Abstract
Urethane is often used as a sole anaesthetic agent for non-recovery studies in laboratory animals. However, the use of urethane is controversial, in part, because the electroencephalogram after urethane administration is similar to the electroencephalogram recorded from unanaesthetized animals. Here, we assessed the minimum alveolar concentration (MAC)-sparing effects of urethane by measuring the effect of two doses of urethane on the MAC of isoflurane in male Sprague Dawley rats. Isoflurane MAC was measured before and after intravenous administration of urethane at 1.0 g/kg (Group G1, n = 6) and 1.5 g/kg (Group G1.5, n = 6), or an equal volume of 0.9% saline (Group Gs, n = 6). Baseline isoflurane MAC was not statistically different between groups (isoflurane concentration: 1.47 ± 0.08%, 1.40 ± 0.19% and 1.42 ± 0.12% for G1, G1.5 and Gs, respectively). Intravenous injection of saline did not alter isoflurane MAC (post-saline MAC: 1.43 ± 0.11%). After urethane administration, isoflurane MAC decreased in a dose-dependent manner (new MAC G1: 0.19 ± 0.06%; G1.5: 0.03 ± 0.01%; P < 0.05). The isoflurane MAC after 1.5 g/kg urethane was not significantly different from room air isoflurane concentrations (0.01 ± 0.01%), demonstrating a 100% MAC reduction at this dose. In conclusion, high-dose urethane (1.5 g/kg intravenously) was suitable as a sole anaesthetic agent to prevent gross purposeful movement during the conditions of the study, whereas low-dose urethane (1.0 g/kg intravenously) was not.
Urethane is widely used as a sole agent to immobilize laboratory animals because a single dose (often between 1.0 and 1.5 g/kg) can provide immobility for several hours with minimal effects on cardiovascular and respiratory function. 1–3 However, the electroencephalogram after urethane administration continues to show spontaneous cyclic and evoked changes that are similar to the electroencephalogram recorded from unanaesthetized animals when they cycle between awake and slow wave sleep. 4–6 Consequently, the use of urethane as a sole anaesthetic for invasive surgical procedures is controversial. Given that we continue to see the use of urethane for non-recovery studies on laboratory animals, there is a need to investigate its anaesthetic properties.
The potential anaesthetic properties of a drug can be evaluated by comparing the test drug with a standard inhalation anaesthetic, such as isoflurane. These studies initially use an inhalation anaesthetic, at the minimum alveolar concentration (MAC) required to prevent gross purposeful movement in response to a standardized noxious stimulus. The test drug is then administered and the MAC remeasured. Many intravenous anaesthetic agents and adjuncts can partially (e.g. dexmedetomidine) 7 or completely (i.e. 100% MAC reduction, e.g. thiopental) 8 replace the amount of inhalation anaesthetic required to provide general anaesthesia in rats. Complete MAC reduction suggests that a drug may be useful as an anaesthetic agent when administered alone. To our knowledge, the effects of urethane on the MAC of any inhalation anaesthetic have not been determined in rats or any other species. The purpose of this study was to determine the effects of urethane on the MAC of isoflurane in adult male rats. We hypothesized that intravenous administration of urethane would dose-dependently decrease isoflurane MAC in adult male rats.
Materials and methods
Animals
The Institutional Animal Care and Use Committee at the University of Pennsylvania approved this study (protocol number 802174). Experiments were conducted on 18 adult (3–5 months) male Sprague Dawley (SD) rats (Harlan colony 217, Indianapolis, IN, USA) with conventional microbiological status. The animals were supplied by van in filter boxes and acclimatized for 10 days in the animal care facility of the institute in 48.3 cm × 26.6 cm × 20.3 cm cages (2–3 per cage depending on animal size) on a Bed-O'Cobs bedding (manufactured by The Andersons, Inc, Maumee, OH, USA). Room temperature and relative humidity were regulated at 22°C (±2°C) and 50% (±20%), respectively. Lighting consisted of artificial lighting between 07:00 and 19:00 h. The experiments were conducted over a period of 61 days during daytime using the methods described below. For all animals, anaesthesia was induced between 08:00 and 11:00 h. Animal diet consisted of ad libitum pellet (LabDiet 5010 Autoclavable Rodent Diet) and ad libitum chlorinated tap water. Animal groups included 1.0 g/kg urethane (G1, n = 6), 1.5 g/kg urethane (G1.5, n = 6) and a control saline group (Gs, n = 6). All animals were weighed on the morning of the experiment prior to anaesthesia (triple beam balance 700/800 series Part #730-00, Ohaus®, Pine Brook, NJ, USA).
