Abstract
The laboratory mouse is the most used animal model in biomedical research. Several artificial reproductive techniques, such as revitalization of cryopreserved strains, rederivation after hygienic contaminations and the production of transgenic mouse models, require the transfer of preimplantation embryos to surrogate mothers. Pseudopregnancy is essential in recipient females and is induced by mating with sterile males. Commonly, surgically vasectomized males are used for this purpose. As an alternative, genetically modified mouse strains have been identified, in which homozygous infertile males are sexually active. Here, we investigated the suitability of genetically infertile Gapdhstm1Dao males under routine laboratory conditions with respect to plug rates, pregnancy rates and frequency of born offspring after embryo transfer. Our results showed no significant differences for these aspects between Gapdhstm1Dao and vasectomized CD2F1 males. In addition, we evaluated the efforts to obtain a defined number of sterile males either by breeding of sterile mutants or surgical vasectomy, and addressed the impact of both options on animal welfare. In conclusion, infertile males of the Gapdhstm1Dao line are a reliable alternative to vasectomized males for the induction of pseudopregnancy, and can contribute to the refinement of the procedure by avoiding surgical interventions.
Introduction
Embryo transfer is an essential method for reproductive technologies in mice, i.e. for generation of genetically modified strains, for saving of lines with problems in fertility or raising of litters, for in-vitro fertilization, for rederivation of hygienically contaminated strains or for revitalization of cryopreserved embryos.
Availability of pseudopregnant surrogate mothers is an essential requirement for the performance of a successful embryo transfer. In mice, the establishment of a pregnancy requires a vaginal-cervical stimulation. During copulation the male produces a vaginal plug which is formed by secretes of male accessory glands. This copulatory plug not only seals the vagina but also induces permanent pressure to the cervix.1,2 The vaginal plug indicates that intromission and ejaculation on the male has occurred. In mice, the induction of pseudopregnancy needs an ejaculation and cannot be achieved by male intromission or mechanical stimulation of the cervix alone. 3
Recipient females for embryo transfer have to be mated with infertile males to induce uterine differentiation resulting in the receptive state for the implantation of the transferred embryos. Commonly, infertile males for the induction of pseudopregnancy are produced by vasectomy under general anaesthesia. The severity of the intervention is classified as a ‘moderate’ procedure. After surgery, sterility has to be confirmed by test mating, which can be started after a recovery period of at least three weeks to ensure that no stored sperm cells were left in the ductus deferens. Alternatively, genetically modified strains in which the genetic modification leads to male infertility in the homozygous carriers have been reported for this purpose in recent years which could be useful for the induction of pseudopregnancy.4–6 For example, the suitability of Prm1-EGFP overexpressing males was tested and discussed for the production of pseudopregnant surrogate mothers. 6 To the best of the authors’ knowledge, however, our study is the first that covers not only the induction of pseudopregnancy, but also several other important aspects of the breeding and use of sterile mutant males.
In the presented study, we assessed the suitability of infertile Gapdhs™ 1 Dao knockout males for large-scale production of pseudopregnant surrogates under routine laboratory conditions. 4 In Gapdhs™ 1 Dao mice the glyceraldehyde-3-phosphate dehydrogenase spermatogenic gene (Gapdhs) was deleted by gene targeting via homologous recombination in TC-1 embryonic stem cells. The mating behaviour of homozygous Gapdhs knockout males is comparable to wildtype males including the production of a prominent vaginal plug. Due to low ATP levels, sperm from Gapdhs-/- males have a significant decreased motility which results in complete infertility. We compared plug rates, pregnancy rates after embryo transfer, and the portion of transferred embryos that developed to term (birth rate) after mating of B6CBAF1 hybrid females with either surgically vasectomized CD2F1 hybrid males, or infertile Gapdhs mutants with CD1 outbred background. In addition, we calculated/evaluated the required efforts to obtain a defined number of sterile males, i.e. the breeding effort, cage space, effort for genotyping and surgery. Moreover, we also addressed animal welfare aspects of both approaches with respect to the 3R guidelines.
Material and methods
Animals and husbandry
All animals were kept in an individually ventilated caging system (IVC, BioZone, Chester, UK) in cages with a ground area of 440 cm 2 under controlled standardized conditions: temperature 21 ± 2℃, relative humidity 50 ± 5% and artificial light (14 h light, 10 h dark, light on at 06:00 a.m.). Sterilized commercial softwood granulate bedding was used (Lignocel, Altromin, Lage, Germany). Autoclaved commercial pellet diet (Altromin 1314) and autoclaved water were provided ad libitum.
