Abstract
Ad libitum (AL) supply of standard chow is the feeding method most often used for rodents in animal experiments. However, AL feeding is known to result in a shorter lifespan and decreased health as compared with restricted feeding. Restricted feeding and thus limiting calorie intake prevents many health problems, increases lifespan and can also increase group uniformity. All this leads to a reduced number of animals needed. So-called standard chows are known to be prone to variation in composition. Synthetic foods have a more standard composition, contributing to group uniformity which, like diet reduction, may decrease the number of animals necessary to obtain statistical significance. In this study, we compared the effects of AL versus restricted feeding (25% reduction in food intake) on standard chow versus synthetic food of three different suppliers on body weight (BW), growth, several blood parameters and organ weights in growing female Wistar rats over a period of 61 days. Diet restriction led to a decreased growth and significantly reduced variation in BW and growth as compared with AL feeding. AL feeding on synthetic diets caused a significantly higher BW gain than on chow diets. Due to experimental design, this same effect occurred on food restriction. Blood parameters and organ weights were affected neither by diet type nor by amount. Incidentally, variations were significantly reduced on food restriction versus AL, and on synthetic diets versus chow diets. This study demonstrates that food restriction versus AL feeding leads to a significantly reduced variation in BW and growth, thereby indicating the potential for reduction when applying this feeding schedule.
The type of diet used and an ad libitum (AL) or restricted feeding schedule can affect the results of an animal experiment. Many laboratory rodents are fed under AL conditions. While this not only results in a shorter lifespan and a decreased health, e.g. an increase in obesity and cardiovascular and renal degenerative disease as well as tumour incidence, it can also result in a potentially confounding variable when compared with restricted feeding conditions (75% of AL intake). 1–3 In general, diet composition and feeding schedule are often overlooked or not taken into account as controllable variables in the experimental design. By restricting the animals’ diet and thus calorie intake, many health problems can be avoided and the variation in body weights (BWs) reduced, thus constituting more uniform groups. 3–7 By food restriction, a reduction in variation of experimental results can be expected, 8–13 which will enhance reproducibility and possibly decrease the number of animals needed to obtain statistical significance.
The type of diet and diet composition can also be used to improve standardization. Most rats are fed on standard diets, the so-called ‘standard chows’. These diets are produced on the basis of natural ingredients. Because of a variation in production, season and the combination of raw materials, the composition of these diets is variable. 13–15 The variation exists between brands as well as between different production batches of the same brand. This may result in a deviation of nutrient levels from the supplier's reported average levels for these nutrients. This variation in nutrient levels may influence the metabolism of the animal, 15 which in turn may influence the animal's performance and the reproducibility of experiments. This variation can be reduced by using synthetic or semi-synthetic diets that are composed of extracted pure forms of natural ingredients and therefore represent a more constant diet composition. The AIN-93 composition is the golden standard with respect to synthetic diets for rodents. 16,17
The extent to which the homegeneity of a group of animals will be improved by using restricted feeding methods and/or purified diets has so far been documented sparsely. In this study, we investigated the effect of food restriction versus AL feeding and, for the first time, the comparison of three different synthetic diets versus three different chow diets in young female Wistar rats was performed. Wistar rats are an outbred strain widely used in toxicological and other studies. Our hypothesis is that synthetic diets and restricted feeding will increase group homogeneity as compared with chow diets and AL feeding, respectively. This was tested by comparing the difference in variations in BW, relative organ weight and several blood parameters (expressed as the coefficient of variation) between AL- and diet-restricted-fed rats on standard chow or synthetic diets of three different suppliers.
Animals, materials and methods
Animals and housing
Experimental group composition
During the entire experimental period (day −9 until day 61) rats were socially housed during the day and individually during the night, in order to make sure that each individual rat would eat the intended amount of food. All cages contained a layer of gold flakes (Bioservices, Uden, The Netherlands) as bedding and a Rat Retreat as shelter (Bioservices). Cages and bedding were replaced weekly. Due to the limited space in the animal unit, some rats were socially housed in groups of three rats instead of groups of two. During the adaptation period from day −9 until day 0 the rats were fed AL on the intended experimental type of diet given. Tap water was also provided AL.
