Abstract
The single housing of laboratory rats may be recommended in some situations such as hypothesis-driven or test-specific studies, during electroencephalogram recording of phases of sleep and after surgical procedures. However, as single housing of laboratory rats has been shown to be stressful, modification of the housing environment is needed to improve the welfare of these animals. This experiment was carried out to investigate the long-term effects of environmental enrichment on some behavioural, physiological, pathological and psychological measures of welfare. With two batches of animals, 24 rats were housed singly in either enriched cages (EC) (n = 12 cages) or unenriched cages (UC) (n = 12 cages). Behaviour was sampled every week and so was body weight and weight gain over a six-week observation period. Behaviours of the rats in the elevated plus-maze were recorded on the seventh week, whereas organ weights were recorded postmortem. The results revealed that long-term single housing of rats in super-enriched cages increased levels of indicators of good welfare including sleep, exploration, movement and feeding behaviour, body weights, weight gains and the relative weights of the thymus gland and spleen, and decreased levels of indicators of poor welfare such as stationary behaviour and the relative weight of adrenal glands. Thus, enrichment of conventional cages of newly weaned singly-housed laboratory rats with multiple physical structures appeared to improve their ability to control the environment and to promote their species-specific behaviour; changes that can ultimately result in good welfare.
Laboratory rodents account for the majority of animals used in scientific experiments worldwide. Over 10 million rodents were used in Europe in 1991, the first and only year for which European figures are available, the majority were rats and mice (31% and 68%, respectively). In the UK where accurate figures are collected annually, 2.18 million (87%) of the total of 2.66 million animals used in 2004 were rodents, this included rats (19%) and other animals. 1 The number of rats used in research is likely to increase with the increased use of transgenic animals. In general, housing rats in laboratory conditions constitutes a confined, non-stimulating, barren environment for them. This restrictive housing situation may compromise the welfare of the animals and can cause stress (defined here as the adaptive response to an event that places special physical and/or psychological demands upon an individual and which may overtax its control system and reduce its fitness 2 ) in various ways. For example, the restriction of behaviour may act as a stressor itself, when highly motivated behaviours are thwarted. 3,4 Moreover, restrictive conditions limit the ability of the animals to predict and control their physical and social environment, and can therefore be stressful. 5,6
Therefore, as rodents spend a major proportion of their lifespan in the laboratory cage, improving this environment may not only improve their overall wellbeing 7,8 by improving their ability to cope with the environment, but also the accuracy of experimental results. 9,10 This in turn is likely to provide a valid animal model for research 11 and can ultimately result in a reduction in the number of animals used.
Environmental enrichment defined as ‘the modifications of the environment resulting in an improvement in the biological functioning of the captive animals’ 12 is an important tool of improving housing conditions of laboratory rodents. Experiments have demonstrated wide beneficial effects of environmental enrichment on group-housed laboratory rodents. 13–16 However, despite this consensus over the effects of environmental enrichment in rats, very few studies have considered how enriching laboratory cages of singly-housed rats by adding multiple physical structures may affect their welfare.
Although group housing is the recommended housing situation for laboratory rodents, 17,18 it may not, under certain circumstances, be achievable. For example, in hypothesis-driven or test-specific studies (e.g. application of isolation-induced aggression) single housing of the subjects may be necessary. It may also be a part of an experimental paradigm to study the effect of deprivation of social stimuli or to induce stress. 19,20
Other reasons for individual housing of rodents could be after surgical procedures involving the implantation of equipment (such as chronic cannulas for serial blood sampling 21 or electrodes for EEG recordings) and sutures which can be damaged by other animals. Animals may thus need to be housed singly or removed from their groups into the operating chamber or cage, and returned after the stipulated time of recording or surgery has finished. Both these solutions are problematic from the welfare perspective: single housing has been shown to be stressful 22,23 while removing and returning animals disturb the social structure of the group which may be stressful and likely to disrupt the social memory of rodents. 24
It is, however, important to distinguish between true isolation and individual housing of laboratory rats. In the latter situation, the rats are not devoid of receiving sensory information from other rats in the same room by means of olfaction, hearing and vision. Nevertheless, individual housing deprives animals of social stimuli, which in turn can cause profound alterations of behaviour. 25 It could therefore be interesting to look at the effect of enriching housing conditions for singly-housed animals.
There have already been some studies that looked at the effect of environmental enrichment on some behaviours of singly-housed rats such as exploratory behaviour and general activity, 26 on the interest of rats towards enrichment items 27,28 and also on the development of their brains in the enriched environment. 29
It has also been shown that environmental enrichment can reduce the response of the hypothalamic–pituitary–adrenal (HPA) axis, including lower corticosterone responses and smaller adrenal weights, to stress (saline injection) in singly-housed rats. 21 However, an elevated level of basal corticosterone 30 and increased weight of adrenal glands 30,31 were also reported in laboratory rats after prolonged housing in enriched cages. Research has also attached some beneficial psychological effects of housing in enriched conditions in terms of amelioration of behavioural deficits and reduction of indices of stress in standard conditions of captive environment in singly-housed laboratory rats. 32,33
However, the fact that none of these studies looked at how long-term enrichment can affect the ability of the singly-housed animals to cope with their environment by looking at their behavioural, physiological, pathological and psychological responses to the housing condition highlights the need for more research. This experiment was therefore carried out to study the long-term effects on multiple measures of welfare of enriching cages of singly-housed rats, by adding various physical structures to the cages that are thought to stimulate rat-specific behaviour.
In the current experiment, a multiple welfare indictor approach was used to investigate the effect of environmental enrichment provision on the welfare of laboratory rats. This approach involved the use of various indicators of welfare including behavioural measures (home cage behaviour), psychology (tests of anxiety such as elevated plus maze), performance (body weight and weight gain) and pathology (changes in weight of internal organs). 5 The use of a multiple welfare indicator approach has been shown to be effective in measuring welfare in juvenile laboratory rats. 34 The elevated plus-maze (EPM) test is commonly used to assess anxiety-like behaviour in laboratory rodents. Anxious animals were found to prefer, and were more active in, the closed arms over the less secure open arms; behaviour which is indicated by less time spent on, and low frequency of entries too, the open arms. 35–37
Weighing internal organs is a simple procedure that may be very instructive in providing good information about whether the animal has been experiencing stress. Although always necessarily an invasive postmortem procedure, it may sometimes be less of an ethical concern as the animals might be autopsied routinely after the termination of the experiment, 38 therefore in such case the stress-related pathological changes can be detected without using invasive procedures in live animals. An additional benefit of using organ weights is that they are likely to be more permanent than measures of hormone levels, i.e. undynamic variables that once changed remain changed.
The increase in the weight of the adrenal gland (adrenal hypertrophy) is generally thought to result due to the increased activity of the gland, particularly the cortex (cortical hypertrophy). 38 This has been suggested to happen under the frequent stimulation and the increased activity of the adrenocorticotrophic function of the pituitary gland, which results from the stimulation of the HPA axis during chronic stress. 39 The reduction of the thymus weight (that is referred to as thymus involution) is also considered to happen under the effect of increased adrenal activity (the level of corticosteroids). 22,39
Materials and methods
General animal housing and husbandry
This experiment was carried out with two batches of animals using 24 rats. Within each batch there were six replicates of each experimental treatment (see later). The subject animals were newly weaned male rats, of the Wistar Hannover (outbred) strain, weighing 35–50 g on arrival (Al-Alamia Company, El-Gharbia, Egypt). The rats were four weeks of age on arrival and were fed on pelleted food (Rat chow®, Oil and Soap Manufacturing Company, El-Gharbia, Egypt) ad libitum. Tap water (2 bottles fitted in each cage) was also provided ad libitum and renewed everyday. Water bottles were changed every week.