Whenever possible, mice were kept in sibling groups. The microbiological status was examined as recommended by FELASA and absence of listed microorganisms, 7 except Pasteurella pneumotropica, Helicobacter sp. and norovirus, was confirmed.
The experiments were in accordance with the German Animal Welfare Legislation and approved by the local Institutional Animal Care and Research Advisory Committee and permitted by the Lower Saxony State Office for Consumer Protection and Food Safety (reference number 42500/1H and 13A404).
Mice of the Gapdhs™ 1 Dao line, kindly provided by Dr D O’Brein, University of North Carolina, USA, were on a mixed 129S6;B6 background. After backcrossing to C57BL/6N the mating habits of males deteriorated considerably. To achieve better reproductive performance the mutants were backcrossed for four generations to Crl:CD1 (ICR) outbred mice. Animals of the N4 backcross generation were intercrossed to produce homozygous females which have been used henceforth to maintain the colony by mating with heterozygous males. Apart from the infertility of homozygous Gapdhs™ 1 Dao knockout males, no further pathological findings have been described. The knockout males produce normal sperm count, behave like wildtype males when mated and produce a prominent vaginal plug. Spermatozoa from Gapdhs-/- males show significantly decreased motility, probably due to very low ATP levels, resulting in complete infertility. 4 In contrast, Gapdhs-/- females as well as heterozygous Gapdhs+/- males and females do not differ from wildtype mice in their reproductive success. DNA for genotyping of mutant offspring was isolated from ear punches taken for marking the animals. Details for genotyping can be found under: https://www.mmrrc.org/catalog/sds.php?mmrrc_id=36555. The observed inheritance pattern of the mutation was according to Mendel’s laws.
CD2F1 males were generated by mating BALB/cJHanZtm (C) females and DBA/2JHanZtm (D2) males. F1-males were vasectomized at an age of 6 weeks.
B6CBAF1/Rj females were purchased from Janvier (www.janvier-breedingcenter.com) at an age of four weeks. They were used as embryo transfer recipients at an age of 8–10 weeks.
Anaesthesia and analgesia for surgery
Anaesthesia was applied by intraperitoneal (i.p.) injection of a mixture of 80 mg/kg ketamine 10% (Ketanest®, Bayer Leverkusen, Germany) 2 mg/kg xylazine 2% (Rompun®, Bayer, Leverkusen, Germany), and 0.7 mg/kg midazolam (Rotexmedica GmbH, Trittau, Germany). As soon as the toe pinch reflex had disappeared, 1 mg/kg meloxicam (Metacam®, 2 mg/ml, Boehringer Ingelheim) for pain relief and 0.05 mg/kg Atropin (Atropinsulfat, 0.5 mg/ml, B. Braun) to prevent bradycardia were injected subcutaneously (s.c.). The eyes were covered with eye ointment (Bepanthen, Bayer, Leverkusen, Germany) to protect the cornea. Post-surgery the cages were placed on a warming plate (25–30℃) until full recovery of the animals.
Vasectomy
CD2F1 males were vasectomized by cauterization of both ductus deferentes, as described in detail elsewhere.8,9
Briefly, the anaesthetized male was placed in a dorsal body position and the abdomen was opened by an incision at the linea alba 0.5 cm cranial from the pelvis. The fat pad surrounding the right testes was grasped with forceps and pulled out until the ductus deferens was visible. A small piece of the spermatic duct (∼0.3 cm) was removed by cauterization (Small Vessel Cauterizer, Fine Science Tools GmbH, Heidelberg, Germany). The procedure was then contralateral repeated. The testicles were shifted back and the abdominal cavity was closed in two layers with sutures. Two to three weeks later the males were test mated to ensure their sterility before using them for mating to induce pseudopregnancy.
Embryo transfer
For the induction of pseudopregnancy B6CBAF1 females were mated overnight either with Gapdhs-/- or surgical vasectomized CD2F1 males. The males were used once a week. Vaginal plug positive mice were used for unilateral embryo transfer, performed as described in detail elsewhere. 9 Briefly, after anaesthesia females were placed in a ventral body position. A longitudinally incision in the lumbar area was made. Next, the abdominal cavity was opened and the ovary with oviduct was carefully pulled forward. The bursa ovarica was opened and two-cell embryos were transferred into the oviduct. The reproductive tract was gently placed back into the abdominal cavity, the peritoneum sutured and the skin closed with wound clamps (Michel suture clips 7.5 × 1.75 mm, Aesculap, Tuttlingen, Germany). Birth was expected approximately 19–20 days later.
