Abstract
Individual housing of laboratory mice may increase vulnerability to surgical stress, and interfere with postsurgical recovery. To analyse the effect of housing conditions on recovery, pair- and single-housed female C57BL/6J mice underwent a minor laparotomy +/− analgesia, anaesthesia only or no treatment. Animals were monitored using non-invasive methods during the immediate postsurgical period to assess pain and general impairment. While no appearance or posture abnormalities were observed postexperiment, home cage behaviours were affected distinctly. Discriminant analysis identified self-grooming, locomotion, climbing and resting as mainly responsible for experimental group separation. Behavioural rhythmicity was disrupted, and behaviours related to wellbeing, such as nest building, climbing and burrowing, decreased. Behavioural pain signs (e.g. press) increased. Most behavioural alterations showed a gradation between treatments, e.g. burrowing latency ranged from an intermediate level following anaesthesia only and surgery with analgesia, to pronounced prolongation after surgery without analgesia. Significantly lower burrowing performance after surgery without analgesia in individually-housed animals indicates better recovery in pairs. Social interaction in pairs – an important component of normal behaviour (64%) and a potential indicator for direct social support – was nearly absent (0.3–0.5%). While anaesthesia and surgery resulted in clear changes in behaviour, differences between housing conditions were minor. Hence, despite a tendency towards better recovery in pairs, we found no distinct negative effect of individual housing. In conclusion, both housing conditions are acceptable during the period immediately following minor surgery, though social housing is always preferable in female mice.
Laboratory mice, just like their wild ancestors, are social animals and are highly motivated to interact with each other. 1 A stable and harmonious social context seems to be important for mouse wellbeing. While providing harmonious groups may be difficult in sometimes aggressive male mice, the advantage of housing female mice in groups is clear since the environment in which animals are housed influences not only animal wellbeing but also experimental results. 1 Despite the common practice of social housing in animal facilities, mice are often separated for scientific or practical reasons such as for monitoring purposes or surgical procedures.
The findings of studies analysing the actual effects on scientific results of individual housing of mice, and the possible adverse impact on wellbeing, have been ambiguous. Some studies found no effect of individual housing of different duration compared with social housing on endocrine stress indicators, 2 physiological indexes 3 or behavioural tests. 2 On the contrary, several other studies have shown distinct effects of individual housing compared with social housing, as evidenced by changes in sympathetic neurotransmission, 4 basal heart rate 5,6 and thymus weight. 6 Other alterations are disruptions to circadian activity patterns 5 and effects on memory, emotionality and anxiety, as well as a tendency to show hyperactivity in behavioural tests. 7–9
Although the proximate effects of individual housing may not always be evident, it might affect the way animals respond to stressors. Even if normal behaviour and general condition are unaffected, some studies have shown that individually-housed or isolated animals were more sensitive to stress, with mice housed in stable groups recovering faster from mild stressors, 6,10 leading to the suggestion that individual housing may hamper the animal's ability to cope with, and increase vulnerability to, stressful episodes.
Surgery and the postsurgical recovery phase represent stressful episodes for mice. Hence, individual housing may exacerbate an animal's vulnerability to surgical stress and may interfere with postsurgical recovery. Two studies seem to support the hypothesis that social housing has a recovery-promoting effect. Pham et al. 11 found that, after laparotomy and caecal manipulation, single-housed mice exhibited greater self-administration of analgesics than social-housed mice and therefore seemed to experience more pain postsurgery. In another study, faster recovery was observed in socially-housed mice following major surgery. 12
Here, we aimed to analyse the potentially beneficial effect of social support on postsurgical recovery by comparing single-housed with pair-housed female C57BL/6J mice after minor surgery. To assess the impact of surgery and different housing conditions on wellbeing we used a range of non-invasive behavioural measurements that can be applied in the animals’ home cage without provoking additional stress. Pain signs, 13,14 burrowing performance, 15 home cage behaviours 12 and classical indices like clinical symptoms, overall appearance and body weight should allow recognition not only of postsurgical pain but also impairment of general condition, thus providing a broad picture of the animal's recovery.
We hypothesize that signs of pain and impaired wellbeing should be reduced in socially-housed mice if this housing condition is beneficial to postsurgical recovery.
Materials and methods
Ethics statement
The animal housing and experimental protocols were approved by the Cantonal Veterinary Department, Zurich, Switzerland, under licence no. ZH 120/2008, and were in accordance with Swiss Animal Protection Law. Housing and experimental procedures also conform to the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes (Council of Europe no. 123 Strasbourg 1985) and to the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Resources, National Research Council, National Academy of Sciences, 1996).
Animals
Sixty-four C57BL/6J and 32 C57BL/6J-TyrC-Brd female mice were obtained from our in-house breeding facility at the age of 6–8 weeks. We used pairs of black-coated C57BL/6J strain and its coisogenic albino mutant C57BL/6J-TyrC-Brd for better optical distinction of the observed animals. The mice were free of all viral, bacterial and parasitic pathogens listed in the FELASA recommendations. Health status was confirmed by a health surveillance programme throughout the experiments. 16
Mice were housed in groups of three to six animals for at least three weeks prior to testing in our animal room. Animals were kept in Eurostandard Type III clear-transparent plastic cages (425 mm × 266 mm × 155 mm) with autoclaved dust-free sawdust bedding (80–90 g per cage) and one nestlet™ (5 cm × 5 cm), consisting of cotton fibres (Indulab AG, Gams, Switzerland) as nesting material. They were fed a pelleted and extruded mouse diet (Kliba No. 3436, Provimi Kliba, Kaiseraugst, Switzerland) ad libitum (provided in a food hopper continuously throughout the entire duration of the experiment) and had unrestricted access to sterilized drinking water. The light/dark cycle in the room consisted of 12/12 h (lights on 15:00, lights off 03:00) with artificial light (approximately 40 lx in the cage). The temperature was 21 ± 1°C, with a relative humidity of 50 ± 5%, and with 15 complete changes of filtered air per hour (HEPA H 14 filter). The animal room was insulated to prevent electronic and other noise. Disturbances, e.g. visitors or unrelated experimental procedures in the animal room, were not allowed.