Housing conditions
Rats were singly housed in cages (manufactured by Al-Alamia Company) supplied with bedding (saw dust) and nesting (a handful of shredded paper) material. Cages were cleaned once a week in which rats were re-housed in clean cages with new bedding and nesting material. The room was maintained under a 12:12 h light:dark schedule with the white light on between 02:00 and 14:00 h and continuous dim red light (two 60 watt bulbs, Serma Electrical, Cairo, Egypt) enabling observation during the dark period, at a constant temperature (20 ± 1°C).
Following acclimatization, rats were arbitrarily allocated to one of the following two conditions for seven consecutive weeks:
‘Enriched cages’ (EC): Standard polypropylene cages (48.5 cm length [L] × 33 cm width [W] × 21 cm height [H]) that were supplied with retreats (20.5 cm L × 15.7 cm W × 11.5 cm H; Guinea pig huts, red-tinted, Lillico, Surrey, UK), nylabone (regular size, original flavour [36 g], Lillico), crawl ball (115 mm, with 3 × 58 mm holes, red-tinted polycarbonate, Lillico), ladders (9-step wooden ladder 35.5 cm, local pet store, The Bird Planet, Kafr El-Sheikh, Egypt) and nestlets (5 cm × 5 cm sterilized cotton fibre pads, Lillico). ‘Unenriched cages’ (UC): Standard polypropylene cages (48.5 cm L × 33 cm W × 21 cm H) that were not supplied with any additional cage structures.
Data collection
Behaviour
Sampled behavioural categories (see ethogram, Table 1) for all individual rats were scanned by one experienced observer. The observer entered the experimental room 10 min before the scheduled start of the observation to allow the rats to habituate to his presence. Observations were carried out every week in two sessions per day (representing one observation week) for the two housing conditions. The first session took place during the light phase (white light was on); starting at 12:30 h and ending at 13:30 h. The second session was carried out while the white light was off (during the dark phase of the day); starting at 14:00 h and ending at 15:00 h.
Ethogram for recorded behaviours
The behaviour of the rats in each of the 12 cages was recorded in realtime using an instantaneous sampling method with 4 s intervals between each consecutive focal animal. Each sample interval was prompted by an audio cue via headphones, and the behaviour recorded onto a check sheet. Each session therefore yielded 75 scans per rat. This meant a total of 150 scans per rat per day (observation week), and a total of 900 scans per rat over the entire experimental period (6 observation weeks). The behaviour of each individual rat was sampled and its position within the cage (underneath food hopper or in the open part of the cage) and state (contacting or away from enrichment) were also recorded.
EPM test
At the seventh week and after behavioural observations were completed, a 5 min EPM test was conducted for each animal from the two housing conditions. The EPM test is widely used in pharmacological research to analyse the level of anxiety in laboratory rodents, and is based on the natural conflict between the tendency of the animal to explore a novel environment and the aversive properties of a brightly lit open area. 35–37 The maze had two open arms and two closed arms (115 × 10 cm). The closed arms had 50 cm high walls. The plus-maze was elevated 100 cm above the floor. The maze was arranged in a manner such that arms of the same type were opposite each other, connected by a central area (15 cm × 15 cm). In order to keep the rats from falling, the open arms were surrounded by a 0.5 cm high edge. All rats were tested individually in the light phase of the light/dark cycle on the same day between 09:00 and 12:00 h. The order of testing was counterbalanced between the two housing conditions to control for possible effects of time of the day on behaviour. Each rat was placed in the middle of the apparatus with its head facing an open arm, and its behaviour was video recorded for 5 min. 40 The arms of the plus-maze were wiped with ethyl alcohol (Pharma One, Cairo, Egypt) after each individual rat was tested. The total numbers and durations of entries into closed and open arms, latency to the first entry into closed and open arms (seconds), frequency of rearing and grooming behaviour and the number of head dips were recorded. Analysis was performed by an experienced observer who was unaware of which housing conditions each animal belonged to.
Performance and pathology
Throughout the six-week observation period rats were weighed weekly. Rats were picked from their cage and weighed using equilibrated scales (Sartorius, AG, Gottingen, Germany). At the end of the seventh week of the housing period rats were rendered unconscious using chloroform, and were euthanized by cervical dislocation. Immediately after euthanasia, the weight (g) of each individual rat was recorded using a digital scale (Oertling, OB033, UK). Each rat was then dissected and selected internal organs, including the thymus gland, spleen and adrenal glands, were removed and stored on ice in sterile balanced salt solution. They were subsequently dried, trimmed and weighed (mg).
Statistical analyses
Behavioural and performance data
We used a repeated-measures general linear model (GLM) with week (weeks 1–6) and session (sessions 1–2) as within-subject factors because the behavioural (ethogram) and performance data (body weight and weight gain) had been collected from the same cages at two different time points every week. Treatment (EC and UC) was included as a between-subjects factor. SPSS (version 12.0 for windows) was used for all statistical analyses. The average percentage of scans spent in performing each behaviour was calculated by dividing the total number of scans for each behaviour variable by the total number of scans for each individual rat in each session (75 scans), and each figure was then multiplied by 100.
The relative weight gain (%) was determined by dividing the value of the absolute weight gain (difference between the body weight measure in a particular week and the body weight measure in the previous week) by the value of the body weight in the previous week, and then the resultant figure was multiplied by 100. Data were checked for normality and homogeneity of variances to test for the suitability of using parametric tests. Performance data showed normality whereas behavioural data showed normality after square root transformation. All data are presented as estimated marginal means (EMM) ± SE.
EPM and pathology data
Data met the assumptions of parametric statistics (normality, homogeneity of variance, linearity). Relative durations of time spent in open (open/total × 100) and closed arms (close/total × 100), and latency to the first entry to open and closed arms were determined for each housing condition. Relative frequency of entries into open (entries to open arms/total arm entries × 100) and closed (entries to closed arms/total arm entries × 100) arms, and frequency of rearing and grooming behaviours and head dip were also recorded for each group. The organ weights were expressed as a ratio of the body weight (relative weight for each organ). Differences between the rats of the two housing conditions in behaviours of the EPM test, final body weight and the relative weight of internal organs were tested using an independent t-test.
Results
Behaviour
Main effects
Several behaviours showed an effect of housing condition, average percentage scan: sleep (F 1,21 = 6.81, P < 0.05); moving (F 1,21 = 9.62, P < 0.05); stationary (F 1,21 = 38.64, P < 0.001) (Figure 1); bedding-directed behaviour (F 1,21 = 26.24, P < 0.001) (Figure 2); under hopper (F 1,21 = 691.57, P < 0.001) and in-the-cage (F 1,21 = 691.57, P < 0.001) (Figure 3). The values of sleep behaviour, movement activities and being in-the-cage were higher in the EC, whereas those of stationary, bedding-directed behaviour and being under hopper were higher in the UC.
