Abstract
Laboratory rats are commonly fed ad libitum (AL). Moderate dietary restriction (DR) decreases mortality and morbidity when compared with AL feeding, but there are several obstacles to the implementation of DR. Traditional methods of restricted feeding disrupt normal diurnal eating rhythms and are not compatible with group housing. We have designed a novel method, the diet board, to restrict the feeding of group-housed rats. Animals fed from the diet board had 15% lower body weight than the AL-fed animals at the age of 17 weeks. The welfare effects of diet board feeding were assessed by comparing the stress physiology of diet board fed animals with that of AL-fed animals. Diet board feeding was associated with higher serum corticosterone levels and lower faecal secretion of IgA, suggesting the diet board causes a stress reaction. However, the AL-fed group had larger adrenal glands with higher adrenaline and noradrenaline content than the diet board animals. No gastric ulcers were found in any of the animals at necropsy. The diet board thus appears to cause a stress reaction when compared with AL-fed rats, but no apparent pathology was associated with this reaction. The diet board could help to solve the health problems associated with AL feeding, while allowing the rats to be group-housed and to maintain their normal diurnal eating rhythms. The diet board can also be seen as a functional cage furniture item, dividing the cage into compartments and thus increasing the structural complexity of the environment. In conclusion, the diet board appears to possess refinement potential compared with traditional methods of DR.
Laboratory rodents are commonly fed ad libitum (AL), i.e. food is always available in unlimited quantities. The reasons for this are practical, as it is the least labour-intensive feeding method. However, AL feeding may result in serious health problems in the animals, and obesity is very common especially in outbred stocks of rats and mice. Moreover, AL feeding is associated with increased mortality and morbidity when compared with many types of restricted feeding or dietary restriction (DR). Numerous health benefits have been associated with DR in rodents; e.g. it prevents obesity and reduces the incidence of neoplasia, 1–3 endocrine disturbances, 3,4 degenerative kidney changes 2,5 and other common diseases. 2,5 Indeed, DR has been shown to promote longevity in a variety of species. 6–10
AL feeding may increase the number of experimental animals needed for research, and thus conflicts with the aim of reduction. 11 The short lifespan of AL-fed rodents results in more animals being needed for long-term safety evaluations. Moreover, the high and unpredictable incidence of spontaneous tumours in AL-fed rodents complicates the interpretation of research results and makes it necessary to use larger experimental groups. 2,4,12–14 Furthermore, controlled food intake significantly decreases interindividual variation in rodents, 6,15–17 which means that fewer animals are needed to achieve the required statistical power.
Although the disadvantages of AL feeding are widely recognized, DR has not been implemented into the routine care of laboratory rodents. Moreover, when DR is used, the animals are often exposed to adverse housing conditions such as social isolation and disturbed diurnal eating rhythms. DR is most commonly achieved by single housing and by offering the animals only one daily ration of food, usually during the daytime, but this is far from optimal for the wellbeing of the animals. Individual housing, i.e. social isolation, is a potent stressor for rats, 18,19 and does not comply with the current European regulations. 20,21 Rats usually eat in several short bouts during the dark phase. 22 If a limited amount of food is offered to a rat during the light phase, it will consume the whole portion at once, which leads to disruption of the diurnal behavioural and endocrine rhythms of the animals. 23,24
The rationale for this most common method of DR is purely practical, as it is more convenient to feed the animals during working hours, and individual housing ensures that each animal receives a calculated amount of food. When food resources are limited in group-housed animals, the hierarchy of the animals has a strong influence on the amount of food consumed by each individual. Even cannibalism has been reported in group-housed rats with DR. 25,26
Several attempts have been made to address these issues. Diet composition can be modified to provide health benefits even if the food is available AL. 27,28 One sophisticated, yet expensive, method of solving the problem is the feeding station (Laboratory Feeding Systems, Denmark), which uses microchips to identify each animal in the group. Each animal has its own feeding regime programmed into the system.
Our research group has come up with a potential solution to this dilemma, the diet board, which combines DR with the possibility of group housing the animals and of maintaining their diurnal eating rhythm. 29,30 The diet board is a simple wooden board with food pellets embedded into it (Figure 1). The animals have to gnaw the wood in order to obtain food. The diet board is left in the cage all the time, giving the animals the option to eat whenever they wish. The diet board is large enough to allow the rats to eat simultaneously from different parts of the board without having to compete for food, and as an extra furniture item, it increases the legally mandated complexity of the cage environment. 20,21 We have already characterized the growth patterns of rats fed from the diet board and its effects on a battery of clinical chemistry values and their variation. 29 The aim of this study was to explore the welfare aspects of the diet board, concentrating on the stress response.
