Abstract
Stress can influence a number of physiological processes including adult neurogenesis, metabolism, cardiovascular function, immune function, neurophysiological function, endocrine function and inflammatory processes following injury. In testing drugs which may be used to treat various diseases or injuries, reducing stress associated with chronic drug delivery to animal models should then be an imperative, which led us to design a reliable voluntary oral drug delivery method. Various drug combinations were tested versus vehicle controls in four different rat stocks or strains (Wistar, Fisher, Long Evans and Sprague Dawley) with our voluntary oral delivery system. Oral medications were placed into a store-bought sugar cookie dough ball (∼4 g), thoroughly integrating the dry drugs with the dough. This method has worked consistently to deliver the medication (complete ingestion) in four different stocks or strains of rats, with reliabilities ranging from 98.6% to 100%. The percentage of rats in each stock or strain that have at any time during the study had incomplete ingestion of the drugs ranged from 1% in Sprague Dawley, approximately 4% in Wistar and Fisher, to approximately 16% in Long Evans. Both serum and brain samples were analysed for high-performance liquid chromatography (HPLC) detection of one of our administered drugs: 5 mg/kg fluoxetine. HPLC analysis shows that serum levels are detectable 2–4 h after ingestion, but not 24 h after ingestion. Brain samples however, showed detectable levels of both fluoxetine and norfluoxetine more than a week following ingestion of a single dose, with higher norfluoxetine levels seen following a month of daily administered drugs.
Injury-induced adult neurogenesis has been studied in both the lateral brain ventricles (subventricular zone) and the dentate gyrus of the rat. Following stroke in adults or hypoxia ischaemia in neonates, the neurogenesis in the lateral ventricles is increased and newly born neurons migrate to the site of injury, rather than following the normal path along the rostral migratory stream to the olfactory bulb. 1,2 Adult neurogenesis has also been shown to be inhibited by stress and depression. 3–5
When testing post-stroke medications that might enhance neurogenesis, it became important that we deliver the medication in a stress-free manner. Chronic intraperitoneal injections have been shown to significantly increase corticosterone levels (stress hormone) in rats when given once daily for 14 days. 6 Corticosterone levels have also been shown to be increased following oral gavage. 7 Recently, voluntary oral administration of drugs has been tried in both mice and rats in an effort to reduce stress in chronic dosing. 8,9 Since we used generic and brand name forms of the drugs which are given orally to humans, we only had to make sure that the food that we used as drug vehicle has not been shown to interact with the drugs and alter their efficacy. Since the two main drugs we have administered have been given to humans for over 20 years, there was sufficient information available about any possible food interactions with the drugs.
Very few experimental protocols allow the animal to eat their medication in a chronic dosing experiment, due to problems with reliability of ingestion of the full dose or ability to control the amount of drugs received (fixed dosage in mg/kg) if the drugs are in water or normal feed. A variety of palatable substances were tested which could be used to induce the animal to eat the full drug dose in a reliable manner. This was made more challenging because one of our drugs, fluoxetine (Prozac™; Eli Lily & Co, Indianapolis, IN, USA), is very bitter tasting and is normally encapsulated. In these studies, the dosage was adjusted to the animal's body weight, so individual drugs were weighed out for each rat and then fully mixed into its vehicle and labelled for each individual rat. We provide data on reliability of this method of chronic drug delivery in four different stocks or strains of rats. In our most recent work, we have also detected serum and brain concentrations of fluoxetine and its active metabolite norfluoxetine at various times following drug ingestion in Sprague Dawley and Long Evans rats using reverse phase high-performance liquid chromatography (HPLC). 10,11 .
