Abstract
Certain special biological characteristics of Japanese quails (Coturnix coturnix japonica) could enable quicker and less expensive laboratory experiments than similar studies using mice or chickens. Moreover, due to some physiological similarities with humans, quails have been recognized as an ideal laboratory model for experiments in some fields such as immunology, endocrinology, and reproductive biology. The aim of this study was to acquire normal ultrasonographic images of reproductive organs of female Japanese quails in order to facilitate laboratory experiments on reproductive biology and to determine a non-invasive in-life alternative to carcass analysis. Thirty female Japanese quails of different ages were used for ultrasound scanning of ovaries, follicles, ova, oviducts and eggs. In addition, five quails were euthanized for experiments using a direct standoff imaging technique. It was possible to identify different sizes of follicles ranging from small white follicles (0.14 cm) to large yellow follicles (2.57 cm), post-ovulatory follicles (POFs), ova at various stages of development, oviducts, and also eggs within the oviducts. These findings demonstrate that ultrasound scanning of female Japanese quail reproductive organs might be a beneficial diagnostic tool for applications such as studying reproductive physiology, anticipating the beginning or the end of the breeding season, detecting any pathological disorders easily, and identifying non-productive females which could lead to more cost-efficient laboratory procedures.
Since the description of Japanese quail as a research model in 1959 by Padgett and Ivey, this truly diverse and efficient animal model has been used in laboratory studies. 1 The biological characteristics of quails can enable faster turnaround and more cost-efficient experiments than the use of other species in similar types of studies. Quails can be maintained at lower cost in terms of husbandry resources and laboratory space. Furthermore their shorter generation time, due to their short maturation period of eight weeks, their massive egg production, and their quick embryonic development of 16 days, means that experiments can have a quicker turnaround time. 2
Due to these unique characteristics and certain physiological similarities with humans, quails have been recognized as an ideal laboratory model for experiments in some fields such as immunology, endocrinology, and reproductive biology.3,4 The present study was focused on the similarities of the quail’s reproductive biology to that of humans.
The reasons for considering quails as an ideal model for this important field of study are that they are a well-known model for studying the hypothalamic and limbic circuits which control the reproductive axis,5,6 and have been recognized as an example for studying photoperiodism and the circadian control of brain function, levels of circulating sex hormones and reproductive activity.7–9 Another important reason is that the female lays one egg per day, so it could provide an ideal model of the follicular function mechanism and the rupture and repair mechanisms. 10
To better understand and more efficiently study the quail’s reproductive biology, we have to know the structure and positioning of this organ within its body. In the juvenile, the ovary lies in cranial parts of the body cavity which have a triangular shape, and is brownish red in color. The organ in young quails weighs 0.06–0.09 g.
11
Increment of the ovary happens after maturation, and it weighs approximately 10–12 g in Japanese quails.
12
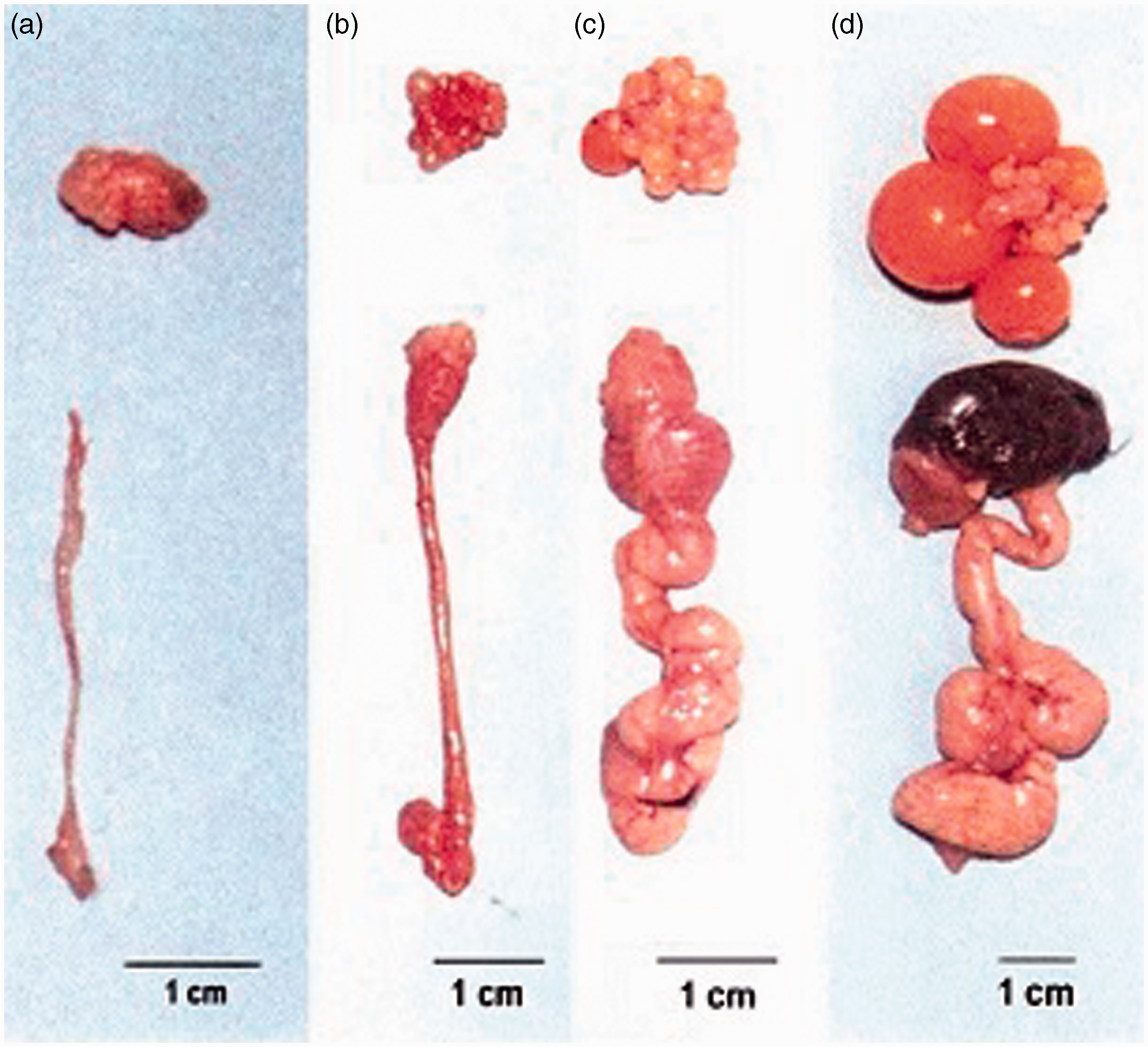
This increment occurs because of the development of large yellow yolky follicles (Figures 1 and 2).