Experiments
Experimental housing and set-up
During the whole experimental period animals were housed under standardized conditions as described above with the burrowing test set-up in addition. As burrowing apparatus, a plastic bottle (standard opaque water bottle, 250 mL, 150 mm length, 55 mm diameter) filled with 138–142 g of food pellets identical to those of the animal's normal diet was used. An additional empty bottle of the same dimensions was provided to serve as a shelter.
Experimental design
Mice were housed either in pairs of one C57BL/6J and one C57BL/6J-TyrC-Brd mouse, or individually (one C57BL/6J). Each pair- or single-housed mouse was observed directly after an experimental procedure. Eight pairs and eight individually-housed mice were allocated randomly to one of four experimental groups: (1) surgery + anaesthesia (mice underwent anaesthesia and surgery without analgesic treatment); (2) surgery + anaesthesia + analgesia (mice underwent anaesthesia and surgery with analgesic treatment), (3) anaesthesia only or (4) no treatment.
Experiments and data acquisition
For acclimatization, animals were housed either in pairs or individually for three days under standardized conditions in cages containing the experimental set-up as described above. Animals had no prior experience with behavioural testing.
In pair-housed mice, both animals underwent experimental procedures. The experiment began at 13:00 h with a subcutaneous injection of 2 μL/g body weight of phosphate-buffered saline (PBS) for the surgery + anaesthesia and anaesthesia-only groups. In the surgery + anaesthesia + analgesia group, 5 mg/kg body weight of the analgesic carprofen (Rimadyl™, Pfizer Inc, New York, NY, USA) was diluted in PBS and injected as 2 μL/g body weight. The animals were transferred 45 min later in transport cages to the nearby operating theatre. Mice were anaesthetized with sevoflurane (Sevorane™, Abbott, Baar, Switzerland) as mono-anaesthesia. The anaesthetic gas was provided with a rodent inhalation anaesthesia apparatus (Provet, Lyssach, Switzerland); oxygen was used as carrier gas. After induction of anaesthesia in a Perspex induction chamber (8% sevoflurane, 600 mL/min gas flow), animals were transferred to a warming mat (Gaymar, TP500, Orchard Park, NY, USA) set at 39 ± 1°C to ensure constant body temperature, and anaesthesia was maintained via a nose mask (4.9% sevoflurane, 600 mL/min gas flow). Eye ointment was applied, the fur was clipped and the operating field disinfected with ethanol (70%) in all animals. Mice in both surgery groups underwent a one-side sham embryo transfer. The incision in the abdominal muscle wall was closed with absorbable sutures (Vicryl™, 6/0 polyglactin 910, Ethicon Ltd, Norderstedt, Germany), and the skin was closed using skin staples (Precise™, 3M Health Care, St Paul, MN, USA). Surgery was completed within 6–8 min in both surgery groups. Anaesthesia lasted 14–16 min in all groups. Animals were allowed to recover for 15–20 min on the warming mat before being transferred back to the animal room for subsequent behavioural observation.
Observation began at 15:00 h by returning each mouse from its transport cage to its home cage containing the re-filled burrowing test apparatus or, in the case of non-treated mice, just refilling the test apparatus and by starting digital video recording.
Data analysis
Home cage behaviours
Ethogram of home cage behaviours and pain signs
Pain signs
The number of aberrant behaviours occurring in pain states, e.g. press, stretch or stagger/fall and rear up (Table 1 13,14 ), was assessed for 15 min at one hour after the start of observation. 18
Burrowing performance
The burrowing test determines burrowing performance and can be used as a simple method to assess postsurgical impairment in mice. Good performance in this test is defined as short latency to remove items from a tube-like apparatus (burrowing). 15 Burrowing was defined as the removal of more than three pellets from the apparatus within 10 s. The latency to burrow per cage was measured. Measurement of latency was continued for 24 h if the animals did not start to burrow within the 6 h of behavioural analysis.
Clinical investigation
Animals were weighed at 15:00 h, 24 h before, and 24 and 48 h after experiment and observed for 20–30 s before, during and after weighing. According to a routinely used scoring system documenting the general condition of an animal, 19 abnormalities of body condition (e.g. sunken flanks), fur condition (e.g. ruffled coat), eyes (e.g. discharge), breathing (e.g. irregular) and posture (e.g. hunched back) were registered, and wound healing, spontaneous behaviour and movement were assessed.
Statistical analysis
Statistical analyses were performed using SPSS 20.0 software (IBM, Armonk, NY, USA).
All data were tested for normal distribution and homogeneity of variance. If necessary, data were log (X + 1) transformed to meet assumptions of statistical tests.
Mean and standard deviation (SD) of latency to burrow were calculated. Kaplan–Meier survival analysis was performed to examine the distribution of time to effect (latency to burrow). To test whether latency to burrow differed statistically between experimental groups or housing conditions, a log rank significance test was performed.
Mean and SD of durations of home cage behaviours, numbers of resting bouts and pain signs were calculated. In pair-housed mice, data of only one mouse (i.e. the black coated C57BL/6J) were used for further statistical analysis.
Discriminant analysis was used to determine the effects of surgery, anaesthesia and analgesic treatment on individual and social home cage behaviour; behaviours mainly responsible for group separation were determined. The determined behaviours were further analysed using univariate general linear model (GLM) with experimental group and housing as fixed factors for individual behaviours and experimental group for social behaviours as fixed factor. Post hoc tests (Bonferroni) were used for comparisons between experimental groups.
Activity duration and number of resting bouts were compared between groups using a univariate GLM with experimental group and housing as fixed factors. Post hoc tests (Bonferroni) were used for comparisons between experimental groups.
Significance for all statistical tests was established at P ≤ 0.05.
