Abstract
Embryo transfer of pre-implantation embryos to surrogate dams is a key technique for the hygienic sanitation of strains, cryopreservation, in vitro fertilization, genetic modification and engineering. However, the effects of several parameters, such as the number of transferred embryos, on the success of embryo transfer are not well studied. In this retrospective study, we reanalysed 1320 embryo transfers of two-cell embryos originating from genetically altered donors, which were performed under routine conditions in our facility over a period of 5 years. Of them, 453 embryo transfers were done with freshly collected embryos and 867 transfers were performed with cryopreserved embryos. Despite the fact that the genetic background of the embryo donors was quite heterogeneous, we found that the transfer of ≥ 21 embryos reduced the success of embryo transfers for freshly collected embryos in correlation with the number of pregnancies and born pups, whereas this was not the case for transfer in the cryopreservation group. Most pregnancies were achieved after embryo transfer of 10–20 freshly collected embryos (90.4%), which dropped to 37.5% if more embryos were transferred. The highest pregnancy rates in the cryopreservation group were achieved if 15–17 embryos were transferred (62.9%). Despite the fact that the precise substrains were only rarely defined, we confirmed that beside the number of transferred embryos, the genetic background of the donors had an influence on the success of embryo transfer. Significantly more embryos in a C57BL/6 background developed to term than embryos on a BALB/c background.
Introduction
Efficient transfer of murine pre-implantation embryos into pseudo-pregnant surrogate dams is an indispensable technique in artificial reproductive work in rodents. Embryo transfer is a basic step for the rederivation of hygienically contaminated strains, the revitalization of cryopreserved strains, the generation of genetically modified mice and for in vitro fertilization.
It is well accepted that the development of transferred embryos to term depends on the donor strain. 1 Embryos of some inbred strains are less viable or more sensitive, whereas embryos of hybrid or outbred stocks have a much better developmental rate. 2 It has been demonstrated that freshly collected embryos show a better developmental rate than cryothawed embryos. The reason seems to be ‘freezing injuries’ as described by Pegg and epigenetic modifications as described by Gardner and Lane.3,4 Early stages of mammalian embryos react very sensitively to microenvironmental conditions, which may affect their developmental capacity and result in long-term effects. 5
The embryos from genetically modified mice are often less viable than those of their genetic background. This might be due to unknown side effects of genetic manipulation. 6
Transgenic mouse strains are distributed all over the world and very often lack information about the strains of origin, number of backcrosses to a certain background strain or the used substrains. This missing information might have an influence on the accurate calculation of the optimal number of embryos that have to be transferred to produce viable offspring.
A lot of experience is needed to transfer the ideal number of embryos to achieve reasonable litter sizes.
Whenever possible, the genetic background and kind of embryo manipulation is taken into account for the calculation of the number of embryos that should be transferred to one surrogate dam. However, in practise, the number of transferred embryos often depends on the availability of pseudo-pregnant surrogate dams and the number of transferrable embryos.
To find out if a more generalized predication about the optimal embryo number for transfer can be given, we analysed the outcome of embryo transfers over a time period of 5 years. The transfers were performed in the reproduction unit of the Central Animal Facility of the Hannover Medical School. Here, we analysed the outcome of 453 embryo transfers with 5150 freshly collected and 867 transfers with 8336 cryothawed mouse embryos. The embryos originated from 114 genetically altered strains.
Material and methods
Animals and husbandry
All procedures described in this retrospective study were performed at the Central Animal Facility of the Hannover Medical School (ZTL), Hannover, Germany.
The mice were maintained in individually ventilated caging systems (floor area 440 cm2, BioZone; or 525 cm2, Allentown) under controlled standardized conditions: temperature 21 ± 2 ℃, relative humidity 50 ± 5% and artificial light (14 h light, 10 h dark; light on at 6:00 a.m.). Sterilized commercial softwood granulate bedding was used (Lignocel, Altromin; Lage, Germany). The mice received a commercial pellet diet (Altromin TPF-1314) and autoclaved water ad libitum.
We used (BALB/cHanZtm × DBA/2JHanZtm)F1 females as surrogate dams for embryo transfer (further abbreviated as CD2).
The microbiological status of the CD2 breeding colony was examined as recommended by Federation of Laboratory Animal Science Associations (FELASA) and the mice were free of the listed microorganisms. 7
All embryos were collected in-house from genetically altered strains that were imported to the ZTL from other facilities or that were transferred in-house to a higher hygienic barrier. The strains passed our rederivation programme with and without cryopreservation of two-cell embryos to achieve the microbiological status of our breeding barriers. There had been no other manipulations on the embryos.
In many cases, the genetic background was not clearly defined. That meant a lack of information about the strain of origin, the substrains used and/or number of backcrosses. Figure 1 gives a rough overview of the genetic backgrounds of the strains involved in this study and their proportion. Thus, more detailed denomination of the involved background strains was not possible.
