Abstract
Some laboratory mice gnaw food pellets without ingesting much of the gnawed material, resulting in the production of waste material called ‘orts’. The fact that this food grinding behavior is not seen in all individuals of a particular strain suggests that it might be abnormal, and thus indicate a welfare concern. Furthermore, the increased rate of feed consumption and cage soiling is undesirable from a husbandry perspective. To try to determine possible motivations for the behavior, and identify potential treatments, outbred Crl:CD1(Icr) mice exhibiting food grinding were selected for one of three treatments placed in the feeder: no enrichment, a chewing device, or sunflower seeds. Both enrichment groups showed a significant decrease (P < 0.05) in ort production when compared with baseline measurements, but only mice provided with sunflower seeds maintained the decreased rate of food wastage after the treatment was withdrawn. A relationship between body weight and ort production was also found, in that cages with greater average body weights had lower levels of ort production. This suggests that a simple need to gnaw cannot alone explain food grinding, and that a nutritional motivation may also be involved.
Laboratory mice may exhibit various abnormal behaviors in standard laboratory housing. 1 The ontogeny of abnormal behavior is complex, but usually reflects the animal’s attempt to cope with an inescapable stressor including an unmet need, or the absence of a critical behavioral substrate. Food grinding is an abnormal behavior seen in laboratory mice as well as other rodents kept in the laboratory. 2 It is widely assumed that food grinding does not involve the ingestion of food, 2 but this has not been shown – therefore it is instead better defined as the production of excessive food waste, called ‘orts’. Individual differences in food grinding are highly stable within individuals, families, and lines, indicating either an effect of maternal condition and/or heredity. 3 We hypothesized that motivations for food grinding include the need to gnaw innate to rodents, or a drive to interact with seeds present in a granivorous herbivore such as a mouse. 4 The first hypothesis has not been clearly tested although considerable evidence exists for the second, e.g. in rats. 5 Mice show a species-typical behavior with seeds or nuts in the wild, gnawing and discarding the shell to obtain the kernel within. In this study, we compared two materials; one provided an outlet for gnawing behavior, while the other allowed for the motivation of an appetitive interaction with seeds. We aimed to elucidate which of these motivations was driving the food grinding behavior.
All the work with animals was approved by Charles River’s Institutional Animal Care and Use Committee. Cages showing food grinding were selected from a larger colony. Female Crl:CD1(Icr) (CD1) mice were housed in individually-ventilated solid-bottomed cages (Innovive, San Diego, CA, USA). Cages were bedded with irradiated heat-treated aspen shavings (NEPCO, Warrensburg, NY, USA), mice were provided with nesting enrichment (10 g of Enviro-Dri, Shepherd Specialty Papers, Watertown, TN, USA), and water and feed (Purina, St Louis, MO, USA) were provided ad libitum. The animals were free of a commonly accepted list of excluded organisms. 6 They were not age-matched and were located on various rows and sides of the ventilated racking. One week prior to implementation of any treatment, the amount of food provided was weighed. After seven days, the amount of food remaining was weighed. Ground feed (orts) was sifted out of the soiled bedding into a separate container using a food strainer. This served as a baseline for the amount of orts produced prior to treatment. Feed, orts, and mice were weighed every seven days during treatment. Eight cages containing 3–5 female CD1 mice were randomly assigned to either a control group or one of two treatments for four weeks. Measures were repeated for an additional week after treatments were withdrawn. Treatments were the placement of either a chew object or sunflower seeds in the feeder of the cage. Certified nylon chew objects (I-Chew, ASAP, Quakertown, PA, USA) were wedged into the feeder and examined weekly to determine if the animals had used the gnawing device. Fifty grams of autoclaved sunflower seeds (the fruit of Helianthus annuus; Kaytee, Chilton, WI, USA) were placed in the feeder, mixed with the normal diet, and replenished weekly. Unopened or uneaten sunflower seeds were extracted from the feed or bedding prior to weighing or ort separation so that neither feed weight nor ort weight were affected by inclusion of sunflower seed shells.