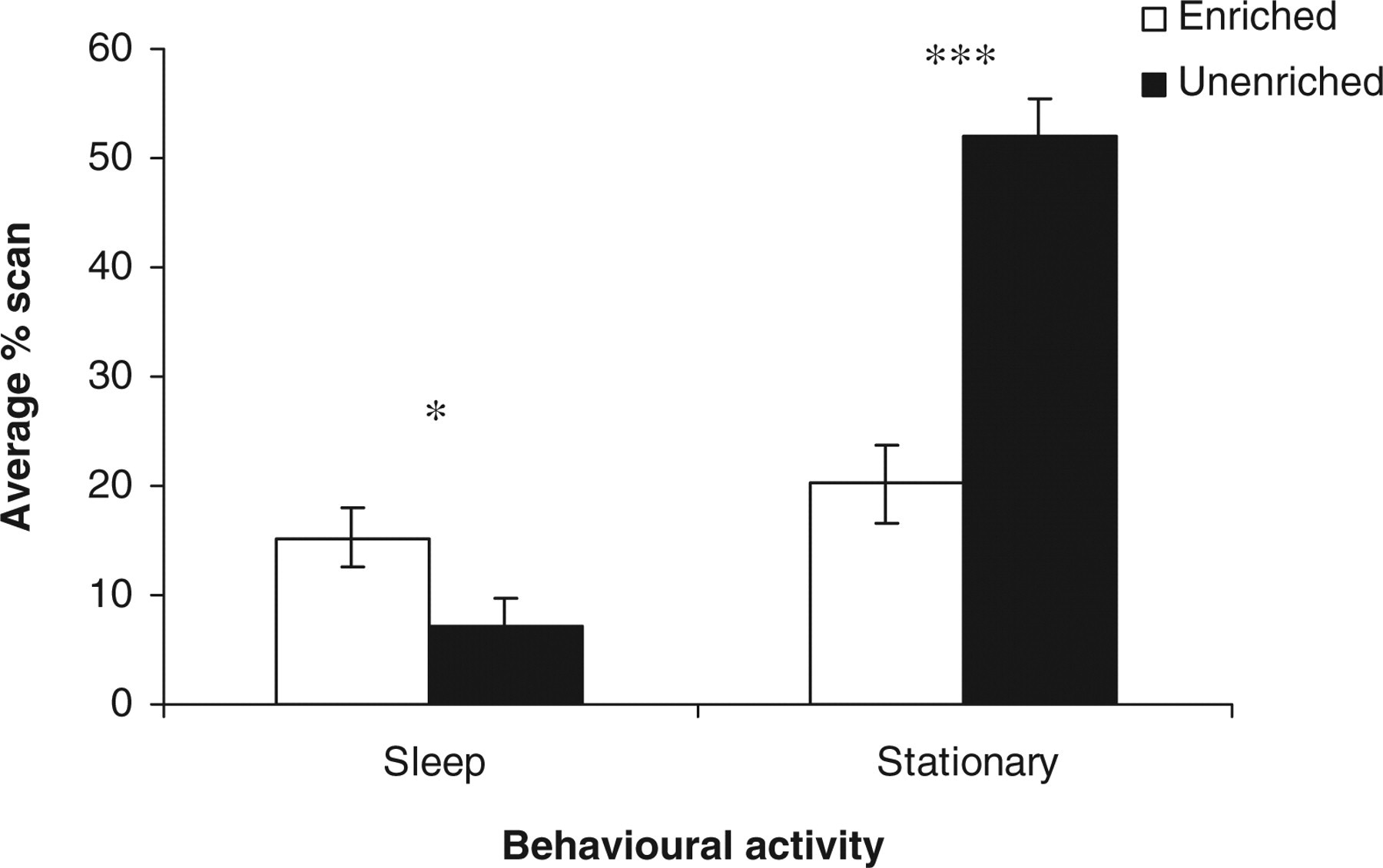
Estimated marginal means ± SE ‘average percentage scan sleep and stationary’ by the rats in the two housing conditions. *P < 0.05, ***P < 0.001
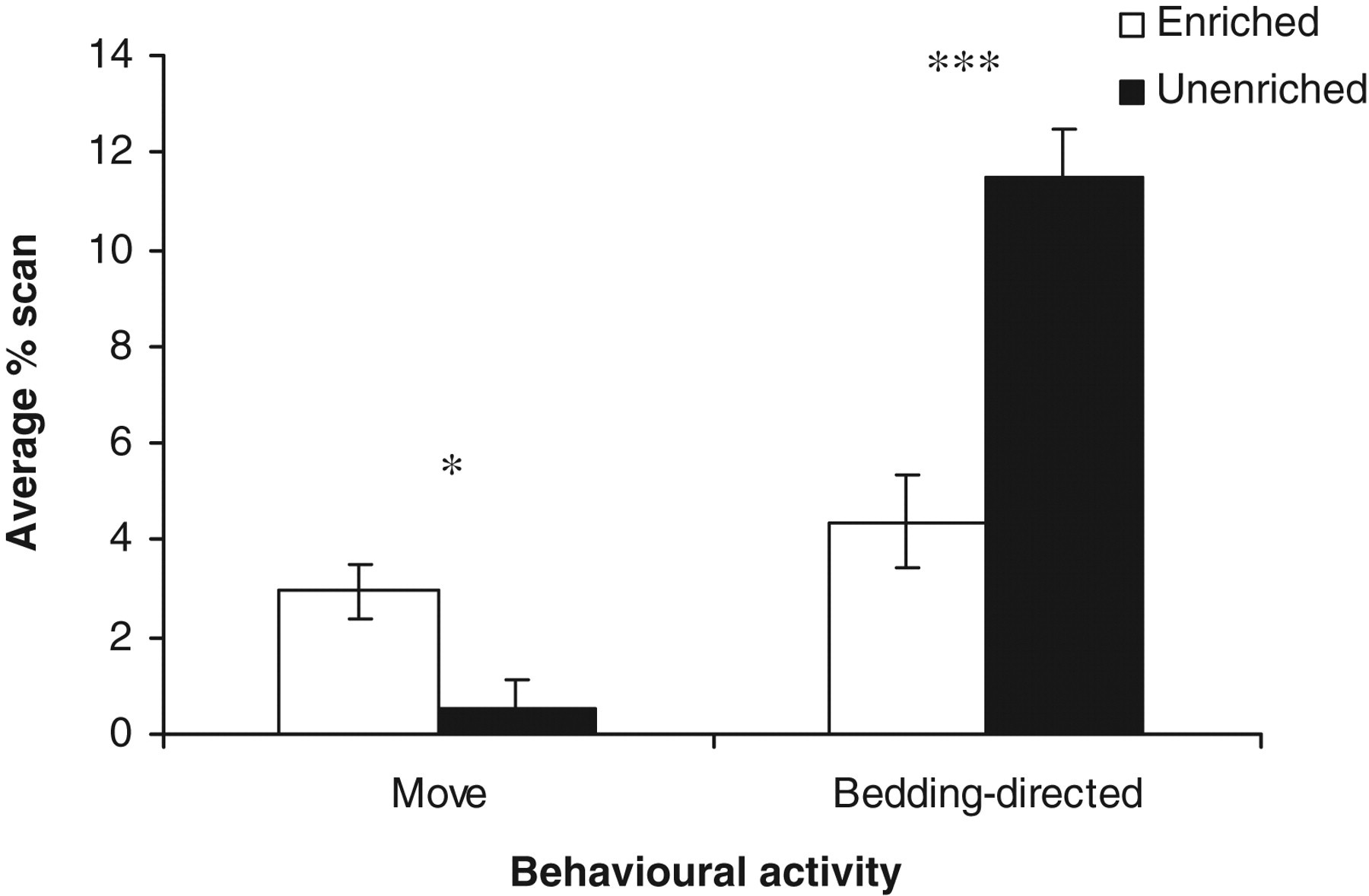
Estimated marginal means ± SE ‘average percentage scan moving and bedding-directed behaviour’ by the rats in the two housing conditions. *P < 0.05, ***P < 0.001
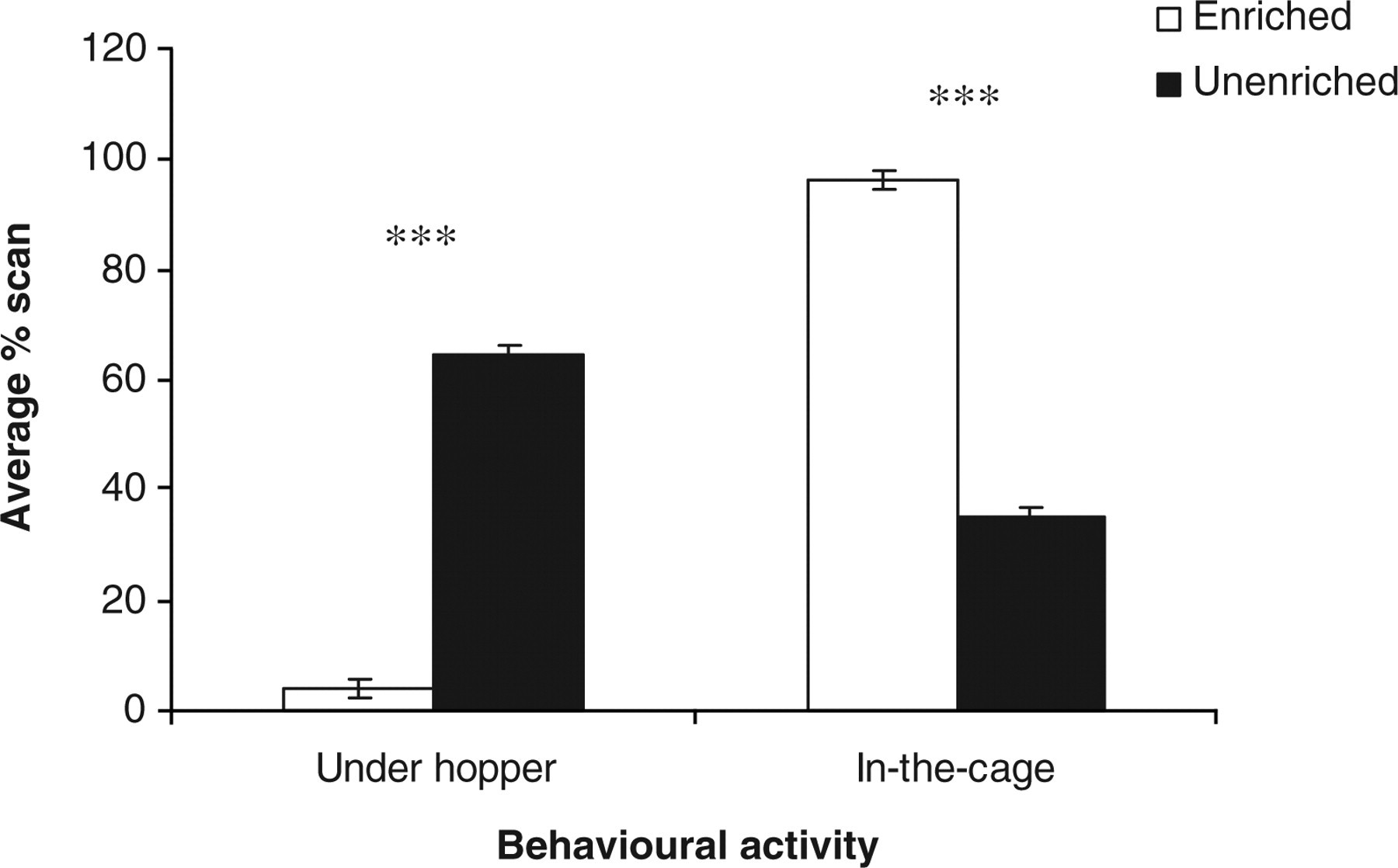
Estimated marginal means ± SE ‘average percentage scan under hopper and in-the-cage’ by the rats in the two housing conditions in the two observation sessions. ***P < 0.001
Interactions
Average percentage scan non-intake maintenance behaviour showed a significant treatment × session (F 1,21 = 5.41, P < 0.05), increasing significantly in the light phase in the EC; and both average percentage scan feeding (F 1,21 = 5.37, P < 0.05) and exploration (F 1,21 = 5.43, P < 0.01), increasing significantly in the dark phase in the EC (Figure 4).
