Abstract
Short-term storage of embryos at low temperature induces developmental arrest of the embryo and would appear to be a valuable aid in embryo-transfer techniques to avoid wasting embryos. Embryo storage at 4°C was examined to allow synchronization with embryo-transfer recipients using the microinjection technique. Superovulation was induced in female Japanese White donor rabbits four days before mating with males. At the same time, control recipients were injected with human chorionic gonadotropin (hCG) to allow synchronization (R1); the hCG injections were delayed by 24 h in the experimental group (R2). DNA constructs for expressing human C-reactive protein or apolipoprotein AII were microinjected into the male pronuclei of the ova. The microinjected embryos were immediately transferred to recipients (R1) or stored at 4°C in phosphate-buffered saline containing 10% fetal bovine serum. After 17–20 h, the stored embryos were incubated at 37°C for one hour, and the morphologically normal embryos were transferred to recipients (R2). In the R1 rabbits, 855 embryos were transferred to 29 recipients, and 72.4% of the recipients became pregnant. Seven of the 84 offspring were transgenic. In the R2 rabbits, 478 embryos were transferred to 16 recipients, and 62.5% of the recipients became pregnant. Two of the 39 offspring were transgenic. There were no differences in pregnancy rate, litter size and transgenic integration rate between R1 and R2. These results suggest that the short-term 4°C storage of microinjected embryos can be a valuable method for synchronization with recipients, and reducing wastage of embryos and the sacrifice of rabbits.
Rabbits have been employed as therapeutic protein bioreactors and human disease models, particularly in studies of lipid metabolism and cardiovascular disorders, owing to their body size and because their lipid metabolism characteristics resemble those of humans. A number of transgenic rabbit strains have been established1–4 since the first transgenic rabbit was created by Hammer et al. 5 in 1985. Though genetic engineering techniques in rabbits lag behind those in mice, novel methods, like iPS cell establishment 6 and the zinc finger nuclease technology, 7 were adapted and in future time make possible to create much needed rabbit models for translational research.
The conventional microinjection technique is still the practical choice for creating transgenic rabbits. 8 Since transgene integration efficiency with the microinjection technique has not improved remarkably,9–11 it is necessary to obtain a number of quality embryos to create transgenic rabbits. To recover as many ova as possible, superovulation is induced in donor rabbits with pregnant mare's serum gonadotropin (PMSG) or follicular stimulating hormone (FSH). PMSG is preferable to FSH (six shots at 12 h intervals) owing to its convenient application (a single shot) and a donor rabbit can yield on average 20–30 ova. 8
The recipient rabbits have to be prepared with an injection of human chorionic gonadotropin (hCG) one day before embryo transfer to be synchronized with the donors’ reproductive cycle. 8 The microinjected embryos are transferred to the recipient rabbits at the most efficient rate (20–30 embryos for a unilateral uterus), and the prepared recipients do not always match the number of embryos to be transferred since some donor rabbits show a hypersensitive (over 100 ova from one rabbit) response to PMSG or no response at all. However, a lack of recipients leads to wastage of embryos and the sacrifice of rabbits. Therefore, methods for storing and temporarily arresting embryo development would be valuable to spare and reuse the microinjected rabbit embryos in the absence of synchronized recipients.
Cryopreservation is a candidate for embryo storage. 12 However, embryo cryopreservation demands rather complex (i.e. vitrification and warming) procedures and is more appropriate for long-term storage. Furthermore, the pronuclear-stage embryos used for microinjection do not lend themselves well to cryopreservation, and a significant loss of viable embryos often results. 13
In an early study, Chang 14 successfully stored fertilized rabbit ova at a low temperature for several days. Later, Hughes and Anderson 15 established a simple method for storing rabbit embryos under conditions that could be easily maintained in an ordinary refrigerator set at 4°C. It has been reported that storing fertilized mouse embryos at 4°C initiates temporary developmental arrest, with the cessation of such molecular events as DNA replication, cytoplasmic polyadenylation of maternal mRNAs, mRNA degradation of a cell cycle-related gene and elevated mRNA expression of zygotic gene activation-related genes. 16 It has been demonstrated that 4°C-stored embryos recommence normal development when returned to 37°C in a 5% CO2 atmosphere; 15 the embryos retain viability and the embryos can be synchronized with later recipients.