Development of quail ovary from completely inactive reproductive system (a) to a fully developed ovary and oviduct (d).
12
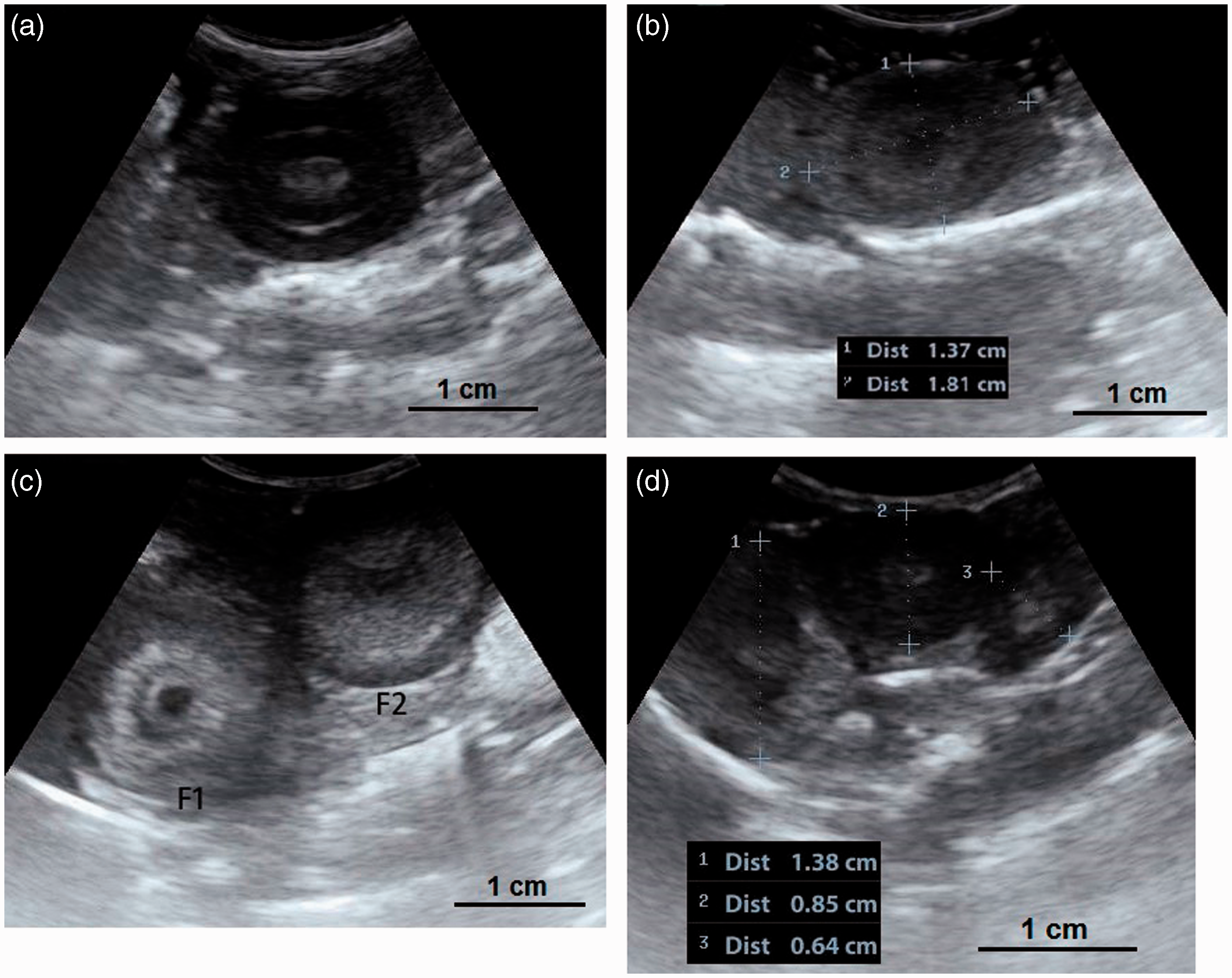
In laying quails, the ovary consists of a hierarchy of large yellow follicles, identified by their sizes as F1, F2, F3, F4, and F5. There are also several follicles which are classified based on their diameters: 4–8 mm (small yellow follicles), 2–4 mm (large white follicles), <1 mm (small white follicles).