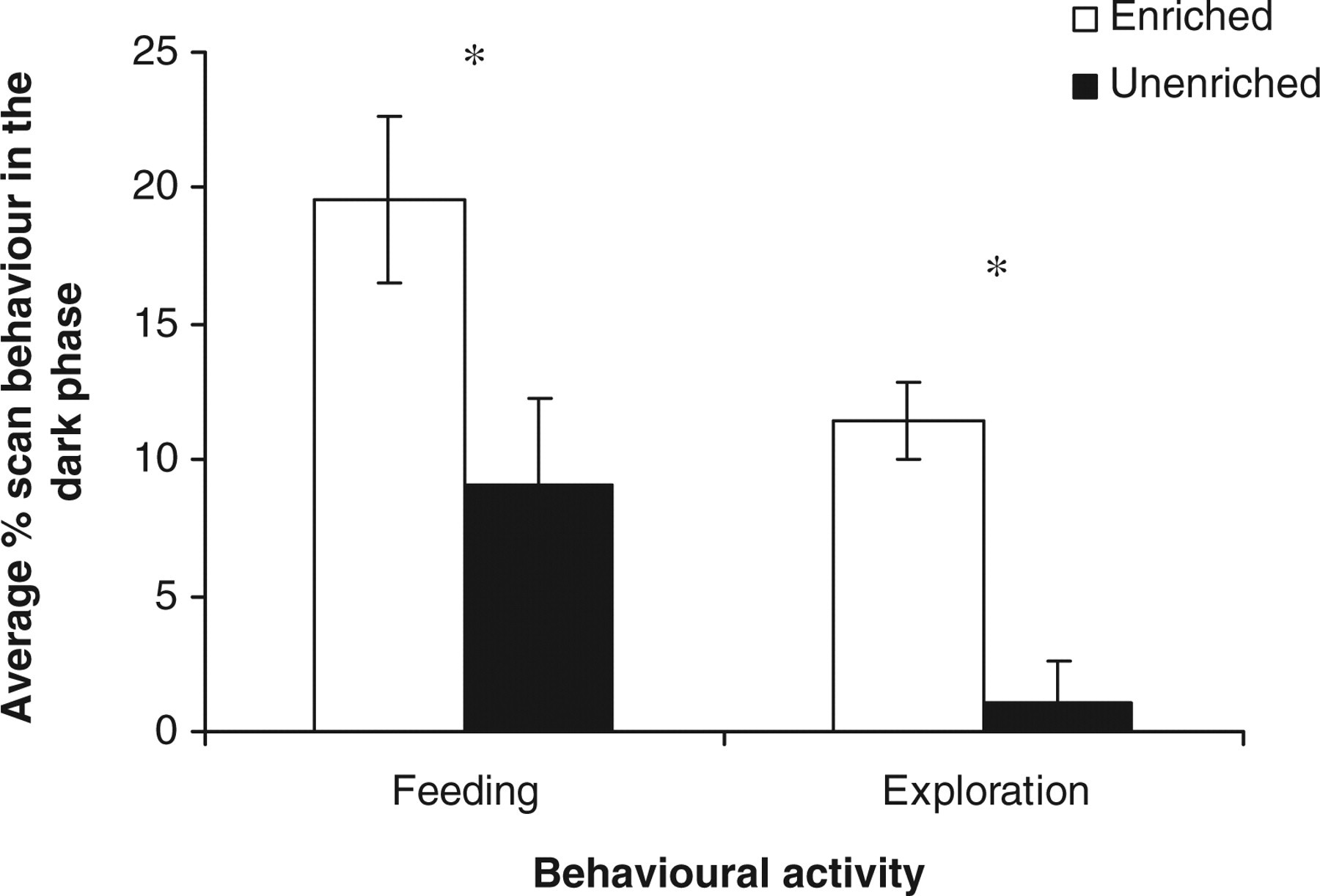
Estimated marginal means ± SE ‘average percentage scan feeding and exploration’ by the rats in the two housing conditions in the dark phase of the light cycle. *P < 0.05
Elevated plus-maze
Housing rats in EC versus UC had a significant effect on their behaviours in the EPM including: relative time spent in open arms (seconds) (t 22 = 3.71, P < 0.001); relative time spent in closed arms (seconds) (t 22 = −3.71, P < 0.001) (Figure 5); relative open arm entry (t 22 = 3.45, P < 0.001); relative closed arm entry (t 22 = −3.45, P < 0.001) (Figure 6); and latency (seconds) to first entry to open (t 22 = −2.78, P < 0.01) and closed arm (t 22 = 5.99, P < 0.001) (Figure 7). There was however no significant effect of the housing conditions on the frequency of both rearing (t 22 = 0.23, NS) and head dip (t 22 = 0.54, NS) in the EPM.
