Abstract
Isoflurane is currently the most common volatile anaesthetic used in laboratory mice, whereas in human medicine the more modern sevoflurane is often used for inhalation anaesthesia. This study aimed to characterize and compare the clinical properties of both anaesthetics for inhalation anaesthesia in mice. In an approach mirroring routine laboratory conditions (spontaneous breathing, gas supply via nose mask, preventing hypothermia by a warming mat) a 50 min anaesthesia was performed. Anaesthetics were administered in oxygen as carrier gas at standardized dosages of 1.5 minimum alveolar concentrations, which was 2.8% for isoflurane and 4.9% for sevoflurane. Both induction and recovery from anaesthesia proceeded quickly, within 1–2 min. During anaesthesia, all reflex testing was negative and no serious impairment of vital functions was found; all animals survived. The most prominent side-effect during anaesthesia was respiratory depression with hypercapnia, acidosis and a marked decrease in respiration rate. Under anaesthesia, heart rate and core body temperature remained within the normal range, but were significantly increased for 12 h after anaesthesia. Locomotor activity, daily food and water consumption and body weight progression showed no abnormalities after anaesthesia. No significant difference was found between the two anaesthetics. In conclusion, isoflurane and sevoflurane provided an equally reliable anaesthesia in laboratory mice.
Rodents are usually anaesthetized by injection of hypnotic, analgesic and muscle relaxant liquid agents. 1 Since continuous intravenous, target-controlled infusion, or the so-called total intravenous anaesthesia, with short-acting drugs such as propofol (e.g. ref. 2), is hard to master in mice, the intraperitoneal or subcutaneous application route is normally chosen in this species. 3–5 Although it would seem easy and practical to induce general anaesthesia with an injection of a single (e.g. pentobarbital) or mixed (e.g. ketamine/xylazine, medetomidine/midazolam/fentanyl) long-acting drug(s), this type of anaesthesia is hard to control. Once the initial dose has been administered, the duration and depth of anaesthesia cannot be adjusted to the specifics of the mouse (strain, age, gender, individual variation, etc.) or the surrounding conditions (time of day, housing conditions, etc.), all of which influence the animals' response to the anaesthetic. 6–10 Thus, despite prior dosage testing, managing injection anaesthesia often remains difficult, i.e. on the one hand anaesthesia is shallow in some individuals, and on the other, the death rate can be unexpectedly high. 3,4,11
Such failures are rarely encountered with inhalation anaesthesia. Modern, commercially available volatile anaesthetics such as isoflurane, sevoflurane, desflurane and others are vaporized in dedicated vaporizers, added to a carrier gas and supplied to the animal via the respiratory tract. Due to their low blood:gas partition coefficients, these compounds provide rapid induction of anaesthesia, are short-acting and are removed from the body in a short time, mostly by respiration. 12 The dosage can be adapted easily and can be titrated to effect for an individual animal. Thus, provided that the animals' vital functions and depth of anaesthesia are monitored, cases of death are unusual because the course of anaesthesia can be easily controlled. Thus, recently developed volatile anaesthetics are used increasingly in laboratory rodents, 1,13 especially since ready-to-use inhalation anaesthesia devices tailored for small rodents are commercially available. The most up-to-date anaesthesia equipment 14–16 normally includes active scavenging systems to prevent the release of waste gas, which is mandatory for protecting personnel and which has been a problem in the past. 17–20
The most common and well-known volatile anaesthetic in laboratory rodents is isoflurane. 1 Sevoflurane, a more modern inhalation anaesthetic, is used in human medicine, 21 but is uncommon in veterinary medicine due to its higher cost. To date, the clinical impacts of isoflurane and sevoflurane have been described mostly in man or in animal species other than mice. This led us to investigate the possible advantageous properties and drawbacks of isoflurane and sevoflurane anaesthesia in laboratory mice. These two substances were compared in a practical setting for their effects during and after anaesthesia from a clinical viewpoint, with the aim of determining their impact on animal physiology and general post-anaesthetic condition.
Materials and methods
Animals
Sixty-four, six-week-old female C57BL/6J mice were obtained from our in-house breeding colony. The mice were free of all viral, bacterial and parasitic pathogens listed in FELASA recommendations. 22 Health status was monitored by a sentinel programme throughout the experiments.