Results
Home cage behaviours
General activity
General activity, determined as the sum of all active behaviours, i.e. all home cage behaviours except resting, was significantly higher in experimental groups in both housing conditions that underwent surgery or anaesthesia compared with no treatment (P < 0.001) (Figure 1a, Table 2). Additionally, the interaction between housing and experimental group was significant (P = 0.006), while housing condition alone was not a significant factor (P = 0.391).
(a) Mean (±SD) duration of general activity. Significant differences were found between the no treatment group and all treated groups (P < 0.001). (b) Mean (±SD) number of resting bouts. For pairs, results of individual and social resting were combined. Significant differences were found between the no treatment groups and both surgery groups (P < 0.001; P = 0.009) and anaesthesia-only animals and surgery groups (P < 0.001, P = 0.036). Significant results are marked with * (P ≤ 0.05) Home cage behaviours Mean (±SD) duration of individual and social behaviours, general activity and numbers of resting bouts. For resting bouts in pairs results of individual and social resting were combined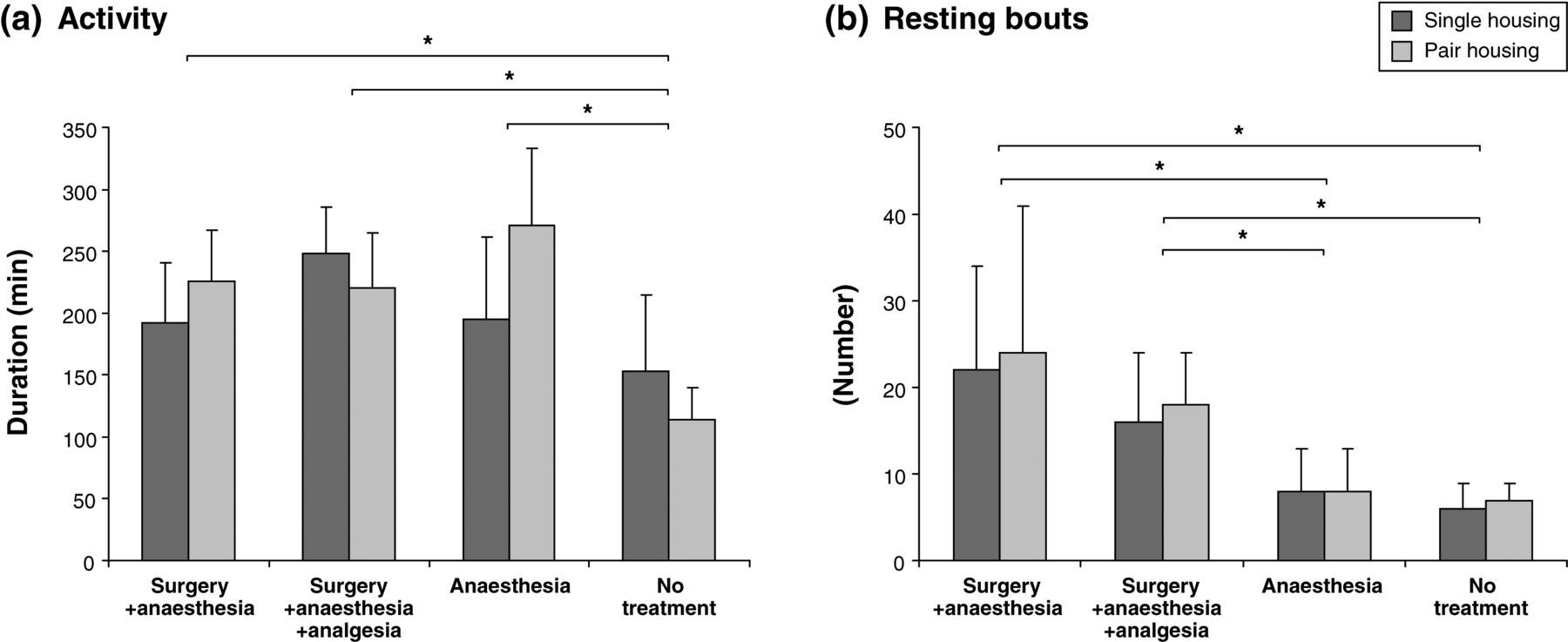
Activity rhythm
The number and temporal distribution of active and passive behaviour bouts was defined as the activity rhythm. In comparison with no treatment, activity rhythms in both housing conditions appeared disrupted following experiments (Figure 2). Non-treated animals, as well as animals that underwent anaesthesia only, showed long resting bouts, disrupted by a few short activity bouts. After surgery, the activity rhythms were highly fragmented into short activity and resting bouts; consequently, the number of resting bouts increased (Figures 1b and 2; Table 2). These differences were significant between no treatment groups and both surgery groups (P < 0.001, P = 0.009), and between the anaesthesia-only and both surgery groups (P < 0.001, P = 0.036). Housing condition and interactions between housing and experimental groups were non-significant factors (P = 0.611, P = 0.952).