From day 0 until the end of the experiment, six experimental groups of 14 rats were fed restrictedly on one of the three purified or one of the three standard natural ingredient (chow) diets. The other six experimental groups were fed AL on either one of the three purified or one of the three standard chow diets. All animals had AL access to tap water acidified with hydrochloric acid (pH 2.5–3). Drinking water was refreshed twice weekly. The food-restricted groups did not have access to food during the day and received a restricted amount of food (75% of the AL food intake of the corresponding food groups) during the night, their natural part of the day for eating, while being housed individually to secure individual food intake. For animal welfare reasons, the animals were housed socially during the daytime. The AL groups had access to food at all times.
A controlled 12 h light–dark cycle (lights on 06:30–18:30 h), temperature (20–23°C) and relative humidity (40–65%) were maintained in the room.
The protocol for animal use was approved by the Animal Ethics Committee of the Radboud University Nijmegen (#99017).
Diet
Diet composition
Diet composition (g/100 g) and energy level of six diets: standard chow and synthetic diet from three suppliers
*Theoretical calculations based on raw products (analysis or manufacturer's data)
†Analytical data based on analysis of finished product
One batch of each of the following diets was used: RM1(P), batch no. 6248 (SDS, Witham, UK); AIN-93M(P), batch no. 32582 (SDS); EF R/M AIN 93M, batch no. 6097147 (Ssniff Spezialdiaten GmbH, Soest, Germany); R/M-H, batch no. 5326832 (Ssniff Spezialdiaten GmbH); AIN-93M (Harlan, Horst, The Netherlands) and 2014 Teklad Global 14% Protein Rodent Diet (Harlan).
The composition of the purified diets was based on the AIN-93 composition, published by the National Research Council, 16,18 with minor adaptations. The AIN-93 diet fulfils the nutrient requirements for rodents as published in 1995 by the National Research Council, except for the vitamin B12 level. The AIN-93 vitamin B12 level was doubled to comply with the minimum requirements as described by the National Research Council. 18 The standard diets were all composed of natural ingredients. The diets were stored in a cool and dry place with a maximum of 7°C until the time of feeding. One batch of each was used throughout the entire experiment.
Measurements
The BW of each animal was recorded twice weekly in the morning when the animals were transferred back to the group cages (Sartorius BP 8100 balance; Sartorius, Goettingen, Germany). All animals were checked for any clinical abnormalities when being weighed.
Food intake in the AL groups was measured during the entire experimental period. Food intake was measured by weighing back the remaining food in all the individual cages and in the AL group cage. By dividing the food intake of the group cage by the number of rats present in the cage the AL food intake per animal was estimated. Seventy-five percent of the AL intake was fed the next day to the diet-restricted individuals in the corresponding experimental groups and it was checked whether the restricted amount of food had been consumed completely. This was always the case.
On days 28 and 59, blood was obtained by orbital puncture from four rats in each group. The selection of rats was performed randomly by using a computer-generated table. Whenever possible, the same rats were used for both blood samples. For six rats it was not possible to obtain blood for a second time due to eye infection. In these cases, three animals from one group cage and one other rat per group cage from the same experimental group were chosen for blood measurement. Blood was taken by orbital puncture under isoflurane anaesthaesia and collected in heparinized tubes (1 mL) at room temperature. Plasma was analysed for the following parameters: Na, K, Cl, Ca, phosphate (PO4), albumin, cholesterol, alkaline phosphatase (AP) and free fatty acids (FFA). For FFA analysis 1 mL of blood was collected on ice. The first FFA blood samples (day 28) were accidentally not collected on ice and therefore could not be used for analysis. Blood samples were analysed by the CCKL-accredited Department of Clinical Chemistry of the Radboud University Nijmegen Medical Centre according to standard procedures as employed for testing of clinical samples.
At the end of the study (day 59 and day 61), internal organs (liver, kidneys, lung and heart) were removed under isoflurane anaesthesia. Adipose tissue was removed carefully and organs were weighed (Sartorius BP 8100 balance). After the organs were removed, the animals were euthanized by cervical dislocation. Due to the large group size, organs were removed and animals were euthanized over a period of two days starting with the four animals per group that were used for blood sampling. The sequence was determined by a randomization scheme, and under code, thus blinded to the treatment modality.
Health
During the experiment, the rats were housed in a unit where Syphacia obvelata (pinworm) infestation was present. It can be expected that the animals in this study have been infested; however, no clinical signs of infection have occurred during the entire course of this study.
During the first week of the experiment (day −9 until day −2) some minor aggressive interactions were observed when placing the rats back to the group cages in the morning. Since this aggression was only observed during the first week of the experiment, it was concluded that the animals had sufficiently acclimatized to this experimental schedule after this first week.