Ort difference from baseline (baseline ort production – time point ort production) was analyzed as a mixed model analysis of variance (ANOVA) using GLM, in JMP 9 (SAS, Cary, NC, USA). Repeated measures and individual differences between cages were accounted for by blocking by cage nested within treatment. Treatment, time point, and their interaction, and mean body weight for the cage at each time point were tested. Age (41–134 days of age) was also assessed as a covariate in the model but was removed because it did not significantly predict ort production. Bonferroni corrected planned-contrasts were performed, post hoc, to determine differences between treatments, time points, as well as differences from baseline ort production.
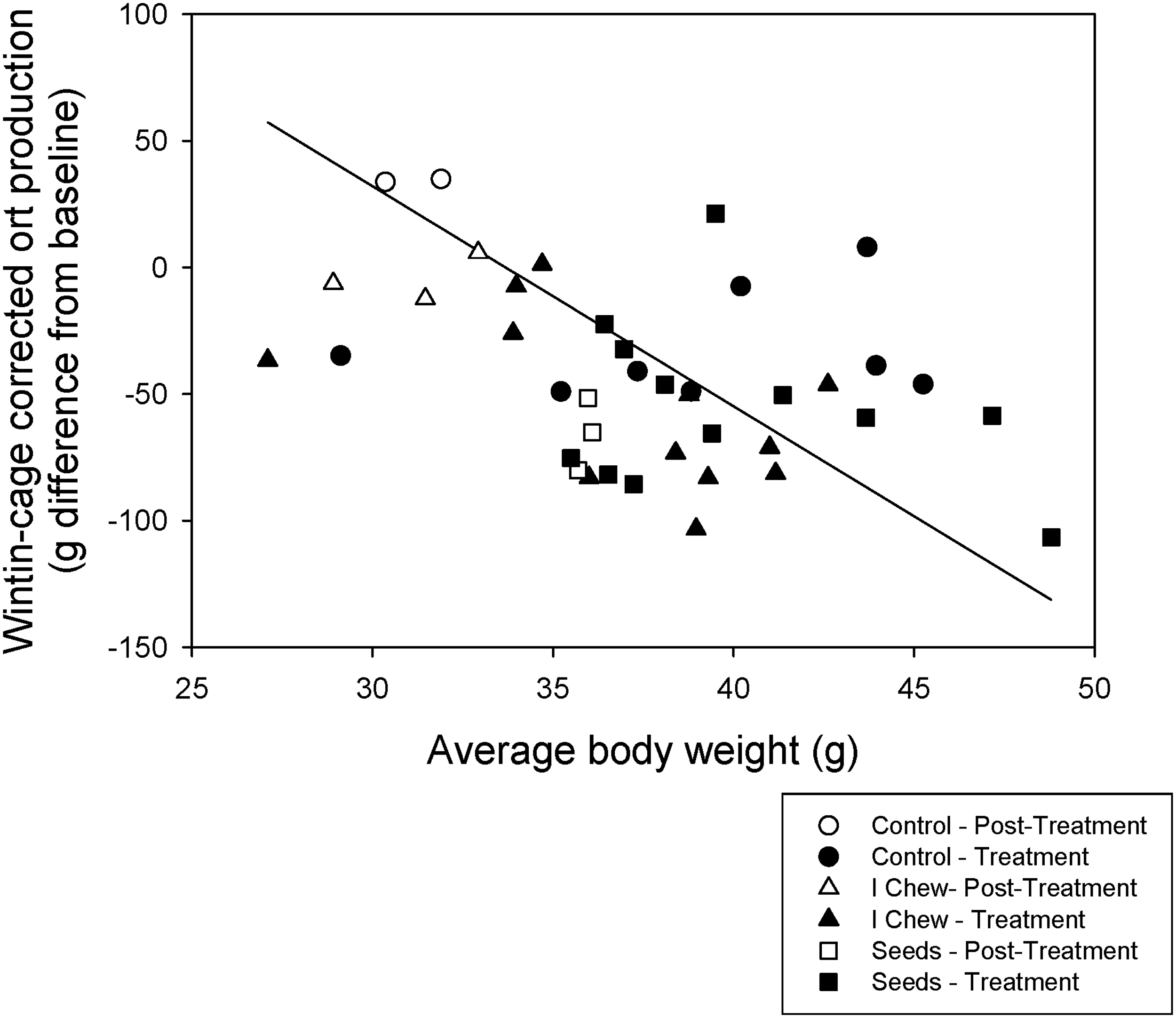
Average baseline ort production for all the cages assessed was approximately 161.4 g/cage over seven days. Both enrichments decreased ort production (P < 0.05; F2,28 = 4.4) significantly during the treatment period (Figure 1). Chew objects, however, did not result in a significant decrease in ort production once the chew object was withdrawn, as ort production returned to baseline levels. Animals treated with sunflower seeds, however, did not return to baseline ort production levels. Within each cage, heavier average body weight decreased ort production (P < 0.001; F1,28 = 18.4; Figure 2). These data suggest that food grinding behavior is successfully reduced in female CD1 mice when provided with a gnawing enrichment. However only sunflower seeds maintained this reduced ort production for seven days after the removal of the enrichment.
