Abstract
Inflammatory bowel diseases (IBD) are chronic relapsing disorders of the gastrointestinal tract. Several mouse models for IBD are available, but the acute dextran sulfate sodium (DSS)-induced colitis model is mostly used for preclinical studies. However, this model lacks chronicity and often leads to significant loss of mice. The aim of this study was to establish a refined and translationally relevant model of DSS chronic colitis in BALB/c mice. In the first part, we compared several standard therapeutic (ST) treatments for IBD in the acute DSS colitis model to identify the optimal treatment control for a DSS colitis model as compared to literature data. In the second part, we tested the two most effective ST treatments in a refined model of chronic DSS colitis. Cyclosporine A (CsA) and 6-thioguanine (6-TG) caused considerable reduction of clinical scores in acute DSS colitis. The clinical outcome was confirmed by the results for colon length and by histopathological evaluation. Moreover, CsA and 6-TG considerably reduced mRNA expression of several pro-inflammatory cytokines in spleen and colon. Both compounds also showed a substantial therapeutic effect in the refined model of chronic DSS colitis with regard to clinical scores and histopathology as well as the expression of inflammatory markers. The refined model of chronic DSS colitis reflects important features of IBD and is well suited to test potential IBD therapeutics.
Introduction
The two major forms of inflammatory bowel disease (IBD), ulcerative colitis (UC) and Crohn’s disease (CD), are chronic relapsing-remitting inflammatory disorders of the gastrointestinal tract.1–3 The etiology is most likely a combination of genetic and environmental factors promoting the disintegration of the intestinal epithelial barrier as well as the dysregulation of a tolerogenic immune response to the intestinal microbiota.4–8 Furthermore, studies in animals indicate that excessive immune cell activation and/or the loss of regulatory responses are causative for a chronic inflammatory reaction. 9 Clinical symptoms of IBD include diarrhea, rectal bleeding, abdominal cramping, and weight loss.10–12 A large number of animal models for IBD have been developed that can be broadly categorized into spontaneous models, genetic models, adoptive transfer models, and inducible models.13,14 Acute and chronic DSS-induced colitis are the two most widely used inducible animal models in IBD research.9,15,16 The severity of disease in DSS colitis models is determined by molecular weight and concentration of DSS, as well as the duration of DSS application. 17 In the original report of DSS-induced colitis, the authors suggested that the intestinal microbiota may be involved in colitis induction. 16 However, another study demonstrated more severe symptoms in germ-free mice after DSS application. 18 This discrepancy might be explained by the different genetic background of the mouse strains, which can also have an influence on disease induction and progression. 19 The model of acute DSS colitis primarily reflects the initial step of epithelial injury in the colon and is characterized by an innate immune response, especially infiltration of neutrophils into the submucosal area. 20 Variants of the chronic DSS colitis model are in most cases performed as cycles of alternating DSS and water phases.21,22 These alternating phases are supposed to reflect the relapsing-remitting nature of IBD and ideally result in a chronic inflammatory response that persists even after DSS removal. 21 However, owing to the repeated challenges with DSS, most variants of the chronic DSS colitis model feature severe body weight reduction with frequent loss of animals. Therefore, we aimed to develop a refined and more translationally relevant chronic DSS colitis model that reflects signs of chronic inflammation and clinical symptoms of IBD without extensive weight loss and death.
Several studies already compared the efficacy of standard therapeutic (ST) treatments in acute or chronic models of DSS colitis.23–25 Because of the differences in sex and genetic background of the mouse strains, composition of the intestinal microbiota and the differing design of the models and treatment regimens, it is difficult to compare the findings and translate them to a newly designed DSS model. Therefore, the first aim of this study was to test the most relevant ST treatments in an acute DSS model. Afterwards, the most effective ST treatments were tested in a refined model of chronic DSS colitis. We established a chronic DSS colitis model resembling typical symptoms of IBD, but without detrimental weight reduction and consequent loss of animals.
Animals, material and methods
Animals
Eighty-five female BALB/cJRj mice (Janvier, Saint-Berthevin, France), 8–12 weeks old, were acclimatized for two weeks. Female mice were used because they are more appropriate for long-lasting experiments using these or similar disease models owing to less aggressiveness of female compared to male animals. Especially older males tend to fight in the cage, which induces stress and can influence immunological parameters of the experiment. Mice were housed as five per cage according to European guideline 2010/63/EU in single, ventilated type-II cages in a temperature- and light/dark cycle-controlled environment with access to autoclaved non-acidified water and standard chow (V1534-300; Ssniff, Soest, Germany)
DSS-induced colitis
For the acute model, mice were given 4% DSS (MW 36,000–50,000 Da, Lot-No. M7191; MP Biomedicals, Santa Ana, CA, USA) in autoclaved drinking water (w/v) for seven days, followed by three days of DSS-free drinking water. Chronic DSS colitis was induced by administration of 2% DSS for seven days, followed by 1% DSS for 10 days and 2% DSS for another seven days. Animals were inspected daily for body weight, stool consistency and colonic hemorrhage (Supplementary Table 1). For the body weight score, the percentage of body weight loss was calculated relative to the body weight at day 0, with 2% corresponding to 0.4 points but a maximum score of five points. The clinical score was calculated as the average of the scores for body weight loss, stool consistency, and blood in stool. The stool-consistency-blood score was calculated as the average of the scores for stool consistency and blood in stool.