In the microinjection technique, the embryos are damaged to some degree by the insertion of the glass capillary into the cytoplasm and nucleus, 17 and damaged embryos may not respond well to storage at low temperature. Thus far, there have been no reports on short-term storage at low temperature in the context of microinjection techniques. The present study describes the short-term storage of embryos at 4°C for embryo transfer after microinjection with recipients with delayed synchronization.
Materials and Methods
Animals
Japanese White rabbits (std: JW/CSK) were purchased from SLC, Inc (Hamamatsu, Japan). All rabbits were kept separately in a room with the temperature and humidity maintained at 24 ± 2°C and 55 ± 15%, respectively. The rabbits were submitted to a regular 12 h light/dark cycle and were given tap water and a standard rabbit chow diet (CRB-1, CLEA Japan, Inc, Tokyo, Japan) at approximately 120 g/day, then ad libitum during the perinatal and nursing periods.
All animal experimental protocols were performed with the approval of the Animal Care Committee of Saga University and conformed with the Guide for the Care and Use of Laboratory Animals issued by the National Institutes of Health.
Recovery of ova from donor rabbits
On the first day, each donor female JW rabbit was intramuscularly injected with 150 units (U) of PMSG (Serotropin, ASKA Pharm, Tokyo, Japan) to induce superovulation. On the fourth day, the donor rabbits were mated with males or artificially inseminated, followed by intravenous injection of 100 U of hCG (Gonatropin, ASKA Pharm), as shown in Figure 1. After mating for 17–19 h, the donor rabbits were sacrificed (at the age of 18–65 weeks) using an anaesthetic overdose (thiamylal sodium; Kyorin Pharm, Tokyo, Japan) and the oviducts were extracted. The ova were recovered by flushing the oviducts with an M2 medium
