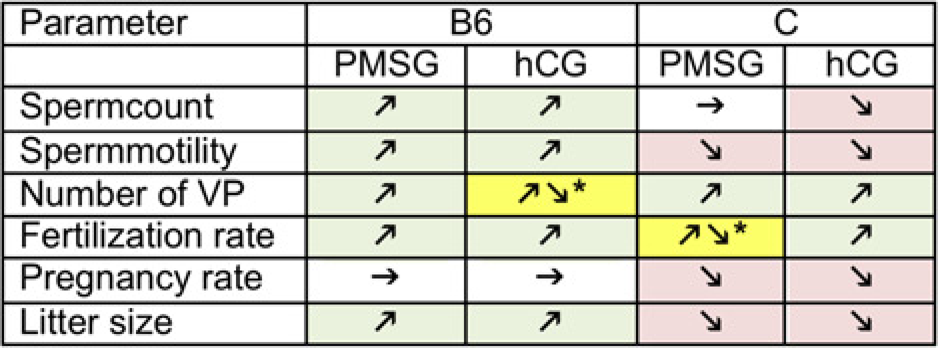
Discussion
It is not rare that the phenotype of a new genetically-modified or mutant-carrying mouse model differs from what is expected. General explanations might be position effect variations or incomplete backcrossing to the strain selected. If the gene is co-expressed in other tissues than the target tissue this might cause pleiotropic phenotypes and might also effect reproduction. 13 This holds true also for strains that have been established by intranuclear injection of DNA.
For our institutional embryo cryopreservation programme of genetically-modified and mutant-carrying strains, we use males homozygous or heterozygous for the gene of interest for timed mating to wild-type females of the respective genetic background as is suggested by the Jackson Laboratory (‘speed embryo cryopreservation’). 14 These females are used as embryo donors. The resulting embryos for cryopreservation are heterozygous, if homozygous males were mated, or heterozygous and wild type if heterozygous males were mated. Particularly, for some strains with the genetic background of C57BL/6N and BALB/cJ we noticed that a great number of the oocytes were not fertilized. The oocyte number was in the range expected (data not shown). Therefore we conclude that the problem was due to a reduced sperm production or quality. This was definitely expected for BALB/c, as this strain is known to have more sperm abnormalities than others. 15
Gonadotropins like FSH and LH are not only important for ovary function but also for testicular function. Therefore, gonadotropins are used in human medicine and in livestock to enhance spermatogenesis. Schuenemann et al. 16 and Bagu et al. 17 suggested that gonadotropin administration increases the number of germ cells in the testis of bull calves. Lately O'Shaughness et al. 18 confirmed that FSH in mice stimulates spermatogenesis through an increase in spermatogonial number and entry of these cells into meiosis.
Encouraged by these data, we tried to enhance sperm count and quality in mouse strains that are predominantly used for the generation of genetically-modified mice.
Our results showed that hormone treatment of male mice had an effect. The results differed for the two strains tested. C57BL/6NCrl mice showed a slight benefit from the treatment in the tested parameters. The number of pregnant females after mating, however, remained constant in this strain. In BALB/cJRj mice the treatment had negative effects for four of the six measured parameters; these effects were not significant. The results could be explained by the fact that we used wild-type animals instead of genetically-modified strains in our study. The used strains are known to be reasonably productive and effects of the hormone treatment might not be as conductive as with genetically-modified strains with a diminished reproductive performance. Another explanation might be that we did not find the optimal treatment regimens. Possible parameters that could influence the effect of hormone treatment are the dosage of the hormones or the age of the males. Nevertheless, our results demonstrate that treatment of male mice with gonadotropins influences the reproduction rate and fertilization ability. The genetic background obviously influences the effect of such treatment. We claim that hormone treatment can enhance male fertility in some strains. These strains have to be identified by further studies.