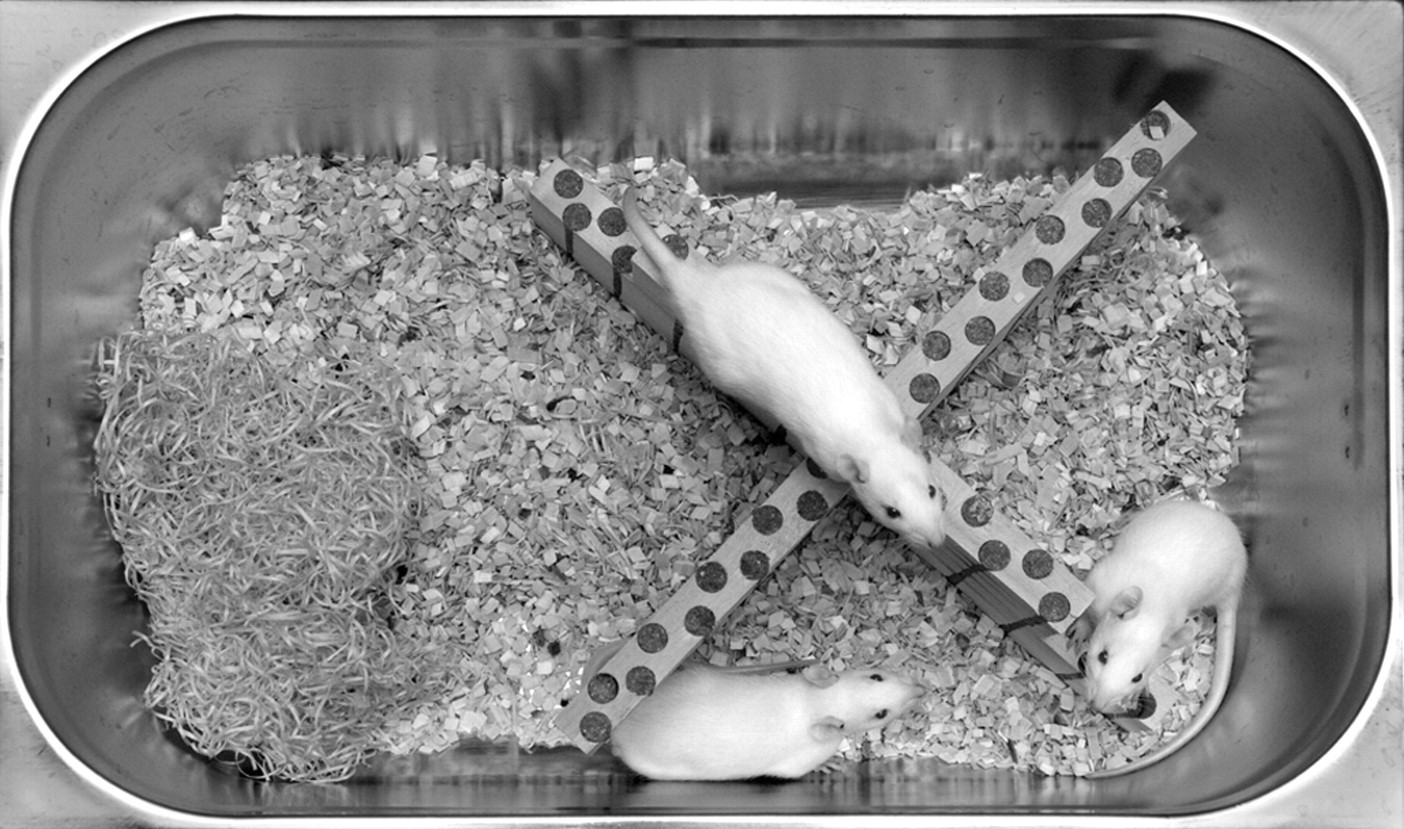
A group of rats in their home cage with the diet board
Materials and methods
This study protocol was approved by the Finnish National Ethics Committee before the study was started at the National Laboratory Animal Center (NLAC), University of Kuopio, Finland.
Animals and housing
Sixty male HsdBrlHan:WIST rats (NLAC, Kuopio, Finland) were used. The barrier of origin was found free of the pathogens listed in the FELASA recommendations for health monitoring. 31 The animals were housed as trios in solid-bottom stainless steel cages with a wire-grid top (48.5 × 28.5 × 20.0 cm, Franke Finland Ltd, Naarajärvi, Finland) in a cubicle. The animals were seven weeks old at the beginning of the study.
The room temperature and relative humidity were maintained at 21 ± 1°C and 55 ± 15%, respectively, and the ventilation in a cubicle provided 15 air changes per hour. Lights went on at 07:00 h and off at 19:00 h. Tap water was offered AL in polycarbonate bottles, and the diet used was autoclaved Lactamin R36 (Lantmännen, Kimstad, Sweden). Food availability is described below. Aspen chips (5 × 5 × 1 mm; Tapvei Ltd, Kaavi, Finland) and aspen wool (Tapvei Ltd) were used as bedding and nesting material; both were replaced with new material at the biweekly cage change.