Embryos
To avoid extra embryo production, we transferred frozen-thawed two-cell ‘control embryos’ from our institutional embryo-freezing programme. For this purpose hybrid two-cell embryos were collected at day 2 post coitus from superovulated BALB/cJHanZtm females mated with DBA/2JHanZtm males. For superovulation an i.p. injection of 5 IU pregnant mare’s serum gonadotropin (PMSG, Intergonan, Vemie Veterinär Chemie GmbH, Kempen, Germany) and 48 h later of 5 IU human chorionic gonadotropin (hCG, Primogonyl-1000, Schering Pharma, Berlin, Germany) was administered. One or two batches with about 20 hybrid embryos were frozen as control with every freezing cycle. Routinely, controls are stored for at least one month in liquid nitrogen after freezing, before they are thawed and transferred to embryo transfer recipients to confirm a successful archiving.
Experimental design
Vasectomized CD2F1 males and Gapdhs-/- males were monogamously mated in parallel overnight to B6CBAF1 females on seven independent experimental days (18–20 matings per group and experimental day). Females in pro-oestrus or oestrus mated during the night. The following morning females were checked for the presence of a vaginal plug (VP) to determine the ‘plug rate’. At the same day we transferred randomized 6–14 frozen-thawed control embryos per female to determine the ‘pregnancy rate’ and the portion of embryos that developed to term (‘birth rate’).
Breeding efforts to generate vasectomized males
The colony index (CI) was used to determine the number of required mating to generate a given number of Gapdhs-/- and CD2F1 males. The CI was calculated by dividing the total number of offspring by the number of mated females per week. 10 Working time for vasectomy (including peri- and postoperative care) and genotyping were evaluated and compared.
Statistics
We used www.biomath.info/power/PowerAnalysis.html to calculate the most appropriate group size. Results were analysed using the GraphPad Prism5 program. Plug rate and pregnancy rate were tested with chi square statistics (Fisher’s exact test). With the assumption of Gaussian distribution for the number of transferred embryos developed to term, we performed two-tailed t-tests to determine statistical differences. The colony indices of Gapdhs-/- and CD2F1 were compared by one-tailed t-tests. Differences of the means with p < 0.05 were judged as significant.
Results
Plug rate
Plug rate, pregnancy rate and frequency of born offspring in the experimental groups.

Males were mated to B6CBAF1 females. Results were determined for every single experimental day. For plug and pregnancy rate a Fisher’s exact test was performed. To compare the rate of born offspring a t-test was performed (results are given as mean ± SD). All differences were not significant.
Females with a vaginal plug were judged as ‘pseudopregnant’ and according to the number of thawed embryos the necessary number of embryo transfer recipients per experimental day was selected (day 0.5 pseudopregnancy was additionally confirmed during embryo transfer by the presence of a swollen ampulla).
Pregnancy rate
Embryo transfers were conducted on five independent experimental days. Fourteen of 17 (82.3%) pseudopregnant females previously mated to vasectomized CD2F1 males became pregnant after embryo transfer. Simultaneously, 16 (80.0 %) of 20 females plug positive after mating to Gapdhs-/- males became pregnant and gave birth. The two-sided Fisher’s exact test (with α < 0.05) showed no significant difference (Table 1).
Embryo development
Sixty of 125 two-cell embryos transferred to 17 recipients developed to term. This results in a frequency of born offspring of 48.04 ± 6.7% for pseudopregnant recipients produced by mating with vasectomized CD2F1 males. An almost identical frequency of 48.7 ± 7.2% was observed for females mated to Gapdhs-/- males. In total 169 embryos were used in for 20 transfers. One recipient of this group which received seven embryos was excluded from the study because of infanticide before the number of pups could be recorded. Altogether, 79 of 162 transferred embryos developed to term in recipients mated with Gapdhs-/- males (Table 1).
The difference in the mean number of transferred embryos per recipient between both mating groups was not significant (8.5 ± 0.4 for Gapdhs-/- and 7.4 ± 0.3 for vasectomized males; p = 0.45) and a possible impact on the presented results can be excluded.
Comparison of breeding efforts to generate vasectomized males
For routine embryo transfers, a group of 40 vasectomized males was permanently available in the Central Animal Facility of the Hannover Medical School. The following calculations refer therefore to the number of 40 vasectomized males.
Colony index
The reproductive performance of a strain is represented by the colony index (CI) and can be used to determine the number of required breeding females to generate a given number of mice.