Distribution of the background strains of embryo donors. The genetic backgrounds were not well defined, thus more detailed denomination was not possible. ‘B6’ can be either one of the C57BL/6 sublines ‘J’ or ‘N’, or a mixture of several sublines (it is same for all the other strains).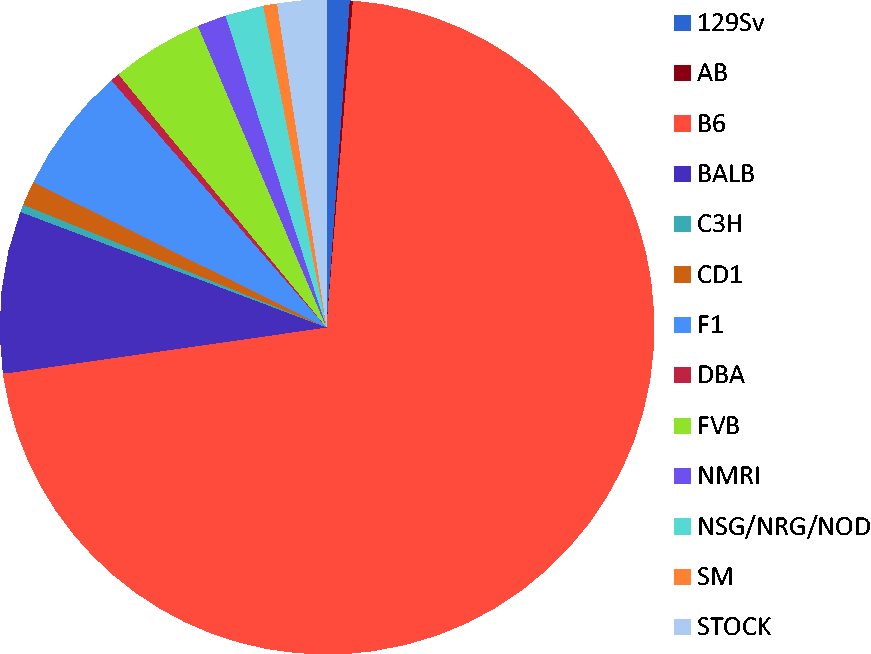
The data presented here are from routine two-cell embryo transfers carried out at the ZTL over a period of 5 years (the participating surgeons were the same).
All procedures were carried out in accordance with German animal welfare legislation, and approved by the local Institutional Animal Care and Research Advisory Committee and permitted by the Lower Saxony State Office for Consumer Protection and Food Safety (reference number 33.19-42502-05-13A404 and 42500/1H)
Embryo collection and evaluation
Six to eight-week-old females of the donor strains were superovulated by injection of 7 IU pregnant mare's serum gonadotropin (PMSG) (Intergonan, Vemie Veterinaer Chemie GmbH, 4152 Kempen, Germany), followed by an injection of 7 IU human chorionic gonadotropin (hCG) (Ovogest, MSD, Germany) 48 hours later and mated immediately. Plug-positive females were killed at day two of pregnancy after CO2 anaesthesia by cervical dislocation. The oviducts were excised, and the embryos were flushed out with phosphate buffer 1 (PB1) and washed several times with fresh PB1. 8
4Morphological integrity was evaluated under a light microscope (Stemi 2000, Zeiss) with 300× magnification. Morphologically intact embryos with two distinct blastomeres and an intact zona pellucida were used directly for embryo transfer, or for cryopreservation without post-harvest or post-thaw culture.
Cryopreservation and thawing of embryos
Embryos were frozen via a modified two-step procedure with 1.5–2 M 1,2-propanediole (PROH, Sigma P1009) as cryoprotectant in PB1 as described previously. 9
For thawing, the cryocontainer was taken from the storage tank and placed at room temperature. After melting, the embryos were flushed into a 35 × 10 mm Petri dish and fresh PB1 was added immediately to dilute the cryoprotectant. The embryos were washed several times with fresh buffer to entirely remove the cryoprotectant.
Embryo transfer
To induce pseudo-pregnancy, CD2 females were mated to vasectomized males with proven sterility. The next morning, plug-positive females were considered as day 0.5 pseudo-pregnant and used as surrogate dams.
Embryo transfer was performed under general anaesthesia achieved by intraperitoneal injection of a mixture of 80 mg/kg ketamine HCl 10% (Ketanest®, Bayer Leverusen, Germany) and 2 mg/kg xylazine HCl 2% (Rompun®). As soon as the toe pinch reflex had disappeared, 0.7 mg/kg meloxicam (Metacam®, 2 mg/ml, Boehringer Ingelheim) and 0.05 mg/kg atropin (Atropinsulfat, 0.5 mg/ml, B. Braun) were injected subcutaneously. The eyeballs were covered with eye ointment (Bepanthen, Bayer Leverkusen, Germany) to protect the cornea. Post-surgery, the cages were placed on a warming plate (25–30 ℃) until full recovery of the animals.