Animal preparation
All rats were anaesthetized only once. General anaesthesia was induced with 5% isoflurane (IsoFlo®, Abbott Laboratories, North Chicago, IL, USA) in oxygen (1 L/min), via a precision out-of-circuit vaporizer (IsoFluromatic™, Foregger®, Smithtown, NY, USA) using a rodent induction chamber (Harvard Apparatus, Holliston, MA, USA). After induction of anaesthesia, isoflurane (2.5%) in oxygen (1 L/min) was delivered by a facemask. Rats were positioned in dorsal recumbency on a heating pad regulated by feedback from a rectal thermistor (TCA-2DFH, Physitemp Instrument Inc, Clifton, NJ, USA). Rectal temperature was maintained between 37.0 and 38.0°C. A 24-gauge catheter (19 mm, BD Insyte™, Becton Dickinson, Sandy, UT, USA) filled with heparinized 0.9% saline (2 IU/mL), was inserted in the left lateral tail vein near the base of the tail and was connected to a fluid pump (Compact Infusion Pump, Harvard Apparatus) to deliver 4 mL/kg/h of lactated Ringer's solution (Hospira Inc, Lake Forest, IL, USA).
The ventral side of the neck and the left inner thigh were clipped of hair and the skin disinfected. The mid-cervical trachea was exposed via surgical cut-down to facilitate tracheotomy and insertion of a tracheal cannula (PE-240 tubing, Harvard Apparatus). The trachea was sealed around the cannula using a silk ligature (no. 2, Deknatel, Research Triangle Park, NC, USA). The rat was continuously ventilated with isoflurane in oxygen via the tracheal cannula using a rodent ventilator (7025 Rodent Ventilator, Ugo Basile, Comerio, Italy). This ventilator is piston-pumped and time-cycled, and creates a non-rebreathing anaesthetic breathing system. Inspired isoflurane concentration was measured between the vaporizer and the ventilator using a side-stream infrared gas analyser (S/5™ Compact Anaesthesia Monitor, Datex-Ohmeda, Helsinki, Finland). Ventilator settings were chosen to maintain an end-tidal CO2 concentration 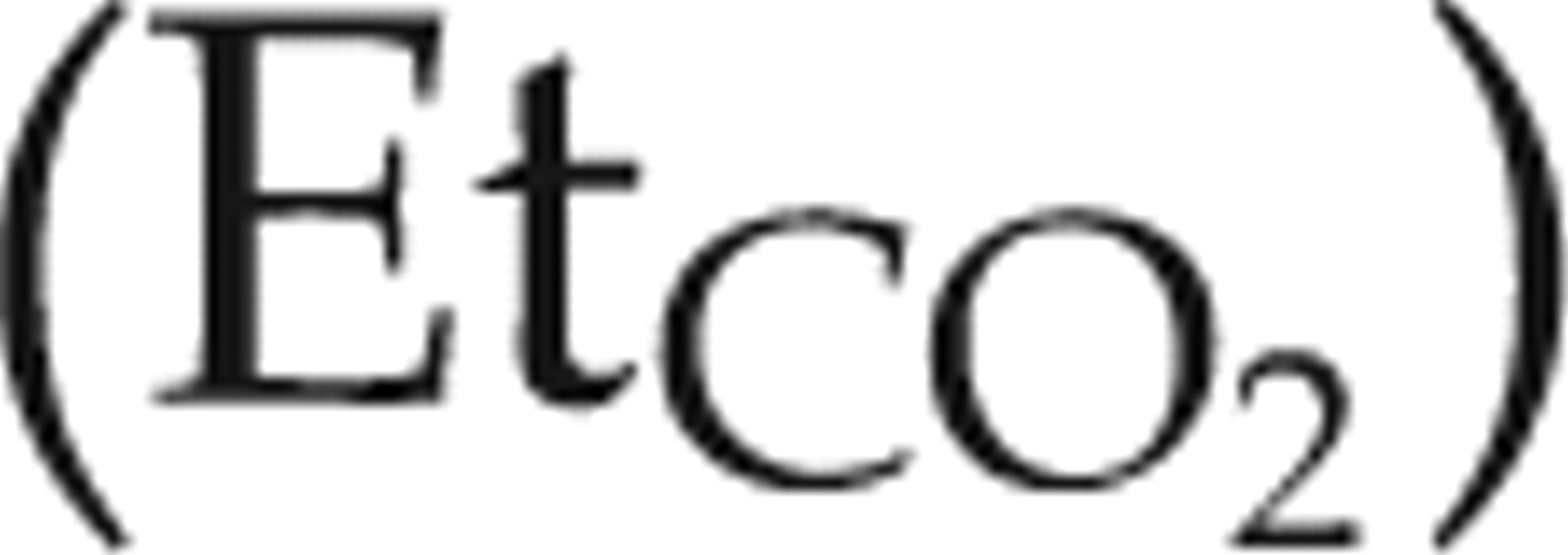
Baseline MAC determination
Following instrumentation, expired isoflurane concentration was set at 1.7% and the MAC of isoflurane was measured using a standard technique (Figure 1). 