The diet board
The diet board was made of two aspen boards (35.0 × 12.2 × 2.7 cm); two corners of each board had been removed (6.0 × 6.0 cm) to ease movement between the four cage compartments. Each board had 20 vertically drilled holes (Ø 12.5 mm) with a 2-3 mm slot visible from the side. The passages were filled with food pellets and fixed in place with an autoclaving cycle (121ºC, 20 min, 220 kPa, Finn-Aqua 121821 D, Steris Finn-Aqua, Tuusula, Finland). The cages of the control animals were equipped with plain autoclaved boards of the same size, but without the drill holes and food. The boards were placed in the form of crosses made by intersecting two boards. Since the boards were made of aspen, i.e. the same material as the bedding, they presumably had the same emission profile, but at lower concentrations than the non-autoclaved aspen bedding material used. 32
Study group and control group
The animals were divided into two groups: DR and AL. The AL group served as controls for the DR animals. The DR animals were fed exclusively with the diet board throughout the experiment. The AL animals were fed AL; food was always available in the food hopper.
Design features
Both the AL and DR groups consisted of 30 rats and were formed in the following manner. Ten litters with a minimum of six male siblings were ordered for the study, and six males from each litter were chosen for the experiment. The male pups were weighed and the six male pups closest to the average weight were chosen. These six siblings were housed together from weaning until the beginning of the study. When the study began, these six rats were placed at random into two cages; one cage with DR and a matching cage with AL feeding. Thus both groups had 10 cages, with three siblings from the same litter in each cage. The location of the cages in the cage rack was randomized to yield an equal number of each group at each horizontal level. This allocation was done on a Friday, when the animals were six weeks old. The animals were also weighed and ear-tattooed on the same day. They were allowed an acclimatization period over the weekend with AL feeding. The study commenced on the following Monday, when the animals were seven weeks old. The animals were weighed again and the autoclaved diet boards and plain boards were introduced into the cages. The boards were replaced by fresh ones weekly at each Monday cage change. The animals entered the study in three cohorts, all following the same week-day routine. The cages were changed twice a week, on Monday afternoon (12:00–14:00 h) and Friday morning (09:00–11:00 h). The rats were weighed twice a week on Mondays and Fridays during each cage change. The study lasted 10 weeks.
Humane endpoints
Three different age-specific humane endpoints (HEPs) were established. From seven to eight weeks of age, the limit was preset to 15% loss of body weight. From eight to 13 weeks of age, failure to gain weight during a two-week period or a 5% loss of body weight during one week was used as the HEP. Thereafter, the limit was set at 10% loss of body weight during a one-week period. Additional clinical signs included in the HEPs for the rats throughout the study were dehydration, unexpected disease, trauma or dental problems.
Blood samples
The blood sampling programme consisted of four bleeds during the study, on the Mondays of weeks 2–5. The samples were taken by piercing a hole in the vena saphena with a 25G needle 33 and the resulting blood drops were collected in a test tube. The sample volume did not exceed 0.5 mL. The collected blood was allowed to coagulate for 10–15 min and was centrifuged at 12,000 rpm for 15 min at room temperature (Eppendorf 5412, Eppendorf, Germany). The serum acquired was placed into plastic tubes and immediately frozen at −20ºC.
Serum corticosterone
Total serum corticosterone (CORT) was analysed from the serum samples with the ImmuChemTM Double Antibody Corticosterone 125I RIA Kit (MP Biomedicals, LLC, New York, NY, USA). The radioactivity was measured with a 1260 Multigamma II gamma counter (LKB Wallac, Sollentuna, Sweden).
Collection of faecal samples
Faecal samples were collected on the Friday of week 10 of the experiment. The animals were placed into a clean normally bedded cage during the routine cage change on Friday morning. After 6 h, all the faecal pellets in the cages were collected and immediately frozen at −20°C.
Faecal IgA
The extraction of IgA from the faecal samples was done according to the method described by Pihl and Hau. 34 The concentration of IgA in the samples was analysed with a Rat IgA Quantitation Kit (Bethyl, Montgomery, TX, USA) using a Multiskan Ex microtiter reader (Thermo Electron Corp, Waltham, MA, USA).
Euthanasia and postmortem examination
After the collection of a terminal blood sample by cardiac puncture under CO2 anaesthesia, the animals were euthanized with 100% CO2. Both adrenals were carefully dissected, weighed and immediately frozen in liquid nitrogen. The ventricle was cut open and examined visually for gastric ulcers. Other abdominal and thoracic organs were assessed visually for macroscopic changes.
Adrenaline and noradrenaline determination
The concentrations of adrenaline (ADR) and noradrenaline (NOR) in adrenal glands were measured using high-performance liquid chromatography with coulometric electrochemical detection. 35 The tissue samples were homogenized in 0.1 mmol/L perchloric acid, proteins were precipitated by centrifugation and the supernatants were extracted with activated alumina. 36 Authentic reference standards and the internal standard, 3,4-dihydroxybenzylamine, were purchased from Sigma-Aldrich (St Louis, MO, USA). The right and left adrenals were weighed separately and absolute weights were used for the statistical analyses. Likewise, the concentrations of ADR and NOR were analysed separately for the right and left adrenals.