Animals were kept in type 3 open-top plastic cages (425 mm × 266 mm × 150 mm, floor area 820 cm2) with autoclaved aspen bedding (80–90 g/cage) (LTE E-001 Abedd, Indulab, Gams, Switzerland). Autoclaved hay (8–12 g/cage) and two Nestlets™ (each 5 × 5 cm), consisting of cotton fibres (Indulab), were provided as nesting materials. A standard cardbox house (Ketchum Manufacturing, Brockville, Canada) was provided. Animals were fed a pelleted mouse diet (Kliba No. 3431, Provimi Kliba, Kaiseraugst, Switzerland) ad libitum and had unrestricted access to sterilized drinking water. The light/dark cycle in the room consisted of 12/12 h with artificial light (approximately 40 lx in the cage) from 3:00 to 15:00 h. The temperature was 21 ± 1°C, with a relative humidity of 50 ± 5%, with 15 complete changes of filtered air per hour (HEPA H 14 filter); the air pressure was controlled at 50 Pa.
Mice were housed in groups, except during the four days before and four days after anaesthesia, when they were housed individually. The first day of single housing served for adaptation to the change in housing conditions; from the second day onwards, the individuals' normal values for heart rate, core body temperature, locomotor activity, body weight, food and water consumption were recorded. To avoid interfering influences, all necessary husbandry and management procedures were completed in the room before starting single housing of mice, and disturbances (e.g. visitors or unrelated experimental procedures) were not allowed. The animal room was insulated to prevent electronic noise.
This study was approved by the Cantonal Veterinary Office (Zurich, Switzerland) under the licence number 111/2007. Housing and experimental procedures were in accordance with Swiss animal protection law and conform to the European Convention for the protection of vertebrate animals used for experimental and other scientific purposes (Council of Europe No. 123, Strasbourg, 1985).
Preliminary transmitter implantation
Prior to the experiments, at age 10 weeks, 16 mice were instrumented with telemetric transmitters. TA10ETA-F20 transmitters (Data Sciences International, St Paul, MN, USA) – which measure heart rate, core body temperature and locomotor activity in freely moving mice – were implanted as previously described in detail. 23 Briefly, under anaesthesia with sevoflurane (Sevorane™, Abbott, Baar, Switzerland), the transmitter body was implanted in the abdomen under aseptic conditions. One wired loop electrode was fixed with silk sutures between the muscles located to the right of the trachea, whereas the other wired loop lead was sutured to the xiphoid process. Muscle layers and skin were closed with resorbable sutures. Postoperatively, buprenorphine (Temgesic™, Reckitt and Colman Products Ltd, Hull, UK) was given at a dose of 0.1 mg/kg body weight, injected subcutaneously twice per day for four days. 24 After transmitter implantation, the mice had a period of six weeks convalescence before the first experiment.
Experimental setting
All experiments were conducted when the mice were aged 16–36 weeks, with body weights ranging from 25 to 30 g. All experiments and weighing procedures were carried out between 15:00 and 18:00 h. Anaesthesia was performed in a separated operating area within the animal room.
Anaesthesia was provided by a commercially available rodent inhalation anaesthesia apparatus (Provet, Lyssach, Switzerland), which was equipped with interchangeable vaporizers for isoflurane (Ohmeda Isotec 5, Abbott) and sevoflurane (Ohmeda Sevotec 5, Abbott). As carrier gas, 100% oxygen was used at a flow rate of 400 mL/min. The anaesthetic gas was introduced into the nose mask (inner diameter 1.2 cm) through a thin tube (outer diameter 0.4 cm, inner diameter 0.3 cm). The opening of the thin tube was at a distance of exactly 0.5 cm from the latex membrane, which had a hole in the centre that fit around the nose of the mouse. The nose of the mouse was placed 2 mm in front of the opening of the inner tube. The nose mask merged into a thick outer tube (surrounding the thin inner tube), which allowed waste anaesthetic gas to be eliminated from the nose mask by a pump-driven filter system (flow rate 400 mL/min). The same principle was utilized for the induction chamber (inflow and outflow 400 mL/min). The concentrations of anaesthetic gases in the nose mask (at 2 mm in front of the opening of the inner tube) and in the induction chamber were measured at the beginning of anaesthesia and then every 5 min using a side stream monitoring device employing infrared technology (Datex-Ohmeda AS/3, Anandic Medical, Deissenhofen, Switzerland). The device was calibrated just before use using the proprietary standard reference gas supplied by the manufacturer.
Determination of minimum alveolar concentration
Forty-eight non-transmitter-implanted mice underwent anaesthesia three to four times in order to standardize anaesthesia by establishing minimum alveolar concentration. Care was taken that the animals had a break of at least two weeks between tests.
Minimum alveolar concentration was determined with a protocol modified from published methods. 25–27 Briefly, after inducing anaesthesia in the chamber for 2 min at maximum concentration of anaesthetic gases (5% isoflurane, 8% sevoflurane), the mouse was taken out of the chamber and placed in dorsal recumbency on a warmed mat (see below). Anaesthetic gas was then applied at the desired concentration via a nose mask, with the mouse breathing spontaneously. After an equilibration time of 10 min, painful stimuli in the form of pinching the tail, the interdigital webbing (pedal withdrawal reflex), the abdominal skin or neck skin were applied every 5 min for the next 30 min. All stimuli were induced by the same investigator by using blunt forceps containing a spacer between its arms. The motor response to a painful stimulus was evaluated as positive or negative, i.e. whether a motor response was visible or not.