9,10 Expired gas samples were manually collected into a 20 mL glass syringe connected to a catheter running inside the tracheal cannula to permit measurement of the expired isoflurane concentration. The catheter tip was positioned at the level of the carina so that the collected gas sampled end-tidal gases. Approximately, 0.75–1 mL aliquots of expired gas were collected at the end of each expiration over 10–15 s (approximating 7–9 breaths). The collected gas was then expelled slowly into the sampling port of an infrared gas analyser (S/5™ Compact Anaesthesia Monitor, Datex-Ohmeda) to obtain an expired isoflurane concentration. Expired gas samples were collected in triplicate to determine a mean isoflurane concentration for any given time point. The anaesthetic agent monitor was calibrated with standard gases (DOT-39 NRC 300/375 M1134, Datex-Ohmeda) at the start of each study day. All the measurements (and calculated values) collected during the pretreatment period were considered baseline values. A noxious stimulus was applied to the tail using a haemostat (straight Rochester Pean Forceps 18.4 cm long, Lipshaw, Germany) clamped around the tail and moved to and fro with continuous movement for 60 s. The force applied to the tail was not measured. However, we standardized the clamp by only closing the haemostat to the first ratchet click and applying the first clamp at 1.5 cm above the tip of the tail (at a segmental level where the diameter of the tail begins to approximate uniformity) and spacing additional clamps at 0.5 cm increments cranial to the previous one. Any purposeful movement (such as a head lift, kicking of the legs and sustained contraction of the abdominal muscles leading to a change in body position) in response to the tail clamp was considered a positive response. A twitch, an increase in HR and/or an increase in BP alone were not considered positive responses. The inspired concentration of isoflurane was then increased (after a positive response) or decreased (after a negative response) by 20% and 15 min permitted to elapse before the tail was re-clamped. MAC was considered to be the midpoint between the lowest expired concentration of isoflurane that prevented gross purposeful movement and the highest expired concentration of isoflurane that permitted gross purposeful movement to the noxious stimulus, and was determined in triplicate.
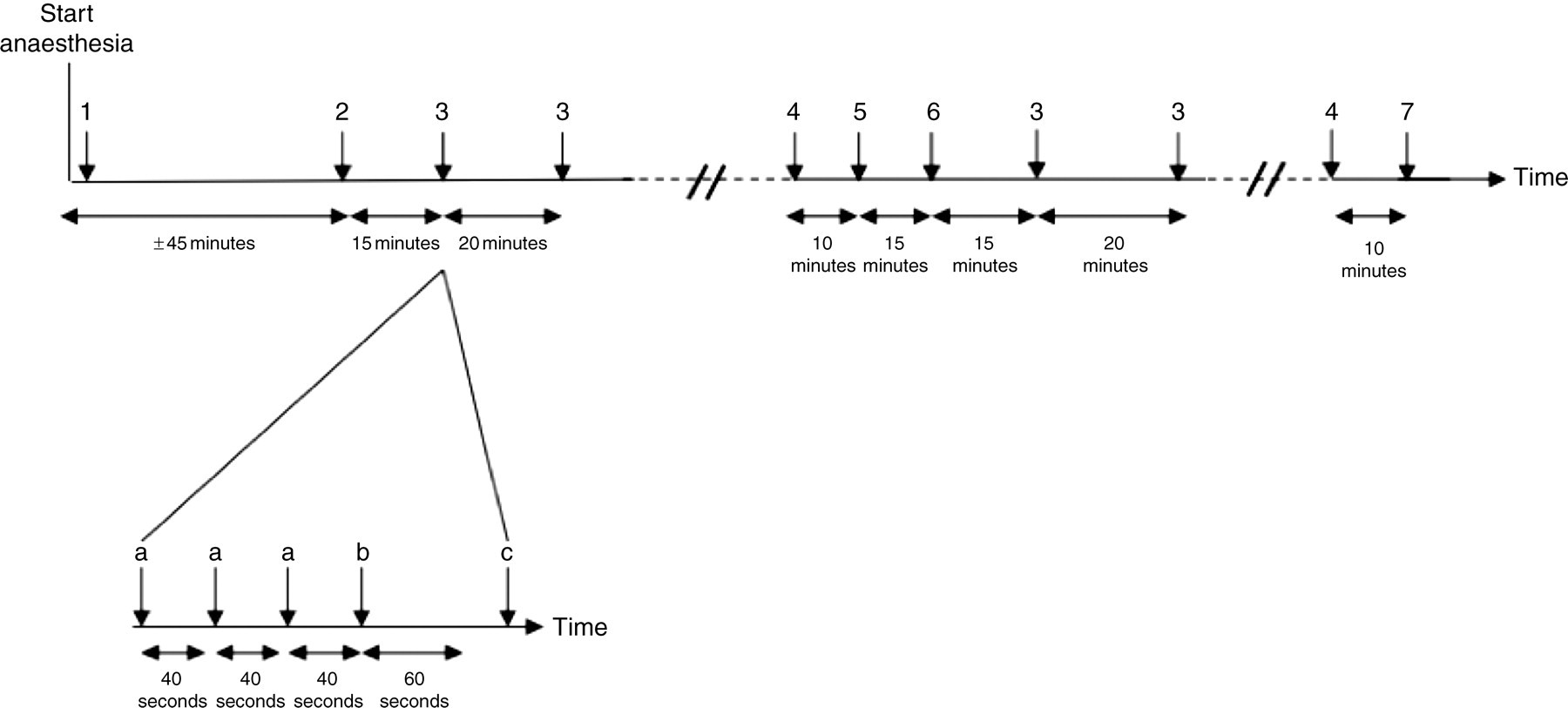
Graphic representation of the experimental methodology used to determine the reduction of isoflurane minimum alveolar concentration (MAC) after urethane administration in adult male Sprague Dawley rats. 1: animal instrumentation; 2: end of surgical preparation (inspired isoflurane to 1.7%); 3: MAC measurements in triplicate (a: gas samples; b: tail clamp; c: change in inspired isoflurane concentration); 4: blood gas analysis; 5: urethane or saline injection; 6: change in inspired isoflurane concentration (urethane groups: inspired isoflurane to 80% of previous MAC; control group: inspired isoflurane to 1.7%); 7: blood and brainstem sampling