12
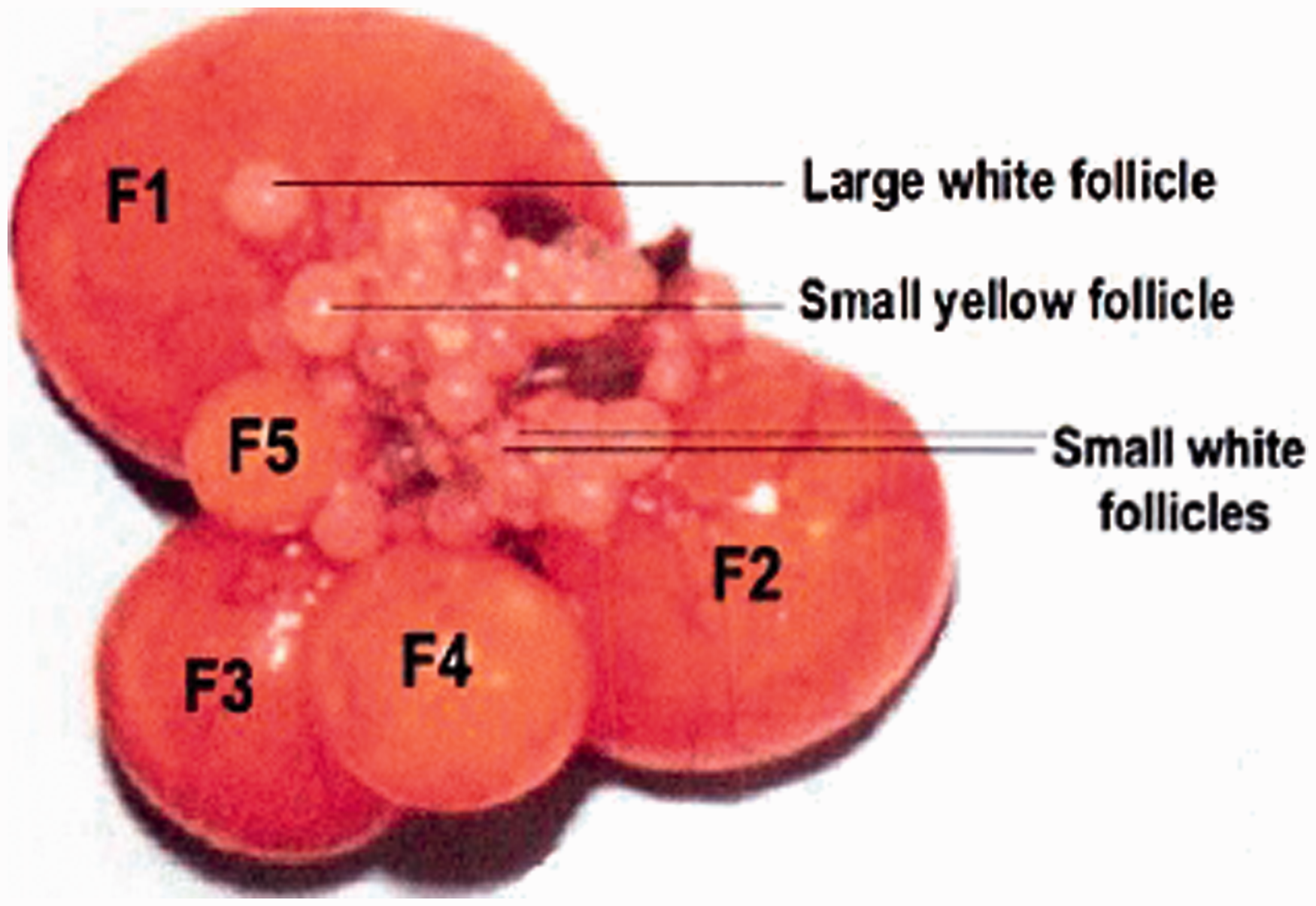
At the point of ovulation, the largest follicle in the hierarchy ruptures through an avascular area of follicular wall named ‘stigma’. After this, the ruptured follicle or post-ovulatory follicle (POF) is easy to identify. Also some follicles are atretic follicles which do not seem to reach the pre-ovulatory stage of development and which regress before the occurrence of ovulation. After ovulation the follicles pass through the oviduct which runs caudally along the ventral side of the kidney and consists of several features including the infundibulum, magnum, isthmus, uterus, and vagina.
Although quails are a perfect model for reproductive biology studies, for numerous reasons, to date no researchers have used non-destructive methods to monitor the reproductive organs of female Japanese quails. Invasive methods like carcass analysis have been used for many years to obtain regular data of reproductive organs in birds.13–15 This method has pros and cons, but the most obvious disadvantage would be limitations in investigating changes in reproductive organ sizes within individuals. Bioethical issues are other serious problems with this method; moreover, there may be a need for sample sizes and time points at which measurements are carried out to be multiplied. Therefore it would be a better idea to use non-invasive methods which can facilitate the study of physiological changes or rheostasis.16,17
Several non-invasive methods have been used to monitor organ sizes in live birds, such as X-ray techniques 18 and magnetic resonance imaging (MRI), 19 which have some advantages and disadvantages. X-ray techniques capture highly detailed radiographs of skeletal systems and are suitable for expert interpretation and detection of pathological changes, but have obvious disadvantages in mapping soft tissue. 20 Although MRI is a good technique for mapping soft tissue contrast in which there are no artifacts such as bones, and is also a safe procedure for technicians; MRI is an extremely expensive technique requiring specially trained personnel and long investigation times. 19 Meanwhile an important disadvantage for ecologically and biologically oriented studies is that the use of X-ray and MRI measurements is confined to laboratory settings. Another choice is ultrasonographic scanning21,22 which has been shown to have few disadvantages such as limited resolution, and more benefits for estimating the size or mass of particular organs. 23 Modern ultrasonographic instruments are portable and can be used easily in laboratories and farms for repeated measurements in the same individual, and are relatively safe and inexpensive. 23 Consequently, the aim of this study was to generate normal ultrasonographic images of different reproductive organs of female Japanese quails in order to facilitate studies using them as a model for reproductive biology in humans.
Animals, materials and methods
The experimental birds were kept, maintained and treated according to the accepted standards for the humane treatment of animals approved by the Institutional Animal Care and Use Committee of Shahrekord University.
The scans were performed on 30 female Japanese quails of various reproductive statuses, aged 4–7 weeks. These birds were collected from three farms around Shahrekord University. To increase our knowledge about the structures that were examined transcutaneously, five birds of apparently different reproductive statuses were marked individually in order to establish easy follow up procedures after euthanasia of the birds.
The examination was performed with a Mindray Z6Vet ultrasonic device (Mindray, Nanshan, Shenzhen, China), which was equipped with a switchable 6–9 MHz microconvex probe. The unit ran on rechargeable batteries, and was relatively small and portable. In order to allow an easy scanning procedure, the birds were first restrained and the abdominal feathers below the keel were easily removed using a slight layer of hair removal spray, and the skin was cleaned with wet cotton. In order to help with handling the bird, an assistant held each quail during the examination procedure, which lasted approximately 10 min. For reducing the influence of food, the birds were starved for about 2 h prior to the procedure.
To maximize the contact between the transducer and the skin, coupling gel was used. The following protocol was performed using transcutaneous ultrasound scanning. Firstly the transducer was placed on the left side of the abdomen approximately in the middle region and several impressions were obtained by making a quick scan through the whole examination area, from dorsal to lateral and from cranial to caudal positions, and vice versa. After scanning the area, a much slower scan was performed within the area to obtain more detailed orientation and information. For quantitative measurements the numbers and sizes of follicles were measured and the sizes of the visible follicles were measured by using freeze frames. Moreover all the ultrasound examinations were recorded on a videotape to facilitate the quantitative measurements.
Macroscopic inspection of ovaries and oviducts was performed with respect to the numbers, sizes and types of follicles. Moreover, capture photographs of some ovaries were taken to record the morphological structures, also an in vitro standoff technique was performed to augment the information about some ovarian structures.
Results
Most of the ultrasound images presented in this section (Figures 3–10) were obtained during in vivo scanning from live quails, with the exception of those shown in Figures 8a and 10a, which were obtained from the euthanized birds in order to determine the normal appearance of POFs and uteri by direct imaging. It should be considered that the influence of air sacs was not observable in the ultrasound images, even in juvenile quails.