Drug treatment
Drug solutions or homogenous suspensions were prepared in phosphate-buffered saline (PBS; 0.15 M). The following test compounds and doses were used in the acute model: cyclosporine A (CsA): 12.5 and 25 mg/kg (Sandimmun® 50 mg/ml; Novartis, Nuremberg, Germany), methotrexate (MTX): 2.5 mg/kg (Methotrexat 500 Infusionslösung; Medac, Hamburg, Germany), 6-thioguanine (6-TG): 1 and 2 mg/kg (Santa Cruz Biotechnology, Dallas, TX, USA), dexamethasone (DXM): 2.5 and 5 mg/kg (Fortecortin Inject® 4 mg; Merck Serono, Darmstadt, Germany), mesalazine (5-ASA): 100 mg/kg (Sigma-Aldrich, Taufkirchen, Germany), and anti-muTNF-α monoclonal antibody (mAb) V1q 21 : 10 and 30 mg/kg (production and purification in-house). Drugs were administered by intraperitoneal (i.p.) injection from days 1 to 10 in a volume of 10 ml/kg.
In the chronic model, CsA (12.5 mg/kg) and 6-TG (1 mg/kg) were administered by i.p. injection from days 1 to 10 and days 18 to 22 in a volume of 10 ml/kg. Control animals received vehicle only.
Dissection
Animals were sacrificed in deep anesthesia using carbon dioxide with a manometer-controlled flow rate of 1 l/min in a 1 l inhalation chamber. After the animals stopped moving they were left for 2 more minutes in the chamber. Death of animals was confirmed by testing the absence of reflexes, heart beat and breathing. The abdomen was opened by a longitudinal incision and a piece of the spleen was isolated and snap-frozen in liquid N2 for RNA extraction. The colon was dissected and the length from caecum to anus was measured. Two parts of the most distal colon were taken for histology evaluation. An adjacent part of 1.0 cm was snap-frozen in liquid N2 for RNA extraction.
Histological analysis
Tissue samples were fixed in 4% phosphate-buffered formaldehyde for 24 hours, dehydrated in grade ethanol, and embedded in paraffin. Three-µm sections were cleared, hydrated, and stained with hematoxylin and eosin (H&E), β-naphtol-AS-D-chloroacetate esterase (CAE) or by Masson Trichrome.
Stained sections were evaluated in a blinded fashion by an experienced pathologist for fibrosis in mucosa and submucosa, infiltration of granulocytes, lymphocytes and macrophages, extent of edema and necrosis (Supplementary Table 2). The histopathological score was calculated as the mean of the grades for these parameters.
For immunohistochemical analysis, blocked tissue sections were incubated with primary antibody (polyclonal rabbit anti-human CD3; # A045201-2; Dako, Glostrup, Denmark) at a dilution of 1:500 for two hours. After washing, samples were incubated with secondary antibody (goat anti-rabbit immunoglobulin (Ig)G, horse-radish-peroxidase (HRP)-conjugated; # 111-035-003; Jackson ImmunoResearch, West Grove, PA, USA) at a dilution of 1:200 for one hour. Sections were incubated with a DAB solution for 5 to 15 minutes and counterstained with hematoxylin.
Isolation of total RNA and quantitative reverse transcription (rt) polymerase chain reaction (PCR)
Total RNA from spleen and colon was extracted using TRI® Reagent (Sigma-Aldrich) according to the manufacturer’s instructions. To remove residual genomic DNA, extracted RNA was treated with RNase-free DNase I (VWR International, Darmstadt, Germany) as instructed in the reagent protocol. One µg of RNA was reversely transcribed using the Transcriptor First Strand cDNA Synthesis Kit (Roche Applied Science, Mannheim, Germany) and random hexamer primers according to the manufacturer’s protocol.
Real-time PCR
Gene-specific primers (Supplementary Table 3) were designed using the Universal ProbeLibrary Assay Design Center (Roche Applied Science). Primers were added to the reaction at a final concentration of 0.4 µM. Five µl of diluted (1:20) complementary DNA (cDNA) were added to 15 µl of master mix. The PCR was carried out using the LightCycler® 480 instrument (Roche Applied Science). Normalized ratios were calculated by the LightCyler® 480 Software 1.5. Target gene expression was related to the expression of two reference genes (Delta-aminolevulinate synthase 1 (
Statistical analysis
All data were analyzed using SigmaPlot 11.0 (Systat, Erkrath, Germany). Clinical data are presented as mean ± SEM. Data for colon length, histopathological score and messenger RNA (mRNA) expression are presented as individual data points. Significant differences between data sets were estimated by Kruskal-Wallis one-way analysis of variance (ANOVA) (if normally distributed) or by one-way ANOVA on ranks (if the normality test failed). In case of normal distribution of data, the data sets were compared by the Holm-Sidak’s post hoc test. If the normality test failed, the Dunnett’s post hoc test was applied. Differences between two groups were analyzed by Student’s
Results
6-TG and CsA reduced clinical symptoms in acute DSS colitis
As a first step, a suitable treatment control should be identified. Therefore, different ST were tested in the model of acute DSS colitis. A clinical score based on weight loss, stool consistency and blood in stool was determined daily. Treatment with CsA and the higher dose of 6-TG significantly reduced the clinical score after day 8 (Figure 1(a) and (b)). In contrast, treatment with DXM aggravated the clinical symptoms starting already at day 3 (Figure 1(c)). 5-ASA, MTX and the anti-TNF-α mAb had no relevant effect on the course of disease (Figure 1(d)–(f)). CsA, DXM and to a lesser extent 6-TG induced substantial weight loss (Supplementary Figure 1(a)–(c)). Since treatment with the higher dose of DXM aggravated weight loss and the clinical symptoms extremely, animals had to be sacrificed preterm at day 9. Even the lower dose of DXM induced severe weight loss indicating that DXM has a detrimental effect in this model. For both CsA and 6-TG, weight loss was rapidly reversed after termination of DSS treatment. In the case of CsA, animals regained weight up to 0–5% below initial weight, which was significantly higher compared to animals treated with vehicle only (Supplementary Figure 1(a)). A similar effect could be observed in 6-TG-treated mice (Supplementary Figure 1(b)). Since weight loss was part of the clinical score, the drug-induced weight loss masked the therapeutic effect with respect to diarrhea and blood in stool. This was particularly evident for the higher dose of CsA, for which the clinical score was higher than in the vehicle control in the first seven days (Figure 1(a)). Therefore, we decided to determine an alternative score representing the average of the scores for stool consistency and colonic hemorrhage, designated as the stool-consistency-blood score. As expected, we saw a greater difference between treated animals and vehicle controls for CsA and 6-TG, indicating that the higher clinical score was mainly due to weight loss in these animals (Supplementary Figure 1(a) and (b)). In DXM-treated mice, the stool-consistency-blood score was similar to the vehicle control, confirming the lack of a therapeutic effect (Supplementary Figure 2(c)). Treatment with anti-TNF-α mAb, MTX and 5-ASA neither resulted in additional weight loss (Supplementary Figure 1(d)–(f)) nor in a consistent therapeutic effect in terms of the stool-consistency-blood score (Supplementary Figure 2(d)–(f)).