15
and transferred to an M199 medium (Sigma-Aldrich, St Louis, MO, USA) containing 20% fetal bovine serum (FBS; Sigma-Aldrich) and 0.1 g/L Experimental protocol for hormone-induced superovulation and timed events of transgenic rabbit production. R1: recipients transferred with embryos following after microinjection; R2: recipients transferred with embryos stored at 4°C for 17–24 h after microinjection. PMSG: pregnant mare's serum gonadotropin, hCG: human chorionic gonadotropin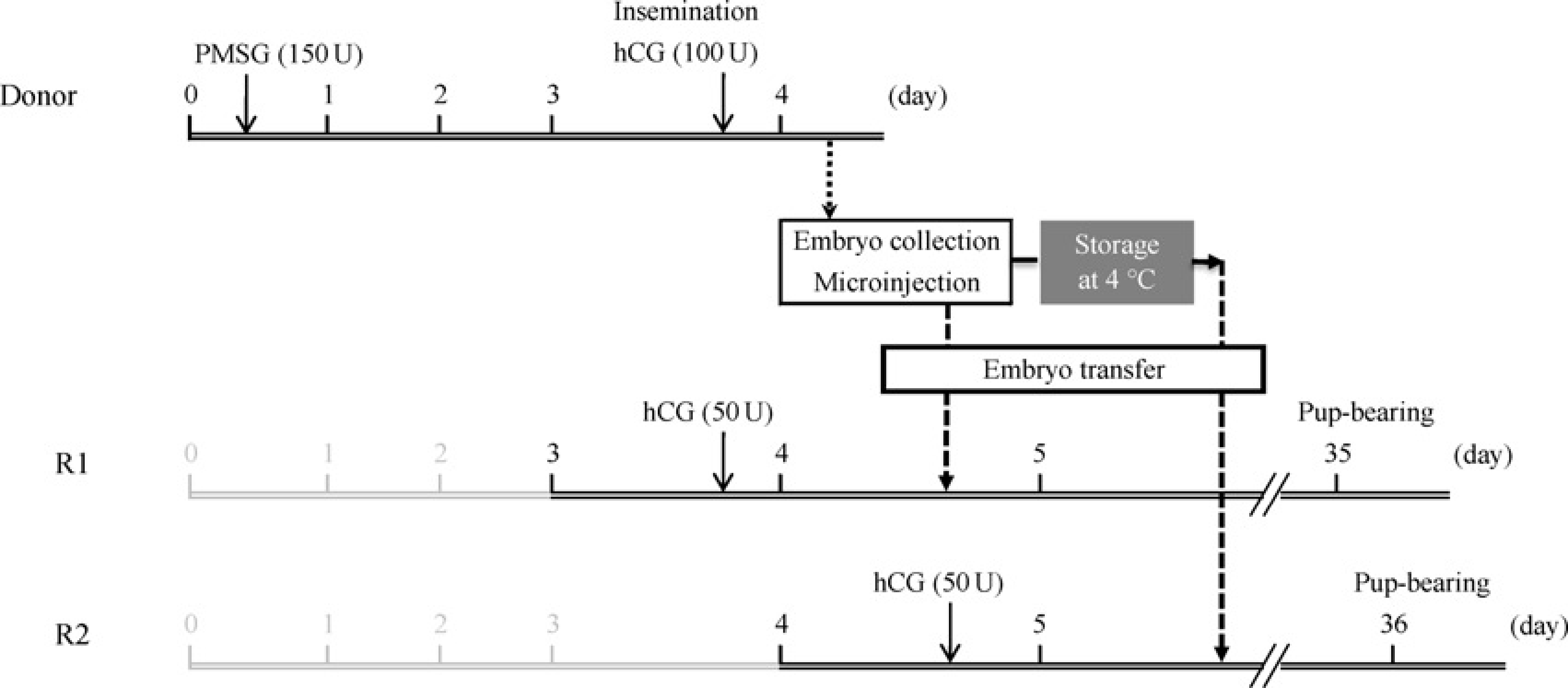
Preparation of recipient rabbits
The control recipient JW rabbits were intravenously injected with 50 U of hCG on the fourth day to allow synchronization with the donors (Recipient 1, R1). The recipients in the experimental group were injected with hCG 24 h after R1 (Recipient 2, R2), as shown in Figure 1.
Embryo manipulation
Complementary DNA for expressing human C-reactive protein (CRP) under the control of liver-specific expression elements from the human apolipoprotein E gene 18 (1.13 kb) or 3 kb human apolipoprotein AII (apoAII) genomic fragment, 19 with four copies of chicken β-globin insulator, 19 was microinjected into male pronuclei of the fertilized ova using the standard procedure previously described.7,20 The microinjected embryos were divided into two groups. The embryos in the control group were incubated for 1–3 h after microinjection and then transferred to the unilateral oviduct of R1 rabbits following the standard procedure (Figure 1).8,21 The other embryos were stored in phosphate-buffered saline (PBS) containing 10% FBS, 5 mmol/L lactic acid and 0.25 mmol/L sodium pyruvate (PBS+) at 4°C for 17–20 h after microinjection. The stored embryos were subsequently incubated at 37°C with 5% CO2 for one hour. Morphologically intact embryos were selected and transferred to R2 rabbits (Figure 1).
