Abstract
Halothane minimum alveolar concentration (MAC)-sparing response is preserved in rats rendered tolerant to the action of dexmedetomidine. It has been shown that halothane and isoflurane act at different sites to produce immobility. The authors studied whether there was any difference between halothane and isoflurane MAC-sparing effects of dexmedetomidine in rats after chronic administration of a low dose of this drug. Twenty-four female Wistar rats were randomly allocated into four groups of six animals: two groups received 10 μg/kg intraperitoneal dexmedetomidine for five days (treated groups) and the other two groups received intraperitoneal saline solution for five days (naive groups) prior to halothane or isoflurane MAC determination (one treated and one naive group of halothane and one treated and one naive group of isoflurane). Halothane or isoflurane MAC determination was performed before (basal) and 30 min after an intraperitoneal dose of 30 μg/kg of dexmedetomidine (post-dex) from alveolar gas samples at the time of tail clamp. Administration of an acute dose of dexmedetomidine to animals that had chronically received dexmedetomidine resulted in a MAC-sparing effect that was similar to that seen in naive animals for halothane; however, the same treatment increased the MAC-sparing response of dexmedetomidine for isoflurane. Isoflurane but not halothane MAC-sparing response of acutely administered dexmedetomidine is enhanced in rats chronically treated with this drug.
Dexmedetomidine, a highly selective α 2-adrenoceptor agonist, was developed as a sedative and analgesic drug for use in intensive care medicine. 1,2 However, it has pharmacodynamic properties that make it useful in anaesthesia. In particular, its application as premedication, analgesia, hypnotics/sedatives to relieve patient anxiety 3,4 and decrease intraoperative requirements for inhalant anaesthetics, 5 opioids and hypnotic induction agents 6,7 is becoming increasingly popular with delivery to a wide spectrum of patients. 8
Prolonged administration of dexmedetomidine could affect the number of available α 2-receptors and the induced responses after its acute administration. It has been demonstrated that tolerance to both the hypnotic and analgesic effects of α 2-agonists develops after chronic treatment with dexmedetomidine. 9,10 However, the halothane minimum alveolar concentration (MAC)-sparing effect of dexmedetomidine is preserved in rats after chronic administration of the drug. 11
The hypnotic 12,13 and antinociceptive 14 responses to dexmedetomidine are mediated via activation of central α 2-adrenoceptors in the locus coeruleus in rats, where it decreases the activation of neurons. 15 On the other hand, the precise mechanism via which inhalant anaesthetics exert their anaesthetic effects is not yet known. It has been shown that halothane and isoflurane have differential effects on excitatory and inhibitory receptors in the central nervous system, 16,17 and they also act at different sites of the spinal cord to produce immobility. 18
The aim of the present study was to determine whether there was any difference between halothane and isoflurane MAC-sparing effects of acutely administered dexmedetomidine in rats treated chronically with a low dose of this drug.
Materials and methods
Animals and husbandry
Twenty-four female specific pathogen free (SPF) Wistar Hannover rats (Harlan Laboratories Models, SL, Barcelona, Spain) with a mean body weight of 200 ± 10 g (range 190–210 g) were allowed to acclimatize for at least one week before the studies took place and they did not receive any medication. Animals were housed in groups of six in U-TEMP polyetherimide cages (1145T Sealsafe®, Tecniplast SpA Varese, Italy), with a bedding (HBK 1500–3000, Harlan Laboratories Models, SL) and free access to food (Global diet 2014 for rodents maintenance, Harlan Laboratories Models, SL) and water, and maintained on a 12 h light/12 h dark cycle (light on at 07:00 h) under controlled environmental conditions (50–70% relative humidity, 20 ± 2°C temperature). All the studies were performed in the morning (09:00–12:00 h). All animals were handled according to the guidelines set in the ‘Guide for the Care and Use of Laboratory Animals’ published by the National Institutes of Health. The institutional animal care and use committee approved the study.
Experimental design
Animals were randomly allocated into four groups of six animals: two groups received a chronic (5 days) intraperitoneal dose of 10 μg/kg of dexmedetomidine (Precedex®, Abbott Laboratories, Rocky Mount, NC, USA) (treated groups) and two other groups received a chronic (5 days) intraperitoneal administration of a volume of saline solution (0.9% NaCl solution, B/Braun Medical SA, Barcelona, Spain) equivalent to 10 μg/kg of dexmedetomidine (naive groups).