Blood gas analysis
Five minutes after baseline MAC was measured, arterial blood samples (0.5 mL) were collected via the arterial cannula in a heparinized syringe and pH, PCO2 and PO2 were measured immediately using a blood gas analyser (iSTAT, Heska Corp, Loveland, CO, USA). Blood gases were corrected for rectal body temperature. After blood withdrawal, the arterial cannula was flushed with 0.5 mL heparinized saline.
Post-treatment MAC determination
After the arterial blood sampling, the rats received one of two randomly and blindly administered doses of urethane (Part# U2500-500G, Sigma-Aldrich, St Louis, MO, USA): 1.0 and 1.5 g/kg, or a non-blinded equal volume of 0.9% normal saline (4.0 mL). The urethane doses were selected based on the standard range of doses found in the literature. 1,11–16 All treatments were administered intravenously over 10 min via the tail vein catheter. Urethane was dissolved in 0.9% saline so that the final volume was 4.0 mL. The investigators were not blinded to the administration of saline in the control group because our methodology required an immediate reduction in inspired isoflurane concentration of 80% below baseline (based on preliminary studies using urethane-treated rats). A similar reduction in isoflurane concentration in the saline-treated rats would likely have caused awareness, which would have been unethical. Appropriate precautions for minimizing exposure to carcinogenic agents were used to handle and prepare the urethane solution. 17
After an additional 15 min had elapsed, MAC was remeasured. In the urethane groups, the noxious stimulus was applied and, if the response was negative, inspired isoflurane concentration was decreased by approximately 0.1% every 15 min until a positive response was obtained. Thereafter, the noxious stimulus was reapplied every 15 min until a total of 2 h had elapsed after the urethane injection. If a response was positive, inspired isoflurane concentration was increased by approximately 0.1% until a negative response was obtained. Five minutes after the second MAC had been determined, arterial blood samples were collected and analysed for blood pH and gases as described above.