Primary stage of a developing ovary. At this stage the follicles are very small and may be indistinguishable as round complete small follicles in the ovary or in ultrasound images (arrows: borders of the ovary; frequency 9.0 MHz; depth 2.8 cm).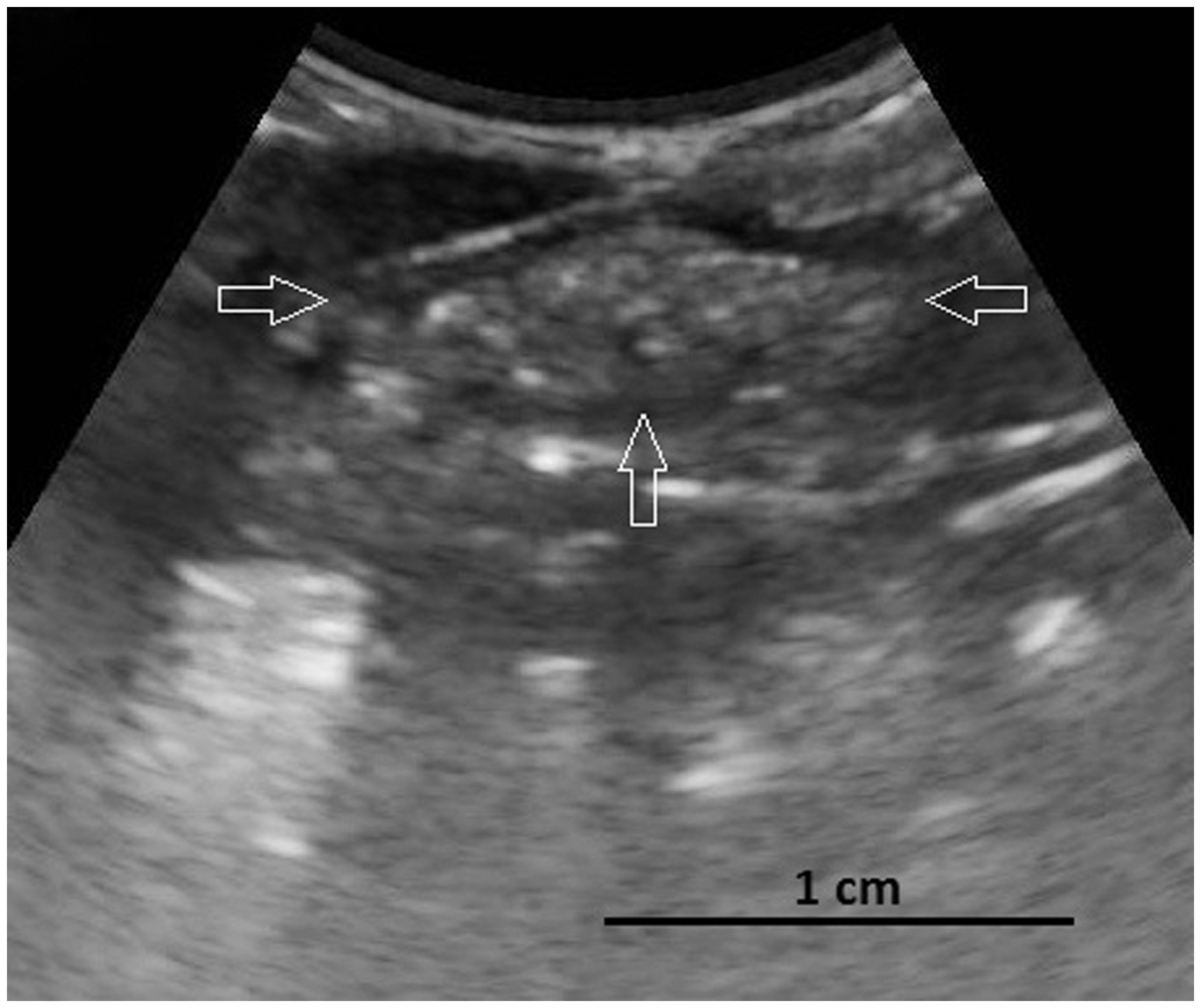
The ovarian follicles visualized varied in stage, number, and size (based on the largest obtained diameter in centimeters). Large follicles, small follicles, POFs, and developing ova were among the ovarian follicles imaged. The diameter of the visible follicles ranged from 0.14 cm in the case of small follicles, to 2.57 cm in the case of developing ova. Developing ova had the biggest diameters while small follicles had the smallest.
Discussion
For more than 50 years Japanese quails have been recognized for some of their unique characteristics such as short lifespans, short maturation times, and short embryonic development. 2 These beneficial characteristics, in addition to some physiological similarities with humans, have resulted in this bird being considered as an ideal animal model for studies in certain fields of medicine such as immunology, endocrinology, and reproductive biology.3,4
Studies have focused on the similarities of a quail’s reproductive biology with humans. Female quails lay one egg per day which makes them ideal for studies of women’s follicular mechanism or malfunction. 10 The female Japanese quail is also well recognized as a model for studying hypothalamic and limbic circuit photoperiodism5,6 and circadian control of the brain function, which could be a good representation of the levels of circulating sex hormones and reproductive activity.7–9
Several methods have been used to evaluate the reproductive function of birds.18,21,22,24 The most commonly used method in laboratories is carcass analysis which is not a preferred choice due to bioethical issues, limitations in investigating changes in the size of organs in each individual, and the need for sample sizes and time points at which measurements are carried out to be multiplied. 25 Hence non-invasive methods would be better options for evaluating these structures. Among these non-invasive methods, such as X-ray, MRI, and ultrasonography, the last one might be the best choice because it is portable, relatively safe and inexpensive. 23
The results of the present study show that transcutaneous ultrasonography, an easily performed non-invasive technique, is a very feasible method for imaging the reproductive organs of a female Japanese quail. For better performance, it is advisable that an assistant should prepare the abdominal region about 10 min before the examination using hair removal spray, and the bird should be handled carefully to avoid distressing the quail.