With the assumption of a sex ratio of approximately 1:1 we need 80 pups to produce 40 CD2F1 males. In contrast, only 25 % of the male offspring were homozygous for the Gapdhs knockout mutation, due to the use of heterozygous breeding males. Therefore, 160 pups were necessary to produce 40 males with the appropriate sterile phenotype.
Expenditure to produce 40 vasectomized or mutant males.

The difference between the average litter size of CD2F1 and Gapdhs mice was significant; two out of 64 litters analysed did not produce Gapdhs-/- mice; n.a., not applicable.
Expenditure of time and severity classification of animal treatments
Expenditure of time for vasectomy, i.e. duration of surgery, postoperative care over the next days (control of movement, food- and water uptake, wound healing etc.) was calculated to be 60 minutes per male. Taking of an ear biopsy, followed by PCR analysis for genotyping, takes about 30 minutes per male.
Severity classification of the treatments was made according to Directive 2010/63EU, Annex VIII. Embryo transfer had to be classified as ‘moderate’ with less than seven days of postoperative pain. A licence for this procedure was obligatory. Ear biopsy was classified as ‘mild’ and postoperative pain was considered for less than one day. No licence was necessary because the primary purpose of ear punching was the identification of the animal.
Discussion
In the present study we assessed (i) whether sterile males generated by a genetic modification can be reliably used to induce pseudopregnancy in females like commonly used vasectomized males, (ii) whether the use of these mutant males or surgically vasectomized males is more efficient and (iii) which method should be preferred in the view of animal welfare aspects and the 3R principle.
The results did not reveal any significant differences in plug rate or pregnancy rate between females that were mated to vasectomized males or sterile Gapdhs mutants. Also the portion of transferred embryos that developed to term (birth rate) did not differ. Furthermore, males of both experimental groups showed a good plug forming ability for easy mating control. This is an important finding because the copulatory plug in mice appears to be important for nidation and maintaining of pregnancy. Recently, it was shown that despite sufficient oocyte fertilization fewer females gave birth when the plug forming ability of the male was disrupted. 11 An impact of the genetic background on characteristics of the vaginal plug has also been reported. 12
Importantly, no leakiness in fertility was found for homozygous Gapdhs knockout males, making test breeding, as commonly applied for vasectomized males, dispensable. In contrast, nearly double breeding effort was necessary to provide infertile male mutants. Only 25% of the offspring are homozygous knockout males with the desired phenotype, resulting in more surplus animals compared to the production of F1-hybrid males for vasectomy. However, a male with hybrid or outbred background can be used for mating for several months and therefore a large-scale production of animals to produce sterile males is unnecessary for both options.
Vasectomy, postoperative care and test breeding for F1-hybrids were more time-consuming than the necessary genotyping of mutants, including the collection of tissue samples, DNA extraction and PCR reaction. However, the expenditure of time for surgery was balanced by the need for more breeding cages and associated time for animal care for the Gapdhs line.
Obvious differences between vasectomy and genetically induced infertility exist in the assessment of the degree of severity. According to the Directive 2010/63EU, Annex VIII, a vasectomy in mice needs to be licensed by the competent authority and only authorized and appropriately qualified persons are allowed to do it.
Besides sterility, the Gapdhs™ 1 Dao males showed no other changes in their phenotype. Vasectomy, however, is carried out as surgery performed under general anaesthesia associated with post-surgical pain, scored as ‘moderate’ severity and required appropriate analgesia. Metacam injection was applied already during anaesthesia and gave relief of post-surgical pain for up to 24 h. Vasectomized males recovered well during the observation period without any evidence of abnormal behaviour which would indicate strong and lasting pain for the animals. Furthermore, we never found indications for wound infections. Long term effects of vasectomy in mice have been described. 13 The effects, however, were of histological nature and did not seem to influence wellbeing of the animals. We routinely use vasectomized hybrid males for up to 18 months without any health problems that could be assigned to the surgery. The length of useful life span of Gapdhs mutants for efficient induction of pseudopregnancy has still to be evaluated.
Summary of the evaluated parameters.