For six rats, the blood collection by orbital puncture resulted in eye infections. For two rats, the infection became too severe and it was decided to euthanize them before the end of the study. The other animals recovered quickly.
Statistics
Statistical analysis was performed using commercial software (SPSS 16, SPSS Inc, Chicago, IL, USA).
Difference in group variations (AL versus food restriction; chow diets versus purified diets) was examined by combining P values (Levine's test for equality of variations) in Fisher's combined probability test with a 5% significance level. Effect of suppliers was analysed by one-way analysis of variance.
In order to analyse the coefficient of variation all data were transformed to logN.
Results
Growth
At the beginning of the experiment the BWs did not differ between experimental groups. Already during the first week of the experimental period, the BWs of the groups started to deviate. At day 17 the differences in BWs between AL and dietary-restricted (DR) groups became significantly different (P = 0.03, t-test independent samples). This significant difference remained throughout the experiment. After an initially retarded BW gain of the DR groups during the first week of the experiment, all the groups displayed a similar growth curve with a comparable slope afterwards (Figure 1a). BW gain of all AL-fed animals taken together was significantly higher as compared with DR (P < 0.05, t-test independent samples) (Figure 1a).
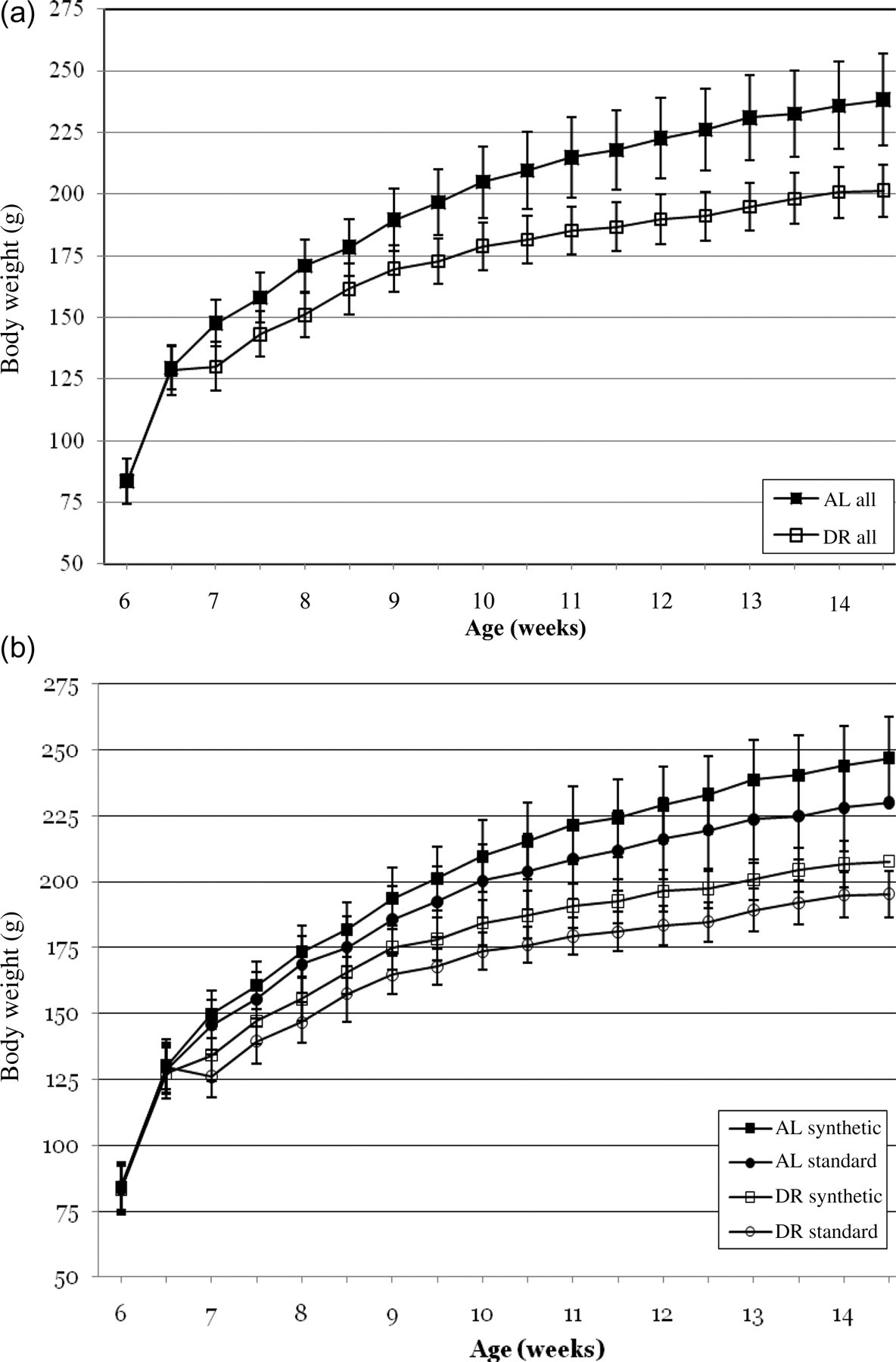