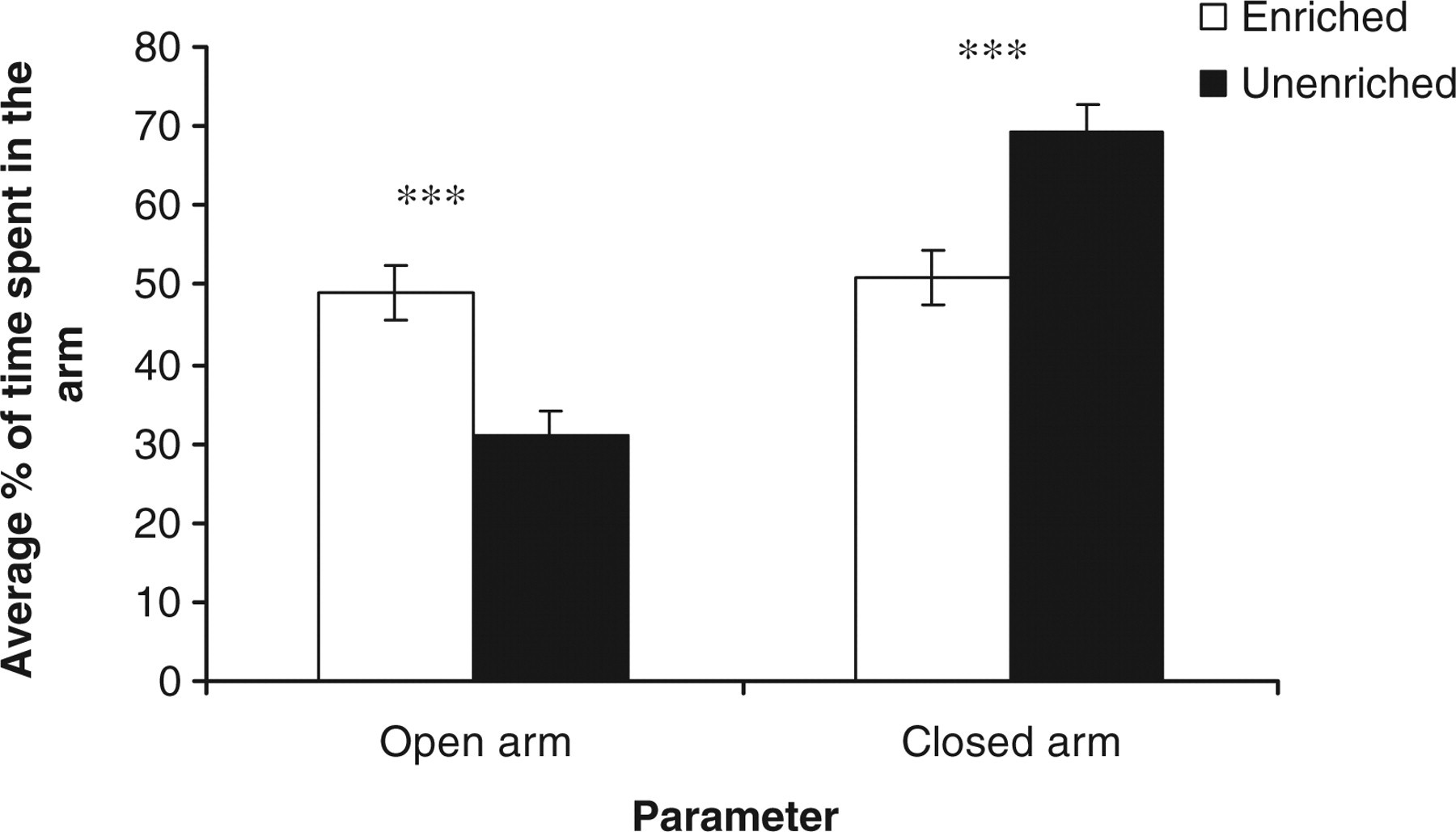
Estimated marginal means ± SE ‘average relative time spent in the open and closed arms of the elevated plus maze’ by the rats in the two housing conditions. ***P < 0.001
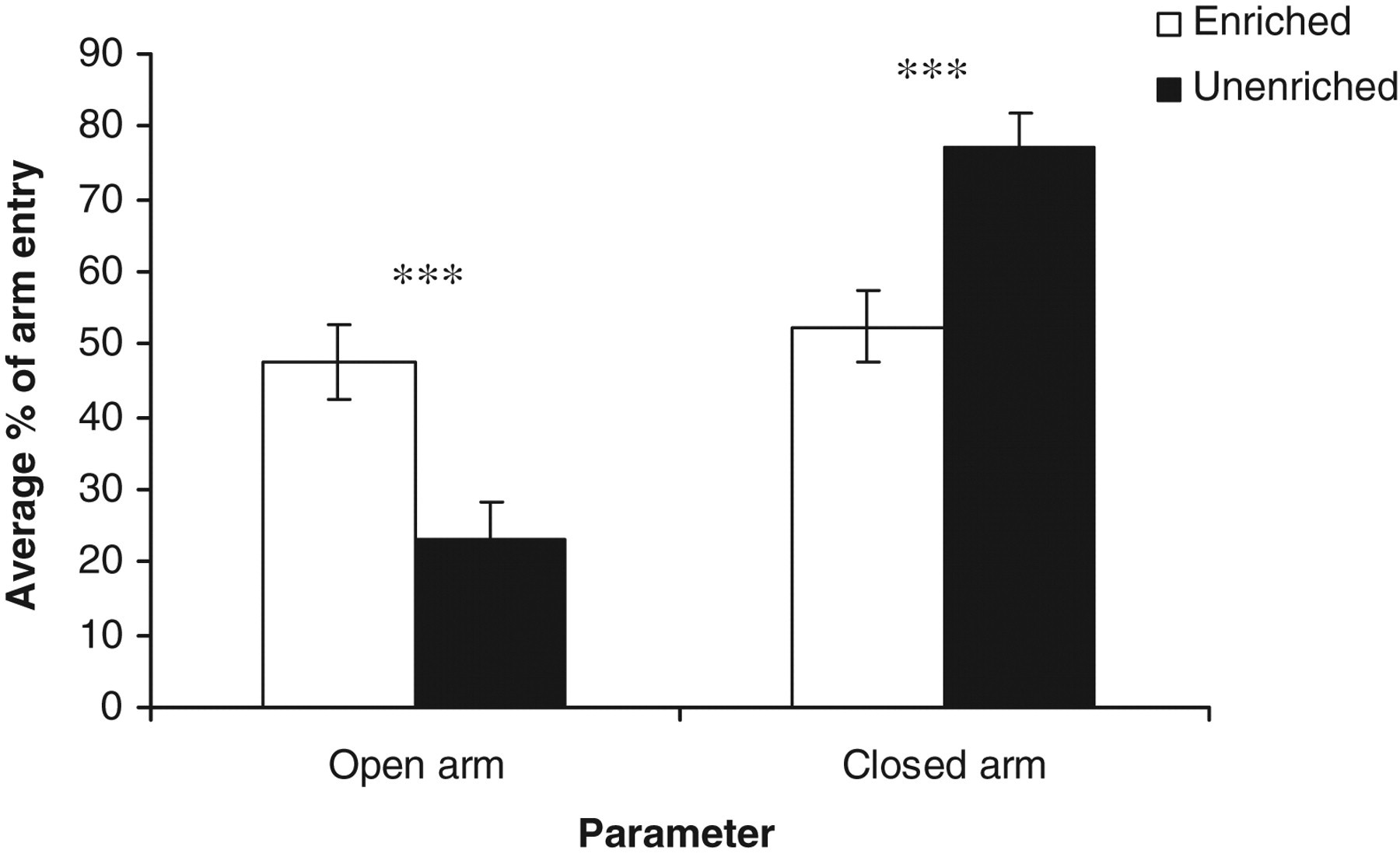
Estimated marginal means ± SE ‘average relative entry to open and closed arms of the elevated plus maze’ by the rats in the two housing conditions. ***P < 0.001
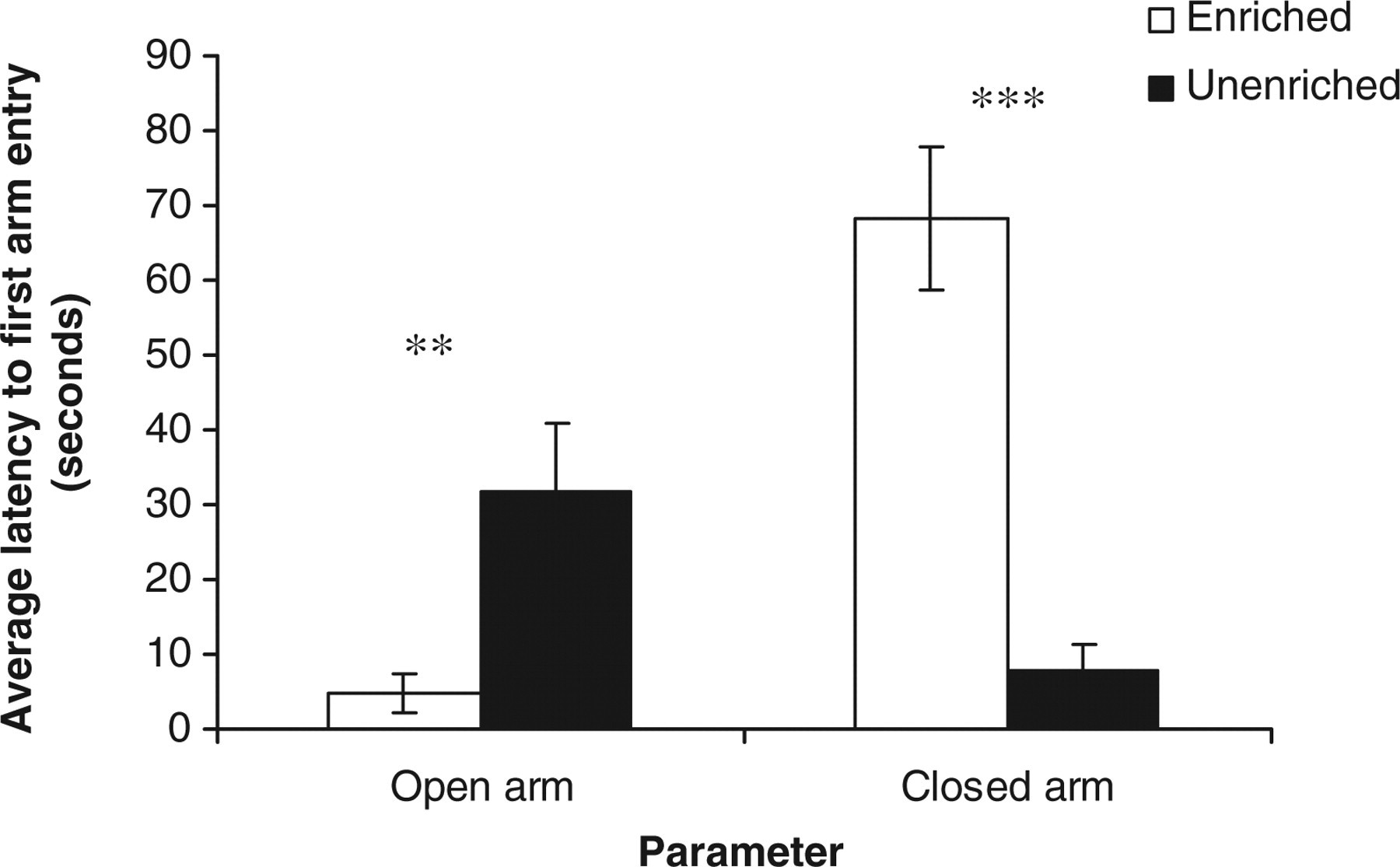
Estimated marginal means ± SE ‘average latency to first entry to open and closed arms of the elevated plus maze’ by the rats in the two housing conditions. **P < 0.01, ***P < 0.001
Performance and pathology
The output of the repeated-measures GLM showed that housing laboratory rats in EC versus UC significantly changed performance parameters measured in this study, including body