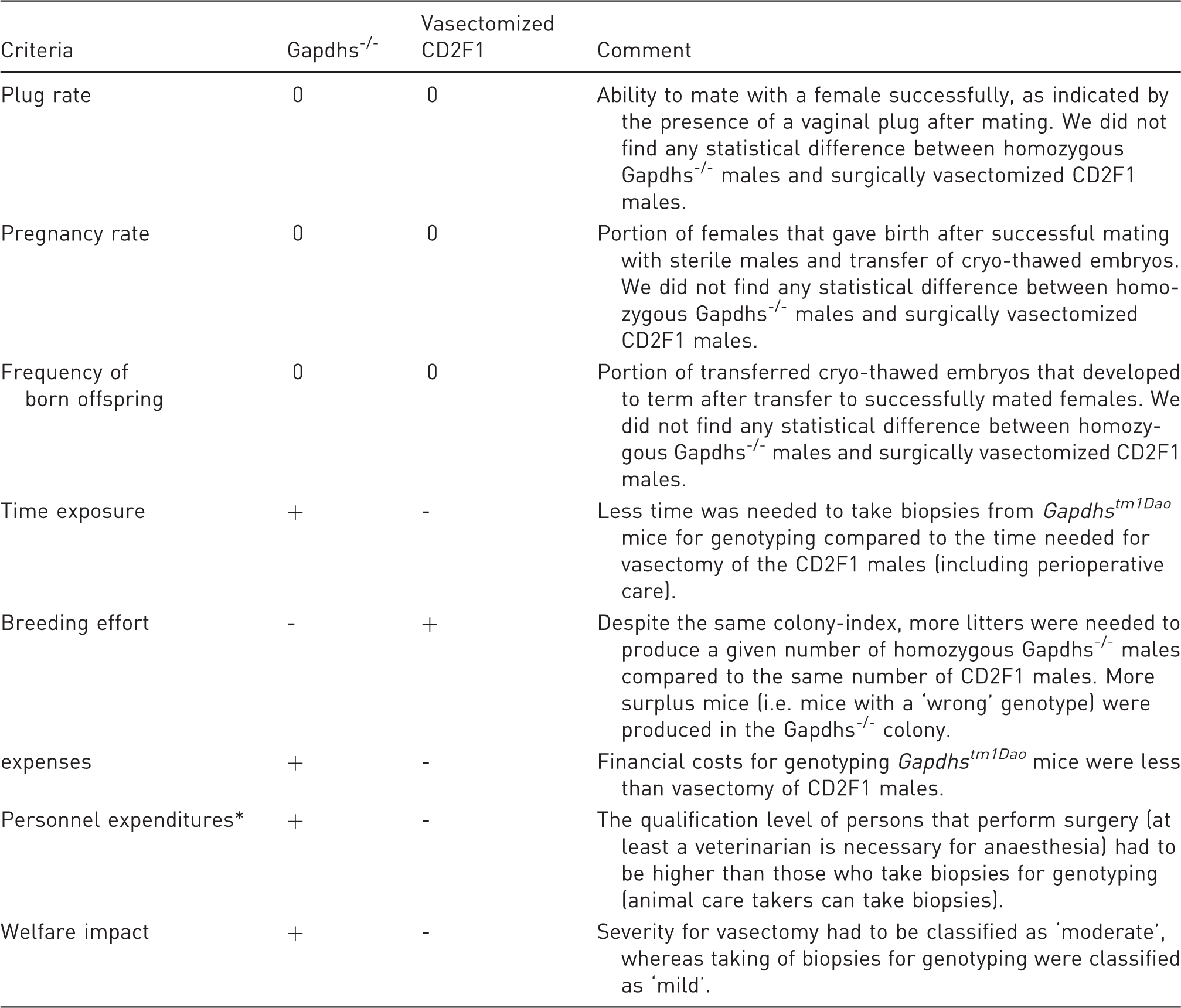
‘0’ no differences were found, ‘+’ more advantageous, ‘-’ less advantageous if genetically vasectomized Gapdhs™ 1 Dao males were compared to surgically vasectomized CD2F1 males. *Personnel expenditures based on the working document on the development of a common education and training framework to fulfil the requirements under the Directive 2010/63/EU of the Expert Working Group. 15
Especially for the production of germ free mice in isolators, the use of sterile Gapdhs males might be a good alternative to vasectomy because surgical intervention under these conditions are laborious and bears the risk for unwanted hygienic contamination. In combination with non-surgical (transcervical) embryo transfer, 14 the use of sterile Gapdhs males would enable research groups, without specific equipment or appropriate persons for surgical interventions, to apply several reproductive techniques which otherwise would require surgical intervention. From the animal welfare aspect, genetically sterile males should be the preferred alternative for the induction of pseudopregnant females, provided that the higher number of produced surplus animals can reasonably be used for other purposes.
Footnotes
Acknowledgements
Gapdhs knockout mice were produced in E.M. Eddy’s lab at the National Institute of Environmental Health Sciences, National Institutes of Health and characterized in the lab of D O’Brein at UNC.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