On the sixth day, naive and treated groups were tested for their isoflurane (Forane®, Abbott Laboratories Ltd, Kent, UK) (1 naive and 1 treated group) or halothane (Fluothane®, AstraZeneca SA, Madrid, Spain) (1 naive and 1 treated group) MAC-sparing response before (basal) and 30 min after an intraperitoneal dose of 30 μg/kg of dexmedetomidine (post-dex).
The animals were placed in an induction chamber to which 4% halothane or 5% isoflurane in a continuous oxygen flow of 3 L/min was directed. Once the animals were anaesthetized, tracheal intubation was performed using a 16-gauge polyethylene catheter (Abbott Ireland, Sligo, Republic of Ireland). With the animal positioned in dorsal recumbency, a flexible, blunt-tip, wire guide was inserted into the trachea with an otoscope and used to direct the endotracheal catheter. The catheter was previously marked and advanced with the tip located 3–5 mm cranially to the carina. After the correct position of the catheter was ascertained by CO2 infrared-absorption analysis (8050, Dräger Medizintechnik GmbH, Lübeck, Germany), it was connected to a small T piece of minimal dead space (<0.2 mL). The proper catheter position of the animals was checked at the end of the study. Oxygen flow to the T piece was adjusted to 1 L/min, and halothane or isoflurane concentration was adjusted as necessary following anaesthetic reflex assessment. During the study, the animals were breathing spontaneously.
The carotid artery was catheterized with a 24-gauge polyethylene catheter (Abbott Ireland) via surgical cut-down. This access allowed for arterial blood sampling and blood pressure measurement via a calibrated pressure transducer (Transtar TM DPT, Medex Medical Inc, Haslingden, UK). Arterial blood pressure and heart rate were recorded continuously (PM 8060 vitara, Dräger Medizintechnik GmbH). Arterial blood gases were measured (1306 pH/Blood Gas Analyser, Instrumentation Laboratory, Barcelona, Spain) periodically during MAC assessment and at the end of the study period to ensure that values were within normal limits for pH (7.30–7.40), pressure of oxygen (PaO2) (>90 mmHg) and pressure of carbon dioxide (PaCO2) (40–45 mmHg). Rectal temperature was also monitored and maintained at normothermia (between 37°C and 38°C) by means of a total temperature management system (Bair Hugger® mod. 505, Augustine Medical Inc, Eden Prairie, MN, USA). Inspired halothane or isoflurane concentrations were further decreased to a value close to the average MACHAL and MACISO for rats (1.2 and 1.4%, respectively), 19 before the first MAC determination.
Intratracheal gas sampling was used to measure anaesthetic gas concentration for determination of the MAC. Inspired and end-tidal halothane or isoflurane concentrations were obtained continuously from gas drawn from a fine tubing (Portex® model 100/383/118, Portex Ltd, Hythe, UK) inserted through the endotracheal catheter over a hole in the T piece and with the tip located at the level of the carina. The proximal end of the catheter was connected to a calibrated infrared absorption analyser (8050, Dräger Medizintechnik GmbH) with a 60 mL/min aspiration flow of the gas sample. After every step change in the anaesthetic concentration delivered by the anaesthetic circuit, at least 15 min was allowed for equilibration maintaining a constant alveolar concentration and an alveolar-to-inspired ratio (F A/F I) of more than 0.95.
The MAC of the inhaled anaesthetic values was established according to the method described previously. 20 A painful noxious stimulus was applied with an eight-inch haemostat clamped (Aesculap®, Aesculap AG, Tuttligen, Germany) to the first ratchet lock on the tail for 60 s (less if the animal moved). The tail was always stimulated proximal to a previous test site. A positive response was considered when a gross purposeful movement of the head, extremities or body, or both, was observed, whereas a negative response was the lack of movement, or the presence of swallowing, chewing or tail flicking. The anaesthetic concentration was then reduced in decrements of 0.1–0.2% until the negative response became positive. The inhaled anaesthetic MAC was defined as the average of the lowest concentration preventing a positive response and the highest concentration allowing a positive response to the supramaximal painful stimulus.