weight (g) (F 1,21 = 111.68, P < 0.001) and weight gain (g) (F 1,21 = 25.98, P < 0.01) (Figure 8), with rats in the EC weighing heavier and gaining more weight every week than rats in the UC.
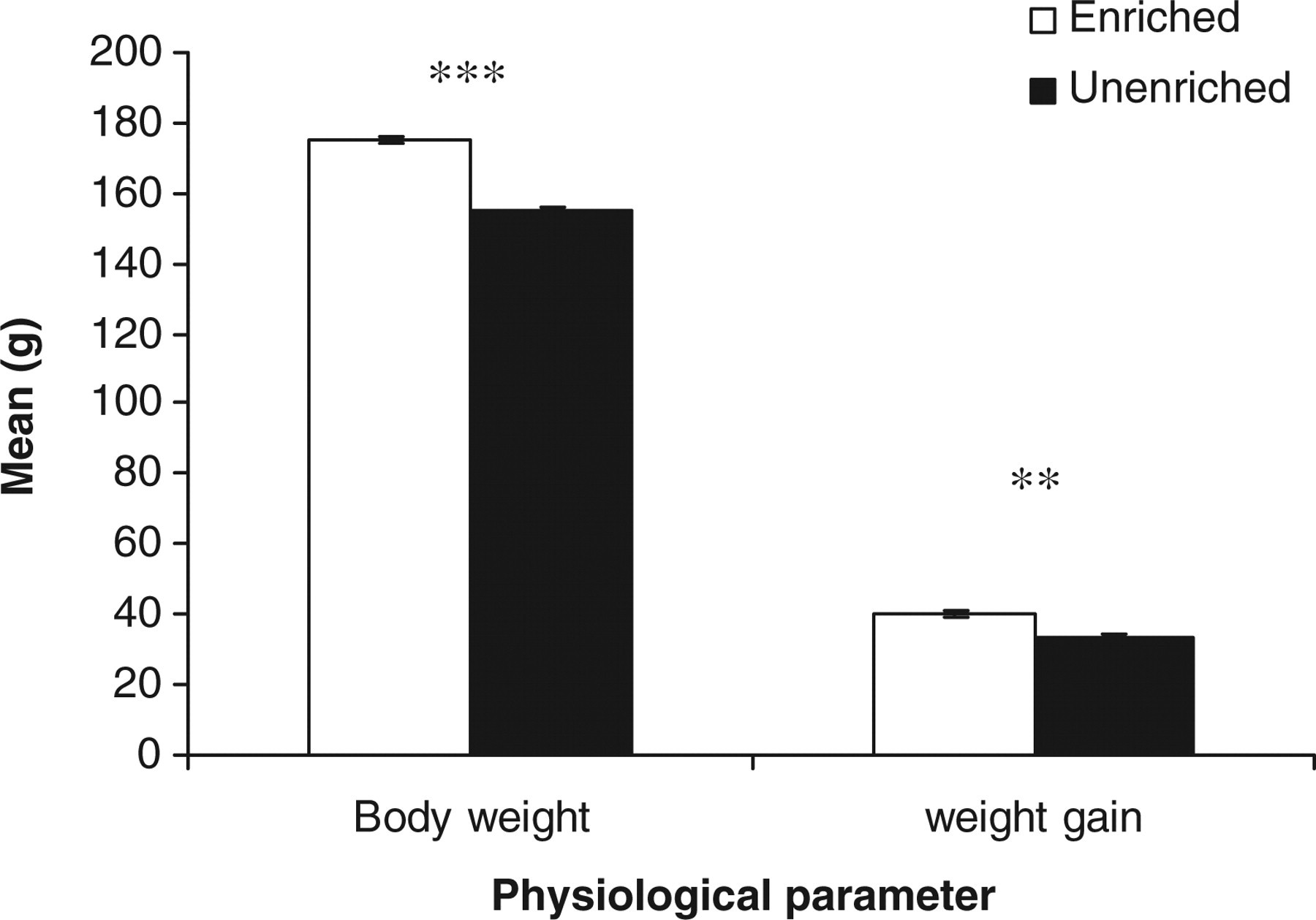
Estimated marginal means ± SE ‘average body weight and weight gain (g)’ by the rats in the two housing conditions. **P < 0.01, ***P < 0.001
Similarly, housing rats in EC versus UC had a significant effect on the pathological measures recorded in this study including: final weight (g) (t 22 = 5.20, P < 0.001); relative adrenal weight (g) (t 22 = −3.14, P < 0.05); relative thymus weight (g) (t 22 = 3.50, P < 0.01); relative spleen weight (g) (t 22 = 3.41, P < 0.05), with the rats housed in the EC weighing more, having heavier thymus and spleen but lighter adrenal than rats housed in the UC (Figure 9).