(a) Growth curves of ad libitum (AL) and dietary-restricted (DR) young female Wistar rats. Per group animals fed on standard as well as synthetic diet from all three suppliers are pooled. Error bars indicate SEM. (b) Growth curves of AL- and DR-fed young female Wistar rats. Per group diets from all three suppliers are pooled. Error bars indicate SEM
All the AL-fed groups (standard chow as well as synthetic diet) showed comparable growth curves throughout the experimental period. This was also true for the restrictedly-fed groups. (Figure 1b). Since no significant differences were found in the BW gain per food type between the three different suppliers (Harlan, SDS and Ssniff standard chow; Harlan, SDS and Ssniff synthetic food), the three groups per food type were considered as one group (Figure 1b). However, AL-fed animals on synthetic food gained more weight as compared with AL-fed animals on standard chow (245.3 ± 15.3 versus 229.0 ± 16.9, P < 0.05). This difference was also observed for animals fed restrictedly (207.1 ± 8.5 versus 195.1 ± 8.7 respectively, P < 0.05).
Variation in BWs
Based on Fisher's combined probability test, DR significantly lowered the variation in BWs as compared with AL feeding from 17 days after the start of DR (Figure 2a). While the variation in BWs of the restricted groups decreased over the experimental period, the variation in BWs of the AL groups remained high and showed a slight increase on the chow diets (Figures 2a and b).
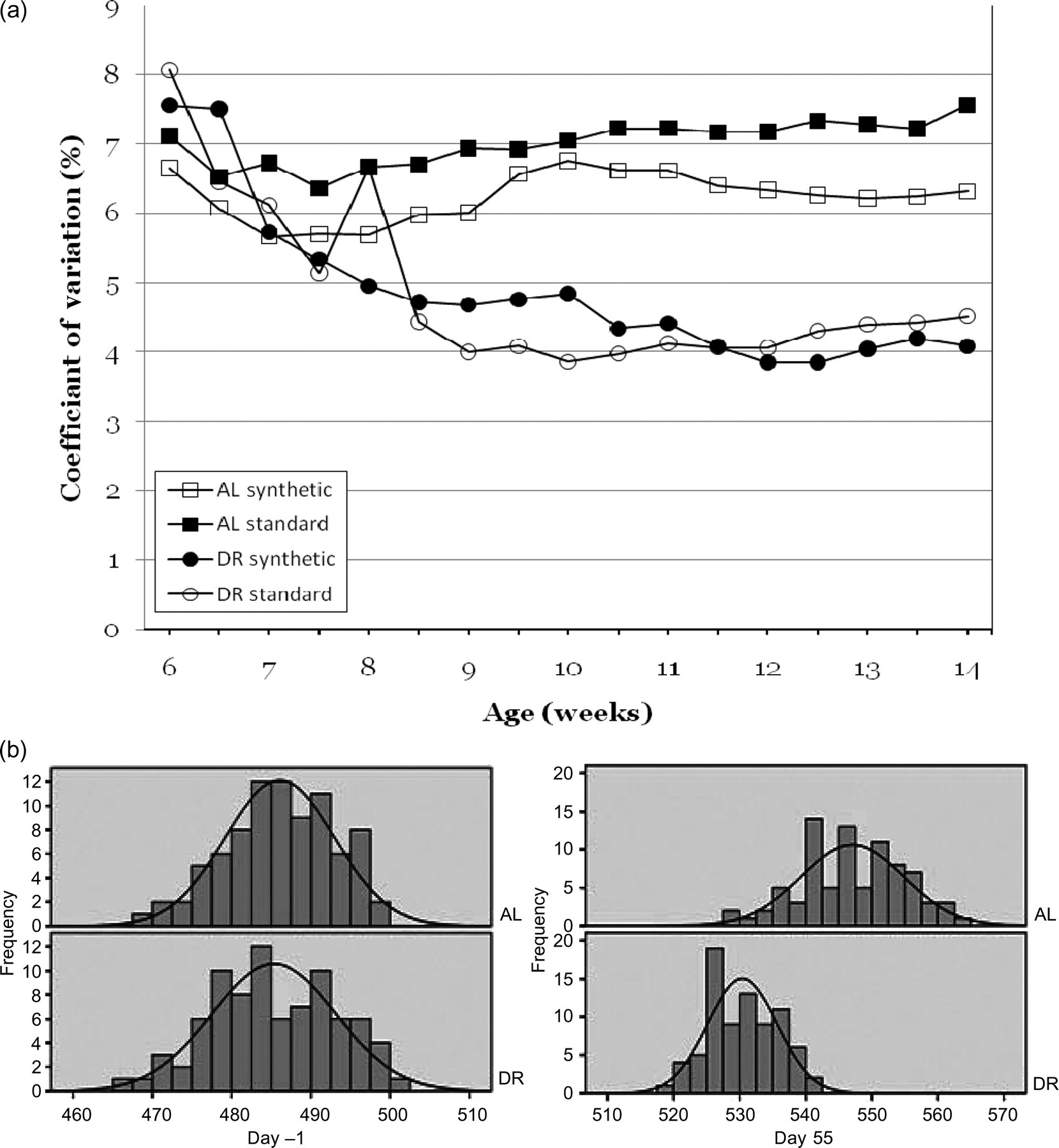
(a) Coefficient of variation in body weights in young female Wistar rats. Per group diets from all three suppliers are pooled. (b) Distribution of rat body weights of ad libitum-fed rats (AL) and restrictedly-fed rats (DR) before the start (day −1) and towards the end of the experimental period (day 55). All groups show a normal distribution. On day 55 DR has caused a significantly lower BW and reduced variation as compared with AL-fed rats
Fisher's combined probability test did not yield significant differences between the variations in BWs of the AL groups fed on standard diets compared with AL-fed groups fed on synthetic diets. Also within the restrictedly-fed groups, no significant differences could be determined between variations in BWs of rats fed on standard and synthetic food.
Food intake
AL food intake as measured during day- and night-time was calculated as the mean intake over three or four days and plotted as a function over time per diet type, averaged for all three suppliers together (Figure 3). Most of the food intake occurred during the night-time when the rats were individually housed (results not shown). During the night-time from day −3 and onwards, the total food intake (grams per day) for standard food was significantly higher as compared with synthetic food (t-test independent samples P < 0.05; results not shown). During the daytime AL food intake for synthetic and standard food did not differ significantly (results not shown; t-test independent samples P > 0.05). From day −3 until the end of the experiment, the food intake in grams per day was almost constant, with only small fluctuations. Food intake on day −8 was significantly lower as compared with the food intake during the rest of the experimental period, which is likely to be the result of transportation stress from the facility of the supplier to our laboratory.