After basal MAC determination, all groups received a 30 μg/kg intraperitoneal dose of dexmedetomidine and 30 min later, isoflurane or halothane MAC-sparing responses were tested again (post-dex MAC). The individual testing for the MAC was blinded to volatile agent and dexmedetomidine treatment. For each animal, MAC was corrected to a sea-level barometric pressure of 760 mmHg.
At the end of each experiment animals were deeply anaesthetized and euthanized with 50 mEq/kg intravenous potassium chloride (UCB Pharma, SA, Barcelona, Spain).
Statistical analysis
Statistical analysis of data was performed using SPSS 15.0 software program (SPSS Inc, Chicago, IL, USA). All data were grouped and summarized as mean ± standard deviation. Data were analysed using a Shapiro–Wilk normality test, and then comparison between naive and treated groups for basal and post-dex was performed by t-test for unpaired data and expressed as mean ± standard deviation. Basal and post-dex mean differences interaction in naive and treated groups was performed by t-test for paired data and expressed as mean difference at 95% of confidence interval. Statistical significance was accepted at P values <0.05.
Results
Chronic administration of 10 μg/kg intraperitoneal dexmedetomidine for five days did not alter the basal MAC for halothane (1.26 ± 0.07% for naive group versus 1.27 ± 0.14% for treated group) or isoflurane (1.51 ± 0.15% for naive group versus 1.40 ± 0.14% for the treated group) (Figure 1).
Effect of five-day administration of 10 µg/kg intraperitoneal dexmedetomidine (treated group) or saline (naive group) on the minimum alveolar concentration-sparing effects of halothane and isoflurane. On the fifth day, minimum alveolar concentration of halothane and isoflurane were determined in each animal, before (basal) and after (post-dex) administration of 30 μg/kg intraperitoneal dexmedetomidine. N = 6 animals per group. Data are expressed as means ± SD. *P < 0.05 compared with the basal state. #
P < 0.05 compared with the naive group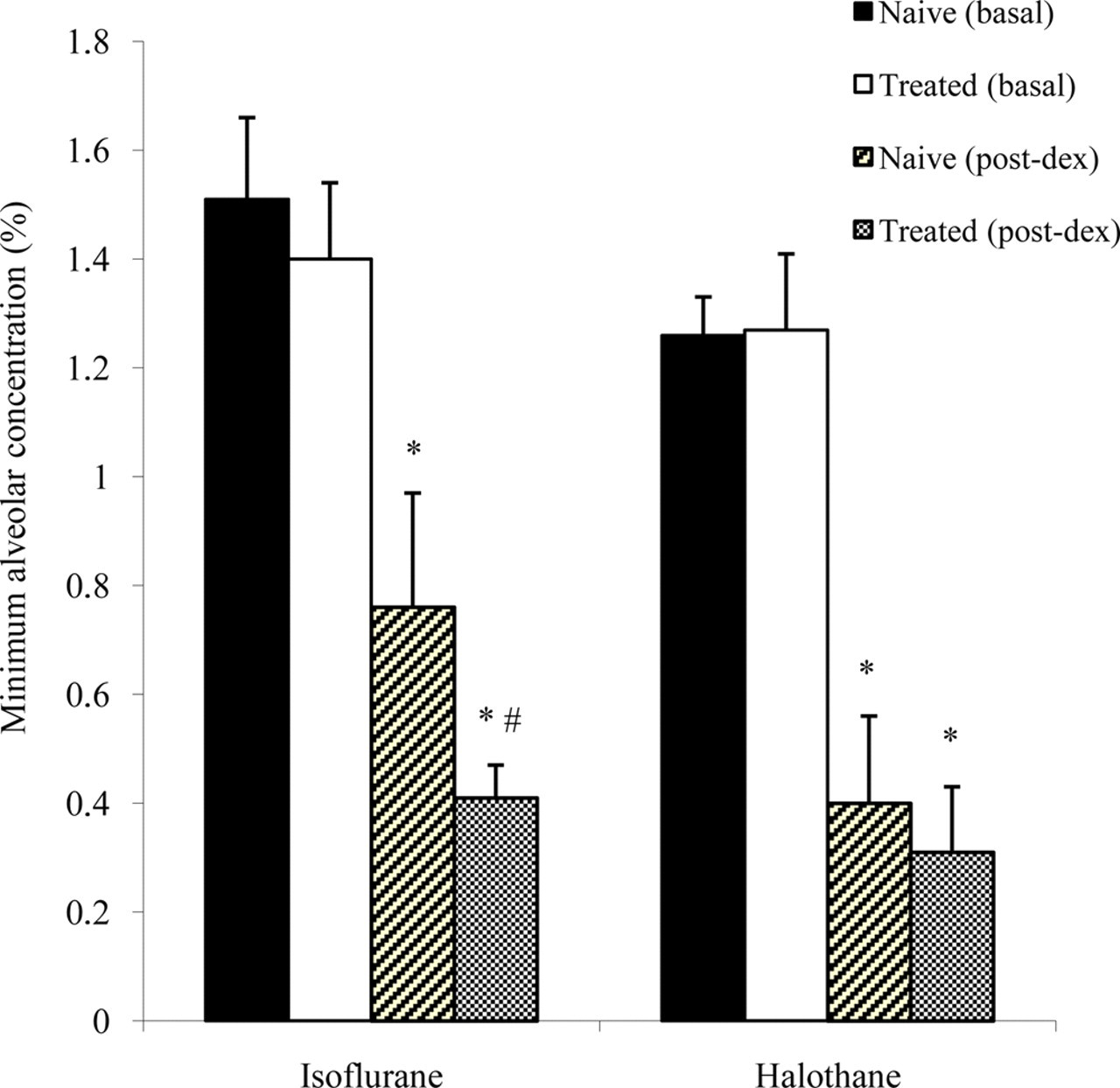
Acute administration of 30 μg/kg of dexmedetomidine significantly reduced (P < 0.05) halothane (0.40 ± 0.16% for naive group and 0.31 ± 0.12% for treated group) and isoflurane (0.76 ± 0.21% for naive group and 0.41 ± 0.06% for treated group) MAC values with respect to their respective basal values (Figure 1).