Activity rhythm. Six hours of representative data of pairs of mice that were not treated, and after surgery + anaesthesia + analgesia. Bars represent the occurrence and duration of observed behaviours (for statistical analysis aggressive grooming was integrated with aggressive behaviour). While both animals showed a clear rhythm, occurring in synchrony with each other, after surgery the rhythm was disrupted into short behavioural bouts. Notably, social resting (i.e. animals in body contact during resting) was absent after experiment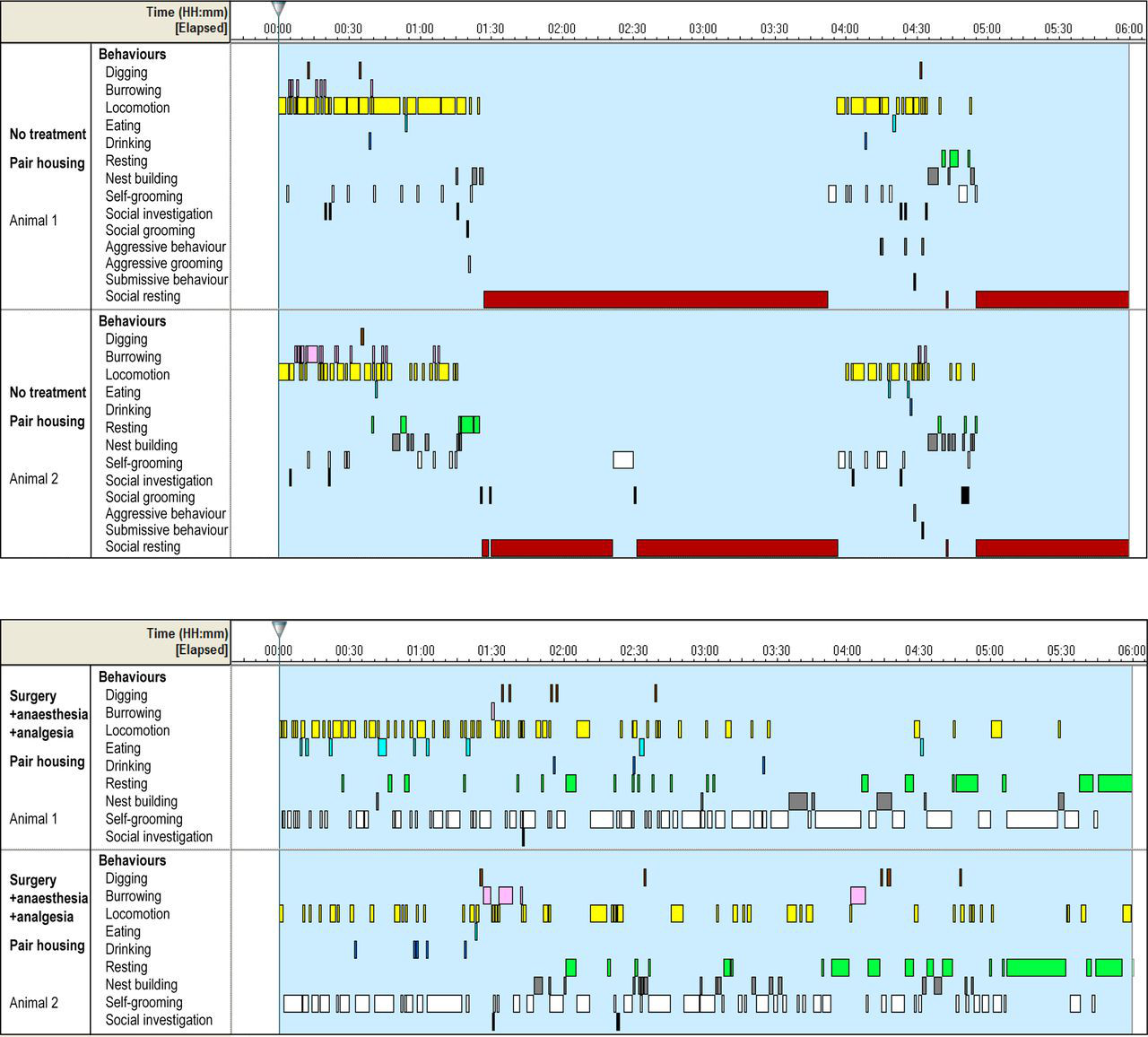
Individual behaviours
In comparison with no treatment, overall resting (for pairs, results of individual and social resting were combined), climbing, burrowing and nest building behaviour decreased, while eating and self-grooming behaviour increased in both housing conditions. Locomotion was equal to, or increased compared with, that in the no treatment group but decreased in the surgery + anaesthesia + analgesia groups. Drinking duration was equal in the surgery + anaesthesia groups while it was increased in the other groups under both housing conditions (Table 2).
Figure 3 shows scatter plots of the discriminant scores generated by the analysis for single- and pair-housed animals. The home cage behaviours analysed contributed to significant group separation. Discriminant analysis of single-housed mouse behaviour found the first function responsible for significant separation of experimental groups (Figure 3a; Wilks’ lambda; function 1, P = 0.010). This function explained 73.7% of the variance. Overall resting, locomotion and self-grooming made the largest contribution to group separation, but climbing and eating also contributed. In pair-housed mice, the first two functions were responsible for significant separation of the experimental groups (Figure 3b, Wilks' lambda; function 1, P < 0.001; function 2, P = 0.001). Function 1 explained 71.9% of the variance. Here, self-grooming and locomotion were responsible for group separation. Function 2 contributed 20.8% to separation; here, separation was based on drinking duration. Under both housing conditions, more self-grooming, eating and drinking correlated with anaesthesia or surgery, while resting, nest building, burrowing and climbing were more prevalent in animals that were not treated.