Embryo transfer was performed unilaterally or bilaterally if more than 15 embryos were transferred into the oviduct, as described in detail elsewhere. 9
Depending of the number of transferred embryos, we allocated the transfers to seven groups: G1 (3–5 embryos), G2 (6–8 embryos), G3 (9–11 embryos), G4 (12–14 embryos), G5 (15–17 embryos), G6 (18–20 embryos) and G7 ( ≥ 21 embryos).
Parameters analysed
Correlation of pregnancy rate (number of females that gave birth) and number of transferred embryos. Frequency of offspring (portion of transferred embryos that developed to term and were weaned): calculated as the ratio weaned pups/transferred embryos. Success rate: calculated as the ratio of transferred embryos/weaned pups. The success rate should give an estimation of how many embryos had to be transferred to achieve the birth of at least one live pup.
Statistics
Results were analysed using the GraphPad Prism5® programme version 5.01. A chi-square test was used to test if the number of transferred embryos correlated with the pregnancy rate.
One-way analysis of variance with post hoc Tukey’s multiple comparison tests was performed to determine statistical differences for the frequency of offspring and success rate. Differences of the means with
Results
Embryo transfer with freshly collected two-cell embryos
Correlation of pregnancy rate and number of transferred embryos
Pregnancy rates (in %) after embryo transfer with freshly collected and cryothawed two-cell embryos.
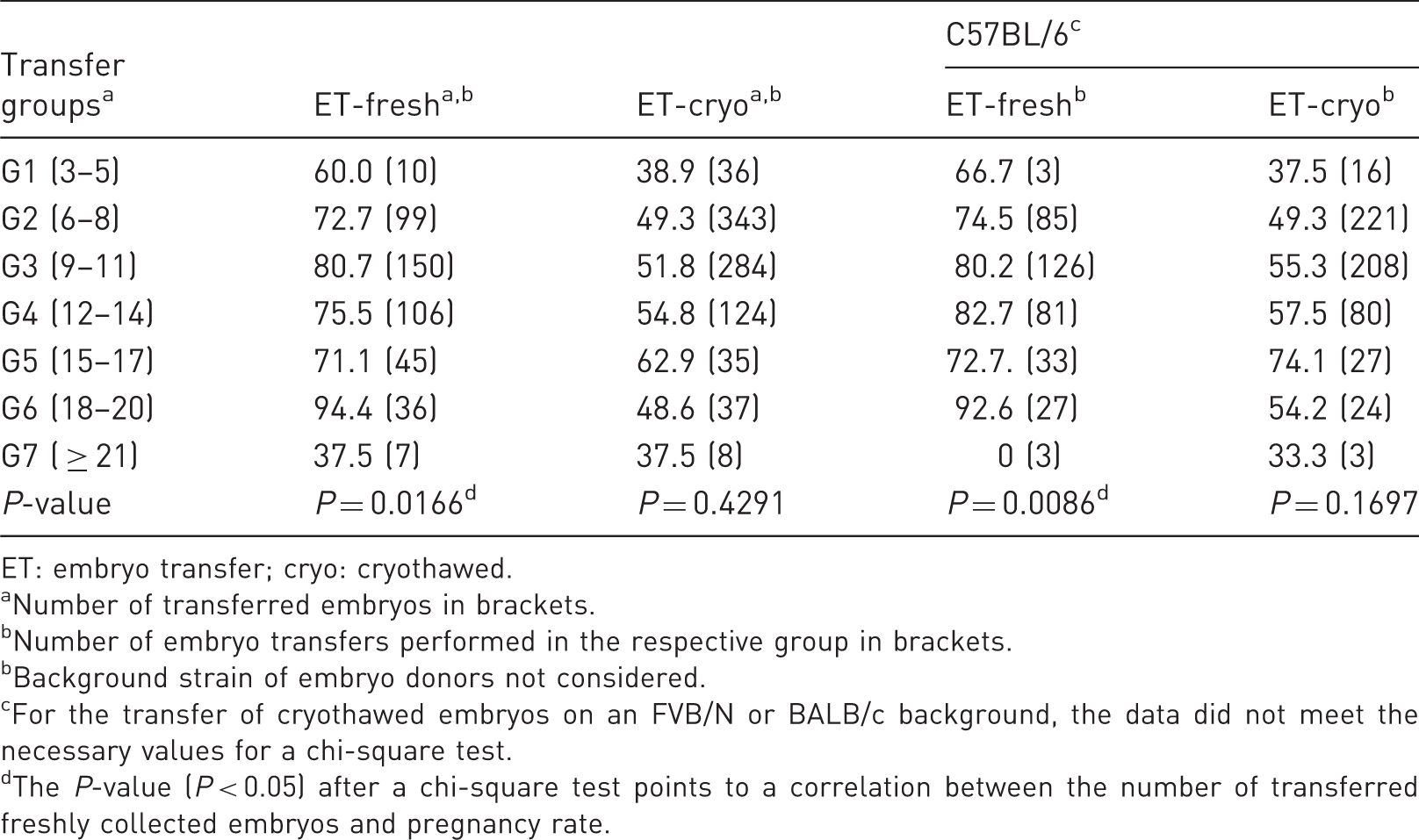
ET: embryo transfer; cryo: cryothawed.