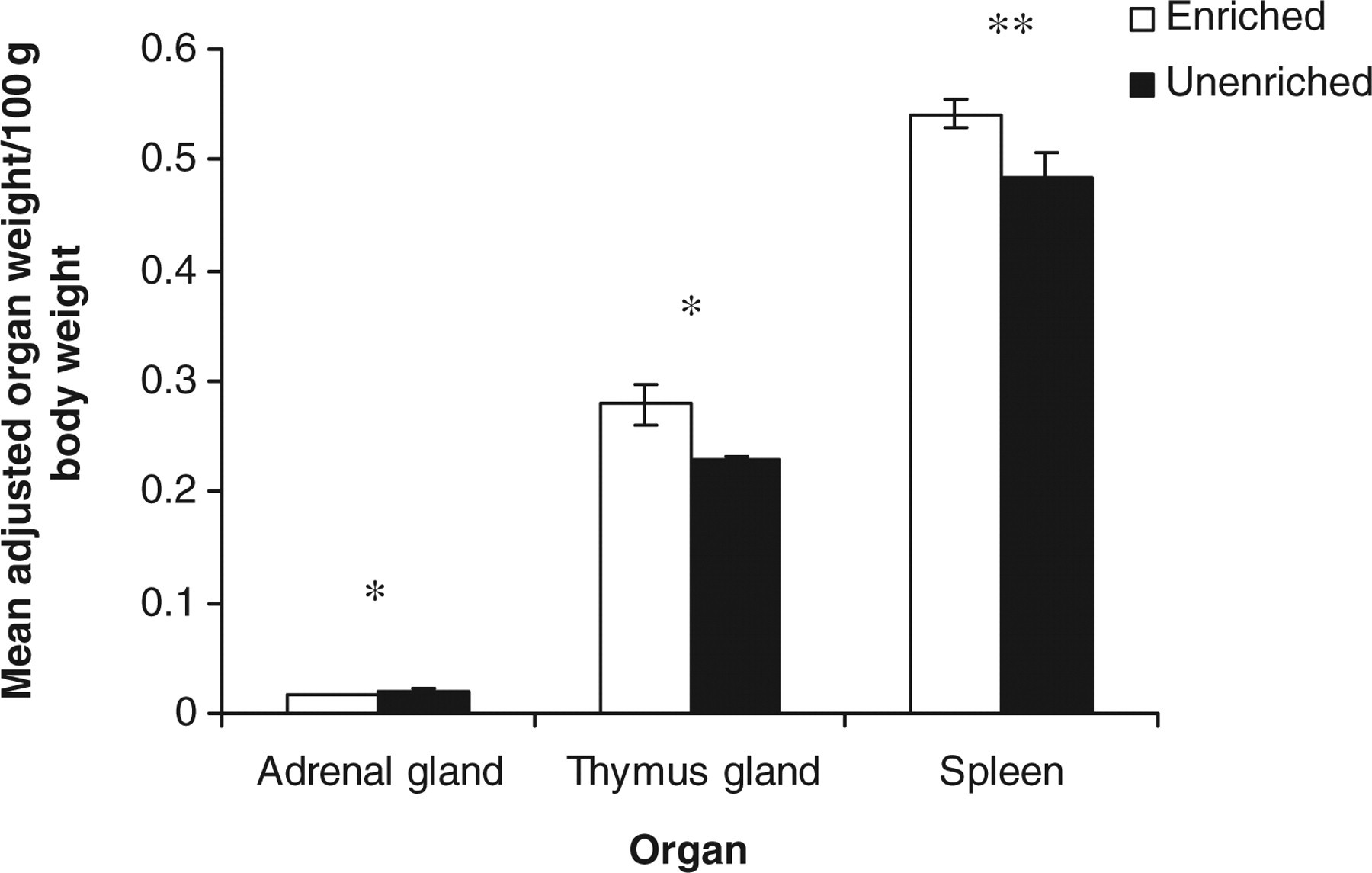
Estimated marginal means ± SE ‘average relative adrenal, thymus and spleen weights (g)’ by the rats in the two housing conditions. *P < 0.05, **P < 0.01
Discussion
Our results demonstrated clear differences between rats in the different housing conditions. Rats housed in the enriched conditions displayed higher levels of sleep, movement activities, exploration and both intake (feeding) and non-intake maintenance (self-grooming) behaviours and lower levels of stationary and bedding-directed activity compared with rats in the unenriched conditions.
High levels of sleep behaviour may indicate good welfare in laboratory rats. 41 Research on laboratory rats has shown that chronic stress can affect both sleep quantity and quality. Typically, in a chronic stressful situation, total sleeping frequency and duration decrease, with sleep liable to have more interruptions. 42,43 It has been shown that physical stress reduces both sleep quality and quantity. 42,44 Chronic psychological stress (e.g. subordination) also appears to reduce sleep quantity. 25
This high level of sleep displayed by rats in the EC could be due to the increased level of their movement and exploration but also due to the increased activity directed towards the enrichment objects. It could also be due to the ability of rats in the EC to control their environment by avoiding the disruptive effect of the white light. It has been shown that unavoidable light constitutes a stressful condition for a nocturnal animal (laboratory rats) and that it results in a marked decrement of both types of sleep (rapid eye movement sleep and short wave sleep). 45 The provision of multiple physical structures to the cage might have allowed the rats to use some of these structures (such as the retreat and crawl ball) to hide from the direct effect of the white light.