BP and HR
Mean arterial BP and HR were measured over a 30 s period preceding each measurement of mean end-tidal isoflurane concentration. Baseline and post-treatment BP were considered to be the midpoint between the mean BP at the lowest expired concentration of isoflurane that prevented gross purposeful movement and the highest expired concentration of isoflurane that permitted gross purposeful movement to the noxious stimulus, and were determined in triplicate before and after the urethane or saline injection, respectively.
Blood and brain urethane concentration measurement
Serum and brainstem urethane concentrations were next measured at the end of each experiment to investigate the relationship between urethane concentration and the effects of urethane on isoflurane MAC. Several studies have indicated that inhalants produce immobility via central mechanisms below the level of the cerebral hemispheres, 18–20 possibly at the level of the spinal cord. 21 Thus, we did not sample cerebral cortices to determine central urethane concentrations in our study. Given the need to remove and freeze neural tissue rapidly for accurate drug analysis, and the easier and faster dissection into the cranium compared with the vertebral canal, we chose to sample the caudal brainstem and not the spinal cord to represent subcortical urethane concentrations.
At the end of the experiment, inspired isoflurane concentration was increased to 3%. Ten minutes later, 5 mL of arterial blood was collected from the arterial catheter, and the brainstem was immediately removed via a craniotomy. Serum and brainstem were immediately frozen in liquid nitrogen and stored at −80°C. Serum and brainstem urethane concentrations were measured by the Pennsylvania Animal Diagnostic Laboratory System Toxicology Laboratory at the University of Pennsylvania, using an API 4000 LC/MS/MS (liquid chromatography [LC] purification, and tandem mass spectroscopy [MS/MS]) with 100 ppm urethane-ethyl-
Data analyses
The presence of normality and equal variance was tested before parametric analyses were performed. The effect of administering urethane on isoflurane MAC, cardiovascular, respiratory, and arterial pH and blood gas measurements were tested using a repeated measure ANOVA (JMP Statistical Software, SAS, Cary, NC, USA). Factors included dose of urethane and time post-injection (repeated measures). When significant differences were detected by ANOVA, they were confirmed using the Bonferroni test. Differences were considered significant when P < 0.05. All measurements are mean ± standard deviation (SD).
Results
Mean body weight (Gs: 384 ± 20 g; G1: 364 ± 15 g; G1.5: 350 ± 15 g) and age (Gs: 85 ± 17 days; G1: 81 ± 5 days; and G1.5 80 ± 6 days) were not significantly different between the three groups. Baseline arterial acid–base status and blood gas concentrations, and mean arterial BP and pulse rate, were also similar between the groups (Table 1). Baseline MAC values were not different between groups (Gs: 1.43 ± 0.12 g; G1: 1.47 ± 0.08 g; G1.5: 1.40 ± 0.18 g; Figure 2) and were consistent with isoflurane MAC values reported previously for rats.
22
After urethane administration, MAC in G1 and in G1.5 significantly decreased to 0.19 ± 0.06% (87 ± 3% reduction) and 0.03 ± 0.01% (98 ± 1% reduction), respectively (Figure 2; P < 0.05). The post-treatment MAC in the G1.5 group was not statistically different from isoflurane concentrations in room air gas samples (0.01 ± 0.01%). In each of the three treatment groups, the post-treatment pH was significantly lower than the baseline pH (Table 1; P < 0.05). After urethane administration and establishing the new MAC value, lactate concentration decreased (P < 0.05) and mean BP increased (P < 0.05) compared with the pretreatment value (Table 1). Saline administration had no effect on these variables (Table 1). The post-treatment mean BP and lactate concentration in the urethane groups were significantly different from those in the saline group.
