Abstract
This study was carried out to investigate how pinworm infection in rats affects nutrient digestibility in the hosts. Twenty-four male outbred Wistar rats were randomly divided into two groups of 12 rats each. The rats from the first group (GI) were kept in cages with bedding containing pinworm eggs, and the second (control) group (GII) were kept in a separate room in clean, uncontaminated filter-top cages. The animals were put into individual metabolic cages later. Metabolic trials lasted five days and records of animal weight, food ingestion, and faecal weight were taken daily. Based on laboratory analysis of the feed and faecal nutrient content, digestibility values were determined. On day 15 of the experiment, the animals were euthanized. Although Syphacia muris were found in all rats from the GI group, animals exhibited no clinical signs. In our experiment, S. muris infection reduced the overall digestibility of all measured nutrients (P < 0.01). The most significant differences in digestibility were observed in the case of crude fibre and mineral matter (P < 0.01).
Rats are the world’s most frequently used laboratory animals. They are used for scientific as well as for a variety of commercial purposes. They are easy to keep, and their breeding technology is very sophisticated. Nevertheless, rats suffer from many parasitic diseases, which can influence experimental results.
One of the most common parasites found in breeding rats is the pinworm. Due to their biology, direct development, short embryonic period, and incidence of autoinfection, pinworms are a very prolific group of parasites. In laboratory rats, the most commonly found pinworm species are Syphacia muris and Aspiculuris tetraptera.
Although pinworm parasites of laboratory rodents are considered to be relatively non-pathogenic and infections are generally regarded as symptomless, there have been reports of laboratory rodents with a variety of conditions such as intestinal impaction, intestinal intussusception, mucoid enteritis, necrosis in all layers of the intestinal wall, and rectal prolapse. These conditions are thought to be associated with heavy pinworm burdens.1–6
One area in which rats are often utilized is feed testing. Rats are most commonly used in experiments designed to test the digestibility of individual nutrients in various feeds. However, it is very difficult to keep rats free of pinworm infection,7,8 and therefore, many of these experiments are conducted with infected animals.
The aim of this study is to investigate how pinworm infection affects nutrient digestibility. We monitored feed intake, nutrient digestibility and feed conversion in laboratory rats with S. muris infection.
Materials and methods
Animals and procedure
Twenty-four male outbred Wistar rats were obtained from the specific pathogen-free (SPF) rat colony of Charles River Laboratories, Sulzfeld, Germany. The animals had an average weight of 89 g (4 weeks old). They were randomly divided into two groups of 12 rats each. The rats from the first (experimental) group (GI) were kept in the cages with bedding containing pinworm eggs for 10 days. Contamination was achieved by adding bedding from the cages of S. muris infected rats to the breeding containers. After this 10-day period, Syphacia eggs in all the animals of GI were detected using a cellophane-tape test. The second (control) group (GII) was kept in a separate room in clean, uncontaminated filter-top cages for 10 days; the beddings of GII group were thoroughly sterilized.
The rooms were maintained at 22 ± 2℃ and 55 ± 5% relative humidity, with normal 12:12 h light–dark cycles and constant air circulation. All animals were monitored daily for the presence of pinworm eggs using the cellophane-tape test.
Composition of rat diet (ST-1 Bergman).