Laboratory rats are well known to be thigmotactic (edge-users), preferring to spend most of their resting and sleeping time in contact with the surrounding walls of their environment. 46,47 Adding multiple physical structures to the conventional cages might have increased walls and edges in the cage, thereby improving the rats' ability to display more natural behaviour such as sleep. Similar findings of increased sleep behaviour in rats housed in enriched laboratory cages, but in groups, have been previously reported. 48–50
Rats in the EC also displayed higher levels of exploration and movement activities compared with rats in the UC. Research has shown that chronic stress decreases general activity levels and locomotor behaviour 51–53 and exploration. 25,54 These higher levels of movement and exploration by the enriched-housed rats could be due to the increased complexity of their environment. It has been illustrated that, when given the choice, rats prefer high complexity in their environment and that they spend more time active (moving and exploring) in the complex environment. 26 This finding of increased levels of movement and exploration by the rats in the EC agree with those reported previously. 28,49,55,56
Rats in the EC displayed higher levels of both intake and non-intake maintenance behaviour as compared with rats in the UC. Research has reported an inhibition or a reduction in self-grooming after chronic stress; a repeated social defeat, 53 chronic stress by anxiogenic drugs, 57 chronic psychological stress (predatory stress) 58 and also in subordinate animals after long period of grouping. 59 Similarly, a reduction in food intake has been found after chronic stress. 52,60,61 This high level of self-grooming activity in the enriched-housed rats may be due to the higher amount of sleep in these animals. Self-grooming was reported to be the second activity of the laboratory rat that occupies the longest duration of their time budget after sleep. Indeed, it is the most time-consuming activity of the laboratory rat's awake time. 62,63 Self-grooming was reported to take place after sleeping, but also occurs when an animal prepares for sleep. However, the high level of feeding displayed by the enriched-housed rats could be due to the higher activity levels performed by these animals.
Rats in the EC exhibited lower levels of bedding-directed behaviours than rats housed in the UC. This relative increase in the level of bedding-directed behaviours in the UC could be due to the fact that rats in these cages did not have enough cage structures (objects) to interact with. The only available cage structure in these cages was the bedding substrate, therefore these conventional cages limit the available options of the rats for interaction. The finding of reduced bedding-directed behaviours in groups of rats housed in enriched cages is similar to that reported in previous experiments. 49
The finding that rats in the EC were present more frequently in the open part of the cage and less frequently underneath the hopper compared with rats in the UC could be due to the increased compartmentalization of the EC by the provision of multiple physical structures into them. This might have provided various resources for the rats to hide from the disruptive effect of the white light, particularly in the light phase of the dark/light cycle, and intensified their thigmotactic nature. This might, in turn, have improved the ability of these animals to exert better control over their environments compared with their counterparts in the UC. Good ability of animals to cope with, and to control, their environment is a necessary requirement for good welfare. 6
Our results showed that rats experiencing the EC explored the open arms of the maze for a longer time and the closed arms of the maze for a shorter time as compared with those experiencing the UC. The EC rats also entered the open arms more frequently and the closed arms less frequently, and showed short latency to open arm entry as compared with the UC rats. Taken together, the results of the EPM indicate that increasing the extent of enrichment of conventional cages of laboratory rats appear to decrease the level of anxiety they experience.
Similarly, it has been demonstrated that rats kept in enriched housing conditions were less fearful, adapted quickly and more adequately with a changing environment (entered more squares in open field tests and showed lowest emergence latencies), and had a better problem-solving ability (made fewer total and repeated errors) compared with conventionally-housed rats. 33 Interestingly, these differences in emotionality between enriched and standard housed laboratory rats have been known for a long time. It has been reported that rats housed individually in enriched cages (supplied with various structures), but able to see one another through the exposed sides of the cages, showed less behavioural indices of emotionality in the open field test (scored higher in ambulation and rearing) as compared with unenriched-housed littermates. 32
In accordance with our results, it has been argued that laboratory rats housed in conventional laboratory cages are ethologically, physiologically and psychologically aberrant and cannot be considered as normal animals. 64 More importantly, reduced external validity of the research and therefore the benefit gained from the research has been shown to arise when laboratory rodents are housed in standard laboratory cages. 9
Single housing of laboratory rat has been shown to be stressful. 23,65 Experiments have demonstrated that individual housing enhances anxiety-like behaviour. 66 However, there are also data that have indicated that individual housing per se did not increase the anxiety-like behaviour. 67 Thus, simply, individual housing per se of laboratory rat may not be stressful 68,69 but housing them in standard laboratory cages for long term may be stressful.
Our results showed that rats in the EC had higher weights and weight gains than rats in the UC. Moreover, the EC rats had higher relative weights of spleens, thymuses and lower relative weights of adrenal glands as compared with the UC rats. The increased weights and weight gains in the EC rats could be due to their increased feeding, but could also be due to their increased sleep behaviour. One of the many theories that have been proposed for the function of sleep is the protective theory that is: the function of sleep is to protect the organism from excessive wear and tear. 70,71 This finding indicates that long-term housing of juvenile laboratory rats in unfurnished laboratory cages appears to decrease their ability to cope with the environment. Body weight and weight gain have been reported to decrease after chronic physical 60 and social 53,59 stress.
An increase in both body weight and weight gain has been demonstrated in both rats and mice housed in groups in enriched conditions as compared with their non-enriched conspecifics. 13,72,73 Whereas other studies reported a reduction either in body weight, 74 weight gain 50 or in both weight criteria 75 in enriched-housed rats. It is however interesting to note that this reduction in either body weight, weight gain or both in the rats housed under enriched conditions was due to the fact that these animals consumed much less food compared with those housed in unenriched conditions.
In accordance with the direction of the behavioural, psychological and performance data, the findings of the changes in the weights of the internal organs could also indicate that long-term housing of rats in the UC appeared to decrease their ability to cope with and to exert control over the environment. Rats housed in EC had lower adrenal weights and higher weights of both the spleen and thymus than their counterparts housed in UC. The findings of decreased adrenal weights and increased weights of the spleen and thymus in the rats that experienced the enriched housing conditions are similar to those reported in a previous work on the effects of environmental enrichment on laboratory mice housed in groups. 13
Under the conditions of this study, the type of cage modification implemented in this study was found to be affordable, practical to use, clean and easy to replace, did not compromise the physical health of the rats, nor did it prevent ease of checking the animals. The modification regime provided all the required physical features of enrichment items suggested by previous studies. 27,76–78 Importantly, this particular type of cage modification provided the rats with ample opportunities to cope with and to exert control over their environment; characteristics that resulted in improved welfare in the animals experiencing it. It has been suggested that for an efficient environmental enrichment programme to improve the welfare of the animals experiencing it, the enrichment should enhance the expression of desirable behaviours such as species-specific behaviours and decrease undesirable behaviours such as abnormal behaviour, or do both. 56,78,79 As, adding some physical structures to the laboratory cages should not be considered enrichment until it produces good long-lasting changes in welfare, 80 the regime used in this study appeared to have met this requirement and can therefore be called enriching.
In summary, long-term single housing of rats in super-enriched cages increased levels of indicators of good welfare and decreased levels of indicators of poor welfare. The findings of this experiment showed that laboratory rats housed in enriched cages demonstrated improved welfare and better ability to cope with their environment compared with those animals housed in conventional laboratory cages. The results, more importantly, demonstrated that when single housing of laboratory rats is necessitated their laboratory cages should be enriched with multiple physical structures in order to improve their welfare. These findings thus strongly support the need of the current conventional housing systems of laboratory rats, particularly singly-housed rats, for re-evaluation to help provide a better environment for the animals that can in turn result in an improvement in their welfare.
Footnotes
ACKNOWLEDGEMENT
The authors would like to thank the Department of Animal Husbandry, Faculty of Veterinary Medicine, Mansoura University for funding the study and the two anonymous referees for their constructive comments on the manuscript.