Acute dextran sulfate sodium (DSS) colitis: Clinical score. Acute colitis was induced in BALB/c mice by feeding of 4% DSS in drinking water for seven days followed by three days of pure drinking water. Animals were treated from days 1 to 10 with different doses of cyclosporine A (CsA) (a), 6-thioguanine (6-TG) (b), dexamethasone (DXM) (c), anti-tumor necrosis factor (TNF)-α monoclonal antibody (d), methotrexate (MTX) (e) or mesalazine (5-ASA) (f) as indicated. Animals treated with phosphate-buffered saline (PBS) represent the vehicle control. The clinical score was evaluated daily and is shown as mean ± standard error of the mean for 5–10 animals per group. *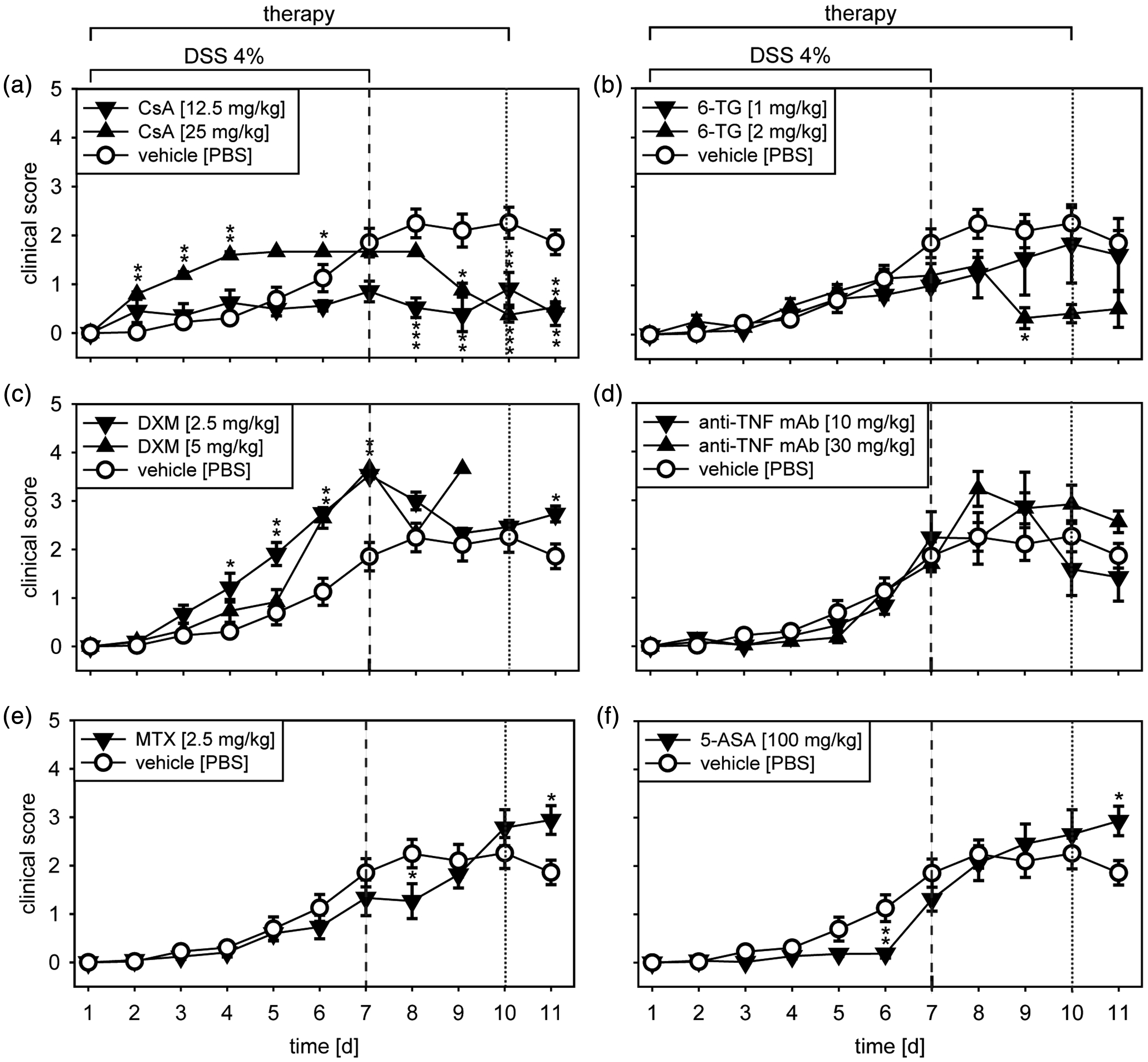
6-TG and CsA reduced inflammation in the colon in acute DSS colitis
DSS-induced epithelial injury and the local inflammatory response resulted in scarring of the tissue and a consequent colon shortening. The colons of CsA- and high-dose 6-TG-treated mice were significantly longer compared to the vehicle controls, indicating a therapeutic effect due to a reduced inflammatory response (Figure 2(a)). The low-dose treatment with the anti-TNF-α mAb also showed a significant effect. In contrast, DXM induced further reduction of the colon length, indicating an extensive inflammatory response.