Number of transferred embryos in brackets.
Number of embryo transfers performed in the respective group in brackets.
Background strain of embryo donors not considered.
For the transfer of cryothawed embryos on an FVB/N or BALB/c background, the data did not meet the necessary values for a chi-square test.
The
We also found a correlation between the background strain and pregnancy rate (
For embryos on a C57BL/6 background, the pregnancy rate correlated with the number of transferred embryos. It was highest after transfer of 18–20 embryos (92.6%). For the transfer of embryos on a FVB/N or BALB/c background, the data did not meet the necessary values for a chi-square test.
Frequency of born offspring
Mean number of transferred embryos, number of born and weaned pups ( ± SEM), and frequency of offspring (% ± SEM) without considering the genetic background.
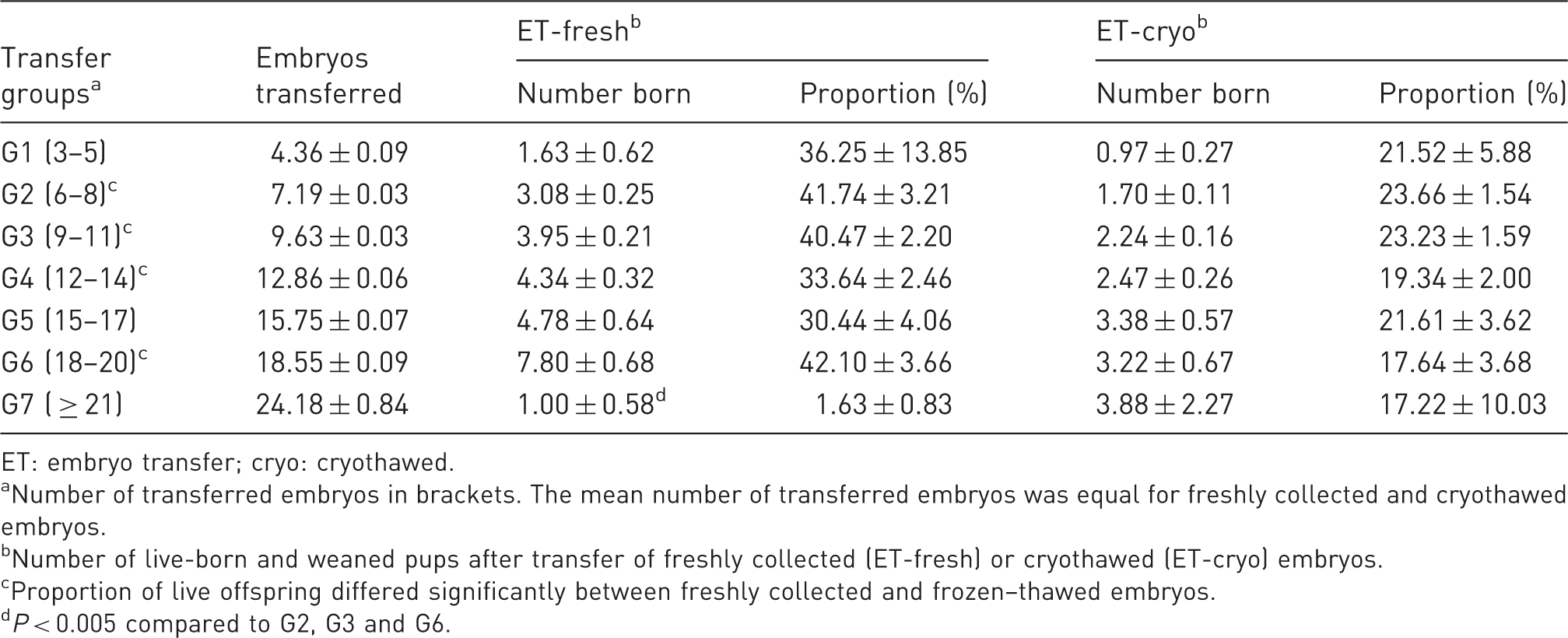
ET: embryo transfer; cryo: cryothawed.
Number of transferred embryos in brackets. The mean number of transferred embryos was equal for freshly collected and cryothawed embryos.
Number of live-born and weaned pups after transfer of freshly collected (ET-fresh) or cryothawed (ET-cryo) embryos.