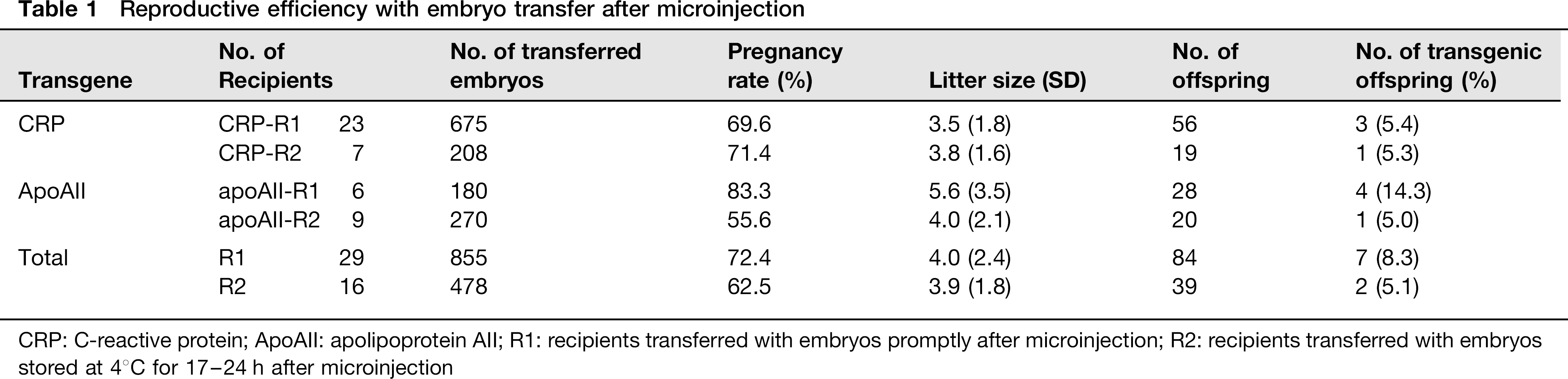
CRP-expressing DNA construct was injected into 883 ova, and 675 were transferred to 23 R1 (CRP-R1, 29.3 ± 3.7 embryos/head) and 208 to seven R2 rabbits (CRP-R2, 29.7 ± 1.8). In all, 450 ova were microinjected with apoAII-expression DNA construct; 180 embryos were transferred to six R1 (apoAII-R1, 30 embryos for each) and 270 were transferred to nine R2 (apoAII-R2, 30 for each) rabbits (Table 1).
Reproductive efficiency with embryo transfer after microinjection
CRP: C-reactive protein; ApoAII: apolipoprotein AII; R1: recipients transferred with embryos promptly after microinjection; R2: recipients transferred with embryos stored at 4°C for 17–24 h after microinjection
Analysis of offspring
The numbers of pregnant recipients and offspring were counted upon delivery, which was around the 30th day after embryo transfer. The genomic DNA was extracted from a piece of tissue from the offspring, and successful integration was analysed using the polymerase chain reaction (PCR). A pair of primers was designed to detect human CRP (sense, 5′-CAGAACTCCACGATCCCTGA-3′; antisense, 5′-GGGGTCTAAGACTGCATGAAC-3′) or apoAII (sense, 5′-GTGGAGAGCCTGGTTTCTCA-3′; antisense, 5′-TGTGTTCCAAGTTCCACGAA-3′); the corresponding PCR product lengths were, respectively, 550 and 595 bp. The PCR program comprised predenaturation at 94°C for 5 min, 35 cycles of 94°C for 30 s, 60°C (CRP) or 58°C (apoAII) for 30 s and 72°C for one minute; this was followed by a final elongation at 72°C for 10 min. The reaction products were analysed using electrophoresis on agarose gels (Figure 2).
Typical examples of polymerase chain reaction genotyping. Characteristic bands for human CRP (a) or apoAII (b) transgene are revealed within DNA construct used for microinjection (pC) and genome DNA from transgenic (Tg) but not non-transgenic (–) rabbit
Statistical analysis
We employed Student's t-test for the number of transferred embryos and litter size and Pearson's χ2 test for pregnancy and transgenic integration rates to test for significant differences (P < 0.05) between R1 and R2 or CRP and apoAII recipient rabbits using PASW Statistics 18.0 (IBM, Armonk, NY, USA).
Results
In total, 1500 ova obtained from 68 donor rabbits were microinjected. After 1–3 h incubation, 96.8% of the microinjected embryos were morphologically intact and were used in subsequent procedures. The 1333 microinjected embryos – predominantly single-cell and two-cell stages – were transferred to R1 and R2 rabbits.
The pregnancy rates of the CRP-R1 and CRP-R2 rabbits were 69.6% and 71.4%, respectively. The litter size (SD) of CRP-R1 was 3.5 (1.8) and that of CRP-R2 was 3.8 (1.6). The pregnancy rates of apoAII-R1 and apoAII-R2 rabbits were 83.3% and 55.6%, respectively (Table 1). The litter size of apoAII-R1 was 5.6 (3.5), and that of apoAII-R2 was 4.0 (2.1).