Minimum end-tidal alveolar isoflurane concentration (means ± SD) required to prevent gross-purposeful movement during baseline conditions (Pre) and after intravenous administration (Post) of saline (Gs), urethane 1 g/kg (G1) or urethane 1.5 g/kg (G1.5). Urethane dose-dependently decreased isoflurane MAC. *Significantly different from the baseline value within that group; †significantly different from the post-drug measurement across groups Arterial blood gas, pH and cardiovascular measurements in isoflurane anaesthetized adult male Sprague Dawley rats during baseline conditions and after administration (post-treatment) of saline (Gs), urethane 1 g/kg (G1) or urethane 1.5 g/kg (G1.5) BP: blood pressure; Pa: arterial partial pressure All values are mean ± SD *Significantly different from the baseline value; †significantly different from the saline group at this time point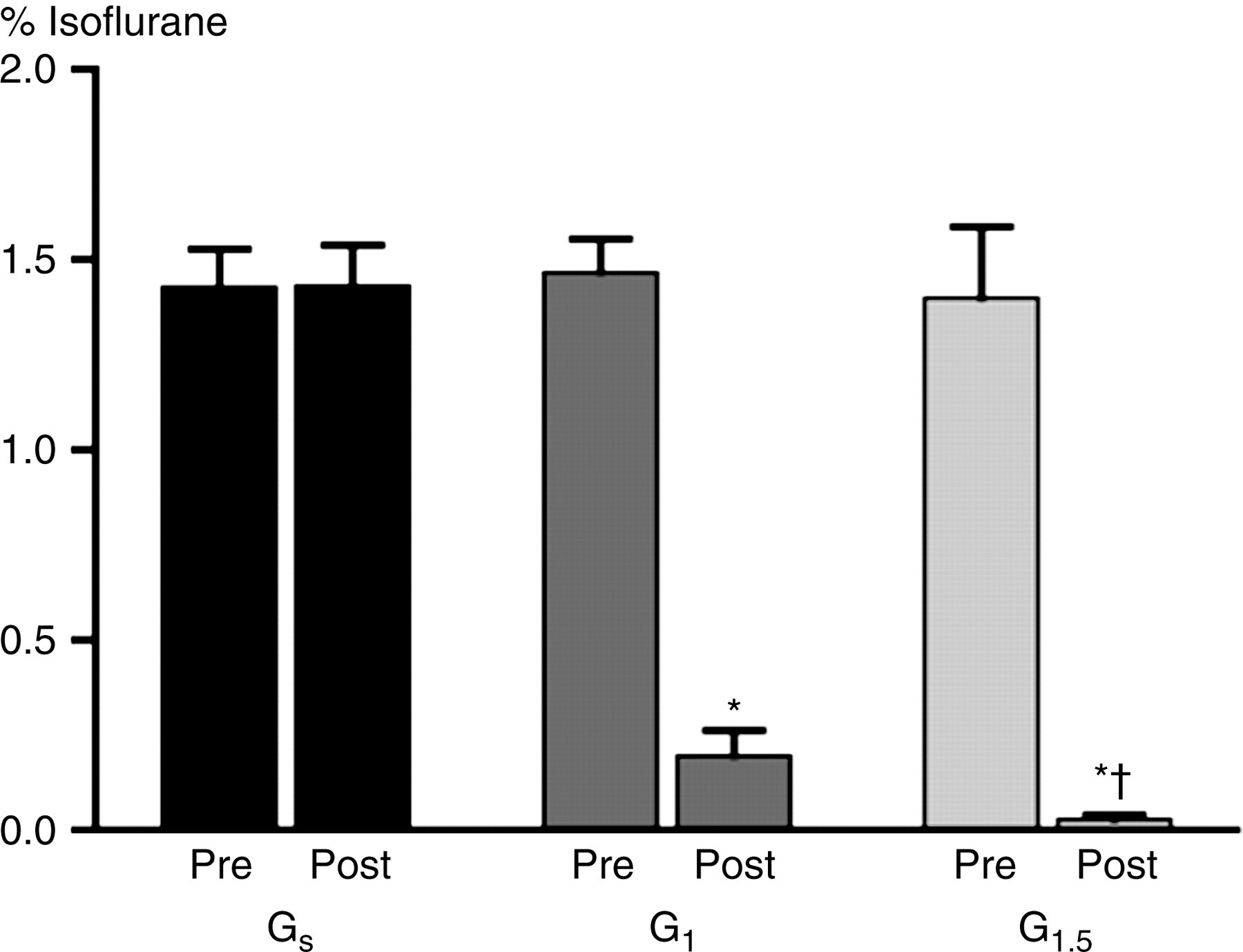
As expected, the serum and brainstem urethane concentrations were higher in rats that received 1.5 g/kg urethane compared with those that received the lower dose (Figure 3; P < 0.05). When both treatment groups were analysed together (i.e. n = 12), urethane concentration in either tissue was correlated to the magnitude of MAC reduction (Figure 3; serum: R
2
= 0.66; brainstem: R
2
= 0.56; P < 0.05). However, within a treatment group (n = 6), there was no relationship between the concentration of urethane and MAC reduction.