Scatter plot of discriminant scores assigned to individual mice of each housing condition in the different experimental groups: (a) single-housed mice and (b) pair-housed mice. The significance of each function in separating groups, and their percentage contribution to between-group variance are shown on each axis. Additionally, behaviours are displayed together with their correlation with each function (increase or decrease)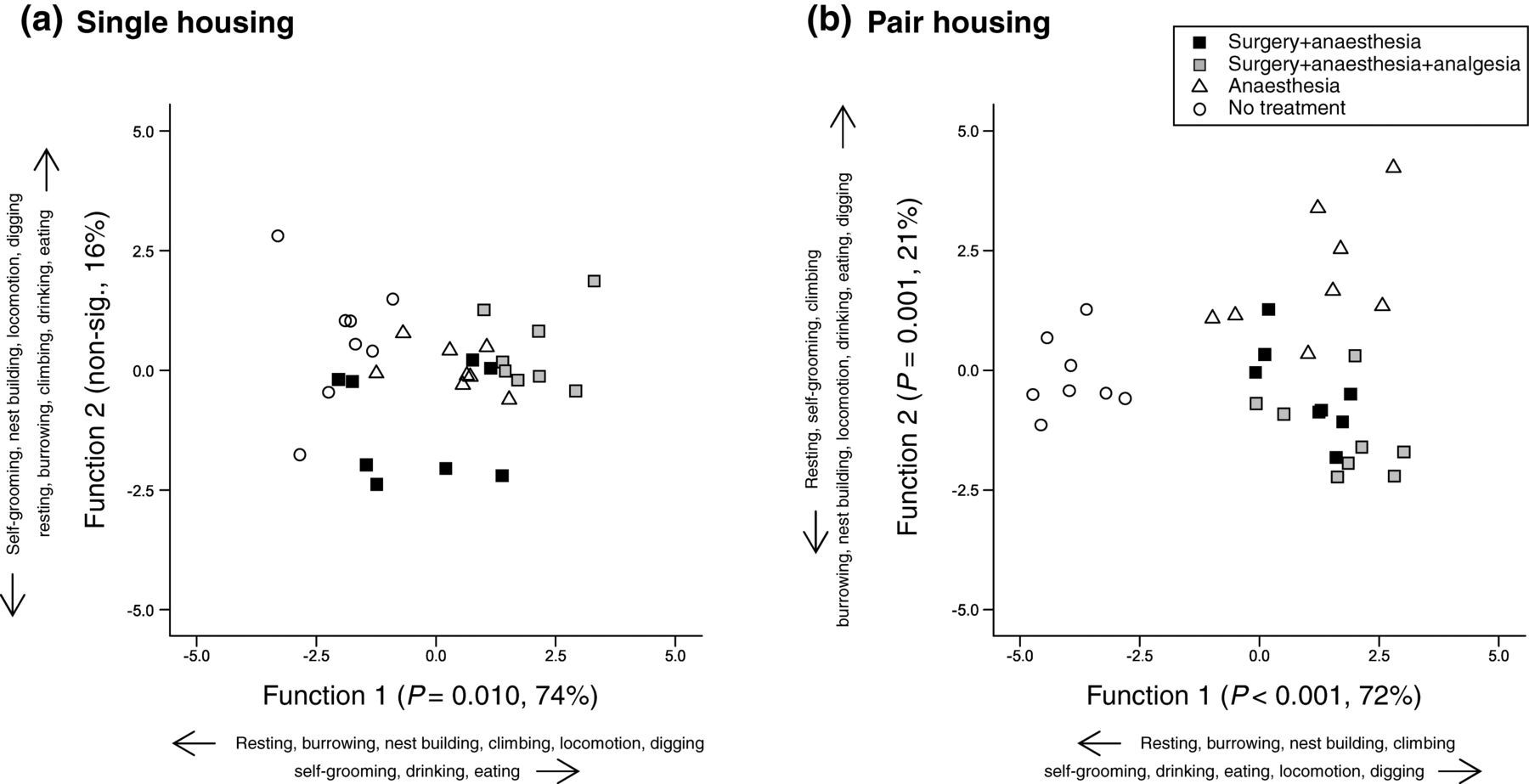
GLM was performed with behaviours that were mainly responsible for experimental group separation to test for significant differences between treatments and housing conditions in these behaviours. Significant differences between no-treatment animals and experimentally-treated animals were found in overall resting (surgery + anaesthesia: P = 0.001; surgery + anaesthesia + analgesia: P < 0.001; anaesthesia: P < 0.001; Figure 4c), climbing (surgery + anaesthesia: P = 0.041; surgery + anaesthesia + analgesia: P = 0.001; anaesthesia: P = 0.002; Figure 4b) and self-grooming (surgery + anaesthesia: P = 0.003; surgery + anaesthesia + analgesia: P < 0.001; anaesthesia: P < 0.001; Figure 4d). Additionally, significant differences were found in locomotion and self-grooming duration between surgery + anaesthesia + analgesia and surgery + anaesthesia as well as anaesthesia-only groups (P = 0.042, P = 0.002 and P = 0.015, P = 0.011; Figures 4a and d). Drinking duration was significantly different in animals that underwent anaesthesia only compared with surgery + anaesthesia and non-treated animals (P = 0.001, P = 0.003). Significant housing effects were found only in locomotion and eating duration (P = 0.025, P = 0.004). A significant interaction between housing and experimental condition was found in resting and drinking durations (P = 0.010, P = 0.046).
Mean (±SD) duration of individual home cage behaviours mostly responsible for experimental group separation. (a) Locomotion: significant differences in locomotion duration between surgery + anaesthesia + analgesia and surgery + anaesthesia as well as between surgery + anaesthesia + analgesia and anaesthesia groups (P = 0.042, P = 0.015) were found. Additionally, significant housing effects could be found (P = 0.025). (b) Climbing: significant differences between no-treatment animals and experimentally-treated animals were found in climbing (surgery + anaesthesia: P = 0.041; surgery + anaesthesia + analgesia: P = 0.001; anaesthesia: P = 0.002). (c) Resting: significant differences between no-treatment animals and experimentally-treated animals were found (surgery + anaesthesia: P = 0.001; surgery + anaesthesia + analgesia: P < 0.001; anaesthesia: P < 0.001), and a significant interaction between housing and experimental condition (P = 0.010). (d) Self-grooming: significant differences between no-treatment animals and experimentally-treated animals were found in self-grooming (surgery + anaesthesia: P = 0.003; surgery + anaesthesia + analgesia: P < 0.001; anaesthesia: P < 0.001). Additionally, significant differences in self-grooming duration between surgery + anaesthesia + analgesia and surgery + anaesthesia as well as anaesthesia groups (P = 0.002, P = 0.011) were found. Significant results are marked with * (P ≤ 0.05)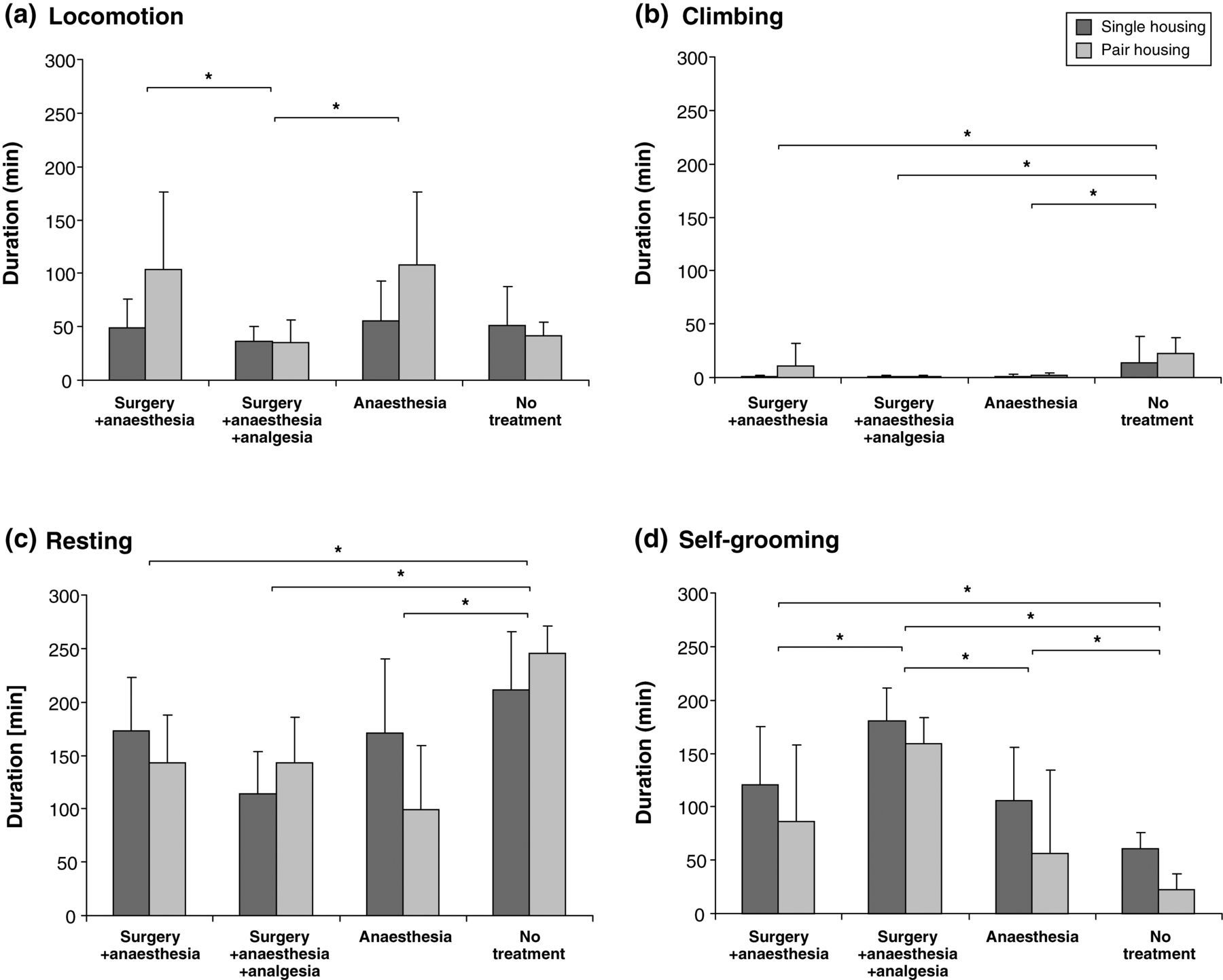