Data processing and statistical analyses
The appropriate size of the experiment was estimated with the Resource equation method; 37 the degree of freedom for error was 17. The data were analysed with linear mixed models using all the collected data. Log-transformation of the variables was employed when deemed necessary. A random litter effect was included in the models in order to accommodate for the possibility of correlated outcomes among siblings. Repeated measures from the same subjects were similarly analysed applying linear mixed models including time, treatment and their interaction as fixed effects into the model. In addition, siblinghood was included as a random effect in the models in order to account for possible dependencies due to the hierarchical nature of the data.
The results of normally distributed variables are presented as arithmetic means and standard deviations. Model-based estimates for difference of the means and their 95% confidence intervals (CI) are also reported. For variables that were not normally distributed, the results are presented as model-based estimates of the geometric means, their ratios and their 95% CI. Geometric means and 95% CI are used to express the results of serum CORT graphically. Box plots from the raw data are used as graphic representation of the rest of the parameters. The statistical software package used to process and analyse the data was SPSS 14.0 for Windows (SPSS Inc, Chicago, IL, USA).
Results
Serum corticosterone
CORT concentrations in blood serum were analysed at experimental weeks 2–5 (Figure 2, Table 1). The values were log-transformed before statistical analysis because their distribution was not normal. The interaction between time and treatment was not significant (P > 0.05) in the linear mixed model for repeated measures. The average levels of serum CORT over time differed significantly between the DR and AL groups, with DR rats displaying higher values. The model-based estimates of average geometric means for the groups were 30.41 ng/mL (95% CI 16.37, 56.64) for the DR animals and 11.22 ng/mL (95% CI 6.03, 20.89) for the AL animals. The ratio of the geometric means was 2.72 (95% CI 1.67, 4.41, P < 0.0001).
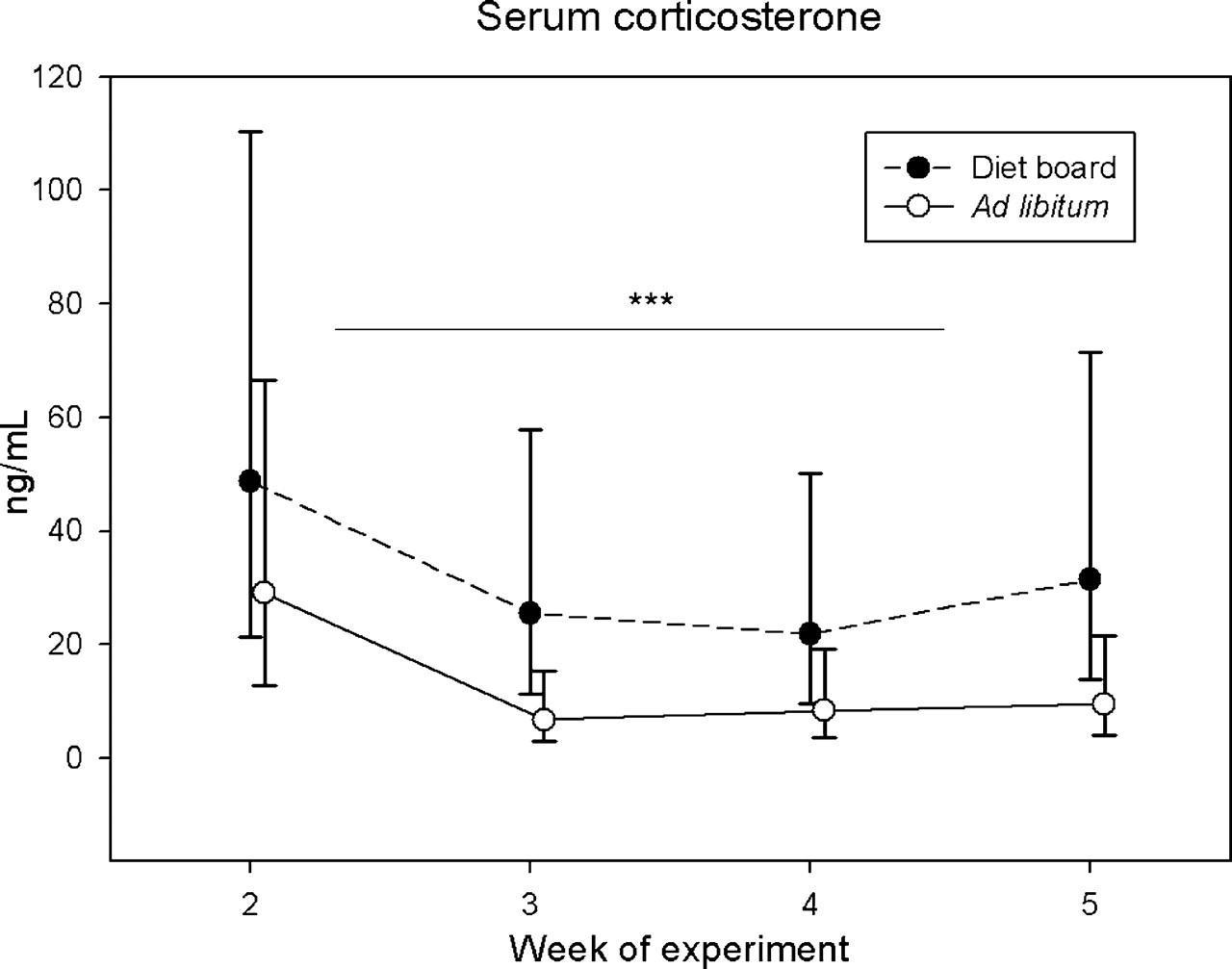
Serum corticosterone results at study weeks 2–5. The results are presented as geometric means and the 95% confidence intervals. The asterisks indicate the statistical significance of the average differences between the diet board (n = 30) and the ad libitum (n = 30) groups tested with a linear mixed model for repeated measures (***P < 0.001)
Physiological values and distributions
Adrenal weights, ADR and NOR
The right adrenal gland exhibited no detectable differences (P > 0.05) in weight, ADR or NOR concentrations, whereas the left adrenal gland of the AL group showed higher values in all variables. The mean weight of the left adrenal gland was 0.0323 ± SD 0.004 g in the DR group and 0.0346 ± 0.0042 g in the AL group (model-based difference −0.002 g, 95% CI 0.004, 0.000, P = 0.024). The ADR concentration was 49.37 ± 2.33 μmol/adrenal in the DR group and 55.12 ± 2.34 μmol/adrenal in the AL group (model-based difference −5.74 