Materials and methods
Animals
Drugs administered to rat stock(s) in sugar cookie dough balls
Pilot studies
Seven different foodstuffs were tested for use as a vehicle for oral administration of our drugs. In the first test, equal amounts of the substance were left in the rat's cages overnight and then we evaluated which foods the rats had completely eaten. Out of the seven foodstuffs tested, the rats completely ate brunschweiger, Pillsbury sugar cookie dough, Pillsbury peanut butter cookie dough and Pillsbury chocolate chip cookie dough. The rats either did not eat Bioserv Transgenic Dough Diet, Napa Nectar (Systems Engineering, Napa, CA, USA), or Fruit Loops (Kellog, Battle Creek, MI, USA) or failed to eat even half of these foods. There is the possibility that if we had allowed more time for the animals to acclimatize to these foods that they would have been useful as vehicles for voluntary oral delivery, at least for short time periods. However, we were most interested in high reliability of ingestion for prolonged periods of time (32 consecutive days), so we chose to focus on those foods which the rats ate completely with a single exposure. In evaluating these substances for further use, the consistency of the sugar cookie dough made it ideal for weighing and mixing in the dry drugs without losing any of the dough on gloves. In addition, the simplicity of the sugar cookie dough ingredients gave it an advantage, so it was chosen for the rest of the study.
Figure 1a shows the method used to make our sugar cookie dough balls containing drugs. The dough balls were prepared in 60 mm glass Petri dishes, because rats will eat part of either plastic weigh boats or plastic 60 mm Petri dishes if they contain cookie dough. First we made a dough ball approximately 4 g in weight. We placed this in the Petri dish and made an indentation in the ball with a gloved finger. The drugs were weighed out on glassine weighing paper and then put into this indented region. We then brought two sides together, sealing the drugs inside the dough. The dough was then picked up, mixed thoroughly and reshaped into a ball, and placed back in the Petri dish. We then put a disposable small hexagonal weighing boat with the rat's identification number over the ball: this was removed before we gave the dough to the rat. Figure 1b shows