Social behaviours
In non-treated pair-housed mice, social behaviours occurred often (64% of total observation time); these were mainly socio-positive or socio-neutral behaviours like social grooming and social resting. Social behaviours decreased strongly in experimentally-treated groups (0.3–0.5%) while individual behaviours increased.
When discriminating individual and social resting in pair-housed mice, duration of individual resting was higher in all experimental groups compared with no-treatment animals, while social resting all but disappeared, and overall resting (individual resting + social resting) was lower (Table 2).
In the discriminant analysis of social behaviours, function 1 explained 99.7% (Wilks' lambda; P < 0.001) of the variance. Social resting was the only behaviour responsible for group separation and was therefore further analysed with GLM. Social resting duration was significantly longer in non-treated mice compared with other groups (each P < 0.001).
Also, the apparent synchronization of activity rhythm in pairs was lost after experiments compared with non-treated mice (see representative example in Figure 2).
Pain signs
While fall never occurred during our observations, press, stretch and stagger occurred only in treated animals but never in non-treated animals. These behaviours were very rare, low-frequency incidents and showed a high individual variability that precluded reliable analysis. Even when combining the measurements press, stretch and stagger as a composite scale, no significant difference in the number was found between experimental groups or housing conditions (data not shown). Rear up was observed significantly more often in non-treated mice compared with other groups but no significant difference in the number of observations was found between other groups or housing conditions (data not shown).
Burrowing performance
In both housing conditions, surgery without pain treatment resulted in longer latencies than surgery with analgesia, while anaesthesia resulted in the smallest increase in latency compared with no-treatment animals.
Kaplan–Meier analyses showed significant differences between non-treated animals and the experimentally-treated groups under both housing conditions (P < 0.001). Pair-housed mice showed also significant differences when surgery + anaesthesia and surgery + anaesthesia + analgesia groups were compared with the anaesthesia group (P = 0.03, P = 0.014), but differences were significant in single-housed mice only when comparing surgery + anaesthesia and anaesthesia groups (P = 0.003) (Figure 5). Comparing both housing conditions, latencies were shorter for pair-housed mice compared with single-housed mice, with an exception in non-treated animals, which was significant after surgery + anaesthesia and anaesthesia only (P = 0.050, P = 0.021) (Figure 5).