Acute dexmedetomidine administered to animals receiving chronic treatment resulted in a MAC-sparing effect similar to that seen in the naive animals for halothane (0.96 [confidence interval [CI] 95% 0.86–1.06] versus 0.88 [CI 95% 0.65–1.08], P = 0.30); however, acute administration of dexmedetomidine to the animals receiving chronic treatment resulted in a significantly greater MAC-sparing response than that observed in the naive group for isoflurane (0.98 [CI 95% 0.83–1.14] versus 0.77 [CI 95% 0.63–0.91], P = 0.027) (Figure 1).
Measured physiological data (mean ± SD) at the time of the determination of isoflurane or halothane minimum alveolar concentration after five-day administration of 10 μg/kg intraperitoneal dexmedetomidine (treated group) or saline (naive group)
N = 6 animals per group. Data are expressed as means ± SD. *P < 0.05 compared with the basal state
On the fifth day, minimum alveolar concentration of halothane and isoflurane were determined in each animal, before (basal) and after (post-dex) administration of 30 μg/kg intraperitoneal dexmedetomidine
PaCO2: partial pressure of carbon dioxide; PaO2: partial pressure of oxygen
At the time of the MAC determination there were no differences in pH, PaO2, PaCO2, mean arterial blood pressure and temperature among the studied groups; only heart rate for halothane and isoflurane was statistically lower after acute administration of intraperitoneal dexmedetomidine in both the naive and treated dexmedetomidine groups (Table 1).
Discussion
Chronic administration of dexmedetomidine does not alter the isoflurane or halothane MAC. Acute dexmedetomidine reduces halothane and isoflurane MAC in both chronically-treated and non-treated dexmedetomidine groups; however, isoflurane but not halothane MAC-sparing response of acutely administered dexmedetomidine was significantly greater in rats after chronic administration of the drug.
The hypnotic 12,13 and antinociceptive 14 responses to dexmedetomidine are mediated via activation of central α 2-adrenoceptors in the locus coeruleus in rats, where it decreases activation of neurons. 15 However, dexmedetomidine also acts at the level of the spinal cord, where dexmedetomidine directly inhibits nociceptive processing. 21,22 This action is mediated by α 2A-adrenoceptor subtypes 23 mostly located presynaptically on the primary afferent neuron.