Proportion of live offspring differed significantly between freshly collected and frozen–thawed embryos.
The frequency of viable offspring (portion of embryos that developed to term and were weaned) in G7 (≥21 embryos) was inferior to that of the other groups (Table 2 and Figure 2). These differences were significant compared to the G2 (6–8 embryos), G3 (9–11 embryos) and G6 (18–20 embryos) groups. The most pups were born and weaned in G6, but the differences were not significant (except if compared to G7). Four out of 348 litters were lost. In G1, two litters were lost because of one stillbirth and cannibalization of another litter. In G2 and G4, one litter was cannibalized each.
Frequency of born and weaned offspring (ratio of embryos that developed to term after embryo transfer). G1: 3–5 embryos, G2: 6–8 embryos, G3: 9–11 embryos, G4: 12–14 embryos, G5: 15–17, G6: 18–20 embryos and G7: ≥ 21 embryos. The transfer of freshly collected embryos was worst in G7. The difference was significant compared to G2, G3, G4 and G6. The outcome after transfer of cryothawed embryos was less compared to the transfer of freshly collected embryos from G1 to G6. Differences were significant for G2, G3, G4 and G6.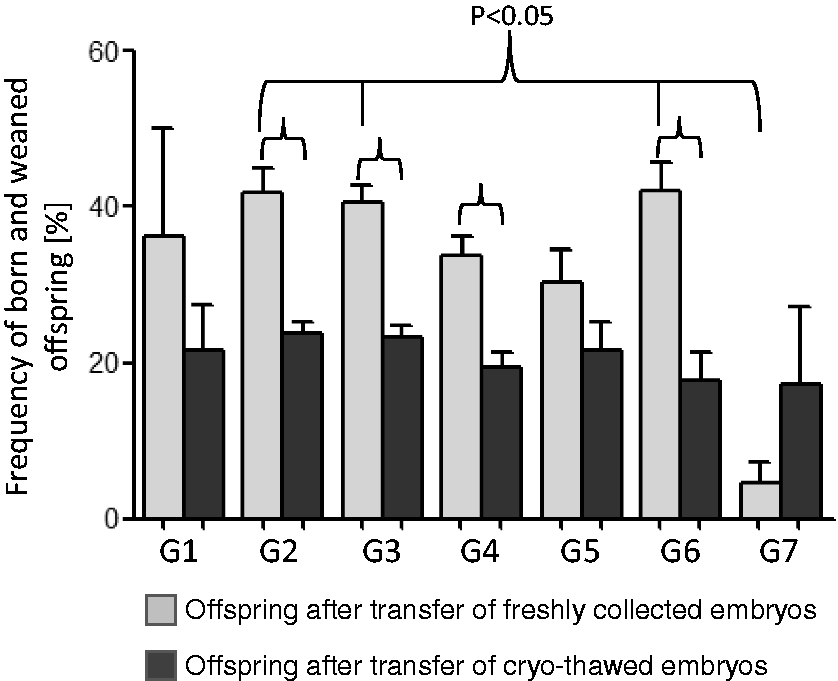
Mean number of transferred embryos, number of born and weaned pups ( ± SEM), and frequency of embryos (% ± SEM) on C57BL/6, FVB/N and BALB/c backgrounds.
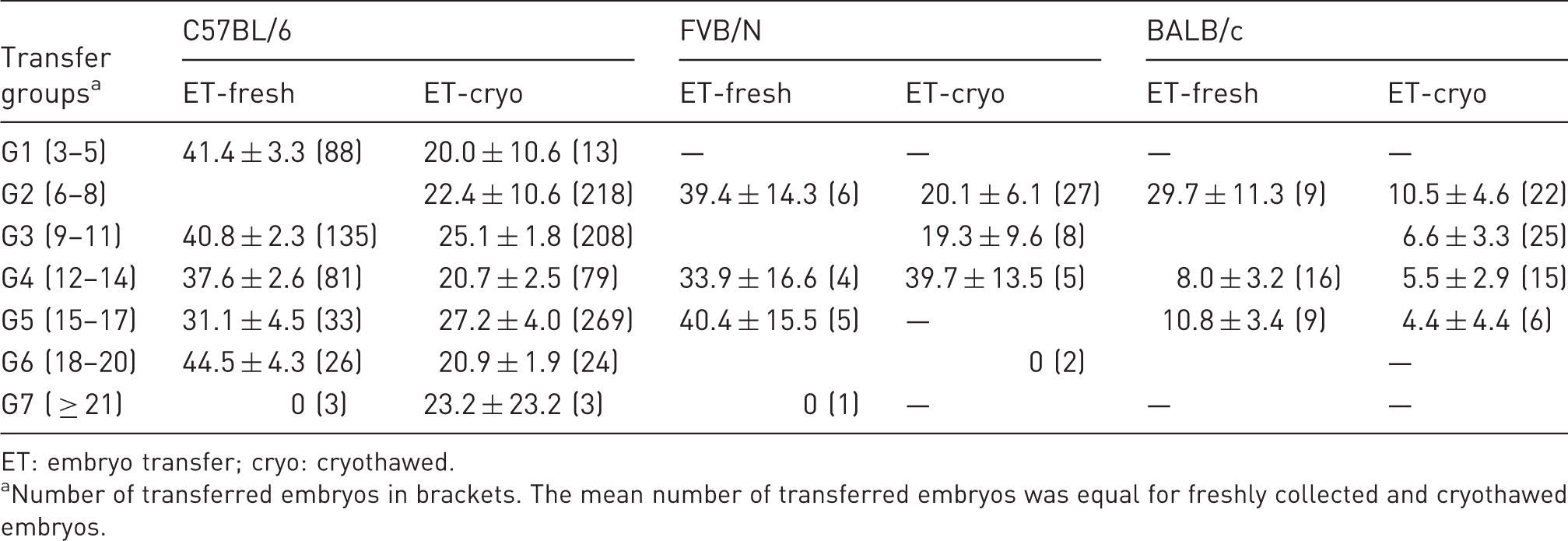
ET: embryo transfer; cryo: cryothawed.