one of our Fisher rats eating her cookie dough ball containing drugs approximately 3 min after it was placed into her cage; most rats ate their complete ball of drugs within 5 min of drug delivery. Although most of the cookie dough was consumed quickly, the occasional rat prefered to eat the ball of dough slowly over time. The cookie dough was left in the cage overnight, and the Petri dishes were only picked up the next day right before the next day's drugs were given. We rarely saw any uneaten cookie dough at that time, and we searched both bedding and dish for any evidence of this. We counted each time there was uneaten sugar cookie dough left the following day as a count against reliability of the method. Since we typically delivered drugs for 15–32 consecutive days, each rat was scored for whether they ate or missed part of their drugs for that day, including the vehicle control rats. Drug ingestion reliability was determined by the following equation, which provides a percentage of complete ingestions of the drugs: Method for voluntary oral administration of drug delivery. (a) Picture showing method of making dough balls containing medication. Starting from the far left and moving right: (1) a dimple was made in the cookie dough ball; (2) the drugs (see Table 1 for specific drugs given) were weighed and put into the dimpled region using glassine weighing paper; (3) the cookie dough edges were sealed by pressing together, making sure the drugs were locked inside; and (4) the dough was picked up and reshaped into a ball. (b) Picture of a Sprague Dawley rat eating the cookie dough ball containing drugs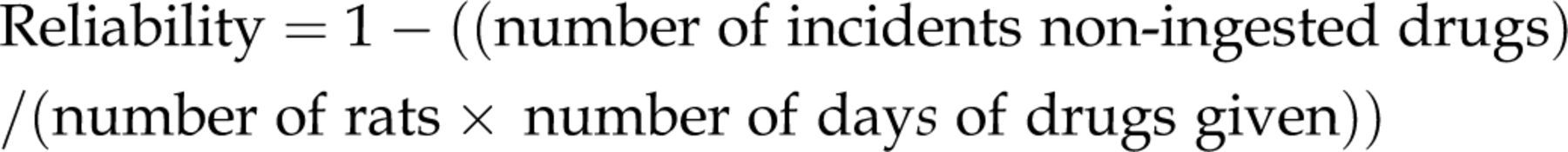
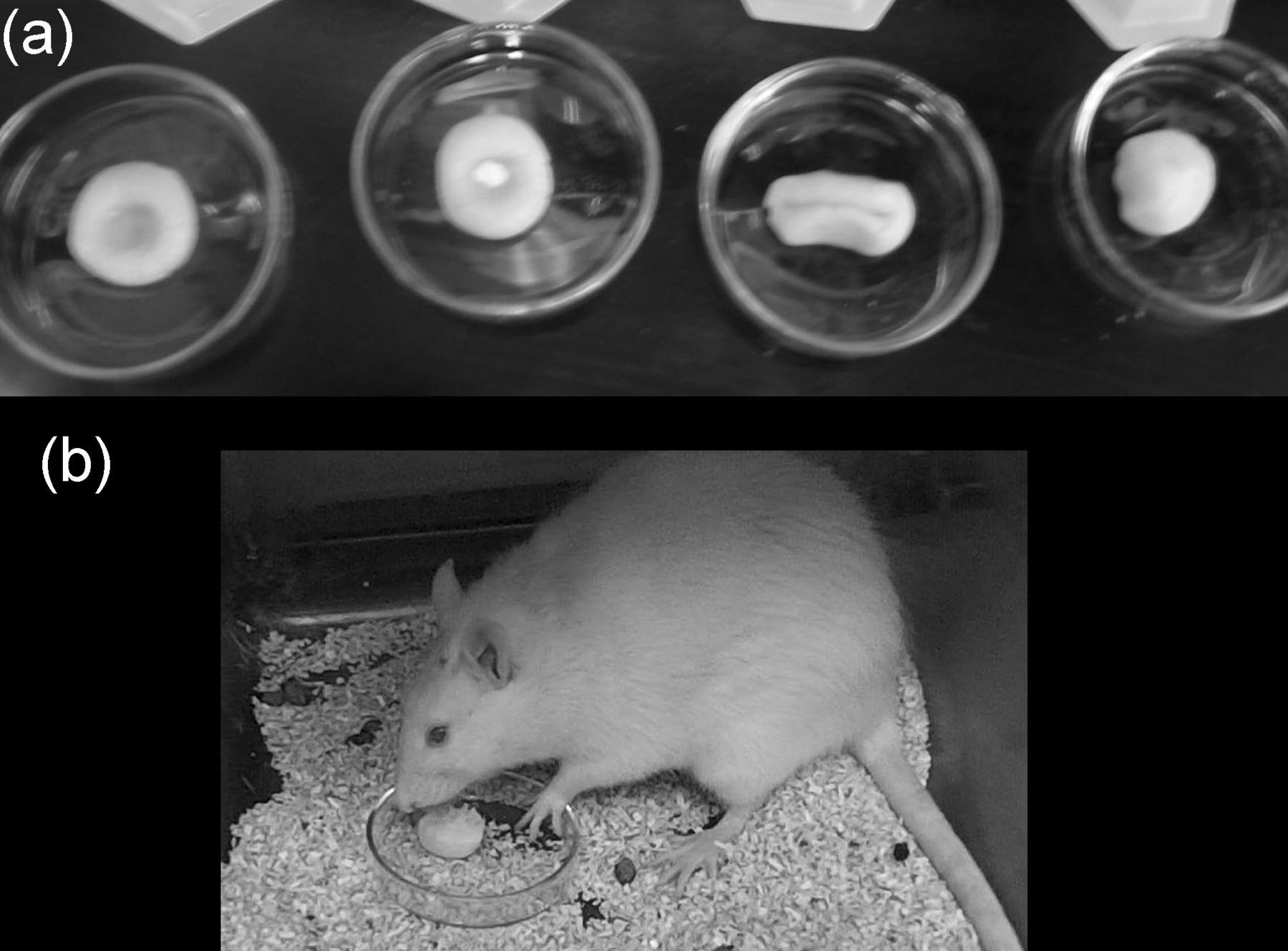
Prior to delivering any drugs to the animals, they were given plain sugar cookie dough (no drugs) for three days: this allowed the animals to get over their aversion to novel food prior to starting the drugs. We typically gave the animals their dough balls at approximately the same time we would give them their drugs: within the time frame from 11:00 to 13:00 h. All animals were on