Several studies demonstrate that the spinal cord mediates most of the ability of inhaled anaesthetics to produce immobility; 24–26 furthermore, halothane reduces noxious-evoked movement at least partly via depression of dorsal horn neurons, whereas isoflurane suppresses movement by an action at more ventral sites in the spinal cord. 18 Other results suggest that the immobility produced by inhaled anaesthetics need not result from their capacity to suppress transmission through dorsal horn neurons. The locus coeruleus has been suggested as another site of halothane anaesthetic action, where there are significant changes in neurotransmitter content and metabolic activity after halothane anaesthesia. 27,28
Dexmedetomidine reduces minimum alveolar anaesthetic requirements for halothane and isoflurane in a dose-dependent manner in rats; 29–31 so that a dose of 100 μg/kg intraperitoneal halothane could be discontinued for up to 30 min without eliciting a purposeful response to tail clamping, 29 and isoflurane MAC could be reduced by approximately 90%. 30 The reduction of halothane MAC caused by dexmedetomidine is mediated through α 2-adrenoceptors; 29 however, α 2-adrenoreceptors do not or minimally mediate the capacity of isoflurane to produce immobility. 32 Different side interaction between dexmedetomidine and both inhaled anaesthetics could probably explain the stronger interaction seen between dexmedetomidine and halothane, 33 since this α 2-adrenoceptor agonist exerts its antinociceptive effect by reducing dorsal horn activity. 21,22
It has been demonstrated that tolerance to both the hypnotic and analgesic effects of α 2-adrenergic agonists develops after chronic treatment with dexmedetomidine. 9,10 However, the halothane MAC-sparing response of dexmedetomidine persists in rats rendered tolerant to the hypnotic and analgesic properties of the drug. 11 It was explained by a comparatively larger receptor reserve than is needed for the hypnotic and analgesic responses in the locus coeruleous, 11 a discrete brain region with an unusual abundance of spare α 2-adrenergic receptors, 34 such that it is unaffected by the reduction in receptor reserve elicited by the same drug treatment regimen. 11
We have observed the same results for halothane in rats after chronic administration of a much lower dose of dexmedetomidine than that used by Rabin et al.; 11 but the isoflurane MAC-sparing effect of acutely administered dexmedetomidine is enhanced in rats chronically treated with the drug. A potential explanation of this may be that halothane MAC was reduced by about 68% while isoflurane MAC was reduced to about 51% by the same dexmedetomidine dose may be the result of a floor effect, i.e. halothane MAC was more resistant to further reduction by dexmedetomidine. When comparing interaction effect in halothane groups, the power may not be sufficient to detect a difference with the calculated size of the groups, although we had a not insignificant effect size of 0.617. A new, different interaction between dexmedetomidine and both inhaled anaesthetics could explain our findings. Although dexmedetomidine and isoflurane suppress nociceptive neurotransmission in rat spinal cord via the same neurotransmitters, there is no evidence that they act at the same cellular site(s), 35 so it is possible that dexmedetomidine acts on some subset of sites acted on by halothane, such as the locus coeruleous, 27,28 but not isoflurane. Unfortunately, our study did not allow us to determine the mechanism of action.
The measured MAC values of an anaesthetic can be altered by differences in animal physiology. During our MAC determinations, there were no differences in arterial blood pressure, pH, PaO2, PaCO2 and temperature among the studied groups; only heart rate for halothane and isoflurane was statistically lower after acute administration of intraperitoneal dexmedetomidine in both the naive and treated dexmedetomidine groups; however, this does not matter with reference to the conclusions because halothane and isoflurane would have the same limitation. Alpha2-adrenoceptor agonists have been described to produce bradycardia and a biphasic effect on blood pressure depending on the plasma concentration. At high concentrations the peripheral effect dominates whereas at low concentrations the central one does. 36
In summary, our study demonstrates that isoflurane but not halothane MAC-sparing response of acutely administered dexmedetomidine is enhanced in rats after chronic administration of the drug.