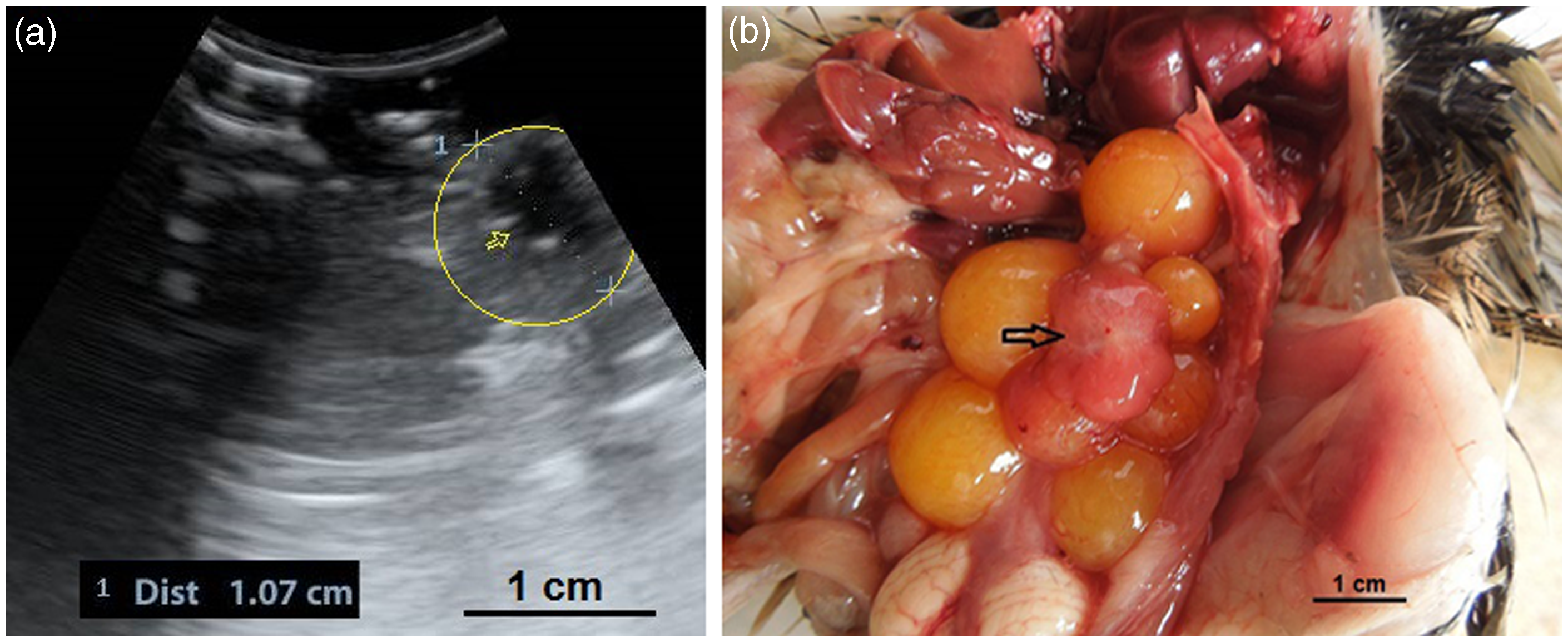
In the present study it was feasible to count the number of follicles ultrasonographically which should help the researchers to either increasing or decreasing follicular activity, or anticipate the start or the end of egg production season. Also it would allow the identification of non-productive birds. It was possible to detect various structures of ovaries from diffuse hyperechoic ovarian stroma which consist of numerous small follicles to different sizes of follicles and POFs (Figures 2–8). Figures 3 and 4a show the juvenile ovary in the near field of transducer without any influence from air sacs. It is suggested that the small body mass of the Japanese quail and the minimal space among air sacs and ovary might confound these findings. As described in other birds,26,27 four types of follicles could be observed in the hierarchy of follicles: small white follicles, large white follicles, small yellow follicles, and large yellow follicles which are designated by their position and identified as F1, F2, F3, up to Fn follicles. Meanwhile, small follicles are positioned at the lowest level among the follicles in the hierarchy of follicles. Some follicles were observed as annular structures of larger than one centimeter, which consist of a hyperechoic core in the center and were encircled by alternating hypo- and hyperechoic rings (Figure 7). These ultrasonographically unique ring structures have been reported by some other researchers who have observed the rings in the follicles of chickens (Gallus gallus) and ostriches.26–28 It was suggested that these rings represent alternating layers of yellow yolk of various composition which are deposited first when maturation of follicles begins and which contain less water and protein. So it could be concluded that, as in other species such as chickens and ostriches, the inner annular appearance of the ovarian follicles is a reflection of the hyperechoic core of a white yolk which is encircled by vitellus aureus (dark stratum of the yolk) and vitellus albus (light stratum of the yolk) and corresponds to the latebra. Moreover, in some cases the anechoic or hypoechoic walls are not smooth, probably due to interference from adjacent structures. After rupturing of the largest follicle, the remaining structure turned into a POF which has been described as a sac-like follicle. Due to a lack of information about the normal appearance of POFs in quails, finding POFs in live birds by ultrasonography was difficult, so the normal sonographic appearance of POF was recorded using a standoff technique. With this technique it was readily identifiable as an irregular round to oval structure with a length of 1.07 cm which consisted of a central anechoic cavity in quails (Figure 8).