Acute dextran sulfate sodium (DSS) colitis: Colon length and histopathology. Acute colitis was induced and animals were treated as described in Figure 1. (a) Post mortem, the colon was dissected and the colon length was determined for 3–10 animals per group. (b) The histopathological score was determined based on hematoxylin and eosin (H&E)-stained sections of the distal colon for 3–10 animals per group. (c) Exemplary H&E-stained tissue sections (3 µm) of the distal colon; scale bar: 500 µm. **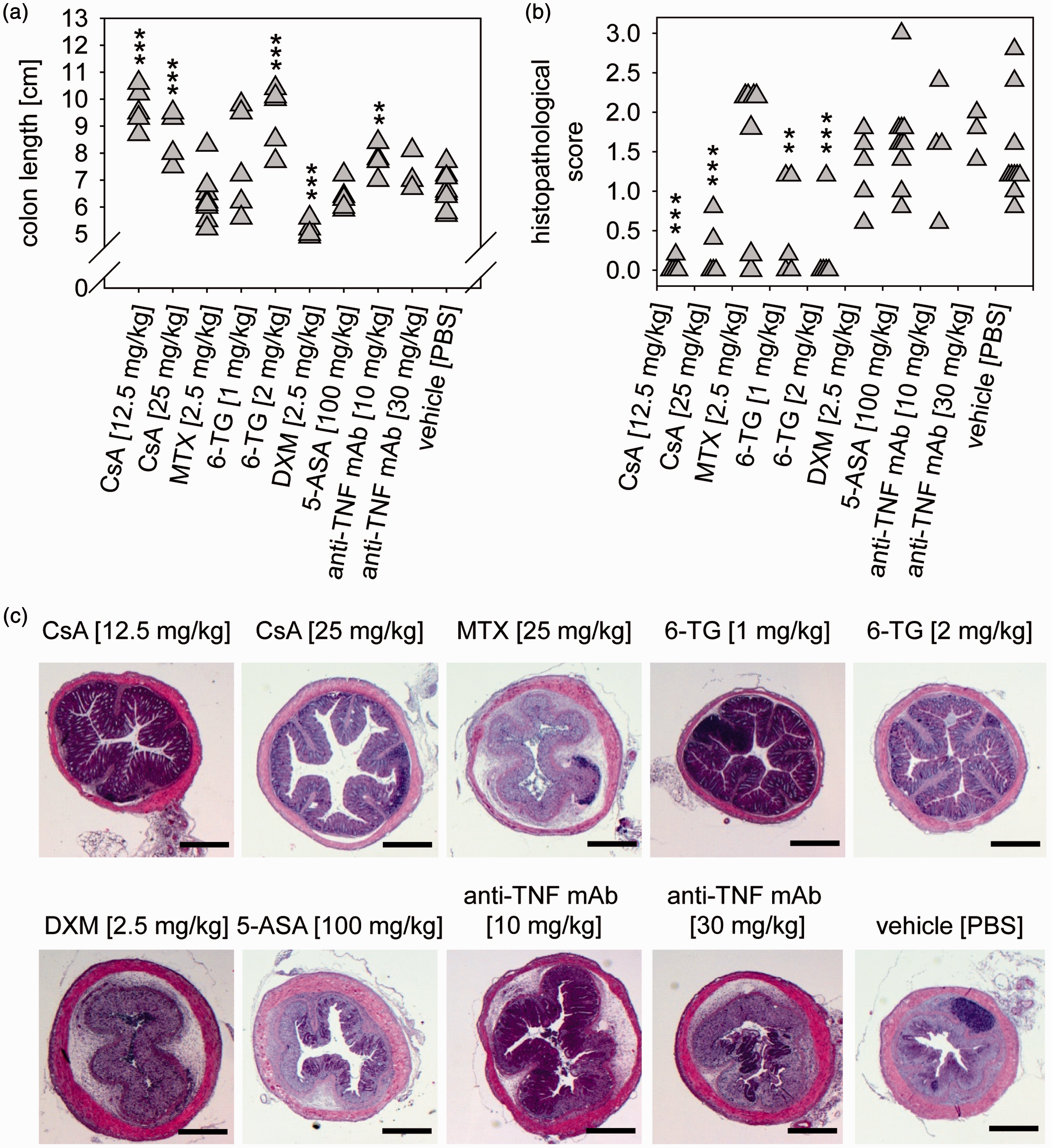
An important parameter for the evaluation of tissue degeneration and inflammation in the colon is the histopathological score based on H&E-stained sections. In contrast to chronic DSS colitis, we nearly never found active necrosis or regenerative surface epithelium in the acute DSS model. There was minor to moderate inflammation characterized by the infiltration of both granulocytes and lymphocytes. In the vehicle control group, we observed moderate inflammation together with massive signs of degeneration particularly evident in the mucosa. These observations were less pronounced in CsA- and 6-TG–treated animals (Figure 2(b) and (c)). The most pronounced inflammation was noted in animals treated with DXM, while MTX showed the highest scores for mucosal/submucosal fibrosis and edema. Treatment with 5-ASA or anti-TNF-α had no effect on the extent of tissue degeneration or the infiltration of inflammatory cells.