The effect of urethane dose on serum (squares) and brainstem (circle) urethane concentration at 2 h post-injection and the relationship between urethane concentration and the magnitude of MAC reduction. All values are means ± SD. Urethane tissue concentration increased as the dose of urethane increased. MAC reduction was correlated to serum and brainstem urethane concentrations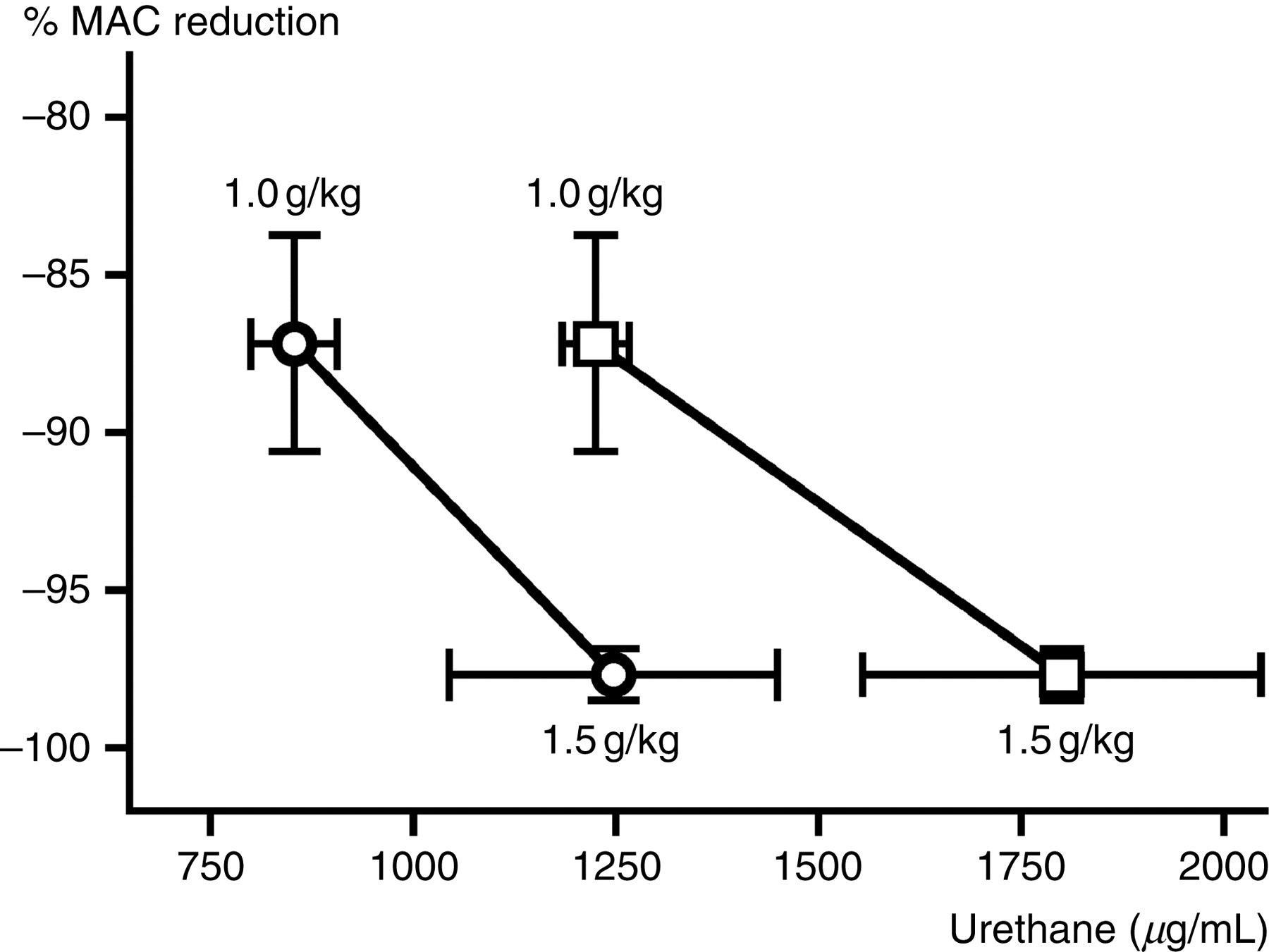
Discussion
We demonstrated that urethane dose-dependently decreases isoflurane requirements to prevent gross purposeful movement under the conditions of our study. The highest dose of urethane was sufficient to provide 100% MAC reduction (when compared with room air samples), which suggests that urethane may be sufficient as a sole anaesthetic agent in laboratory rats. However, given that urethane is mutagenic and carcinogenic, and represents a hazard for both the patient and the anaesthetist, 17 the use of urethane on a routine basis cannot be recommended. With appropriate environmental health and safety measures in place, our study does support the use of urethane for non-recovery surgical procedures where minimal cardiovascular and respiratory depression are necessary conditions of the experimental investigation.