Kaplan–Meier analysis of latency to burrow. In both housing conditions, no-treatment animals had significantly shorter latencies compared with all other groups (P < 0.001). Analyses showed significant differences for pairs when surgery + anaesthesia and surgery + anaesthesia + analgesia groups were compared with anaesthesia group (P = 0.03, P = 0.014), but a significant difference only in single-housed mice when comparing surgery + anaesthesia and anaesthesia groups (P = 0.003). Latencies were mainly shorter for pairs compared with single-housed mice, which was significant after surgery + anaesthesia and anaesthesia (P = 0.021, P < 0.001)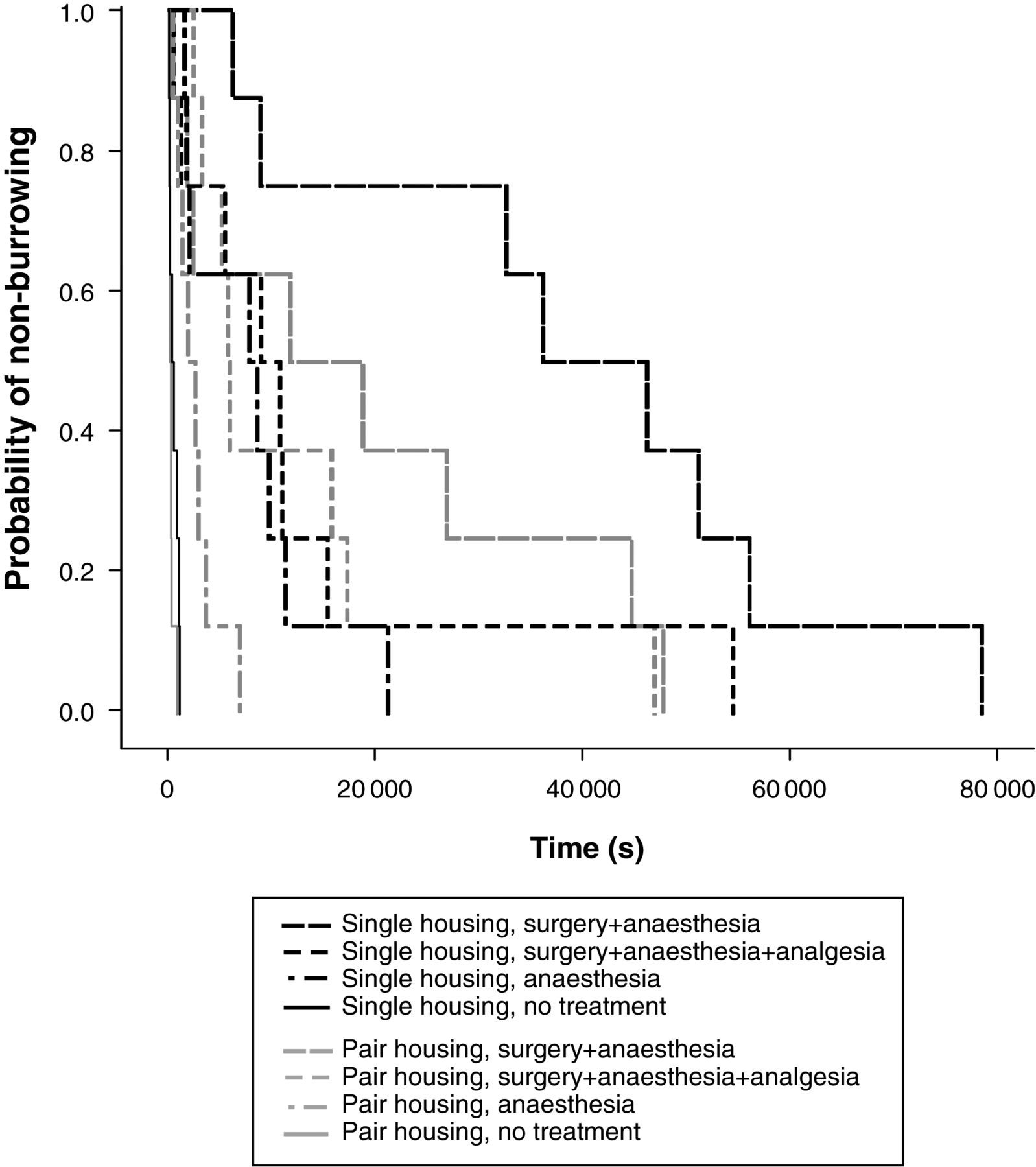
Clinical investigation
No abnormalities in appearance, posture or spontaneous movements were detected. No complications in wound healing were observed, nor any manipulation of the wound by the cage mate in pair-housed mice. No changes in body weight compared with one day before experiments occurred at either one or two days after experiments (data not shown). Clinical investigation revealed unaltered general condition scores in all groups.
Discussion
This study was set up to determine whether social or individual housing is more beneficial for postsurgical recovery and wellbeing of female mice. For this purpose, animals were monitored closely during the period immediately after surgery. Behavioural investigations revealed only slight tendencies towards better wellbeing in social housing and no adverse effects (e.g. aggression, wound manipulation by cage mate) were detected. However, social interaction, which forms a large part of the behaviour of healthy females and may be an indicator for direct social support, was nearly absent after experiments. As no pronounced detrimental effect of single housing was seen, and differences between single- and pair-housed mice were only minor, no definite conclusion can be drawn that one housing condition was clearly superior.
Clinical investigations, focusing on changes in appearance, posture and body weight, carried out daily are standard monitoring tools after surgery. Since no abnormalities were detected with these investigations, we suggest that our model has only low impact on condition, health and wellbeing, particularly in comparison with other models of surgery (e.g. Ref. 11).
Pain signs occurred only after surgery or anaesthesia and never in non-treated mice but were very variable and infrequent, with one exception. Rear up was observed significantly more in non-treated mice. Since we identified the signs from video recordings of the home cage and not a designated cage with arrangements for visibility, 13,14 some of these subtle signs may have been overlooked. However, since pain may have been less intensive in our model, pain signs may have been exhibited rarely by our animals. Altogether, no differences between treatments or single- and pair-housed mice could be established with these signs.
The rhythmicity and duration of most home cage behaviours, and the latency to burrow differed clearly between treated and non-treated mice, suggesting a decrease in wellbeing following experiments.