Number of transferred embryos in brackets. The mean number of transferred embryos was equal for freshly collected and cryothawed embryos.
For embryos on an FVB/N background, we had no results for G1 and only one transfer in G7. The results for G3 + G4 and for G5 + G6 were combined. For embryos on a BALB/c background, we had no results for G1 and G7. The results for G2 + G3 and for G5 + G6 were combined (Table 3).
Frequency of offspring (% ± SEM) for the transfer of embryos on C57BL/6, FVB/N and BALB/c backgrounds without considering the number of transferred embryos.

ET: embryo transfer; cryo: cryothawed.
Number of live-born and weaned pups after transfer of freshly collected or cryothawed embryos.
Differences between C57BL/6 and BALB/c were significant (
Success rate (ratio of transferred embryos/weaned pups)
Success rate: mean number of embryos ( ± SEM) that had to be transferred to produce the birth and weaning of at least one pup, given as a ratio of transferred embryos/born and weaned pups without considering the genetic background.
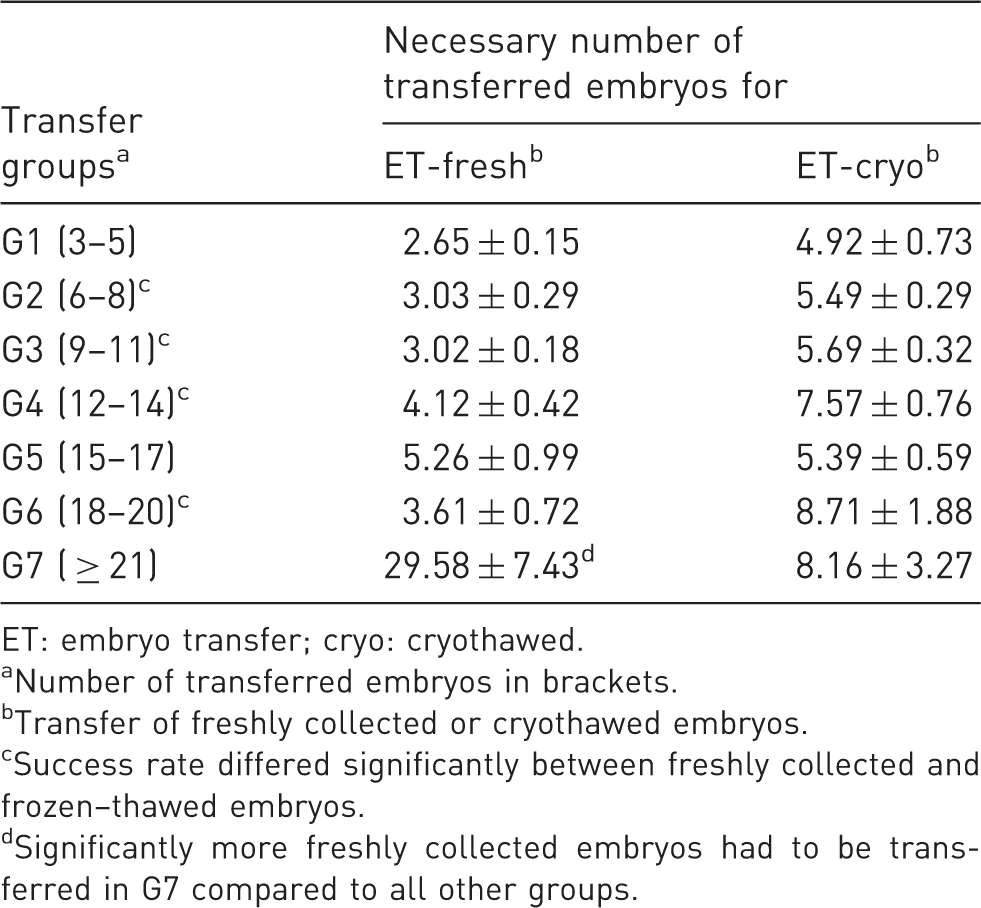
ET: embryo transfer; cryo: cryothawed.