Our results are consistent with a previous study in rats, where the authors report that anaesthetic conditions suitable for moderate painful procedures were obtained for at least 2 h after 1.5 g/kg of urethane. 16 Field et al. 16 administered urethane intraperitoneally at 1.2 and 1.5 g/kg, which was similar to our dose range. However, we chose the intravenous route to ensure consistent uptake of urethane from the site of injection. The authors in the previous study reported that urethane abolished motor responses to toe, tail and abdominal pinch using an alligator clip for at least 2 h after urethane administration, and that this effect was seen at both doses of urethane. In contrast, our lowest dose of urethane (1.0 g/kg) did not prevent gross purposeful movement in response to tail pinch. Thus, minimal dose necessary to completely replace isoflurane as an anaesthetic for surgical procedures in rats may be between 1.0 and 1.2 g/kg.
Using loss of gross purposeful movement to assess MAC reduction can be problematic. Gross purposeful movement has sensory, perceptive and motor components. A drug acting on the motor component (i.e. neuromuscular blockers such as pancuronium) may prevent the motor response to a noxious stimulus but permit pain perception. In this scenario, the calculated ‘MAC reduction’ would be 100%, although the animal would also remain conscious. In our study, rats at MAC had evidence of muscle activity (occasional limb twitches, whisker movement and respiratory efforts against the ventilator). Thus, we do not believe that urethane provided complete MAC reduction by only blocking the motor component to gross purposeful movement.
All rats were anaesthetized only once because a crossover design was not appropriate given the fact that recovery from urethane anaesthesia is prolonged, variable, and often associated with excitement and thrashing (personal observations). Urethane can also be mutagenic, carcinogenic and hepatotoxic resulting in a high incidence of lung tumours. 17
In the present study, arterial pH decreased after drug administration (in saline-alone and urethane treatment groups) is consistent with the effects of urethane in a previous study. 16 Changes in arterial pH may reflect changes in respiratory (i.e. CO2) and/or non-respiratory (i.e. lactic acid) acid–base balance. In our study, PaCO2 did not change after treatment and PCO2 decreased after urethane administration in the previous study. 16 Collectively, these data suggest that accumulation of a non-respiratory acid was responsible for the change in pH after drug administration. In our study, lactate concentration increased in the saline-treated group, which could explain the change in arterial pH over time in this group. On the other hand, lactate concentration decreased after urethane administration. This difference between treatment groups may reflect better tissue perfusion (i.e. subsequent to increased BP) after the new MAC was established compared with the baseline MAC value. Although the cause for the decrease in arterial pH in urethane-treated rats remains unknown, the value remained within the widely accepted normal range (pH 7.35–7.45).
The serum urethane concentration necessary for ‘narcosis’ in rats has been reported to be between 600 and 800 μg/mL. 23 In their study, narcosis was defined as lateral recumbency (without noxious stimulation). The authors also demonstrated that urethane concentration was independent of the tissues analysed (blood, brain, liver or spleen). Boyland and Rhoden 23 also reported that urethane concentration decreased by only 10–20% between 2 and 5 h after urethane administration. Overall, the values for our urethane measurements were consistent with this report; however, we found that the concentration of urethane was consistently higher in serum compared with brainstem. In our study, urethane serum concentrations between 1610 and 2280 μg/mL were sufficient to provide surgical anaesthesia.
Footnotes
Acknowledgements
We extend our appreciation to Sharon Martinez for her technical assistance with the experiments and to Dr Daljit Vudathala who measured the serum and brainstem urethane concentrations. This work was supported by a grant provided by the Department of Clinical Studies-Philadelphia, School of Veterinary Medicine, University of Pennsylvania, Philadelphia, PA, USA.