The total pregnancy rate of R1 rabbits was 72.4% and that of R2 was 62.5%; the litter sizes of R1 and R2 were 4.0 (2.4) and 3.9 (1.8), respectively (Table 1). There were no significant differences between R1 and R2, either in terms of pregnancy rate or litter size.
Three of 56 (5.4%) offspring from CRP-R1 and one of 19 born from CRP-R2 (5.3%) rabbits were transgenic for the human CRP gene, and four of 28 (14.3%) offspring from apoAII-R1 and one of 20 from apoAII-R2 (5.0%) rabbits were transgenic for the human apoAII gene (Table 1).
Discussion
The present study showed that a number of offspring were obtained from R2 as well as R1 rabbits, and there were no significant differences between R1 and R2 or CRP and apoAII in pregnancy rate and litter size. Therefore, the microinjected rabbit embryos stored at 4°C for 17–20 h retained their full viability, and 4°C storage of embryos is certainly a viable option for synchronization with later recipients in embryo transfer following microinjection.
It is known that mammalian embryos suspend development when maintained in vitro below normal body temperatures; 14 temperatures between 0 and 10°C are adequate for short-term storage of mammalian ova to achieve arrested development.22–25 It is important to define the conditions that allow fertilized ova to retain their full viability during storage in vitro. Storage conditions for embryos have been investigated in various mammalian species.22–25 Chang 14 successfully stored fertilized rabbit ova at 0 and 10°C for four days; this method was modified to allow easier embryo storage in an ordinary 4°C refrigerator by Hughes and Anderson. 15
PBS with supplementations has commonly been used as the medium for storing ova at low temperatures.15,22,26 In the present study, PBS supplemented with 10% FBS, 5 mmol/L lactic acid, and 0.25 mmol/L sodium pyruvate was chosen as the low-temperature storage medium following the storage medium based on earlier data by Hughes and Anderson 15 in rabbits. Our results are in agreement with earlier data that the medium was adequate for low-temperature storage of rabbit embryos since the reproductive outcome with 4°C-stored embryos was not inferior to that of controls.
In the microinjection technique, DNA solution is injected into the male pronuclei of the fertilized ova at the pronuclear stage 17–19 h after mating. At that stage, the ova are located in the distal part of the oviduct.27–29 Ideally, the microinjected embryos at an early developmental stage (single-/two-celled) are transferred to the same part of the oviduct from which the ova were recovered. Microinjected embryos have been transferred by injection using a catheter inserted approximately 15–20 mm into the oviduct via the fimbria to achieve localization of the single-/two-celled fertilized embryos.27–29 Embryos stored at 4°C have also been single-/two-celled even after 17–20 h storage, presumably because their development was temporarily arrested by the low temperature.15,16 The same procedure of embryo transfer was applied to both R1 and R2 rabbits in the present study. Though successful vitrification of pronuclear rabbit embryos has been reported, 30 it has also been suggested that further refinement of the vitrification procedure for rabbit embryos earlier than the eight-cell stage is required to improve their cryotolerance. 13 Storage at 4°C is more appropriate for short-term storage of early-stage embryos owing to its high viability and simplicity of procedure.
The temporary developmental arrest was terminated by re-incubation at 37°C with 5% CO2, and the embryos recommenced normal development, as has been reported in other species.22,31,32 Transgenic efficiency is known to be affected by DNA construct; 11 apoAII gene tends to be integrated more efficiently than CRP, though we found no difference between CRP and apoAII in the number of transgenic offspring (Table 1).
To avoid inducing drug tolerance by repeated hCG treatment, the conversion of non-used delayed recipients into donors or mating with vasectomized bucks would be preferable for efficient use of experimental animals.
The short-term storage of embryos at 4°C to arrest development in vitro is effective in successful embryo transfers after microinjection so as to allow synchronization with later recipients. Using 4°C-stored embryos to create transgenic rabbits can improve experimental efficiency by avoiding wastage of embryos recovered from donor rabbits and reducing the number of animals sacrificed.