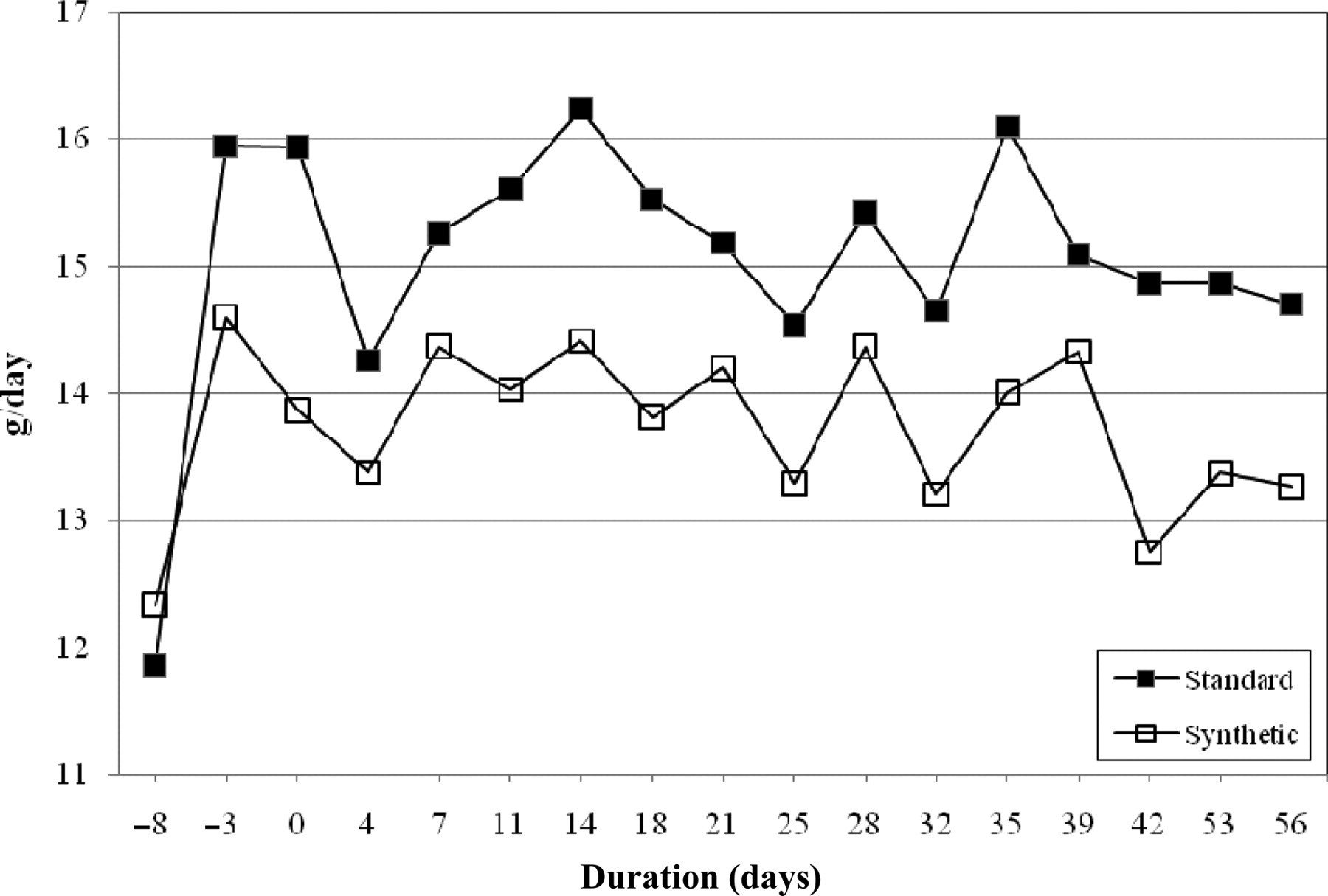
Ad libitum food intake (g/day) of female young Wistar rats on standard chow and synthetic diets. For each type of diet the results from three different suppliers have been averaged
Organ weights
When analysing the absolute organ weights of liver, lung and heart, the AL-fed groups showed significantly higher weights as compared with DR groups. When expressing weights relative to body mass (g/100 g BW), these results were no longer significantly different. With respect to variation, the DR groups showed a significantly lower variation in lung weights (P = 0.01) only. Otherwise no significant differences occurred.
Blood data
The direction of the effect is always such that DR causes a lower variation than AL and synthetic food causes a lower variation than standard chow.
Discussion
Since Wistar rats are widely used in the European pharmaceutical industry during the last decade and because excessive weight gain and early death is a problem when using outbred rats in long-term (toxicology) studies, we used female Wistar rats in this study as a relevant model for the current practice.
As could be expected a 25% reduction in food intake resulted in a reduced BW gain in growing animals. As has been described before, a 25% reduction in food intake in growing animals does not cause any adverse effects and will lead to a decreased weight gain predominantly expressed in reduced fat mass, while organ weights, skeletal size and even muscle mass are not affected. 19,20 Immediately after starting DR, the animals did not gain any weight for several days, indicating a physiological adaptation. Thereafter, the growth curves of animals fed AL as well as DR exhibited a comparable slope. Until the end of the experimental period, the growth curves of restrictedly-fed animals parallelled those of AL-fed animals, but the DR-fed rats never caught up with respect to absolute weight. This is comparable to studies conducted by Kasanen et al., 21 who used a diet board for achieving a reduction of about 15% in food intake. It remains to be determined whether this difference in absolute BW remains throughout the whole lifespan of the animals.
