Abstract
When establishing animal models of viral respiratory infection, the optimal dose and route of delivery are critical to ensure reproducible outcomes. The mouse model for influenza infection is widely used due to the small animal size and simplicity of viral inoculation. During establishment of a mouse model of influenza A infection we observed a marked shift in morbidity when identical influenza A inoculum doses were delivered in less than 35 μL. We show for the first time that mice challenged with a 25 μL inoculum volume readily recovered following infection with an infectious dose of influenza A virus that was fatal when inoculated in 35 or 50 μL volumes.
Due to the relative ease of handling and low costs, the mouse model is widely used to test the immune responses and efficacy of potential vaccines as well as viral pathogenesis studies.1,2 The first consideration when establishing an infection model in mice is how to administer the optimal challenge inoculum. Aerosol delivery requires specialized equipment and the inoculum dose generated may not be taken up uniformly by all of the inoculated animals. Therefore, the preferred method is direct intranasal inoculation as this simple technique can be accurately controlled. 3 However, there are considerable variations in the reported volumes used for intranasal inoculation of challenge viruses. For example, in the influenza research field, volumes of 15, 4 20,5–8 25, 9 30, 10 40 11 and most commonly 50 μL3,12–14 have been reported to induce mortality in mice. It could therefore be expected that the outcome of influenza infection would be relatively consistent when using any of these volumes. In preliminary studies of establishing a mouse model of influenza A infection, 15 we initially chose an inoculum volume of 50 μL as this was the most widely reported. However, our observations showed that this intranasal volume delivered to the mouse caused breathing difficulty and was not well tolerated and required some animals to be resuscitated. In order to refine the procedure to reduce the adverse events, in subsequent experiments we reduced the inoculum volume to 25 μL while keeping the infectious dose constant. Unexpectedly, we observed a marked decline in morbidity that may have been influenced by this reduced inoculum volume (data not shown). To assess the volume-dependent outcome of influenza A infection we conducted a small-scale study that compared identical influenza A inoculum doses, given intranasally, in volumes of 25, 35 and 50 μL.
Ten-week-old female BALB/cARC mice were obtained from the Animal Resources Centre (Western Australia, Australia). Mice were housed in groups of five in HEPA-filtered top, micro-isolator cages (width 15 cm, depth 26 cm, overall floor area 390 cm2) containing autoclaved nestbedding (recycled newspaper product). Clean cylindrical cardboard tubes (ca. 15 × 3 cm) were also provided on the cage floor to facilitate hiding. Sterile water (250 mL) with overhead ball-bearing sippers as well as moist food (changed daily) was provided in a plastic weight tray on the cage floor. The room was maintained at a constant temperature (21–24°C) and humidity (40–60%) on a 12 h light–dark cycle, with lights off at 18:00 h. Mice were lightly anaesthetized via the intraperitoneal route with 0.1 mL/10 g body weight with ketamine:xylazine (10 mg/mL:1 mg/mL) prior to influenza virus challenge (A/Aichi/68 X31 (H3N2)) (GenBank: J02090.1). Prior to embarking on animal studies, we performed validation of the viral stocks. The tissue culture infective dose (TCID50) was calculated using the method of Reed and Muench. 16 The stock X31 virus titre was 9 × 106 TCID50/mL as assessed by plaque-titration on MDCK cells or 104.5 MID50/mL.
Mice were weighed on a daily basis and assessed for clinical signs of infection and sacrificed following the rapid advancement of clinical signs or >25% body weight loss. Assessment of clinical signs was based on visual observations of mice and assigned a score of 1 for each of the following: dull/ruffled coat, change in temperament, reduced food/water intake, weight loss and reluctance to move. Scores with ≥4 required that mice be euthanized. All animal handling procedures and protocols were assessed, approved and carried out in accordance with the guidelines of the Institute of Medical and Veterinary Science Animal Ethics Committee and the National Health and Medical Research Council (NHMRC) of Australia.
Three groups of four micewere challenged while in a nosedown, supine position via the left nostril with 4.5 × 103 TCID50 of influenza A virus in a single bolus of 25, 35 or 50 μL using a fine bore plastic pipette tip (Quality Scientific Plastic #010-R204). A group of four control mice received 50 μL of saline only. Mice which received the 25 μL influenza-containing inoculum lost 20% body weight within four days postinoculation (range: 19–23% of original body weight) but were able to overcome the effects of infection and recovered (Figure 1a). To ensure these mice were recovering we maintained them until a significant reduction in clinical signs was observed. Conversely, mice that received either the 35 or 50 μL influenza-containing inoculums succumbed to infection and were euthanized at >25% body weight loss (Figures 1b and c). Mice that received the saline-only inoculum also displayed immediate and transient signs of respiratory distress upon inoculation but no signs of influenza A infection thereafter (Figure 1d). The levels of influenza viral RNA in the lungs of the virus inoculated mice at autopsy were also assessed by realtime-reverse transcription polymerase chain reaction (RT-RT PCR) as previously described.15,17 Briefly, lung tissues were excised at the time of autopsy, immersed in RNA later (Qiagen), incubated overnight at 4°C and stored at –20°C until used. Viral RNA was extracted from lung tissues with an RNeasy Mini Kit (Cat # 74106; Qiagen GmbH, Hilden, Germany). The influenza A conserved matrix gene was selected as the target for amplification.