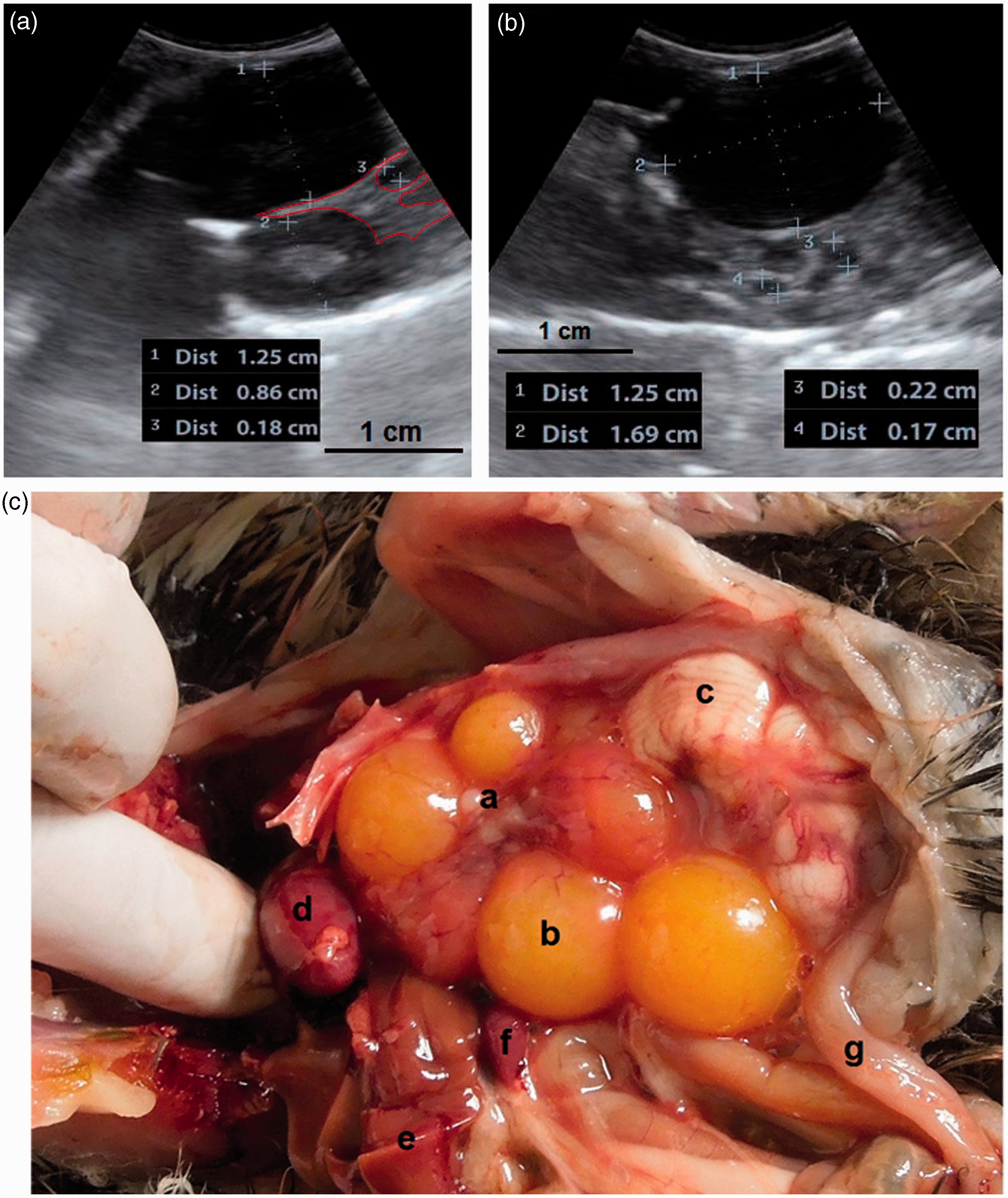
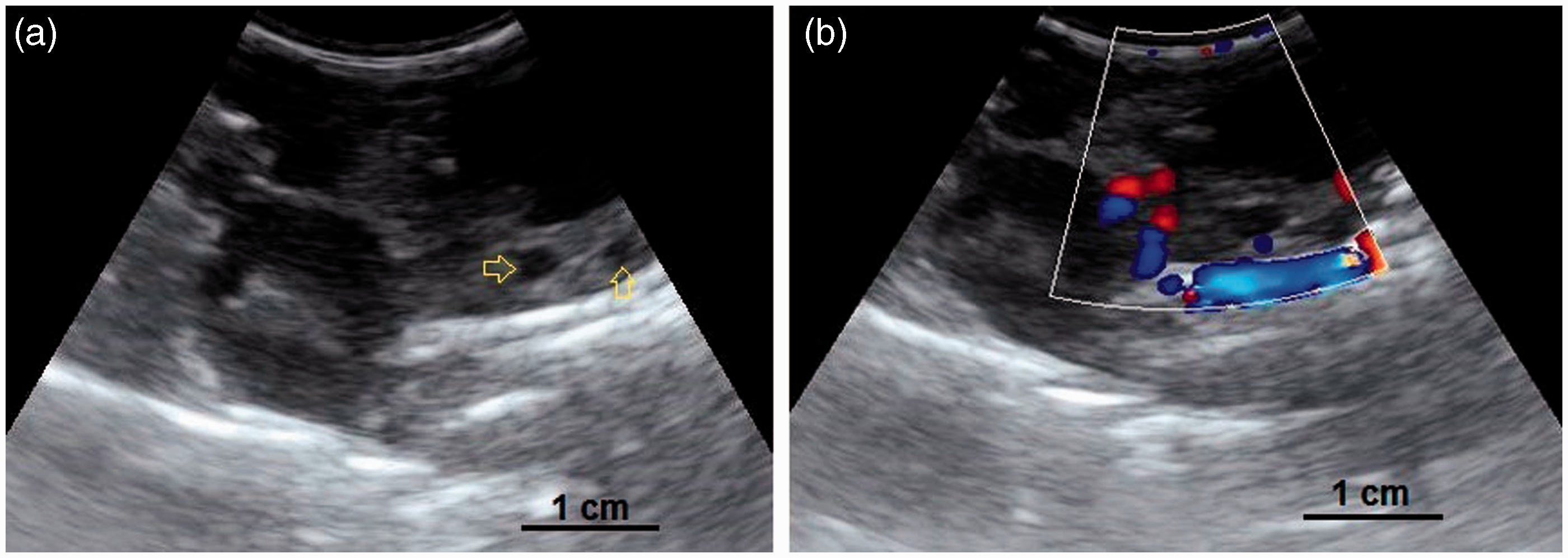
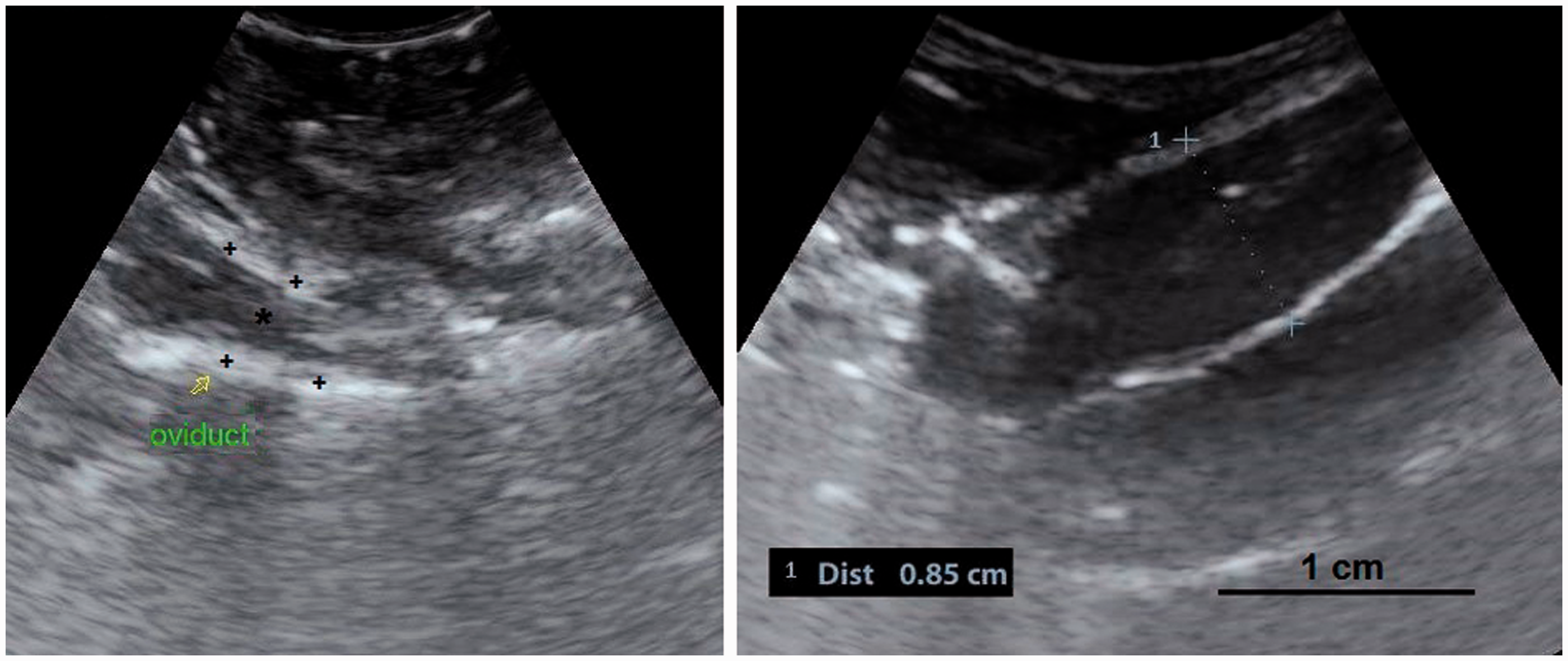
(a, b) A developing ovary in a quail which has recently started the maturation process. This is a later stage than the maturation stage of the ovary in Figure 3 and the follicles are larger. Small white, large white and small yellow follicles with different sizes could be distinguished within the ovary (4a) and have made hypo- and hyperechoic irregular appearances in the ultrasound image (4b). Within the carcass, lung (a); ovary (b); liver (c); proventriculus (d); spleen (e); abdominal air sac (f); kidney (g); oviduct (h) and intestine (i) are shown (arrows: borders of the ovary). (4b: frequency 6.5 MHz; depth 2.8 cm). Overall view with follicles of different sizes. (5a) Two relatively large follicles in a hyperechoic stroma. The red line is the border of the ovarian stroma. (5b) A large follicle and several small ones. (5c) A mature genital tract in a Japanese quail which consists of several follicles of different sizes and a developed oviduct. Within the carcass, white follicles (a); large yellow follicles (b); oviduct (c); heart (d); liver (e); spleen (f) and intestine (g) are shown. (5a: frequency 6.5 MHz; depth 4.6 cm); (5b: frequency 7.5 MHz; depth 4.6 cm). Two-dimensional ultrasonography (a) and color flow imaging (b). The color flow imaging was performed in order to differentiate the small follicles and blood vessels (arrows: follicles). (6a, b: frequency 6.5 MHz; depth 4.6 cm). (a, b) Detailed larger follicles with a hyperechoic central structure and the following hypo- and hyperechoic rings. (c, d) Hierarchy of large follicles in which the F1 follicle is the largest follicle in an ovary. (7a: frequency 6.5 MHz; depth 4.6 cm); (7b: frequency 