μmol/adrenal [95% CI −9.54, −1.94, P = 0.004]). The NOR concentration was 14.08 ± 1.20 μmol/adrenal in the DR group and 17.16± 1.20 μmol/adrenal in the AL group (model-based difference −3.07 μmol/adrenal, [95% CI −4.63, −1.52, P < 0.0001]) (Figure 3, Table 1).
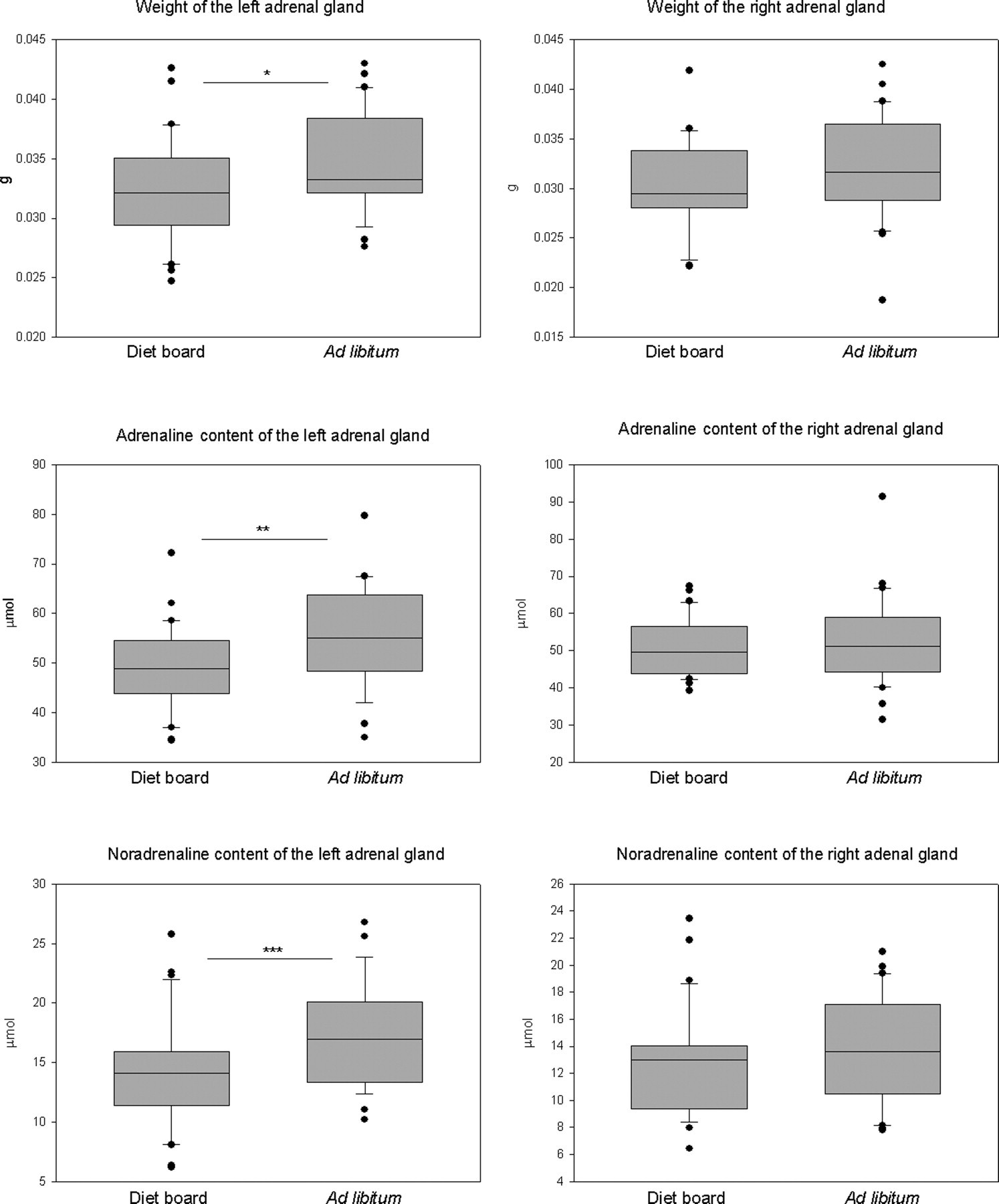
Box plots of the adrenal weights and the adrenaline and noradrenaline contents of the adrenals at the end of the 10-week diet restriction experiment. The asterisks indicate the statistical significance of the difference between the diet board (n = 30) and the ad libitum (n = 30) groups (*P < 0.05, **P < 0.01, ***P < 0.001)
Faecal secretion of IgA
The parameter used in the analyses was the secreted amount of IgA per hour per cage (ng/h). The values were log-transformed before the analysis because their distribution was not normal. The DR animals had a higher rate of faecal IgA secretion. The model-based estimate of geometric means for the groups was 78,900 ng/h (95% CI 45,600, 136,100) in the DR cages and 167,100 ng/h (95% CI 96,600, 289,000) in the AL cages. Average individual secretion rates were 26,300 ng/h/rat and 55,700 ng/h/rat, respectively. The ratio of the geometric means was 0.47 (95% CI 0.32, 0.70, P = 0.002) (Figure 4, Table 1).
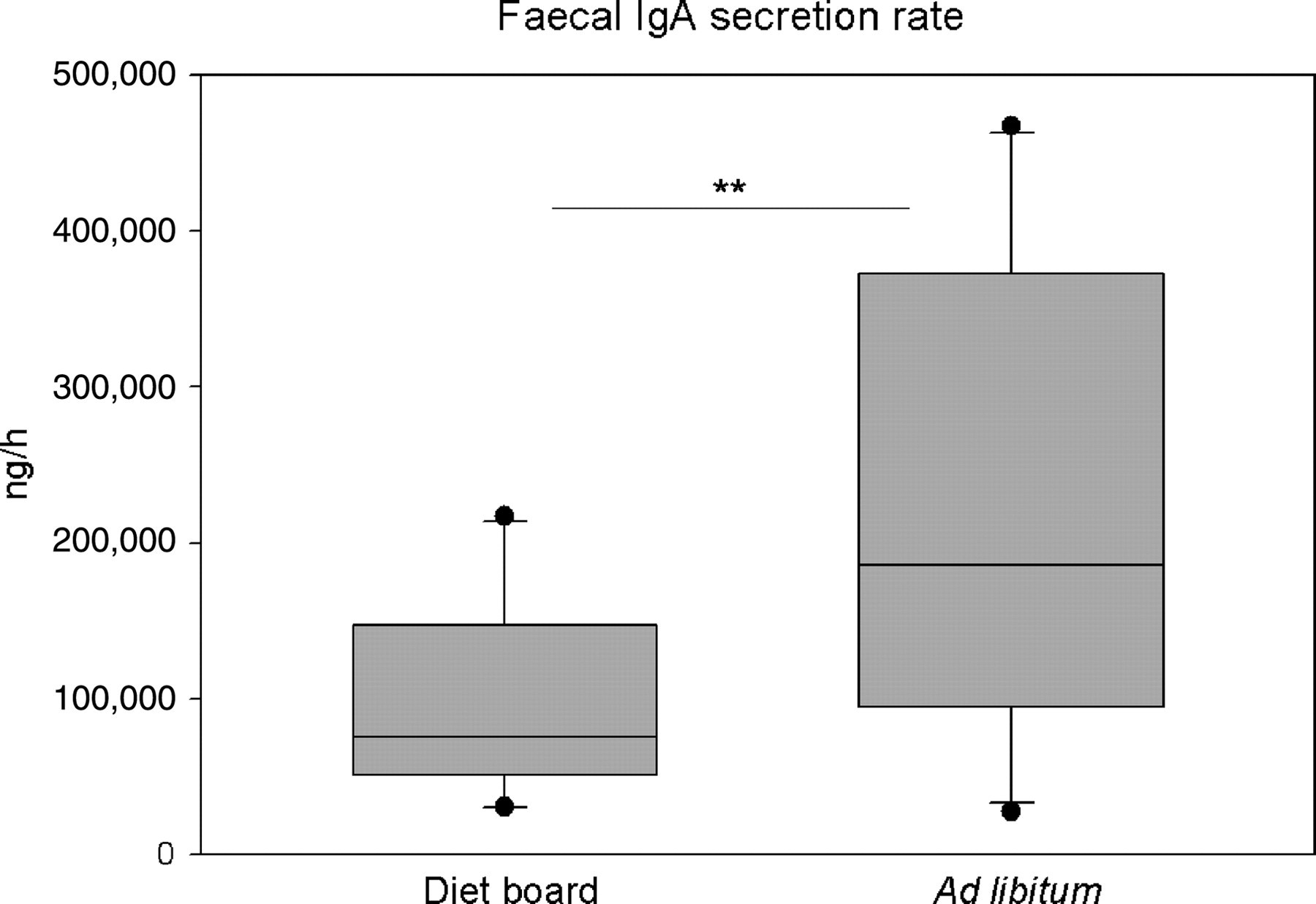
Box plot of the faecal IgA secretion in week 10. The asterisks indicate the statistical significance of the difference between the diet board (n = 30) and the ad libitum (n = 30) groups (**P < 0.01)
Gastric ulcers
No gastric lesions were found in any of the animals in the postmortem examination.