17
RT-RT PCR was performed using a QuantiTect SYBR Green RT-RT PCR kit (Qiagen). Uniform RNA amounts were loaded using a standard curve constructed from normal lung tissue spiked with 10 μL of stock virus. Normal lung RNA was added to obtain uniform total RNA in each reaction. Normalization of RNA added was performed by a separate RT-RT PCR under the same conditions as the influenza specific RT-RT PCR using primers spanning the intron/exon junctions of the mouse β-actin gene. There were no significant differences in the levels of viral RNA in the lungs of the mice that received 35 or 50 μL inoculum (P = 0.1216), whereas the lungs of mice that received the 25 μL inoculum contained significantly lower amounts of viral RNA compared with those inoculated with either 35 μL (P = 0.0258) or 50 μL (P = 0.0082) inoculum. Although this analysis was performed on tissues collected approximately 72 h after the 35 or 50 μL groups were euthanized, it confirmed that recovery was correlated to a reduction in influenza A viral RNA in the lungs of the 25 μL inoculated groups. We subsequently demonstrated that a 35 μL inoculum volume consistently showed typical evidence of influenza infection as judged by immunohistological and pathology examinations of infected lung sections.
15
Groups of four mice were infected by the intranasal route with 4.5 × 103 TCID50 of influenza A/H3N2 (X31) virus in a total inoculum volume of 25 (a), 35 (b) and 50 (c) or 50 (d) μL of saline. Mice were weighed on a daily basis and assessed for clinical signs with a score of 5 being the most severe. Body weight loss is expressed as percentage of original body weight. Dotted lines with open squares represent the average clinical score of the mice. Lung tissue was collected on day 7 (a) and day 5 (b, c). Viral RNA was extracted from the lungs of all mice at autopsy and analysed by realtime-reverse transcription polymerase chain reaction (RT-RT PCR). Bars represent standard error of the mean levels (normalized against β actin) of viral RNA detected (e). Statistical analysis of the RT-RT PCR data was performed using a one-way analysis of variance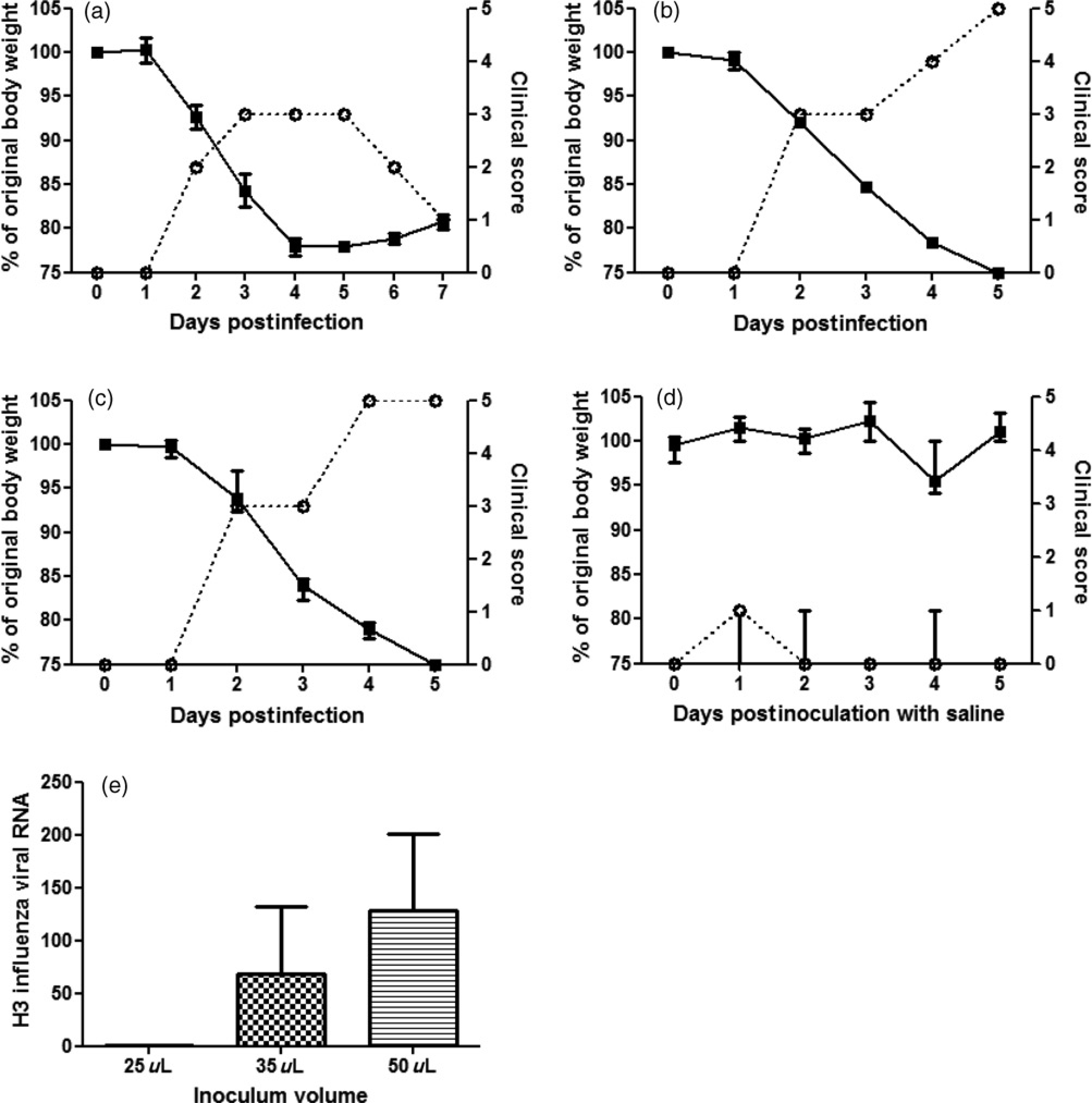
One potential mechanism for the observed differences in outcome is that when using a lower inoculum volume, proportionately more virus may be expelled from the respiratory tract following awakening from anaesthesia. Alternatively, it is probable that the physiological differences in the respective cellular microarchitecture in the upper and lower respiratory tracts contributed to the altered outcome.