Over the scope of this project, we have tested a number of different drug combinations, many of them containing 5 mg/kg fluoxetine as one of the drugs (see Table 1 for complete names and dosage of drugs administered for each rat stock). We detected the fluoxetine and its primary metabolite norfluoxetine in both blood serum and brain homogenate as a test of the drug delivery method (see below for the method). Every group of animals had a control group that received just the sugar cookie dough vehicle with no added drugs (Wister: 6 control rats; Fisher: 17 control rats; Long Evans: 13 control rats and Sprague Dawley: 25 control rats). For the drug combinations, each individual drug was weighed and combined in the centre of the sugar cookie dough vehicle. The following drugs were used either alone or in various combinations, with no more than four drugs in combination: fluoxetine at 5 mg/kg (generic as well as Prozac™), simvastatin at 0.5 mg/kg (generic as well as Zocor™; Merck & Co, Inc, Whitehouse Station, NJ, USA), ascorbic acid at 20 mg/kg (Sigma-Aldrich, St Louis, MO, USA),
Serum collection
We collected blood via cardiac puncture following anaesthesia by an overdose of pentobarbital (euthasol). This was performed immediately after we initiated cardioperfusion with saline in the left ventricle, collecting the blood from the right atria. The blood was collected in 0.5 mL of 0.129 mmol/L sodium citrate, then centrifuged at 13,000 × Chromatograms of fluoxetine (FLX) and norfluoxetine (NFLX) levels in blood serum and brain homogenate. (a) Chromatogram of standards, showing elution time of both NFLX and FLX. (b) Representative chromatogram from blood serum of a rat which received daily drug treatments for 32 days, with the last drug ingestion approximately 3–4 h before serum collection. (c) Representative chromatogram from brain homogenate supernatant. Brains were homogenized approximately 24 h after the last drug ingestion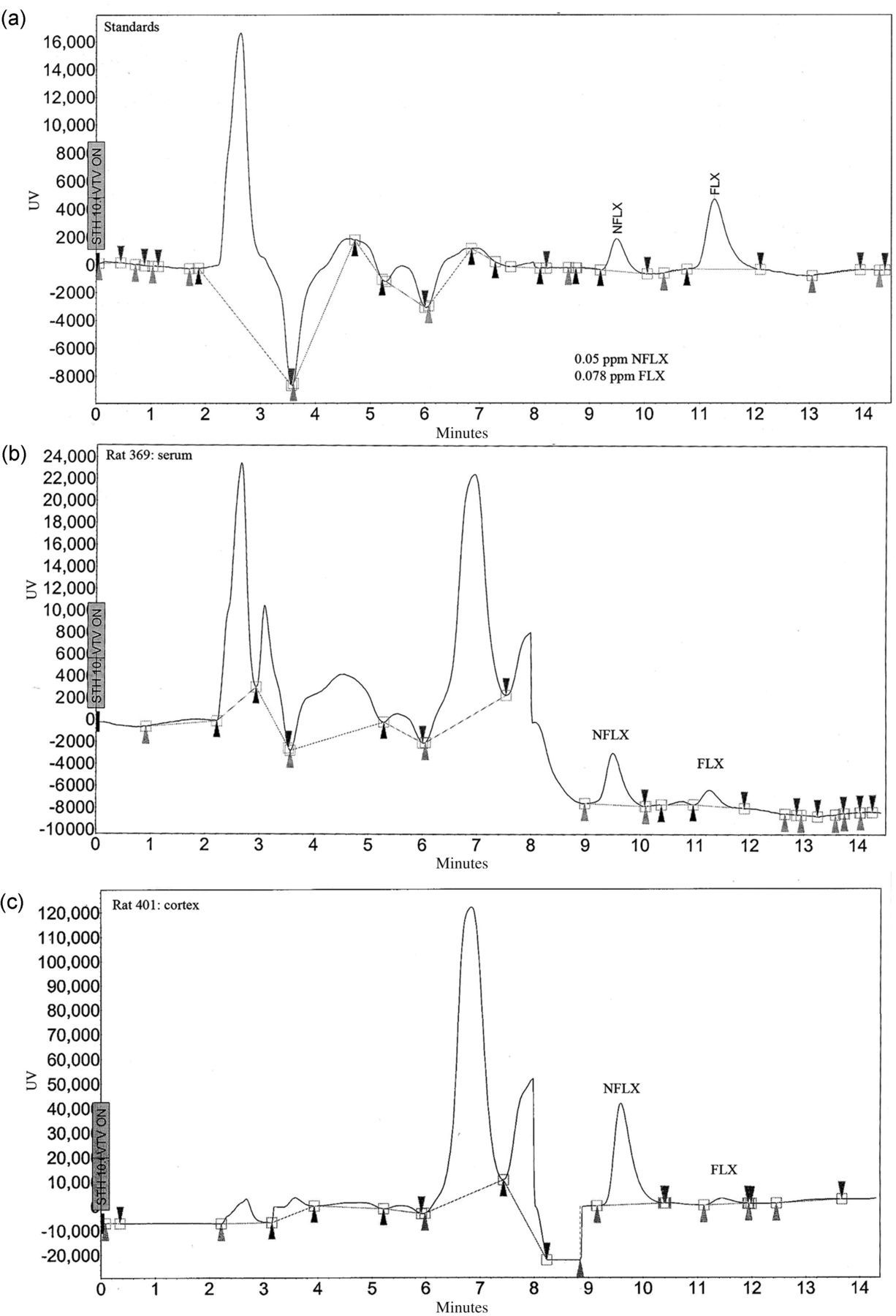
Brain homogenization
Unfixed brains were collected via dissection following euthanasia (pentobarbital overdose via euthasol injection, followed by decapitation) and placed in phosphate-buffered saline on ice. Approximately 100 mg of brain tissue from the cortex was dissected and then homogenized with a Tissue Tearor in 1 mL of water. The resulting homogenate was centrifuged at 13,000 ×
HPLC analysis of fluoxetine and norfluoxetine
The purpose is to separate and quantify fluoxetine and its major metabolite norfluoxetine in serum and brain tissue using reverse-phase liquid chromatography with fluorescence detection, according to the method of Unceta