Discussion
The diet board is a simple, functional method of restricting weight gain in laboratory rats. It does not offer the same control over feeding that is provided by traditional methods of DR; the amount of food to be eaten cannot be preset, nor exactly measured afterwards. Nonetheless, the diet board has potential for major advantages compared with traditional methods of DR: the rats can be group-housed and can maintain their normal diurnal eating and resting rhythms. 29,30 The diet board may be considered a refinement compared with the traditional methods of DR.
The drawbacks of AL feeding in the long term are well known, i.e. obesity and increased morbidity and mortality. 2,4,38 Even though the traditional DR methods can provide valuable benefits for the health of the animals, they may also hamper the overall wellbeing of the animals. 39,40 This study was designed to assess the short-term welfare outcome of the diet board. An outbred Wistar stock was used in this study. Excessive weight gain is a more pronounced problem in outbred stocks than in inbred strains. The Wistar stock is an especially relevant model, since Wistars have become the most commonly used rats in the European pharmaceutical industry during the last 10 years (Haasio K, personal communication).
The body weight of the diet board rats is maintained at ∼85% of the body weight of AL-fed controls. The difference in absolute weights between DR and AL rats is established during the first week; thereafter, the growth curves appear to have the same slopes and the same mutual distance. While there are gross differences in body fat deposits, the diet board retards skeletal growth only minimally. 29 The diet board offers mild to moderate DR, and no adverse effects attributable to the diet board or group housing have surfaced. There is no evidence of fighting or competition for food; the animals frequently eat simultaneously from different parts of the diet board. Furthermore, the absence of difference in the variance of body weights suggests that the DR animals grow as evenly as the AL animals. 29
The average serum CORT concentration was higher in DR rats from experimental week 2 to week 5 (Figure 2, Table 1). Elevated serum CORT concentrations are a central part of an organism's response to stressors, i.e. the stress reaction. 41–43 Evidence of a stress reaction does not necessarily imply a deterioration of welfare. The pathological changes caused by stress are manifested only when the stress reaction is insufficient to help the organism cope with the stressor. DR is clearly a stressor to the organism and is associated with a stress reaction. This stress reaction, or heightened adrenocortical activity level, has been proposed to mediate the positive effects of DR on health and longevity. 44–46 It is not clear whether the higher levels of CORT seen in the diet board rats reflect a beneficial adaptive reaction to DR or a difficulty in coping with it.
Serum CORT follows a diurnal rhythm, with rats showing the lowest values in the beginning of the light phase. 47–50 Stressors have been shown to disturb this diurnal rhythm; in this case, the CORT levels are elevated throughout the day. 48,51,52 The difference between the CORT levels of unstressed and stressed animals is most prominent during the beginning of the light phase, when unstressed animals have low CORT values. In this study, the CORT samples were taken between 09:00 h and 11:00 h to allow the detection of subtle changes in the serum CORT values. Both DR and AL groups had relatively low CORT values. Even though the serum CORT levels were significantly higher in the DR group, the results do not allow the interpretation that the DR group lacked a normal diurnal CORT rhythm, since the whole 24 h rhythm of CORT was not determined in this study.
The absolute weights of the right adrenal glands were similar in both groups, but the AL group had slightly but significantly larger left adrenal glands (Figure 3, Table 1). Stress-associated hypertrophy of the adrenal glands has been reported to consist mainly of the thickening of the cortex. 53 In this study, the structure of the adrenals was not analysed. This result is not consistent with the higher values of CORT observed in the DR group.