Using this protocol, rough minimum alveolar concentration was first estimated within the concentration window (1–3% and 2–4% for isoflurane and sevoflurane, respectively) in which we empirically expected the minimum alveolar concentration to lie. Concentrations were graded in 0.25% steps and 10 animals per concentration were used.
Finally, four concentrations were chosen, and 25 animals per concentration were then tested. Minimum alveolar concentration was then calculated as the mean of the two partial pressures bracketing the response or lack of response in our tested population.
Anaesthesia experiments
Anaesthesia was induced by placing the mouse in a clear Perspex induction chamber (8 × 8 × 8 cm, volume 512 mL), into which either 5% isoflurane (Isoflo™, Abbot) or 8% sevoflurane (Sevorane™, Abbott) was then introduced. After 2 min, the animal was quickly transferred to a nose mask, where anaesthesia was maintained with 2.8% isoflurane or 4.9% sevoflurane, equivalent to 1.5 minimum alveolar concentrations (as established above). Mice breathed spontaneously while lying in dorsal recumbency on a water-filled warming mat (Gaymar, TP500, Orchard Park, NY, USA) set at 38 ± 1°C.
Tail pinch, pedal withdrawal and abdominal skin pinch reflexes were applied at 5 min intervals. All reflex tests were induced by the same investigator by using blunt forceps containing a spacer between its arms. The reflex tests were registered as positive or negative, i.e. whether any motor response was observable or not. Respiration rate was counted from the movement of the thorax wall.
Anaesthesia was stopped after 50 min by removing the nose from the mask and letting the mouse breathe room air. Two minutes later, when the mice had righted themselves from dorsal to ventral recumbency and were able to move, they were placed back in their home cage.
Telemetric data acquisition and analysis
Using the 16 transmitter-implanted mice, telemetric data were recorded in eight mice per anaesthetic. Mice were allocated randomly to the two groups. Telemetric data were recorded with the Dataquest LabPRO program (Data Sciences International). Data collection was initiated by switching on the transmitter with a magnet. Data acquisition started three days before anaesthesia and continued for the four days following anaesthesia.
To establish normal values (3 days before anaesthesia) and to investigate the postanaesthetic effects (4 days following anaesthesia), heart rate and core body temperature were measured every 5 min for 30 and 10 s, respectively. Locomotor activity was recorded continuously and stored at 5 min intervals.
To estimate the acute effects of anaesthesia (i.e. data measured during the 50 min anaesthesia experiment), heart rate and core body temperature were recorded for 4 s every 15 s (four measuring points of 4 s/min) while administering anaesthesia. From these data, the mean values of heart rate and core body temperature were calculated for each minute for each individual. Normal values represent means from the time period 15:00–18:00 h (i.e. the congruent time frame in which anaesthesia was carried out) during the three days prior to the experiment.
For analysis of long-term postanaesthetic effects, we took into account that values vary greatly during a 24 h cycle since mice are active mainly at night. Therefore, the means of the telemetric values for each animal were calculated separately for the night (12 h dark) and day (12 h light) phases. An individual's normal values were established by calculating means from the three days prior to anaesthesia. For each day after anaesthesia, the mean of the dark and light phase was compared with the individual's normal values, resulting in delta values.
Changes in body weight, and food and water intake
Body weight progression and daily food and water consumption were established from transmitter-implanted mice for three days before and three days after anaesthesia. Weights (animal, food pellets, water bottle) were recorded with a precision balance (PR 2003 Delta Range, Mettler-Toledo AG, Greifensee, Switzerland), especially adjusted for use with moving animals. Body weights recorded in transmitter-bearing mice were corrected to take into account the weight of the transmitter (3.6 g). The mean normal weights (from 3 consecutive daily measurements prior to the experiment) were calculated for each mouse, and compared with the weights recorded on each of the three days afterwards.
Acid–base balance and blood gas concentration
Three to four weeks after minimum alveolar concentration determination, the same 48 non-transmitter-implanted mice were used to obtain arterial blood with which to assess the acute side-effects of the anaesthetics used on respiration and acid–base balance.