Reagents and materials
Fluoxetine hydrochloride (FLX) [59333–67–4] was purchased from Calbiochem (La Jolla, CA, USA; Cat. #343290M Lot# D00095949, 25 mg) or from Sigma-Aldrich (Cat. # F132–10 mg, lot 129K2148) and its primary active metabolite, norfluoxetine hydrochloride (NFLX) [83891–03–6], were purchased from Sigma-Aldrich. Sodium acetate buffer (0.050 mmol/L) was prepared from sodium acetate (Fisher Scientific, Inc, Fairlawn, NJ, USA) and glacial acetic acid (VWR brand, West Chester, PA, USA). Borate buffer (0.1 mmol/L) was prepared from boric acid, H3BO4 [10043–35–3] (Sigma, St Louis, MO, USA; B9645–500G) and sodium hydroxide [1310–73–2] (Fisher Scientific). Perchloric acid, 70%, was obtained from EMD Chemicals, Inc, Philadelphia, PA, USA. Solvents were HPLC-grade acetonitrile (Pierce, Rockford, IL, USA) and water purified using a Milli-Q system (Millipore Corporation, Danvers, MA, USA).
Stir bar sorptive extraction (SBSE) was performed using GERSTEL-Twister® sorptive stir bars (GERSTEL GmbH & Co. KG, Eberhad-Gerstel-Platz, Germany; 011222–001–00) obtained from Agilent Technologies (Santa Clara, CA, USA). The stir bars are 10 mm long and are coated with a 0.5 mm film thickness of polydimethylsiloxane. Extractions were carried out in Fisherbrand 21×70 mm amber glass vials (Fisher Scientific, Pittsburgh, PA, USA). Desorptions were performed in Varian 4.0 mL clear glass vials with PTFE/sil septa containing Agilent 400 μL glass inserts (Varian, Inc, Palo Alto, CA, USA).
Instrumentation
Chromatographic separations were carried out on a Varian ProStar HPLC system (Varian, Inc) with Galaxie software (Varian, Inc), a Varian ProStar Model 410 autosampler (Varian, Inc), and a Hitachi Model L-2485 Elite LaChrom fluorescence detector (Hitachi, Pleasanton, CA, USA). Separations of 100 μL injections were achieved on a GRACE Platinum C18 reverse-phase column (250 mm × 4.6 mm, 5 μm particle size; Grace, Deerfield, IL, USA). The mobile phase consisted of a 30:70 (v:v) of 0.050 mmol/L sodium acetate buffer (pH 4.5) and acetonitrile delivered isocratically at a flow rate of 1.0 mL/min. The fluorescence detector was set at 228 nm (excitation) and 284 nm (emission). The retention times for NFLX and FLX are 9.3–9.5 min and 10.9–11.5 min, respectively.