The AL group had significantly higher values of both ADR and NOR in the left adrenal (Figure 3, Table 1). Enhanced secretion of catecholamines from the adrenal medulla is a part of the stress reaction. 41,54–57 It is not clear whether this observation can be interpreted as a stronger stress reaction in the AL group or whether it is just an incidental finding. It has been suggested that DR causes a mild beneficial stress reaction (hormesis) in itself and prepares the organism for other stressors. 58 DR has been shown to decrease the reactions to different stressors. 13,59 We hypothesize that the method of feeding might alter the way an animal reacts to the stressors it encounters. Diet board animals that experience the hormesis of DR might be better equipped to react adequately to the stressors routinely encountered. AL feeding offers no challenge for the innate stress systems of the animal, and so fails to prepare the animal to cope with stressors. AL animals might repeatedly ‘overreact’, causing a visible difference in the adrenomedullary system. On the other hand, AL feeding could be a stressor in itself for the animals, only manifested in a different way than the heightened adrenocortical activity seen in DR animals.
We have previously reported that AL-fed rats exhibited higher levels of serum free fatty acids (FFA) than diet board rats after 10 weeks of the different feeding regimes. 29 Increases in serum FFA have been observed in stressed animals. 60–62 Catecholamines from the adrenal medulla mediate stimulation of lipolysis, leading to the increased levels of circulating FFA. 63–65 Even though DR causes a stress reaction in the body, it has been associated with decreased serum FFA levels when compared with AL feeding. 66–68 This could suggest that the stress reaction associated with DR is limited to the adrenal cortex without activating the adrenal medulla. On the other hand, lower values of FFA in DR animals could represent just a general decrease in the fat metabolism due to reduced food intake and smaller fat deposits in the body.
In rodents, quantification of faecal IgA secretion has been used to assess stress reactions. It can be done with minimum disturbance to the animals, which are then less prone to be affected by the sampling itself. Faecal secretion of IgA is negatively correlated to faecal corticosterone secretion in rats. 69 In rodents, chronic stress (e.g. social stress, social isolation) is associated with decreased IgA secretion into the faeces 70 and into the saliva. 71 Acute stressors are not reflected to such an extent in the IgA secretion. 69 In this experiment, the DR group had a significantly lower rate of faecal IgA secretion than the AL group (Figure 4, Table 1). This could indicate that the DR rats had a chronic stress reaction compared with the AL animals. The secretion was expressed as the amount of IgA secreted per hour per cage. The absolute secretion of IgA was analysed instead of the secretion rate in relation to body weight or the weight of the faeces. It is not known whether the IgA secretion is somehow directly affected by the amount of food eaten.
In the postmortem examination, the stomach of each rat was visually inspected for possible gastric ulcers. Stressful experiences predispose rodents to ulceration of the gastric mucosa, i.e. stress ulcers. 72 DR (80% of AL feeding) has been shown to increase the incidence of gastric lesions in mice. 40 In this experiment, no gastric ulcers or other lesions were seen in any of the rats from either group.
The parameters measured in this experiment all reflect the stress reaction of the body. The overall welfare of an animal cannot be assessed by stress reactions alone, but a chronically stressful environment can be considered a serious threat to welfare. 41,73–75 The results of this experiment show that although the diet board causes a measurable stress reaction in the rats, no stress-related pathology could be observed in the diet board rats. It is not yet known whether the stress reaction associated with the diet board is beneficial or harmful for the health of the animals over time. The true significance of the differences in adrenomedullary activity remains to be resolved. Future studies will concentrate on the effects of the diet board on other aspects of welfare, i.e. home-cage behaviour, performance in behavioural tests indicating anxiety, depression, etc., diurnal rhythms of behaviour and metabolism, and cardiovascular parameters of stress.
The diet board can be considered as a refinement when compared with other methods of DR. It has some obvious welfare advantages over traditional methods of DR. In addition to the aforementioned benefits (possibility of group housing, undisturbed diurnal rhythms, etc.), the diet board offers the animals some control over their environment. They have the option to work for food when they feel hungry. Even when total energy intake remains low, the possibility of trying to find food can decrease the stressfulness of DR. 76
The refinement potential of the diet board may increase with the age of the animals and the duration of the experiments, because the health problems associated with AL are emphasized at older age. When rodents are used in long-term experiments, DR could provide valuable benefits for the health and wellbeing of the animals. However, large numbers of young rodents are used in short-term experiments where obesity and associated ills are not such a prominent issue.
In conclusion, the diet board is a promising solution when DR is needed in laboratory rats. It causes an apparent stress reaction, but no pathology of the parameters examined is associated with this reaction. The diet board could help to solve the health problems associated with AL feeding in a welfare-friendly way, allowing the rats to be group-housed and to maintain their normal diurnal eating rhythms.
Footnotes
ACKNOWLEDGEMENTS
This study was financially supported by the Finnish Ministry of Education, the Academy of Finland, the European College of Laboratory Animal Medicine (ECLAM) and European Society of Laboratory Animal Veterinarians (ESLAV) Foundation, Universities Federation for Animal Welfare (UFAW), the Research and Science Foundation of Farmos and the Finnish Foundation of Veterinary Sciences.