Arterial blood was taken under anaesthesia at time points 10, 30 and 50 min of anaesthesia from eight mice per anaesthetic and time point. Following incision of the anterior neck, dissection of the right common carotid artery and cutting a small hole in the artery using a fine-bladed pair of scissors, arterial blood was collected in a heparinized syringe. Acid–base balance (pH), partial pressure of carbon dioxide (pCO2, mmHg) and standard bicarbonate (HCO3, mmol/L) were determined immediately using a blood gas analyser (AVL Compact 3, AVL List, Graz, Austria). These animals died immediately from the subsequent rapid loss of blood under anaesthesia. The normal values of pH, pCO2 and HCO3 used for comparison had been established previously from the arterial blood of 20 HanRcc:NMRI mice of similar age as those used in the present study. 4
Statistical analysis
All data are presented as mean ± standard deviation. Statistical analysis using SPSS for Windows, version 13.0 was carried out to validate the results of the postanaesthetic effects of isoflurane and sevoflurane. One-way analysis of variance was performed to compare group means of heart rate, core body temperature and locomotor activity at each of the four days after anaesthesia in both anaesthetics with normal values. Post hoc analysis with Bonferroni tests was carried out to identify significant differences between groups; P values ≤0.05 were considered significant.
Results
Minimum alveolar concentration
Mean minimum alveolar concentration was established as 1.85% (±0.15%) for isoflurane and 3.25% (±0.14%) for sevoflurane in the adult female C57BL/6J mice used here. All anaesthesia experiments were conducted with 2.8% isoflurane or 4.9% sevoflurane, under which none of the mice showed a motor response to testing of the pedal withdrawal reflex, tail pinch or abdominal skin pinch.
Acute effects of anaesthesia
All mice were clearly immobilized within one minute after placing in the induction chamber. Monitoring of heart rate, core body temperature and respiration rate during anaesthesia revealed no deviation from normal values in heart rate and core body temperature. In contrast, the respiration rate decreased markedly below the normal values of the resting mouse (Figure 1).
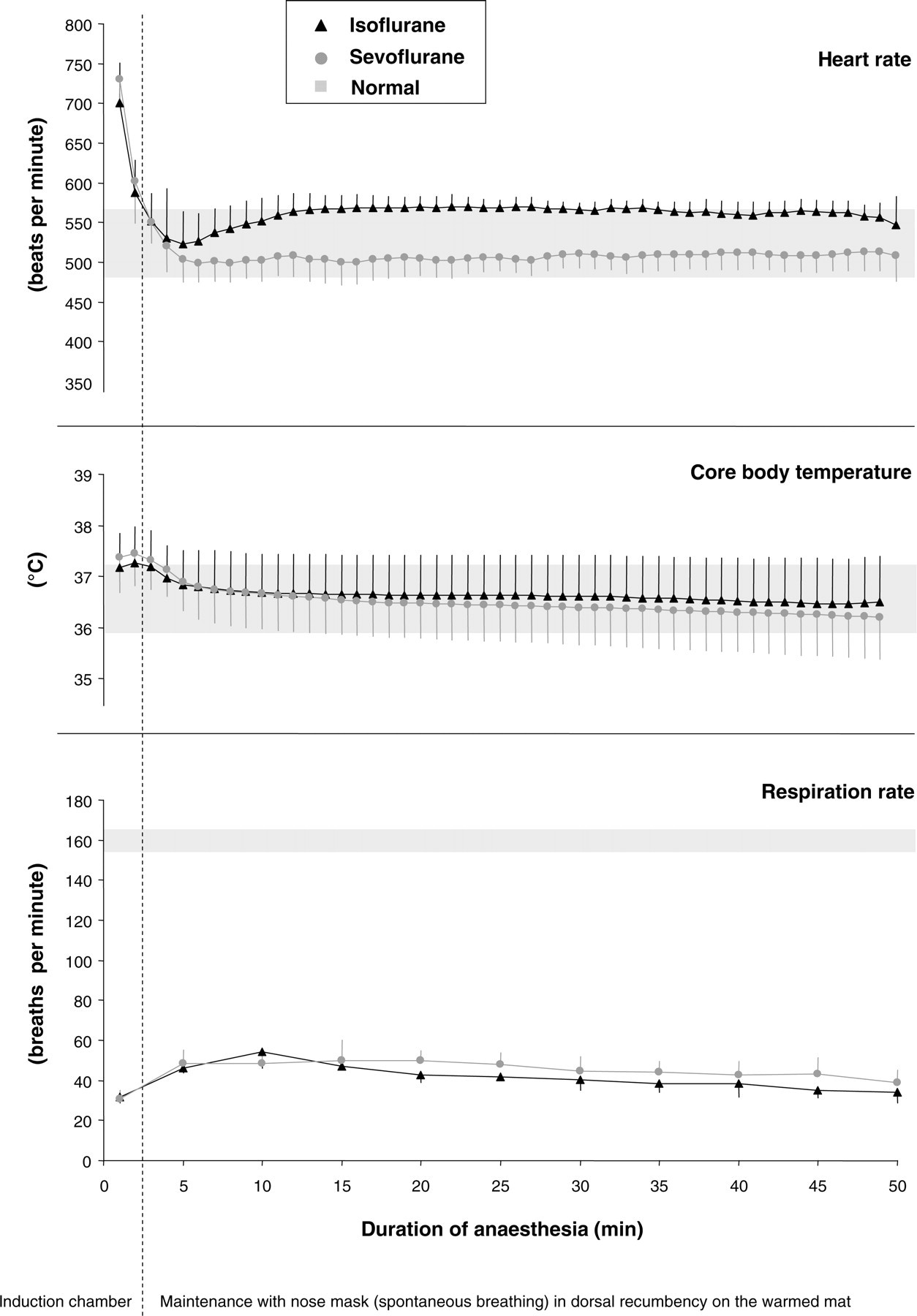
Heart rate, core body temperature and respiration rate during 50 min of anaesthesia with isoflurane or sevoflurane. The grey areas indicate normal values at the corresponding time of day in conscious animals. Data represent the mean values of eight mice; bars represent standard deviation
Acid–base balance and blood gas measurement in the arterial blood showed acidosis (i.e. decrease of pH) and hypercapnia (i.e. increase of pCO2) at 10, 30 and 50 min of anaesthesia (Figure 2).