To further investigate the local as well as the systemic inflammatory response, mRNA expression of the pro-inflammatory cytokines Acute dextran sulfate sodium (DSS) colitis, messenger RNA (mRNA) expression of pro-inflammatory cytokines. Acute colitis was induced and animals were treated as described in Figure 1. Post mortem, relative mRNA expression normalized against housekeeping genes (Alas1 and Hgprt) was determined for 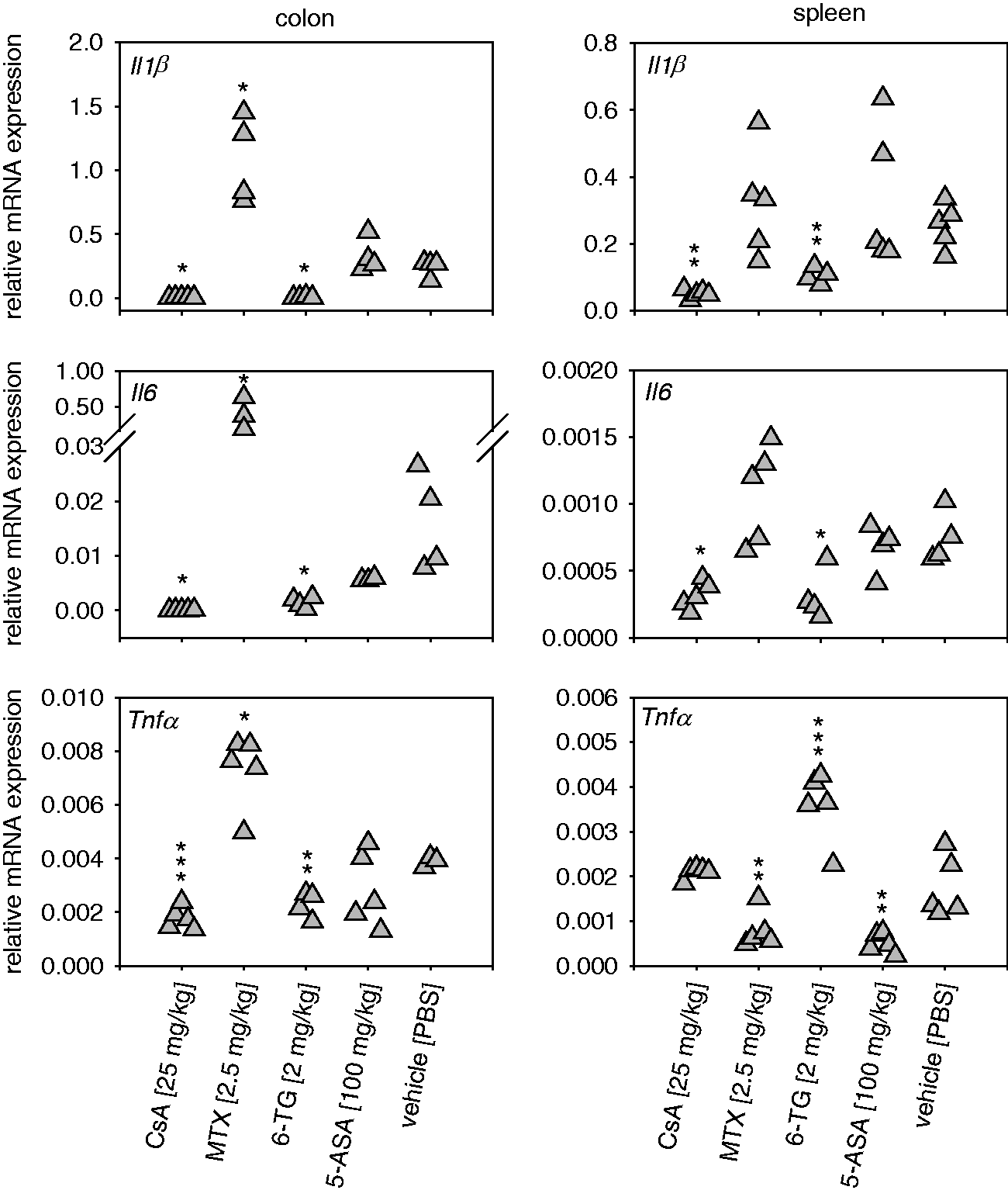
A refined model of chronic DSS colitis
We tested several experimental setups differing in DSS concentration, duration of DSS administration and recovery phases, as well as number of cycles (data not shown). We found that the animals mostly recovered very quickly during pure-water phases without apparent clinical symptoms. Therefore, we tested whether a small amount of DSS given during the recovery phase would prevent complete healing of the mucosa and induce a state of chronicity. In order to establish a disease model for testing of potential IBD therapeutics, we tested the most efficient ST treatments, as deduced from the acute DSS colitis, as treatment controls in the new chronic model. Consistent with the results from the acute model, we observed a significant reduction of the clinical score in animals treated with 6-TG or CsA (Figure 4(a) and (b)). Most important, we observed only a moderate reduction of body weight (0–5%), thereby preventing the loss of animals (Figure 4(c) and (d)). We also determined the stool-consistency-blood score, which consequently showed a more pronounced therapeutic effect for both STs (Figure 4(e) and (f)).