Least squares mean (LSM) and standard error (SE) values of the difference from baseline ort production during and after treatment are plotted on the y-axis. Negative values indicate a reduction from baseline ort production. Gray bars behind plotted data depict the 95% confidence interval (CI) of each category. Significant t-tests (value different from zero or baseline; α corrected for the number of comparisons) are indicated by asterisks. Different letters indicate significant differences between treatments (α corrected for the number of comparisons). A significant contrast (P < 0.05) between the I-Chew and sunflower seed treatment is indicated by ‘†’. Ort production decreases as the average cage body weight within the cage increases. The analysis explicitly controls for cage (and hence baseline differences between cages in body weight). The value of ort production plotted controls for this baseline difference.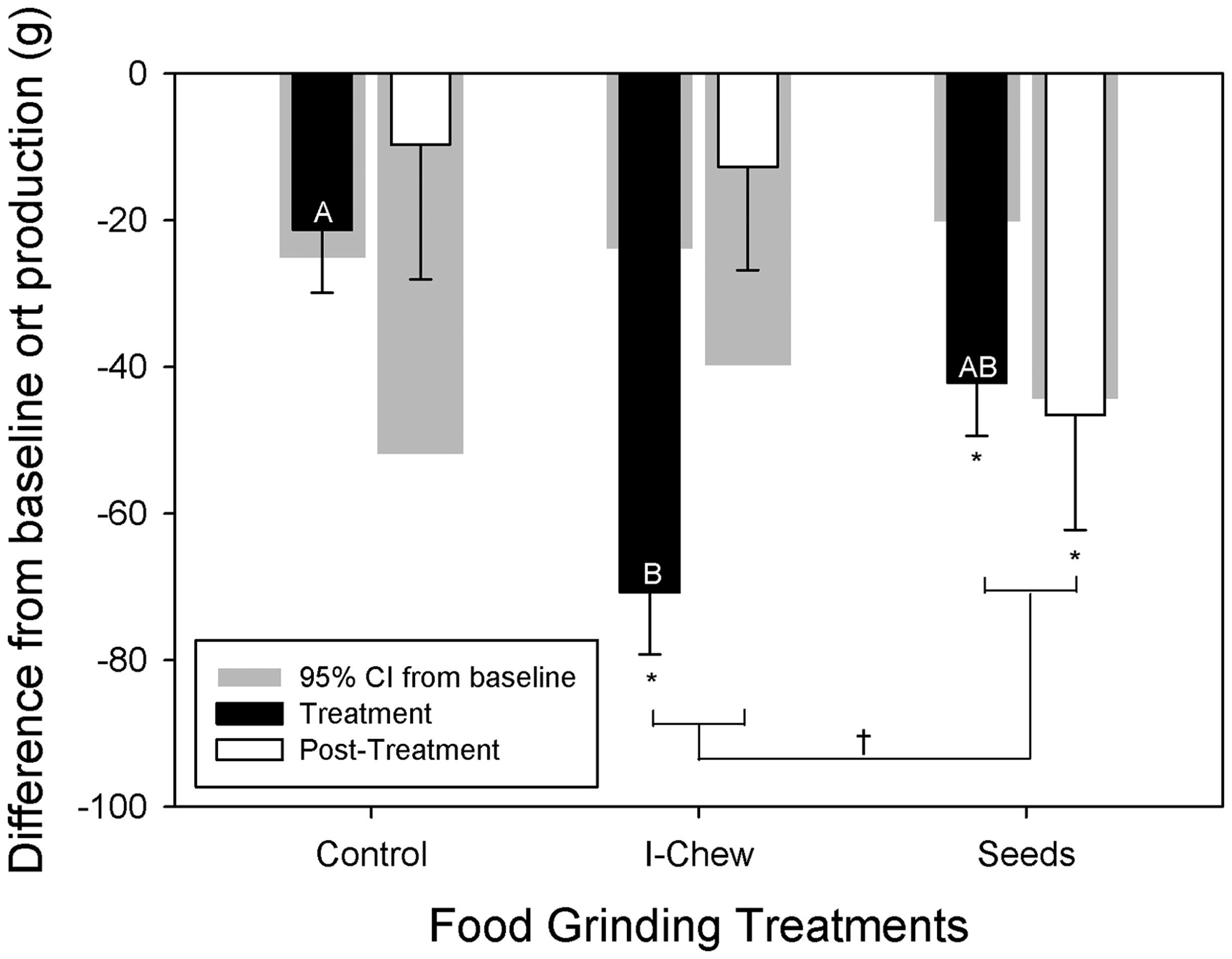
As granivores, rodents also spend time sorting, processing, and manipulating seeds. Since food is usually provided in feeders, the animals cannot manipulate the food pellets as they might seeds, 7 and instead they have to gnaw the pellets as they remain in place, and only become able to grasp them when they become too small to remain within the feeders. Providing rodents with food in a feeder is required by regulation in many countries in order to keep the food from being contaminated with urine or feces, although this requirement is waived for animals known to hoard, such as hamsters and gerbils. This regulatory mandate may be driven by anthropomorphism, however, as rodents use urine marking to note safe, accessible food sources.8,9 Feeding mice on the floor of the cage may allow the exhibition of species-specific behaviors and decrease the incidence of food grinding. We did not examine this premise in this study and, to avoid confusion with a drive to forage, did not provide either the chewing device or the sunflower seeds on the floor of the cage.
The food provided to animals in this study was the caloric restriction (Charles River, Wilmington, MA, USA) US standard which contains approximately 6% fat and 18% protein; while sunflower kernels are 51% fat and 21% protein. 10 Of particular importance in mice, the energy content per gram of orts is lower than that of the food pellets, 11 suggesting that the mice are selectively consuming the higher energy/g components of the diet which is consistent with the increase in food grinding seen at lower temperatures. 3 When mice only have access to chow, their frustrated granivory may be manifested by the discarding of the fiber and carbohydrate components of the chow as orts, as they would seed or nut shells. Perhaps this behavior is elicited when mice face high-energy demands, such as with the thermal stress incurred at typical laboratory temperatures 12 which is reduced at heavier body weights. To further refine and test the hypothesis, an important follow-up experiment would be to provide feed with a nutritional density similar to seeds or nuts, perhaps in a factorial design with both hulled and unhulled sunflower seeds. This would allow further separation of the contributions to food grinding of a behavioral need to gnaw from that for increased nutritional density. Follow-up studies might also be designed to assess treatment for a longer period and to include a greater number of baseline measurements.
Footnotes
Acknowledgements
R Keefe is a student at Colby-Sawyer College, New London NH, USA. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