Method validation
Individual stock solutions were prepared of 550 mg/L of FLX and 490 mg/L of NFLX in acetonitrile. The solutions were stored in the freezer.
Calibration standards for high-performance liquid chromatography analysis of fluoxetine (FLX) and its active metabolite norfluoxetine (NFLX)
Calibration curves were linear over the entire range of calibration with
Stir bar sorptive extraction of fluoxetine and norfluoxetine from serum and brain tissue
Brain tissue was weighed (∼100 mg) and homogenized after adding 1 mL ddH2O. Rat cortex 402 was a control sample to which a spike was added to determine SBSE recoveries. The recovery spike consisted of 10 μL of the 55.0 ppm FLX and 98.0 ppm NFLX mixed standard to yield a final brain spike of 550 ng and 980 ng of FLX and NFLX, respectively.
Prior to use, each Gerstel® stir bar was washed with acetonitrile for 20 min in a 15 mL vial with the magnetic stirrer set at 900 rpm at 75°C, rinsed with ddH2O, and patted dry with a lint-free tissue. One millilitre of 0.1 mmol/L borate buffer was added to each brain tissue sample and a stir bar was added. Each sample was stirred at 900 rpm at 75°C for 45 min and allowed to cool to room temperature. The stir bar was removed with a magnet on the outside of the extraction vial. The stir bar was rinsed with ddH2O and patted dry with a lint-free tissue. For desorption, the stir bar was placed into a 2 mL sample vial with a glass vial-insert into which 0.300 mL acetonitrile had been added. Vials were capped and the analytes desorbed by magnetic agitation at 900 rpm and at 75°C for 30 min. Each vial was cooled slightly before opening to remove the stir bar. The vial caps were replaced and the samples analysed immediately by injecting 100 μL into the HPLC. The samples were also analysed by liquid chromatography-mass sepectometry after being stored at −20°C for three days.
Concentration and percent recovery
The measured concentrations were converted by the following equation to obtain the total nanograms of FLX and NFLX: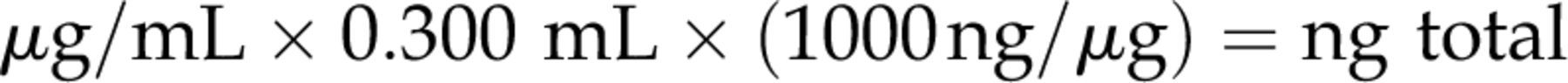
Percent recovery values were determined for Rat 402 (spiked control) by dividing the recovered concentration by the spiked concentration according to the following equation: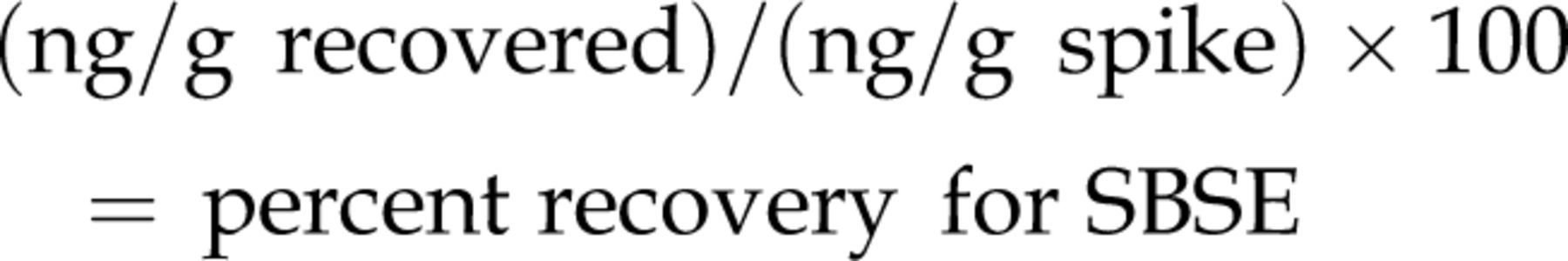
Statistics
The Fisher's exact test variation of chi-square test was used. Significant differences are seen with a
Results
Drug ingestion reliability
Incomplete drug ingestion incidents were noted when any more than 100 mg of cookie dough were found in either the cage or the Petri dish from the previous day
Concentrations of fluoxetine (FLX) and norfluoxetine (NFLX) in brain homogenates
*This control rat ingested food containing the fluoxetine (single dose), approximately one week prior to euthanasia
†Due to an integration error in the high-performance liquid chromatography/fluorescence chromatogram for Rat 402 (spiked control), the result from liquid chromatography-mass spectrometry analysis of the same samples was used in the calculation to determine percent recovery values
Discussion