Keenan et al. 3 described that AL feeding (overfeeding) as compared with DR causes an increased inter-individual variation with respect to BW, survival as well as tumour incidence. AL feeding may therefore be considered as an uncontrollable variable in experiments. Reduction of interindividual variation through DR has already been demonstrated for several parameters 8–11 and has even been proposed as a powerful reduction tool. 12,22,23 The current study indeed shows that the variation in BW becomes significantly reduced by DR as compared with AL from day 17 to the end of the experiment (see Figure 2). So, from a general perspective, this study indeed supports the hypothesis of lowering the number of animals needed in experimental studies. When comparing single food types, the variation in BW of animals fed restrictedly on Harlan synthetic food did not reach significant differences from that of animals fed AL on the same diet towards the end of the experiment (days 48, 52 and 55), although it did reach significance at days 41 and 45. The reason for this remains unclear.
In this study, we also compared two diet types from three different suppliers. As synthetic diets have a more standardized composition than chow diets, it was hypothesized that this could lead to a lower variation in experimental results. This study did not support this hypothesis, although the coefficient of variation in AL chow-fed animals was always higher than on synthetic diets, and even showed a slight increase at the end of the study (Figure 2). AL food intake in grams per day on synthetic diets was lower than on standard chows, which is likely the result from the higher metabolic energy content of the synthetic diets. 14,17,18
With respect to blood parameters no consistent differences could be determined, not in the absolute levels nor in the variation, indicating that a 25% reduction of food intake may not affect physiology or biochemistry to such an extent that it becomes reflected in blood levels of Na, K, Cl, Ca, PO4, albumin, cholesterol, AP or FFA. This also indicates that a 25% DR level in young growing female rats does not cause adverse effects. Also no differential effects of using standard versus synthetic food were observed with respect to overall physiology or biochemistry as reflected in the unchanged blood values of Na, K, Cl, Ca, PO4, albumin, cholesterol, AP and FFA. Standard chow diets have been proven to be subject to more variation in composition when compared with synthetic food; 14,15 therefore, batch-to-batch variation as well as seasonal variation in the composition of single food compounds should always be taken into consideration. When interpreting experimental results, it cannot be ruled out that effects of diet type and composition can interfere with experimental results. It is important when planning experiments to determine which dietary factors can interfere with the specific goals of those experiments. 15
With respect to animal welfare it could be argued that AL feeding leads to obese animals that suffer from malnutrition and that DR can be seen as normalizing the animal's metabolism and render it a better research model with less confounding morbidity.
5
It is important to realize that the method of DR can disrupt circadian rhythms and lead to social isolation, both causing stress and complicating interpretation of experimental results.
24
This can be overcome by use of an automated feeding station. This system has to be designed in such a way that group-housed animals can gain individual access to food based on the registration of food consumption for each individual animal. Implanted microchips can be used to register and regulate food delivery to individual animals in software-controlled feeding systems. Since it has been demonstrated that changes in the temporal distribution of feeding does not affect circadian rhythms,
25
it is unlikely for automated feeding systems to affect the diurnal rhythm. Even a shiftwork situation in which rats are forced to eat and drink during their inactive period, does not affect their underlying diurnal rhythm.
26
Examples of commercially available feeding systems are MBrose, Faaborg, Denmark (
While testing pharmacological compounds in restrictedly-fed laboratory animals may decrease the number of animals needed to reach significant results (based on power calculations) due to a decrease in inter-individual variability, morbidity and increase in longevity, the choice of experimental design will depend on many experimental and practical factors. One may prefer testing certain antidiabetic drugs in AL-fed animals based on the comparison with the targeted patient population. However, as AL feeding will still result in a different individual food intake for each individual, this will add to the variation in results. So even in the case of AL feeding, one may consider standardizing the ‘AL amount’ of food for each individual, in order to fulfil the basic principles for experimental testing.
In long-term tests (e.g. toxicological tests), DR provides a clear benefit because of the increased lifespan and reduced morbidity, making it possible to use fewer animals from the start of this type of experiments. In addition, a reduction in the numbers of animals required to obtain statistically significant results becomes possible due to better standardization. Our study shows that in a short-term experiment, indeed a lower variation in BW is achieved by DR as compared with AL feeding, whereas no such favourable effects have been demonstrated on the other parameters measured in this study.
Footnotes
ACKNOWLEDGEMENT
The ESLAV-ECLAM Foundation is gratefully acknowledged for financial support.