The upper respiratory tract includes the nasal cavity, larynx and pharynx. Additionally, the mucosal surface of the upper respiratory tract provides the body's primary defence against airborne pathogens and particulate matters by enacting non-specific, physical entrapment and removal of foreign material. This function is augmented primarily by the simple, non-stratified cells comprising the epithelial layer which have plasma membranes separated by polarized tight junctions that are impervious to simple diffusion by macromolecules such as viruses. 18 Additionally, the epithelial layer of the upper respiratory tract is covered by cilia and mucus that aid in retrograde removal of intruding foreign material.
By contrast, the lower respiratory tract consists of the trachea, bronchi and bronchioles. 19 Bronchioles lead to the respiratory zone of the lungs where gas exchange occurs via the alveoli. Most of the epithelial layer cells in the lower respiratory tract are pluristratified, not polarized and do not contain tight cellular junctions. 20 Therefore, it is probable that intranasal challenge of mice with a larger inoculum volume distributes more influenza virus deeper into the lower respiratory tract where the physical barrier is less stringent, ultimately resulting in exacerbated disease. Interestingly, in an independent study where a lentiviral vector expressing β-galactosidase was administered in increasing inoculum volumes, increased transduction of the lower respiratory tract was observed. 21 It has previously been demonstrated in a non-infection model using a radioactive tracer to localize distribution of intranasal delivered particles, that the majority of radioactive particles were localized to the upper respiratory tract of mice when inoculation volumes were low (5–25 μL). Importantly, significantly greater amounts of radioactive particles were detected in the lower respiratory tract when volumes of 35 μL or more were administered. 22
In this study we have formally shown for the first time that the inoculum volume, with a constant challenge dose of influenza A virus, significantly alters the morbidity outcome of infection in mice. Importantly, we also report that the use of 50 μL inoculum volumes, as used by the majority of influenza A research groups, causes significant dyspnoea in ~15% of the mice upon inoculation which we concluded was unacceptably high. However, in most cases, gentle abdominal massage of the mice which showed severe dyspnoea, recovery was observed within 2 min. In our studies we have demonstrated that 35 μL inoculum volumes produce less respiratory distress, while maintaining infection outcomes. These results would assist other researchers when establishing small animal models of infection with intranasal virus inoculation.
Footnotes
Acknowledgements
We are grateful to Professor Lorena Brown (University of Melbourne, Australia) for kindly supplying the X31 influenza A virus. Statistical analysis was performed by Thomas Sullivan from the Data Management and Analysis Centre, Department of Public Health, University of Adelaide. This work was supported by the NHMRC of Australia by grant 399298 for urgent research into a potential avian influenza-induced pandemic.