Healthy mice mostly rest during the light phase and show a stable circadian rhythm; disruption of this rhythm might indicate impaired wellbeing. 20 In our study, overall resting duration was decreased in treated animals, accompanied by more and shorter resting bouts, resulting in a dramatic disruption of the activity rhythm in all surgery groups, while anaesthesia alone had no pronounced effects on rhythmicity.
Nevertheless, the consequent increase in general activity, mainly due to self-grooming, seemed greater in mice that were only anaesthetized or received pain treatment after surgery. Thus, the increase in activity was presumably caused mainly by anaesthesia, while mice in pain seem to spend slightly more time in inactivity.
These observations were in accordance with discriminant analysis, which showed a significant contribution of the analysed home cage behaviours to group separation. Behaviours contributing most to this separation were locomotion, resting, climbing, self-grooming and, to a lesser degree, drinking and eating. Less resting and climbing were correlated with surgery and anaesthesia. Climbing duration was shorter in all groups compared with non-treated mice. This indicates that reduced climbing activity in our study was not due to abdominal pain 12 but rather to general impairment after anaesthesia. Eating, drinking and self-grooming were more prevalent in experimental groups that underwent anaesthesia or surgery. Changes in these behaviours may be consequences of the anaesthetic and surgical procedures. The prolonged eating and drinking duration in some conditions, especially in single-housed mice, might indicate increased food and water consumption that may help to reconstitute the animals’ health after an exhausting event. Eating and drinking increased mainly in animals that were only anaesthetized or received pain treatment after surgery, whereas animals without pain treatment did not increase their food and water intake compared with non-treated mice. This low food and water intake is probably a sign of postsurgical pain in these animals. As self-grooming was not increased after surgery without pain treatment compared with the other treated groups it is unlikely to be a specific sign of postsurgical pain. Therefore it could be correlated with the animals’ general wellbeing after anaesthesia as well as increased attention to the shaved operation field, 21 the wound or the eye ointment used.
Nest building behaviour has been identified in several publications as a useful estimate of wellbeing and pain. 12,19,22 Even though discriminant analysis did not reveal it as an important behaviour, we observed a decrease in this behaviour after surgery and anaesthesia. A slight gradation in nest building behaviour was seen in pair-housed mice, with longer durations in animals that received pain treatment after surgery and longest durations in anaesthesia-only and non-treated animals.
To summarize, even though socially- and individually-housed mice did indeed differ in several behaviours, none of these differences were significantly relevant in the experimentally-treated groups.
Burrowing behaviour is a highly motivated behaviour that has been shown to decrease after painful surgical interventions. 15 In our study, burrowing performance ranged from short latencies of non-treated animals to an intermediate level following anaesthesia only, to a pronounced prolongation of latency to burrow after surgery without pain treatment. These findings, in accordance with previous studies, 15 indicate an anaesthesia effect as well as the occurrence of pain in animals after surgery. While in both housing conditions non-treated animals had similar short latencies, following treatments latencies to burrow increased more strongly in single animals compared with pairs. The shorter latencies in treated pairs compared with treated single-housed animals hint at a better performance of pairs in the burrowing test, although both cage mates experienced the experimental procedures. This decreased interest in burrowing activity is likely correlated with better coping in pairs after experiments.
Interestingly, after experiments, pairs showed a dramatic decline in social interactions, with only short and rare bouts of social behaviours. Healthy mice show a strong preference for sleeping together in one place; 23 indeed, social resting was the social behaviour most commonly observed in non-treated mice in our study. The strong decrease in social behaviour was due mainly to an increase in self-grooming and individual resting. However, long-term observation revealed social resting even after major surgery. 12 As expected, pair-housed females displayed only very few socio-negative behaviours; hence, the risk of social stress or injury seems to be negligible in female mice after surgery.
As social behaviours almost disappeared, no actual signs of positive social interaction could be found that could hint at observable social support in the postsurgical phase. But while the proximate effects of housing conditions may not be clearly evident, they may affect how animals respond to surgical stressors in the long term. This is in line with our analysis of burrowing performance and is supported by longer term studies showing that a social partner can act as a support after abdominal surgery 11 or telemetry transmitter implantation. 12 Our behavioural observations are limited by a short observation period of only 6 h. We assume that postsurgical pain and impairment of general condition following minor surgery lasts longer than our observation period, possibly up to 24 h. 19 Therefore, we cannot exclude that any recovery-supporting effects of social housing may have been more obvious in the longer term.
It is important to consider that our animals were housed in harmonious groups before and after the experimental period and were always in acoustic and olfactory contact with other mice. Moreover, we used only female mice, which inevitably hamper the transfer of our results to male mice because group-housed males tend to fight, especially when re-grouped after transient individual housing. However, male mice also show a preference for social contact, preferring to sleep in proximity to a familiar male 23 and seem to profit from social housing after challenging events like stroke or nerve injury. 24,25
In conclusion, although behaviours related to subjective wellbeing of mice decreased after surgery and anaesthesia, most behaviours, except burrowing performance, were too variable to reveal significant differences between housing conditions. From the overall results of our study, we cannot claim that postsurgical social housing is truly superior over single housing. Nevertheless, the observed changes in burrowing performance suggest a tendency towards better wellbeing in pair-housed animals after surgery. Thus, although both housing conditions are acceptable in female mice at least for 6 h after surgery, social housing might be in general preferable. Conversely, single housing has the advantage of allowing accurate monitoring of individuals, 26 which is advised in many situations, e.g. humane endpoint anticipation. Therefore, in our view, the decision on how to house female mice after surgery should be made on a case-by-case basis, considering various aspects of laboratory routine, legislation and – of course most importantly – possible impacts on animal wellbeing.
Footnotes
ACKNOWLEDGEMENTS
This work was sponsored by grants from the Federal Veterinary Office (Bern, Switzerland) and UBS foundations. The authors would like to thank Robin Schneider and the staff of the Central Biological Laboratory for support in housing mice. We thank Professor Kurt Bürki for generously providing research facilities and resources and Professor Burkhardt Seifert and Alexandra Hoffmann for statistical advice.