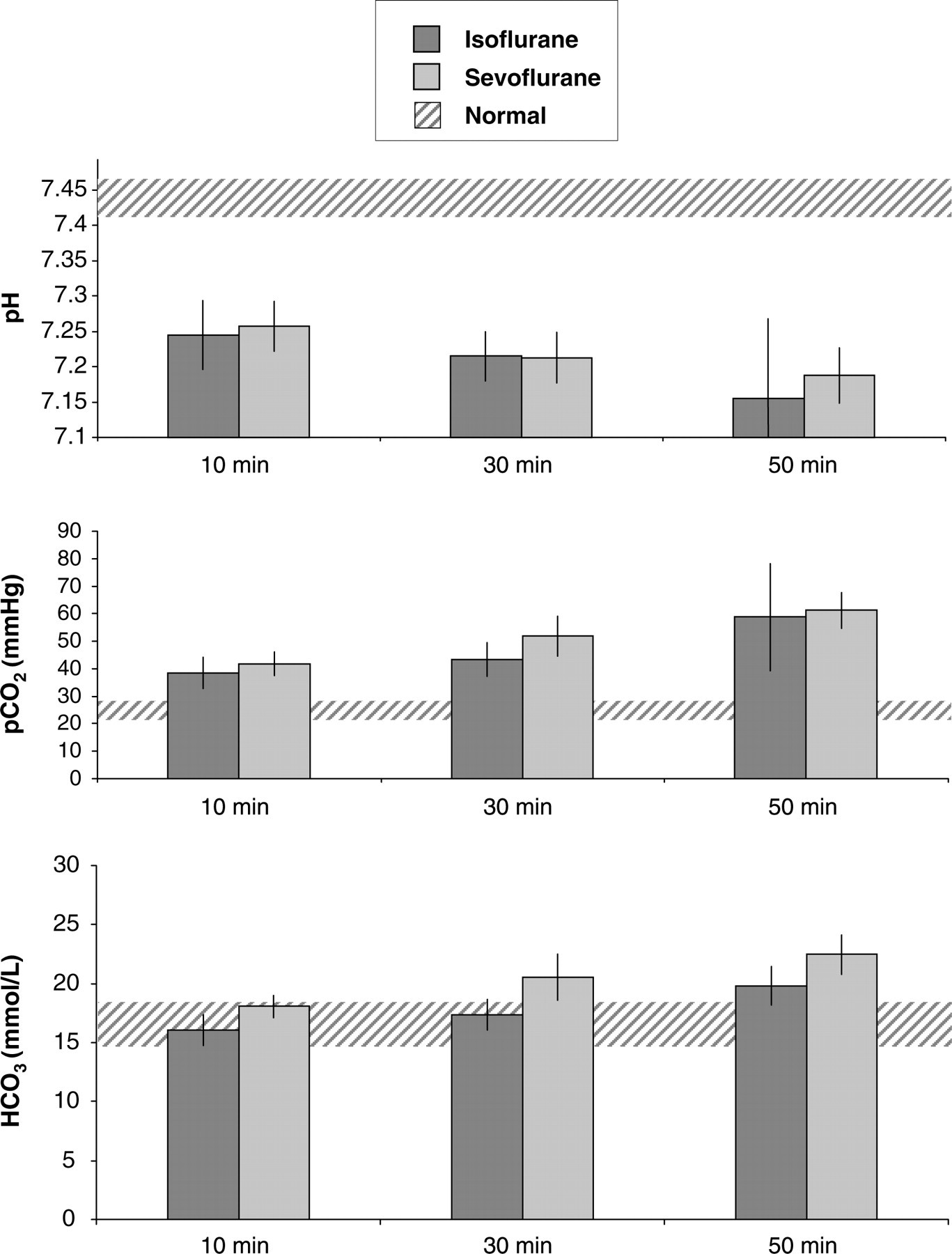
Acid–base balance (pH), partial pressure of carbon dioxide (pCO2) and standard bicarbonate (HCO3) in arterial blood at 10, 30 and 50 min of anaesthesia. Hatched areas indicate the normal values established in a previous study.4 Data represent the mean values of eight mice; bars indicate standard deviation
When anaesthesia was completed, i.e. when the nose mask was removed, animals showed increasing respiration rate and muscle rigour within one minute. Mice turned to sternal recumbency and showed spontaneous movement at 1–2 min after anaesthesia was withdrawn.