9.0 MHz; depth 4.6 cm); (7c: frequency 9.0 MHz; depth 4.6 cm); (7d: frequency 9.0 MHz; depth 3.7 cm). (a) Post-ovulatory follicle (POF) appeared as an irregular round to oval structure with a length of 1.07 cm and a central anechoic cavity (arrow) from examining the carcass. (b) The appearance of POF among other mature follicles in a Japanese quail. (8a: frequency 6.5 MHz; depth 4.6 cm). (a) An ovum with inner structures and rings. Outside an annular anechoic structure corresponds to the albumen and in the center of the yolk is the latebra. (b) An egg with a developing shell. The calciferous material causes a more intense echogenicity of the periphery. Within the egg, central yolk (1) and surrounding albumen (2) are shown. (9a: frequency 9.0 MHz; depth 3.7 cm); (9b: frequency 7.5 MHz; depth 4.6 cm). (a) Oviduct appeared in the carcass as a structure with parallel hyperechoic walls and hypoechoic internal lumen inside the walls. (b) Ultrasonographic image of the oviduct in live Japanese quails. (+) Hyperechoic wall, (*) Hypoechoic lumen. (10a: frequency 9.0 MHz; depth 2.8 cm); (10b: frequency 6.5 MHz; depth 4.6 cm).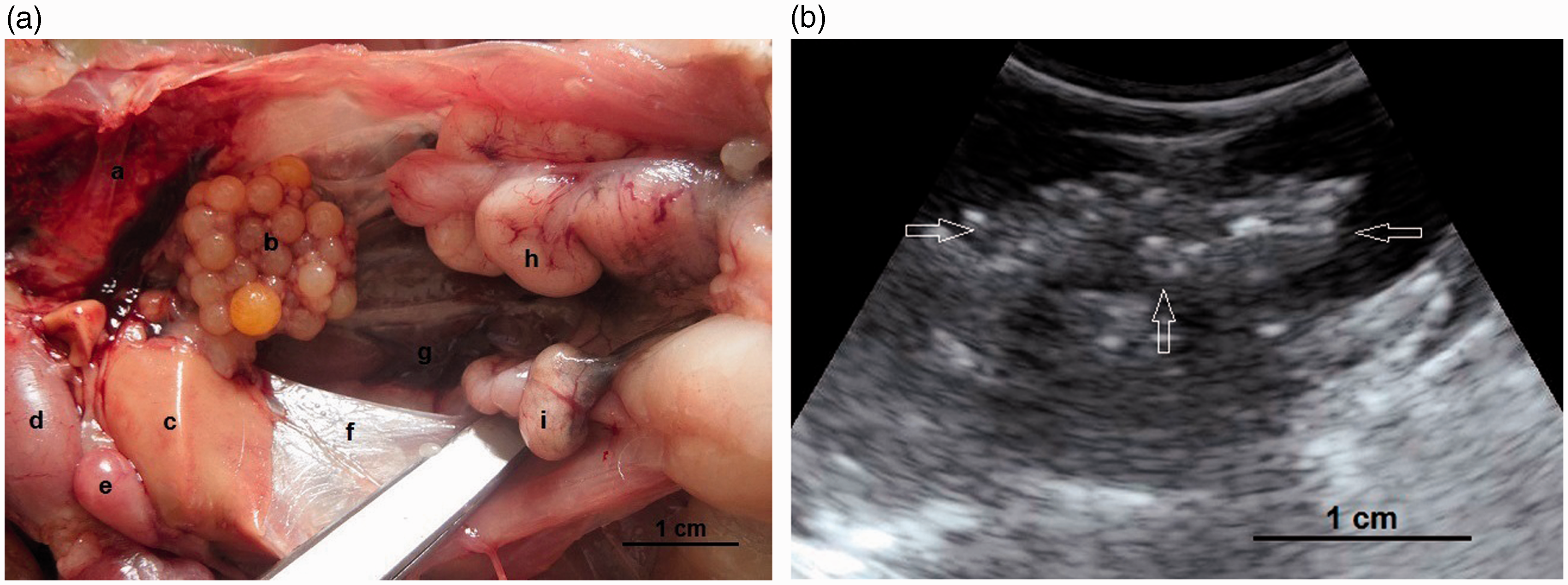
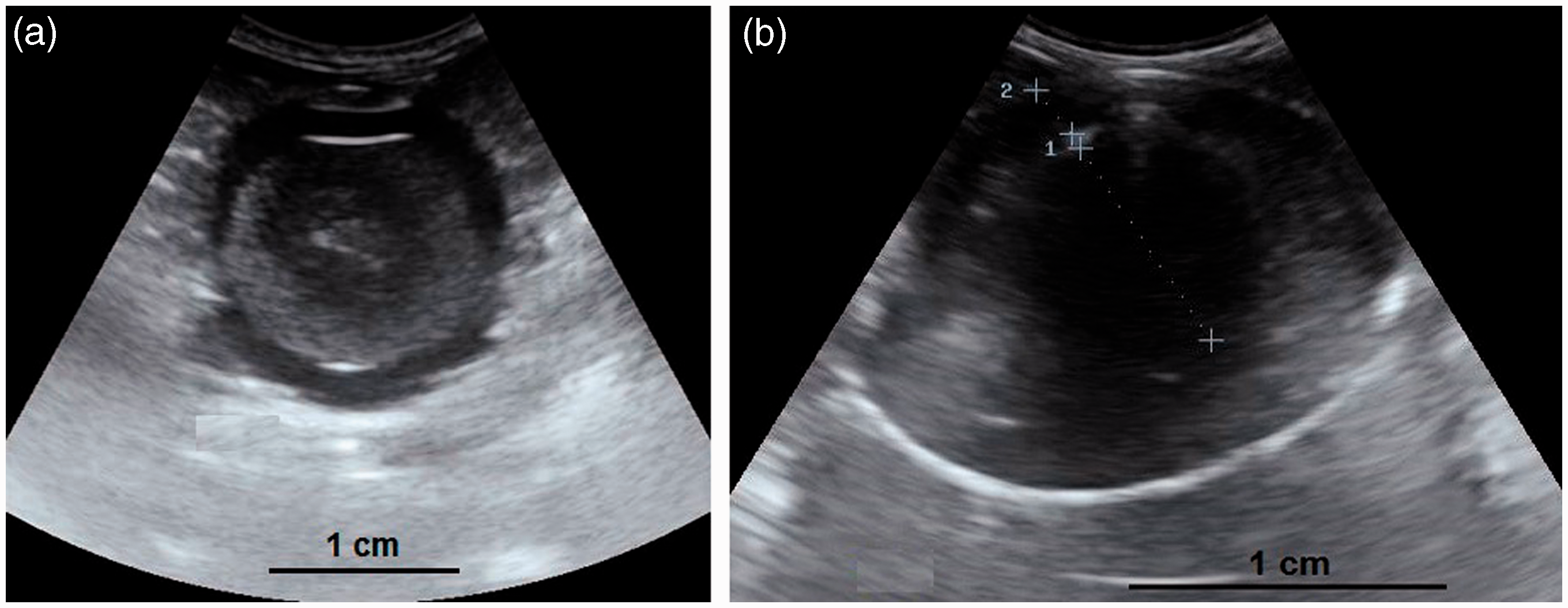
Developments of ova and eggs in different stages are noticed as shown in Figure 9. When the calciferous shell was not deposited completely, the ovum’s inner structures and rings were easily identified (central yolk and surrounding albumen layers) and the ovum was probably located in the magnum or isthmus. On the other hand, when the calciferous shell was deposited completely and had more intense echogenicity, the egg could be assumed to be positioned in the uterus. It should be considered that the progressive formation of eggshell was connected with visible artifacts in the sonogram.