Chronic dextran sulfate sodium (DSS) colitis: Clinical score, stool-consistency-blood score and body weight. Chronic colitis was induced in BALB/c mice by feeding of 2% DSS in drinking water for seven days (dark gray) followed by 10 days of 1% DSS (light gray) and another seven days of 2% DSS. Animals were treated from days 1 to 10 and 18 to 22 with 6-thioguanine (6-TG) ((a), (c) and (e)) or cyclosporine A (CsA) ((b), (d) and (f)) as indicated. Animals treated with phosphate-buffered saline (PBS) represented the vehicle control. The clinical score ((a) and (b)), the body weight ((c) and (d)) and the stool-consistency-blood score ((e) and (f)) were evaluated daily and are shown as mean ± standard error of the mean for five animals per group. *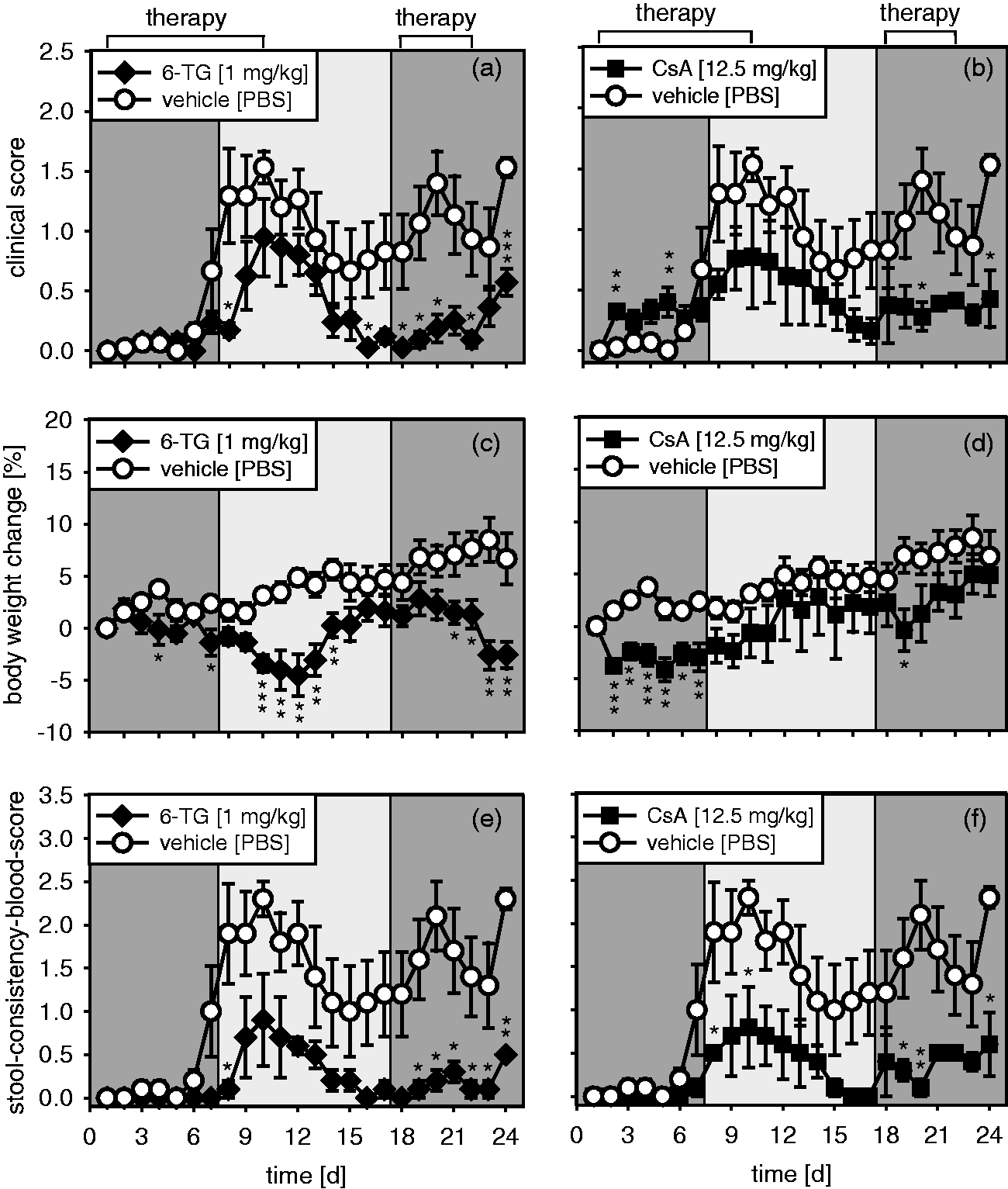
In the histological analysis, we found increased infiltration of neutrophils and T cells into the colon tissue. Furthermore, a superficial necrosis of minor severity was occasionally observed. The cells within the fibrosis resembled epithelioid or myoid cells. Fibers between cells were apparent but not very distinct and abundant, which was confirmed by Massom Trichrom staining.
In contrast to acute DSS colitis, in which the mucosa was often found to be destroyed, a partial recovery was observed in chronic DSS colitis with the mucosal surface being covered by epithelium. However, the mucosal architecture remained altered, i.e. the mucosal crypts were mostly flat but also showed in places mild hyperplasia. Animals treated with 6-TG or CsA showed fairly normal mucosal architecture. We also found massive lymphatic infiltrates in vehicle-treated animals that were less pronounced in 6-TG- or CsA-treated mice (Figure 5(a), upper panel). Furthermore, CAE and anti-CD3 staining revealed a moderate reduction of infiltrating neutrophils and T cells in the mucosal tissue after 6-TG or CsA treatment (Figure 5(a), middle and lower panel). Based on the histological evaluation, we observed a positive therapeutic tendency for 6-TG treatment (Figure 5(b)). In a further experiment with higher numbers of animals, in which 6-TG was used as treatment control, we observed a significant therapeutic effect (Supplementary Figure 3). In addition, a significant therapeutic effect was found for 6-TG and CsA with respect to colon length (Figure 5(c)).