Number of transferred embryos in brackets.
Transfer of freshly collected or cryothawed embryos.
Success rate differed significantly between freshly collected and frozen–thawed embryos.
Significantly more freshly collected embryos had to be transferred in G7 compared to all other groups.
Success rate for the transfer of embryos on C57BL/6, FVB/N and BALB/c backgrounds, without considering the number of transferred embryos.

ET: embryo transfer; cryo: cryothawed.
Transfer of freshly collected or cryothawed embryos.
Success rate for embryos on a BALB/c background was inferior to that of C57BL/6 or FVB/N (for the transfer of freshly collected and for cryothawed embryos).
Embryo transfer with cryothawed two-cell embryos
Correlation of pregnancy rate and number of transferred embryos
A chi-square test revealed that the pregnancy rate did not correlate with the number of transferred cryothawed embryos (Table 1).
The total mean pregnancy rate after transfer of cryothawed embryos was 50.87%, and varied between 37.5 (G7) and 62.9% (G5).
By analysing embryos on a C57BL/6 background, no correlation between the number of transferred cryothawed embryos and pregnancy rate could be seen. For the transfer of cryothawed embryos on an FVB/N or BALB/c background, the data did not meet the necessary values for a chi-square test.
Without considering the number of transferred cryothawed embryos, we saw a correlation between the background strain and pregnancy rate (
Frequency of live offspring
As expected, the number of viable and weaned pups increased with an increasing number of transferred embryos (Table 2).
The frequency of viable and weaned offspring after transfer of cryothawed embryos was lower than that for the transfer of freshly collected embryos (significant for G2, G3, G4 and G6). Within the cryothawed groups, we did not find any significant differences (Table 2 and Figure 2).
Nine out of 435 litters died or were cannibalized, and one litter was lost by leakage of the drinking bottle.
For the transfer of cryothawed embryos on a C57BL/6 background, we did not see significant differences within the transfer groups (Table 3). For FVB/N, we had no results for G1, G5 and G7. For BALB/c, we had no results for G1, G6 and G7. Therefore, the results for FVB/N and BALB/c are not sufficient for a reliable conclusion to be drawn.
Without considering the number of transferred cryothawed embryos, the frequency of born and weaned pups was best after transfer of embryos on a C57BL/6 background and worst after transfer of embryos on a BALB/c background. This difference was significant (Table 3).
Success rate (ratio of transferred embryos/ weaned pups)
The success rate after transfer of cryothawed embryos was inferior to the transfer of freshly collected embryos in all groups (significant for G2, G3, G6 and G7). In contrast to freshly collected embryos, the success rate for cryothawed embryos did not depend on the number of transferred embryos (Table 5).
It is important to mention that the transfer of cryothawed embryos from some genetic backgrounds did not produce litters at all. In order to include them for statistical evaluation, these transfers were combined with those that produced litters.
The embryo transfers with following background strains did not produce litters:
In group G2 (5–8 embryos): NSG ( In group G3 (9–12 embryos): NMRI ( In group G4 (13–16 embryos): NSG and NRG ( In group G5 (17–20 embryos): NSG (
Without considering the number of transferred cryothawed embryos, the success rate for embryos on a BALB/c background was significantly inferior to that on a C57BL/6 or FVB/N background (Table 6).
Discussion
It is a general problem that the backgrounds of genetically modified mouse strains are often not clearly defined. In our study, the genetic backgrounds of the embryo donors of most strains were ‘B6’, which often just meant that the mice were ‘black’. Therefore, it is important to mention that the population of the embryo donors was quite heterogeneous in our retrospective study, and included strains with poor reproductive outcome or with known revitalization-difficulties like BALB/c, immune-deficient strains like NSG or NRG, or with a totally undefined background (‘STOCK’). Therefore, the results presented here for the outcome after embryo transfer of freshly collected or cryothawed embryos might be inferior to results published earlier.10,11
Nevertheless, we thought that retrospective analyses of more than 1300 embryo transfers could give an overall estimation for better planning and performance of embryo transfers, because heterogeneity of the background strains was equal for all groups and could therefore be disregarded here. The number of transferred freshly collected embryos influenced the pregnancy rate. We found that the number of transferred embryos reached a maximum at 20 embryos. While the mean pregnancy rate was 76.82%, it dropped to 37.50% for the transfer of ≥ 21 embryos. This result became even more obvious when we looked at the ‘success rate’ that was defined as the number of embryos that had to be transferred to achieve the birth and weaning of at least one pup. Three to five embryos were needed for the weaning of at least one pup in the transfer groups of > 20 embryos. Almost 30 embryos were necessary for the weaning of one pup in the transfer group of ≥ 21 embryos.