Postanaesthetic effects
Telemetric measurements revealed a significant increase in heart rate and core body temperature for 0–12 h after anaesthesia compared with normal values (i.e. before anaesthesia) with both anaesthetics. In this time frame, locomotor activity also showed a tendency to increase, but this was not significant for either anaesthetic (Figure 3). Statistical comparison of isoflurane versus sevoflurane regarding their long-term effects on heart rate, core body temperature and locomotor activity revealed no difference between these two anaesthetics.
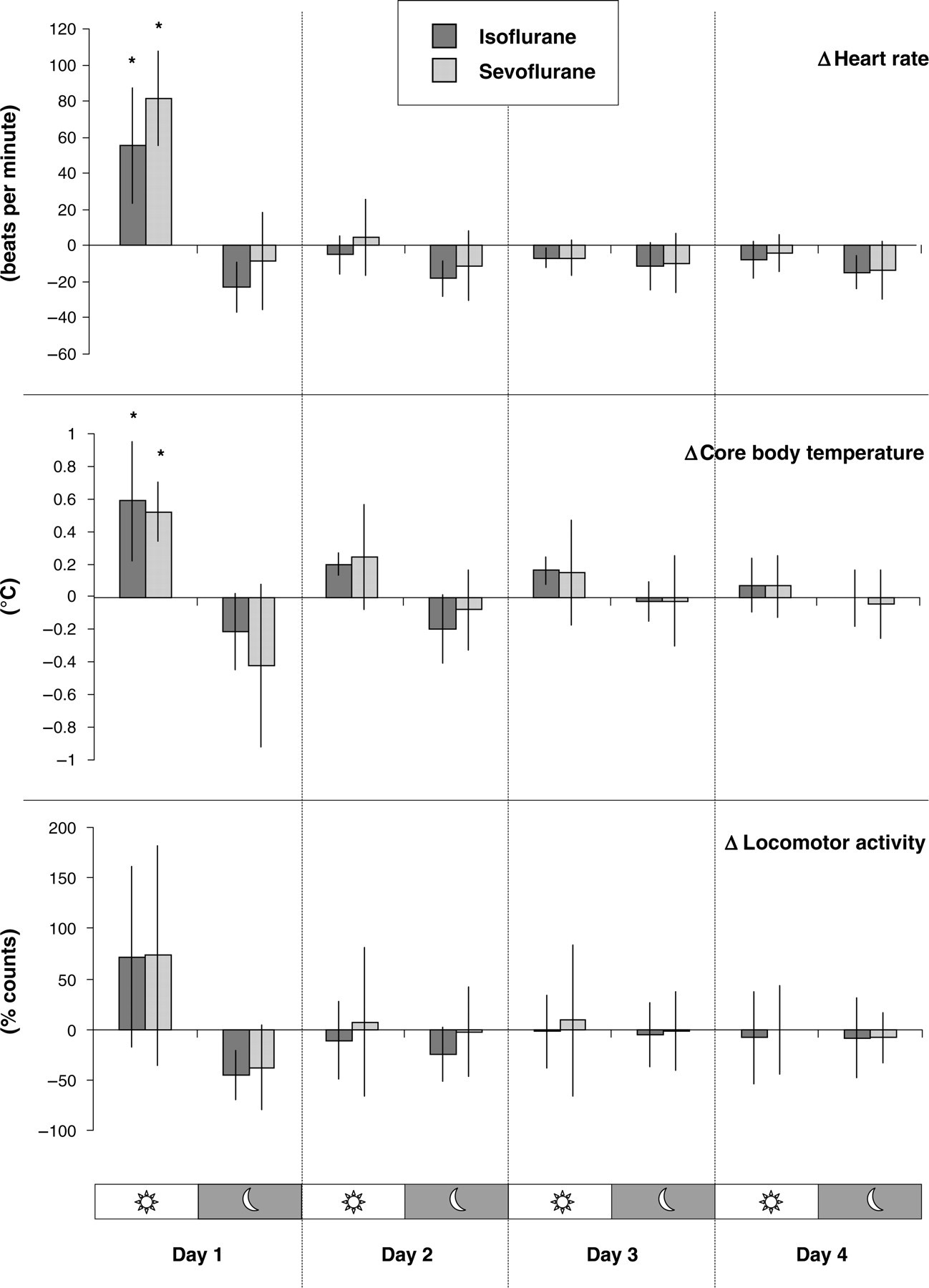
Postanaesthetic measurements of the impact of isoflurane and sevoflurane on heart rate, core body temperature and locomotor activity. Delta (Δ) values represent deviation from normal values (established prior to anaesthesia) at the corresponding 12 h day and night time. Data represent the mean from eight mice; bars indicate standard deviation. Asterisks indicate significance at P ≤ 0.05
Body weight with both anaesthetics was constant in the postanaesthetic phase, i.e. mean body weights varied with ≤0.5% (±0.01%) compared with the normal body weight before anaesthesia. The mean daily food intake showed a decrease of 10% (±0.2%) at the first postanaesthetic day in both anaesthetics. At the second postanaesthetic day, mean food consumption was almost unchanged, with a decrease of 1.5% (±0.2%) in both anaesthetics. At the third postanaesthetic day, food intake was decreased with 5% (±0.1%) in isoflurane and 9% (±0.1%) in sevoflurane. Water consumption showed high interindividual variability. The mean water consumption in both anaesthetics showed an increase of 6% (±19% for isoflurane; ±25% for sevoflurane) at the first day after anaesthesia. Mean water intake ranged from a decrease of 1.5% to an increase of 8.7%, with standard deviations ranging from 12% to 19% at the second and third postanaesthetic days in both anaesthetics. The alterations in body weight, food and water intake were not statistically significant.
Discussion