It was not feasible to determine the appearance of the oviducts easily in laying quails, so a standoff technique in euthanized birds was carried out to capture its normal ultrasonographic appearance, after which some regular images of the oviduct in live birds were recorded. The oviduct was observed as a structure which consists of hyperechoic walls and an internal hypoechoic lumen. It was not possible to identify the five parts of the oviduct without any shell formation. However, during eggshell formation, the uterus could be distinguished from the other parts. It was suggested that the difficulty in determining the structure might be due to the absence of any pathological changes such as purulent salpingitis in the lumen. Also the abdominal region is filled with several large follicles during laying, which makes the oviduct more difficult to identify in mature quails.
The findings presented here demonstrate that ultrasound scanning of the reproductive organs of female Japanese quails might be a beneficial diagnostic tool for studying reproductive physiology, anticipating the beginning or the end of the breeding season, and for determining the ideal time for laboratory procedures such as artificial insemination. Moreover, we presented the normal appearance of oviduct, ovum and eggs which could help laboratory researchers to easily detect any pathological disorders (for instance internal ovulation, egg binding, broken eggshells and purulent salpingitis). This method could also help to identify non-productive females, which could lead to more cost-efficient laboratory procedures.
Footnotes
Acknowledgement
This work was supported with funds granted by the Vice Chancellor for Research at the University of Shahrekord.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