Voluntary oral administration of drugs to rodents has been used before, using Nutella in mice, 8,13 jello in rats, 14 syrup in rats 9 or a syringe containing a sucrose-based solution with and without drugs. 15 It has already been demonstrated that voluntary oral administration of drugs eliminates an increase in stress hormone corticosterone compared with subcutaneous administration of the same drug. 13 When voluntary oral dosing 15 was compared with gavage, for certain drugs which normally were metabolized rapidly in the liver, oral administration allowed for more drug delivery to the bloodstream than the gavage method, possibly through absorption through lingal or buccal membranes. Since four of the drugs we were testing were lipid soluble, which should enhance absorption into lingal and buccal membranes, this gave an advantage to using a voluntary oral administration method rather than oral gavage. In most of the previous studies on voluntary oral administration of drugs, the drugs were not given daily for a prolonged period of time (a 2–4 week period) or evaluated for reliability of ingestion in several different stocks or strains of animals. Here, we demonstrate that purchased sugar cookie dough can be easily combined with dry weighed drugs for a fixed dosage (mg/kg) and reliably ingested by at least three different stocks or strains of rat (Wistar, Fisher and Sprague Dawley) for a prolonged period of time (up to 35 days in these studies). The Long Evans rat showed a significantly higher rate of incomplete ingestion than the other stocks or strains, with one animal (receiving control vehicle) repeatedly refusing to completely consume the vehicle dough ball on eight separate days within a 31-day period, so we would not recommend this rat stock for this method of voluntary oral administration of drugs.
While the sugar cookie dough method works very well with dry drugs, which can easily be incorporated into the dough, the other methods using syrup or a sucrose solution would be preferable for individuals who prefer to use a liquid drug stock. Sugar cookie dough requires less preparation time than incorporating drugs into jello, but again a liquid drug could be incorporated into the jello easily. Some of our drugs have very low solubility in aqueous solutions, so voluntary oral delivery methods which depend on dissolving the drugs were not an option for us.
Detection of fluoxetine and norfluoxetine peaks in brain homogenates can pick up any mistakes in drug administration even following a single dose administration a week before euthanasia, which provides a convenient check against mistakes by laboratory staff. We were only able to detect serum levels of fluoxetine or norfluoxetine soon after ingestion (within 4 h of ingestion) using our method of detection, but not if we waited until 24 h after ingestion. It is possible that the procedure to clear protein from the serum prior to stir bar absorption and extraction increased the signal to noise for fluoxetine and norfluoxetine detection, but lowered the overall amount absorbed. Since we did not analyse recovery from the stir bar from a spiked control with these serum samples, it is possible the serum concentrations may be as much as 50% off, similar to what we observed for the brain homogenate samples with recovery analysis.
Using different drug combinations in the Wistar rats, we have detected a significant increase in neurogenesis in the anterior subventricular zone of the lateral brain ventricles of these older rats, which is reported in a separate paper. We are currently testing various drug combinations delivered by this method for functional recovery following ischaemic stroke. Overall, this method of voluntary oral administration of drugs is reliable for multiple stocks or strains of rats for chronic fixed dosage of drug(s).
Footnotes
ACKNOWLEDGEMENTS
The source of funding for this project was the Wright State University School of Medicine Seed Grant, the LEADER Consortium Advance mini-grant and funds from the Department Chair, Timothy Cope, and Vice President of Research at Wright State University, Dr Jack Bantle.
