Abstract
Cervical dislocation is a commonly used method of mouse euthanasia. Euthanasia by isoflurane inhalation is an alternative method which allows the sacrifice of several mice at the same time with an anaesthesia, in the aim to decrease pain and animal distress. The objective of our study was to assess the impact of these two methods of euthanasia on the quality of mouse oocytes. By administering gonadotropins, we induced a superovulation in CD1 female mice. Mice were randomly assigned to euthanasia with cervical dislocation and isoflurane inhalation. Oviducts were collected and excised to retrieve metaphase II oocytes. After microscopic examination, oocytes were classified into three groups: intact, fragmented/cleaved and atretic. Intact metaphase II oocytes were employed for biomedical research. A total of 1442 oocytes in the cervical dislocation group were compared with 1230 oocytes in the isoflurane group. In the cervical dislocation group, 93.1% of the oocytes were intact, versus 65.8% in the isoflurane group (P ≤ 0.001). In light of these results, we conclude that cervical dislocation is the best method of mouse euthanasia for obtaining intact oocytes for biomedical research.
The mouse is the most commonly used model in biomedical research, particularly for fundamental studies on gametes and embryos. 1,2 Euthanasia of laboratory animals must be performed according to ethical standards, with trained personnel using appropriate techniques, equipment and reagents in order to induce a death as least painful and stressful as possible. 3 However, the euthanasia method should not cause histological or histochemical changes that would adversely affect research results. Cervical dislocation is a conventional technique for mouse euthanasia. Instead of cervical dislocation, we wanted to test an alternative euthanasia method by isoflurane inhalation, which is an anaesthetic agent of the family of halogenated ethers, in the aim to decrease pain and distress in mice. Because our research laboratory conducted biomedical research on oocytes, 2,4 we wanted to assess the impact of these two methods of euthanasia on the quality of mouse oocytes.
We used prepubescent CD1 female mice aged 4–5 weeks (Charles River Laboratory, L'Arbresle, France). The animals were housed in a temperature- and light-controlled room, with free access to food and water. All experimental protocols and animal handling procedures were reviewed and approved by the National Ethics Committee on Animal Experimentation (IRB: 3-4112009). On day 1, mice were injected intraperitoneally with pregnant mare serum gonadotropin (10 IU/100 μL) (Sigma-Aldrich, St Fallavier, France) to induce superovulation. On day 3, ovulation was triggered by adding an intraperitoneal injection of human chorionic gonadotropin (10 IU/100 μL) (Sigma-Aldrich). Sixteen hours after human chorionic gonadotropin, mice were euthanized. Two methods of euthanasia were tested, each condition was replicated six times on two groups of 16 mice: (1) cervical dislocation that causes a sharp section of the spinal cord followed by an instantaneous cardiac arrest, and (2) inhalation of a lethal dose of isoflurane (concentration of 5%) in a closed chamber, which causes death in less than 60 s after a depression of the central nervous system and the respiratory system. Isoflurane was delivered from an Isotec 3 vaporizer (Ohmeda, Steeton, West Yorkshire, UK), with O2 as a carrier. Mice were dissected immediately after euthanasia to collect oviducts. Oviducts were dissected into M2 medium (Sigma-Aldrich). Intact cumulus masses were released from excised oviducts and incubated with hyaluronidase (10 mg/mL). A morphological examination under a binocular microscope (×200) was blindly conducted to assess oocyte quality: appearance of the cytoplasm, intact oocytes, fragmented/cleaved oocytes and atretic oocytes were scored (Figure 1). Statistical differences between the two methods of euthanasia were determined using the χ 2 test. The test was considered positive and statistically significant when P < 0.05. Intact metaphase II oocytes were then used for biomedical research on the oocyte. 2