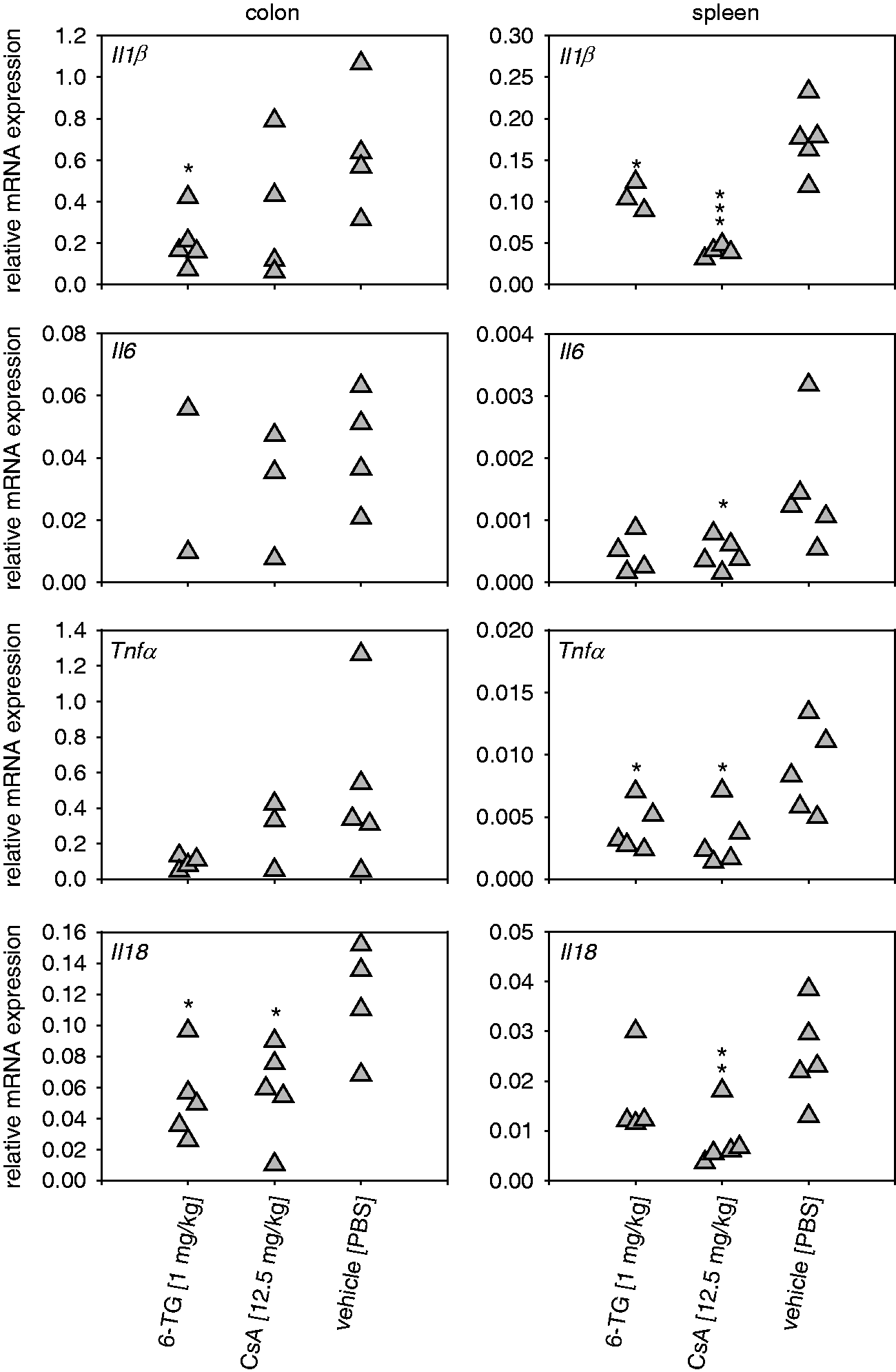
To evaluate markers of local and systemic inflammation, we analyzed gene expression of the pro-inflammatory cytokines Chronic dextran sulfate sodium (DSS) colitis: Histopathology and colon length. Chronic colitis was induced and animals were treated as described for Figure 4. (a) Exemplary photomicrographs of hematoxylin and eosin (H&E) (upper panel), β-naphtol-AS-D-chloroacetate esterase (CAE) (middle panel), or anti-cluster of differentiation (CD)3 staining (lower panel) of tissue sections (3 µm) of the distal colon; scale bar (H&E): 500 µm; scale bar (CAE, anti-CD3): 100 µm. (b) The histopathological score was determined based on H&E-stained sections of the distal colon for five animals per group. (c) Post mortem, the colon was dissected and the colon length was determined for five animals per group. * Chronic dextran sulfate sodium (DSS) colitis: Messenger RNA (mRNA) expression of pro-inflammatory cytokines. Chronic colitis was induced and animals were treated as described for Figure 4. Post mortem, relative mRNA expression normalized against housekeeping genes (Alas1 and Hgprt) was determined for 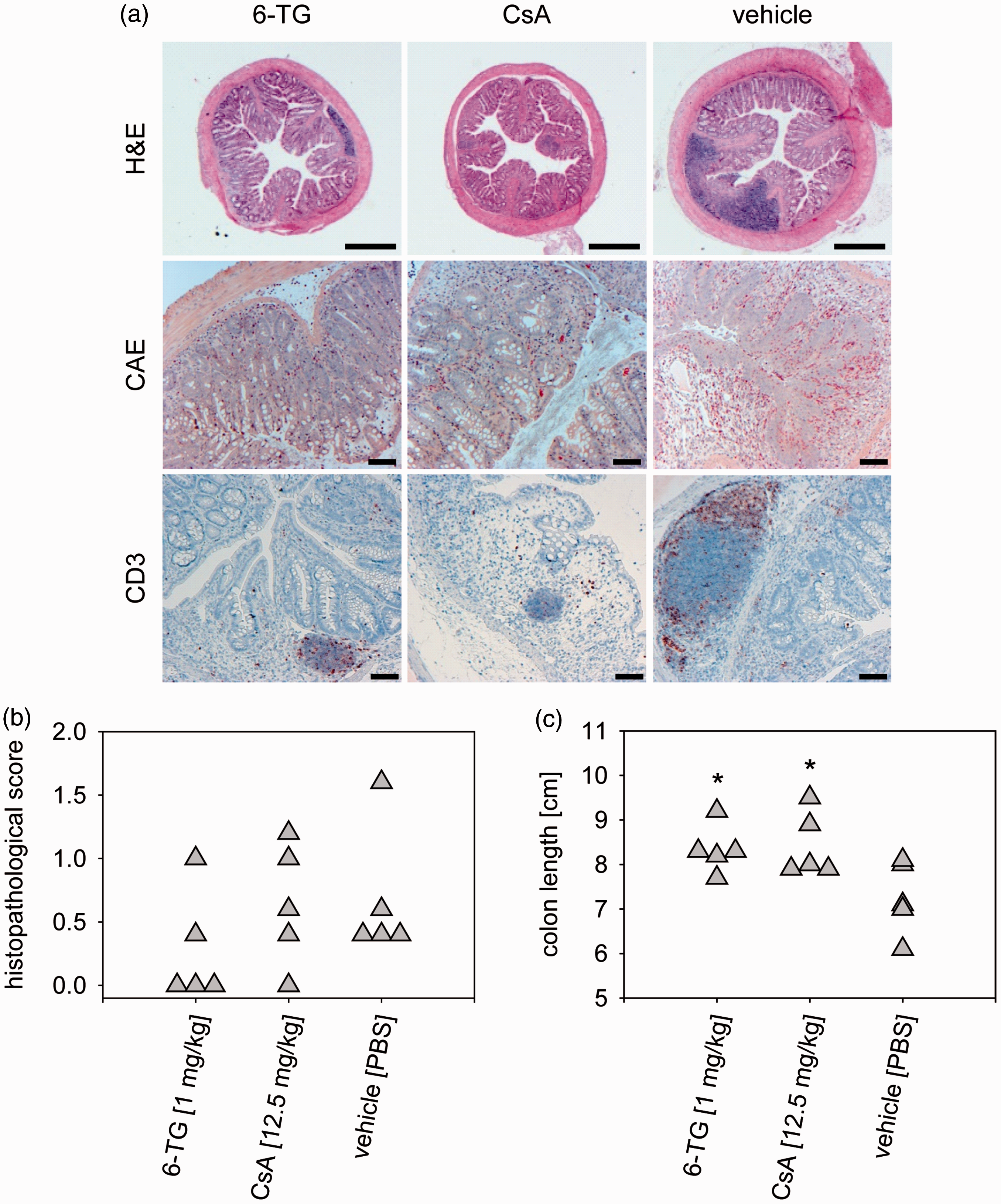
Discussion
First, we studied the efficacy of ST treatments for IBD in a time-saving acute DSS model regularly found in the literature.26–28 Selected drugs have already been tested in acute or chronic DSS colitis models in previous studies.23–25 However, translation of these findings to alternative DSS models may be difficult owing to the functional variance of DSS lots, different study designs and treatment regimens as well as differences in the composition of the intestinal microbiota between animal facilities. Irrespective of these differences, our study was in agreement with results from a previous study revealing that 6-TG and CsA represent effective ST treatments in the acute DSS model.
24
In particular, no ulcerations or infiltration of lymphocytes were observed in 6-TG- or CsA-treated animals. Furthermore, both therapeutics prevented colon shortening as an indicator of tissue injury and reduced the expression of the pro-inflammatory cytokines