In 1996, Johnson et al. postulated a ‘plateau’ for the development of live foetuses if more than a certain number are transferred. 12 Johnson et al. were talking about a ‘uterine capacity’ phenomenon, ‘in which it is thought that beyond a finite number, the uterus does not sustain the development of any more embryos to term’. This might be an explanation for smaller litter sizes than would have been expected, but does not explain our results for the reduced pregnancy rate and extremely small litter size (mean litter size for naturally mated CD2 females is 9.4 ± 1.03 at our institute, data not shown).
Recent studies have shown that trans-uterine migration of embryos does not occur. 13 This was the reason why we decided to perform bilateral embryo transfers if more than 15 embryos were to be transferred, to avoid an ‘overload’ of the uterus horns. However, one could assume that this kind of surgery might cause more stress to the surrogate dam leading to a reduced pregnancy and birth rate. Wiebold and Becker found an unequal function of left and right ovaries, and uterus horns, in Swiss Webster and B6D2F1 hybrid mice. 14 However, Mahabir et al. recently found no difference between uni- and bilateral embryo transfers. 15 These results were also confirmed by our present study for the transfer groups of 15–17 (G5) and 18–20 (G6) embryos, in which we successfully performed bilateral embryo transfers. However, the studies mentioned above cannot explain the poor success rates in our results for the transfer of more than 21 freshly collected embryos.
Another surprising result of our analyses was the fact that >35% of the embryos developed to birth in the transfer group of three to five embryos. Song et al. and Salamonsen et al. previously showed that the placenta can maintain pregnancy by itself only after decidualization.16,17 In mice and rats, this only occurs in the presence of blastocysts. One might assume that three to five embryos are not enough to send this essential signal for the maintenance of pregnancy. On the other hand, our results are in line with recently published data, which showed that the transfer of smaller embryo numbers yielded better success rates. 12
The proportion of embryos that developed to term was reduced after cryopreservation. We can exclude direct damage due to the freezing and thawing process because degeneration is normally directly visible after thawing. Randomly performed viability tests using fluorescein diacetate confirmed our light microscopy evaluation (data not shown). As we transferred only morphologically intact embryos, we assume that the reduced birth rate after the transfer of cryothawed embryos was caused by effects other than freezing injury or damage, possibly epigenetic modification. Tachataki et al. and Larman et al. concluded from findings from investigation of in human and mouse embryo freezing that the freezing process changes the pattern of early gene expression.18,19 The loss of embryos might occur before implantation, as Saenz-de-Juano et al. concluded from their studies on rabbit embryos. 20 Moreover, some strains seem to be more susceptible to freezing injury than others. Findings from Shi and Haaf indicate that the genotype influences the susceptibility to stressors. 21 Mobraaten showed different revitalization rates for different inbred strains 22 and Dinnyes et al. concluded that cryopreservation reduced post-implantation potential. 23 The above-mentioned studies not only give an explanation for the reduced birth rate after transfer of cryothawed embryos, but also explain why some strains are inferior to others.
The data from the literature mentioned above could also explain why the number of transferred cryothawed embryos played only a minor role regarding the success rate compared to the transfer of freshly collected embryos. If freshly collected embryos were transferred the success rate declined with increasing embryo number, while it remained more or less constant for the transfer of cryothawed embryos.
Although we did not have detailed information about used substrains or backcrossed generations for embryos on a C57BL/6, FVB/N or BALB/c background, we analysed these transfers in more detail because these are the most used background strains. These analyses confirmed our results described above. In addition, we confirmed that the genetic background of the embryo donors had an influence on the success of transfer of freshly collected as well as cryothawed embryos. Embryos on a BALB/c background were always inferior to the other strains.
Conclusions
The success of embryo transfers with freshly collected embryos is influenced by the number of transferred embryos. The transfer of ≥21 freshly collected embryos dramatically reduced the success rate. In cryothawed embryos, we could not see such a decrease. Once an embryo survives cryopreservation, thawing and implantation, it is also strong enough to complete development.
When planning embryo transfers:
The genetic background of the embryo donors has to be taken into account. Embryos on a BALB/c background perform the worst compared to other strains. The best results were achieved for the transfer of ≤20 freshly collected or ≤17 cryothawed embryos. The transfer of five or less embryos yielded reasonable success rates.
Footnotes
Acknowledgement
The authors would like to thank Wilfried Kues (Institute of Farm Animal Genetics, Friedrich-Loeffler-Institut, Neustadt-Mariensee, Germany) for critically reading the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interests with respect to the research, authorship and/or publication of this article
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