Prior to these investigations, the anaesthetic procedure and dosage of anaesthetics were standardized based on the specifics of the equipment (e.g. calibrating the gas concentration) and mouse population (e.g. strain, age, gender) used. Therefore, minimum alveolar concentrations were established following the widely accepted method of determining the concentration of anaesthetic gas at which 50% of the animals fail to respond with purposeful movements to the testing of reflexes. The minimum alveolar concentration determined for sevoflurane in female C57BL/6J mice was almost identical to that described for male outbred mice (3.25% in female C57BL/6J versus 3.22% in male CD-1). 28 The minimum alveolar concentration is known to vary considerably between mouse strains, 26 but other factors such as age and gender can also influence anaesthetic potency. Gender differences may explain why the minimum alveolar concentration for isoflurane was found to be higher (1.85%) than values described in the literature for C57BL/6J mice (1.30% 27 and 1.34% 29 ). In former reports, male mice 27 or a mixture of both sexes 29 were used, whereas in our experiments only female mice were investigated. On the other hand, the suggestion that anaesthetic requirements decrease with age, and literature reports of lower minimum alveolar concentrations in younger mice (7–9 weeks 27 , 6–12 weeks 29 ) are in contrast to the 0.5% increase found in our study, in which older mice (16–36 weeks) were used. However, when comparing minimum alveolar concentrations established in different laboratories, technical aspects of how the gas was provided must be considered. The easy-to-use open anaesthesia system used here might include an uncertainty in the absolute values of the gas concentration measurements.
After preliminary standardization of the dosages by establishing the minimum alveolar concentrations, the anaesthetics were then compared with dosages of isoflurane and sevoflurane representing 1.5 minimum alveolar concentrations. For this dosage it is generally postulated that 99.9% of animals will not react to painful stimuli, 30,31 i.e. that the animals have reached surgical tolerance. However, since analgesia was not proved with sophisticated methods such as measurements of the reaction in heart rate, blood pressure or respiration upon a substantially painful stimulus (e.g. skin incision), surgical tolerance cannot be definitively claimed from our study. However, motor reflex responses were suppressed in all animals and none died from the 50 min inhalation anaesthesia with 1.5 minimum alveolar concentrations (i.e. 2.8% isoflurane or 4.9% sevoflurane).