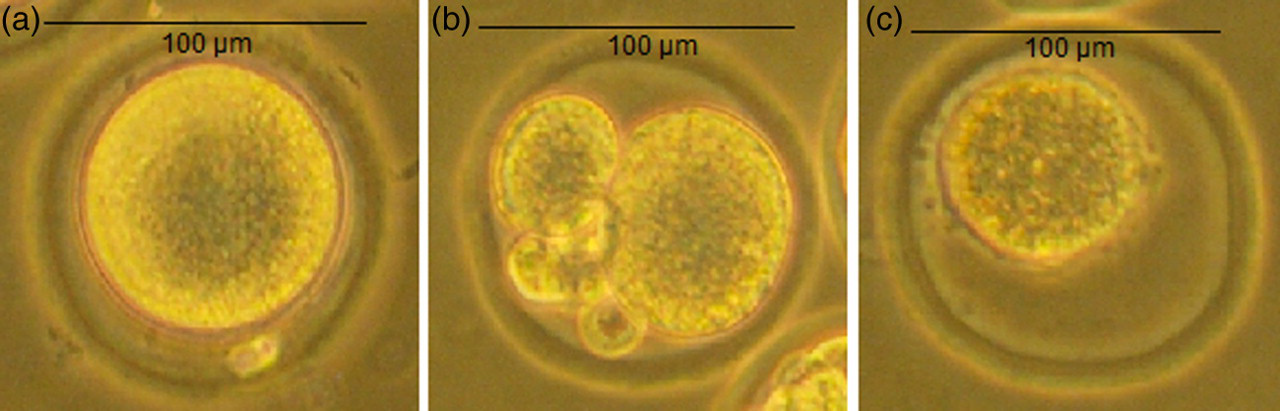
Morphological examination of different types of oocytes under a binocular microscope: (a) intact oocyte; (b) cleaved oocyte; and (c) atretic oocyte. Scale bar, 100 μm. Final magnification ×500
During the mouse dissection, oviduct dissection and cumulus masses removal were easier in the cervical dislocation group. Indeed, the different anatomical structures were easily individualized. In the isoflurane group, the uterine horns and ovaries were the site of microhaemorrhages by vasodilation, which perturbed the oocytes removal in the oviducts. A total of 1442 oocytes in the cervical dislocation group was examined and compared with 1230 oocytes in the isoflurane group. In the cervical dislocation group, 93.1% (n = 1342) of the oocytes were intact, versus 65.7% (n = 809) in the isoflurane group (P ≤ 0.001). The number of fragmented, cleaved and atretic oocytes was significantly higher in the isoflurane group (100 versus 421 altered oocytes, P ≤ 0.001).
The mouse is the most commonly used model in biomedical research. Different methods are used for euthanasia. 5 The use of inhalation anaesthetic agents, like isoflurane, is justified by the ease of the protocol, the relative safety of the procedure, the capacity to sacrifice larger numbers of animals at the same time with an anaesthesia, by requiring a minimum of handling of animals. 6,7 Staff safety is ensured by the use of gas scavenging units, as recommended by the European Commission recommendations for euthanasia. 8 Cervical dislocation is another technique commonly used for euthanasia of mice. Realized by trained personnel, this method offers many advantages. Neither specific equipment is required; it is safe, painless and non-aversive like gas. However, the main drawback of this technique is the inability to euthanize many animals simultaneously, which may be a prerequisite for conducting studies with larger cohorts. 7 Moreover, the realization of cervical dislocation requires a learning curve to induce as less painful and stressful a death as possible. Isoflurane inhalation appears to be an alternative method of euthanasia compared with cervical dislocation. The National Center for Scientific Research in France considers isoflurane inhalation to be the most appropriate method of euthanasia for rodents. To our knowledge, our study is the first to compare these two methods of mouse euthanasia on the oocyte quality. Many parameters can affect the quality of oviduct collection as well as the morphology and viability of oocytes. We have observed that euthanasia with isoflurane had a negative impact on oocyte quality. The occurrence of microhaemorrhagic events into the surrounding tissues hindered the collection of oocytes in the oviducts. In addition, the oocyte morphology was significantly altered in the group using isoflurane euthanasia, with only 65.7% intact oocytes. The exact mechanism of action of isoflurane is still unclear. Absorbed mainly by inhalation, isoflurane is distributed throughout the body via blood. It induced a loss of consciousness by a disturbance of neurotransmission (reversible changes in the neuronal membrane) and caused a dose-dependent depression of respiratory and cardiovascular centres. 9 The consequences of isoflurane on the reproductive system is unknown. Ceyhan et al. 10 reported that exposure to isoflurane in rabbits had negative effects on spermatogenesis and sperm morphology. Conversely, to our knowledge, no study in the literature has investigated the impact of this anaesthetic on the oogenesis.
A good quality of oocytes is the key step of any biomedical research on gametes and embryos. In conclusion of this study, we have observed that mouse euthanasia by cervical dislocation was the best method to obtain intact oocytes in comparison with isoflurane inhalation.
Footnotes
ACKNOWLEDGEMENTS
We thank Michel Skandalovski and Jean-Marc Feuerstein for their precious technical assistance.