Acute DSS models are suitable to study initial events of epithelial injury and invasion of luminal antigens into the mucosa/submucosa. However, they are less appropriate to study the pathophysiology of the chronic inflammatory response. Several protocols for chronic DSS models have been developed that all utilize alternating phases of DSS and pure water to induce chronification of gut inflammation. Using BALB/c mice, we observed that the animals recovered quickly during pure-water phases. Therefore, our aim was to develop a refined chronic DSS colitis model that better established chronicity. To this end, we decided to substitute pure water by a low concentration of DSS to maintain a low-level inflammatory stimulus. Moreover, we lowered the DSS concentration in the exacerbation phases. In our experience, higher DSS concentrations in the exacerbation phase frequently caused dramatic symptoms and even death. Applying this alternative protocol, we demonstrated that animals developed a disease characterized by moderate to strong diarrhea and colonic hemorrhage without extensive weight loss. Clinical symptoms improved slightly during remitting phases but persisted throughout the experiment. In our opinion, this model better reflects the clinical symptoms and the relapsing-remitting course of disease observed in patients. Although we could not demonstrate a significant therapeutic effect of 6-TG or CsA with respect to histopathology in this experiment, we observed a tendency of reduced inflammatory infiltrates. A significant therapeutic effect of 6-TG was confirmed by a further experiment including a higher number of animals (Supplementary Figure 3). Furthermore, 6-TG and CsA treatment prevented inflammation-induced scarring of the tissue as indicated by reduced colon shortening. For transcriptional analysis of crucial inflammatory cytokines (i.e.
In summary, we present a refined model of chronic DSS colitis circumventing the often deleterious weight loss seen in other DSS models but still retaining the most relevant symptoms, i.e. diarrhea, colonic hemorrhage and the histopathological alterations of the colon tissue. With this approach, we are in agreement with another study stating that models with mild intestinal damage would be more suitable to study the effects of potential IBD therapeutics. 37 We have already used the chronic model presented here in several contract research studies testing different potential therapeutic agents (data not shown). We found that the occurrence of clinical symptoms and histopathology were highly reproducible. Therefore, we propose this refined model as an ideal tool for preclinical efficacy studies.
Footnotes
Acknowledgments
The authors gratefully acknowledge technical support by Drs. Christiane Füldner and Elke Ueberham. Moreover, we thank Sarah Leitenroth for animal husbandry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