In the early induction phase of anaesthesia, heart rate peaked at the upper normal level of 700–800 beats per minute (bpm), which we suggest as a normal reaction to removing the animal from its cage and placing it in a foreign environment. During anaesthesia, heart rate was stable within the normal values of the resting mouse (490–550 bpm). This was in agreement with recent publications indicating only slight depression of heart rate for isoflurane anaesthesia. 13,32–35
During 50 min of inhalation anaesthesia, core body temperature could be maintained with a simple, water-bath-driven warming mat. That core body temperature falls due to any kind of anaesthesia is well known, and mice are especially sensitive to hypothermia due to their small size and high body surface area (e.g. a drop to 30–31°C was shown following isoflurane anaesthesia in mice 35 ). Obviously, such hypothermia should be prevented, because it influences physiology and the course of anaesthesia and can ultimately lead to the death of the animal. Thus, warming the animal has been common practice for years, and is particularly worthwhile in long-term anaesthesia in mice. 36–38
In contrast to the almost normal levels of heart rate and core body temperature, respiratory depression, as evidenced by decreased respiration rates (far below the values of the resting mouse), marked hypercapnia and acidosis, were seen to an equal extent with both isoflurane and sevoflurane anaesthesia. Hypercapnia and acidosis associated with isoflurane has also been found by others, 13 but was less intense than with injection anaesthesia with pentobarbital. 34 Other studies also report a marked decrease in respiration rate upon isoflurane anaesthesia, but values reported in the literature 36,38 were not as low as found here with both anaesthetics.
Thus, respiratory depression was the major adverse effect observed with both isoflurane and sevoflane. In general, the influence of inhalational anaesthetics on respiratory function is well known. By inhibiting the control systems of respiration (e.g. feedback control of central respiratory centres, various chemoreceptors, pulmonary reflexes and neuronal input) inhalational anaesthetics alter oxygen supply and CO2 elimination. 39 This is mirrored by aberrations in arterial blood gas levels (e.g. increase of pCO2, decrease of pO2) and thus hampers the ability of the organism to maintain cellular homeostasis. Respiratory depression is often accompanied by acid–base imbalance (e.g. alterations of pH and HCO3), and changes in the depth and frequency of respiration. Since respiratory depression is the most probable emergency situation when using isoflurane or sevoflurane anaesthesia, it may be useful to monitor the respiration rate as an indirect indicator of impaired respiration and thus prevent fatal outcomes under routine conditions. In addition, administering oxygen instead of room air in the recovery phase (i.e. after the volatile anaesthetic ceased) may be beneficial for animals with impaired respiration.
The time required for induction and recovery were almost the same for both anaesthetics (1–2 min). The short recovery time from both inhalational anaesthetics should be highlighted as a distinct advantage compared with injection anaesthesia. The benefits of fast recovery include reducing postoperative complications associated with prolonged inability to correct physiological impairment (e.g. hypothermia, hypoglycaemia, dehydration) that may induce suffering and hamper the rapid return of the animal to its normal state.
Heart rate and core body temperature increased significantly in the 12 h following the 50 min anaesthesia. Locomotor activity showed a tendency to increase only in the first hours after anaesthesia, suggesting that physical activity is not the reason for the elevated heart rate and core body temperature. Alterations in body weight progression as well as daily food and water consumption are known to hint at pain, distress or impaired wellbeing in laboratory animals. 40,41 Postanaesthetic determination of these indices revealed no relevant aberrations, indicating the negligible impact of a 50 min anaesthesia with isoflurane or sevoflurane on body weight, food and water intake. Although it is unclear why the postanaesthetic elevation in heart rate and core body temperature occurred, it appeared to be of no long-term detriment to the animals.
Considering the effects of isoflurane and sevoflurane on physiology and behaviour, we found that both agents exerted a similar impact on the normal state established in the unanaesthetized mouse. Whereas several publications describe the usefulness of isoflurane, there is limited description of sevoflurane in mice in the literature. Henke and co-authors 36 compared induction and recovery times, and respiration rates of sevoflurane with those of isoflurane in the gerbil. Although they found prolonged recovery from isoflurane compared with sevoflurane, they concluded that there is no overall preference for one of these two volatile anaesthetics over the other. Another study compared blood glucose and some specific parameters required in functional PET investigations in mice; sevoflurane was considered superior compared with isoflurane, and the former was consequently recommended for physiological imaging by Flores and co-authors. 42 From our results, neither anaesthetic was clearly superior over the other.
In summary, we conducted inhalation anaesthesia in a routine, cost-effective setting, using commercially available equipment. The anaesthesia experiments were standardized by establishing minimum alveolar concentrations, i.e. the dosage was adjusted to the characteristics of the animals used (female C57BL/J mice, aged 16–36 weeks). Both volatile anaesthetics tested showed short induction and recovery times in an easy-to-manage, standard inhalation anaesthesia procedure. During anaesthesia, the most prominent adverse effect was respiratory depression. Hypothermia, which generally occurs under anaesthesia, was prevented by placing the animal on a warmed mat. After completion of anaesthesia, altered physiological functions, such as elevated heart rate and core body temperature, persisted for approximately half a day. In conclusion, both isoflurane and sevoflurane provided an equally effective anaesthesia with acceptable adverse effects.
Footnotes
Acknowledgement
This work was sponsored by the ECLAM and ESLAV Foundation. The authors would like to thank Robin Schneider and the staff of the central biological laboratory for support in housing the mice. We thank Professor Kurt Burki for generously providing research facilities and resources.