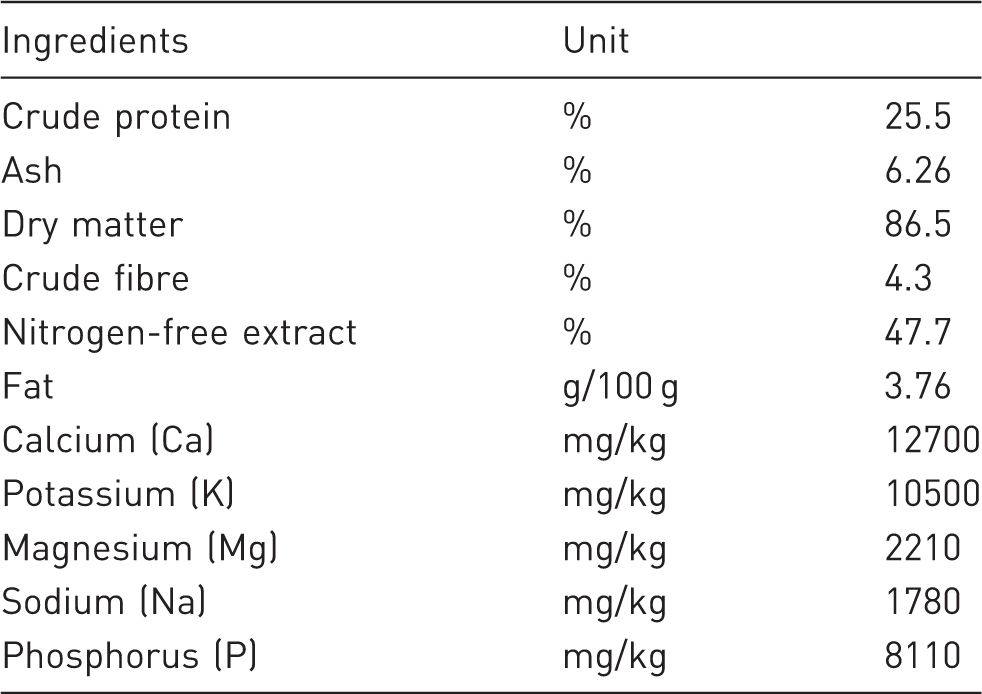
Metabolic trials lasted five days, and records of animal weight, food ingestion, and faecal weight were taken daily. Daily faecal outputs were collected in plastic bags, weighed, thoroughly mixed and stored at − 20℃ for later analysis.
From these measured values, we calculated daily weight gain, feed intake and feed conversion. Based on laboratory analysis of the nutrient content of feed and faeces, digestibility values were determined.
On day 15 of the experiment, the animals were euthanized using an intravenous administration of T-61 solution (Intervet, Boxmeer, The Netherlands). The viscera (stomach, small intestine, colon and caecum) of each rat were collected and processed for worm recovery, enumeration and identification.
All of the experimental procedures were conducted in accordance with Czech legislation (section 29 of Act No 246/ 1992 Coll., on the protection of animals against cruelty, as amended by Act No 77/2004 Coll. and Directive 2010/63/EU on the protection of animals used for scientific purposes and the guidelines and recommendations of the Federation of European Laboratory Animal Science Associations).
Laboratory analysis
The food and faecal samples were lyophilized (LP3 lyophilizer; Jouan, France) and grounded (Cyclotec 1093, Tecator, FOSS, Hillerød, Denmark) in order to pass through a one-millimetre stainless steel screen. The feed and faecal dry matter contents were determined at a temperature of 103℃ (UFB500, Memmert). The Kjeldahl method (Kjeltec 2400, Foss) was used to determine crude protein (CP) levels, and crude fibre was measured using the Henneberg and Stohmann method (Coex 106, VELP Scientifica, Usmate, Italy). Fat digestibility was analysed with the help of the Soxhlet method (SER 146/8, VELP Scientifica). This method is based on the principle of extraction with an organic solvent (petroleum ether). During this extraction the results include fat and any other substances which are readily soluble. The content of other substances, such as carotenoids, chlorophylls, cholesterol and fat-soluble vitamins, were not taken into account. Gross energy was measured using an LS10-A calorimeter (Laget, Prague, Czech Republic), and ash content (mineral matter) was determined at 550℃ (LH 15/13; LAC, Rajhrad, Czech Republic). The ash samples were incinerated, then boiled in 6 M hydrochloric acid and the insoluble residue was filtered through an ashless filter, which was dried and burned. Analyses were performed according to EC No. 156/2006 modified to a specific device.
Values of nitrogen-free extract (NFE) were calculated as follows: 100 – (% moisture + % protein + % fibre + % ash + % fat). Nutrient digestibility was defined as the difference between food intake and faecal excretion expressed as a percentage of the intake: 100 – {[(feed insoluble ash × faecal nutrient)/(faeces insoluble ash × feed nutrient) – excretion)/intake] × 100}.
Data analysis
The mean and standard deviation for each group were calculated and compared using analysis of variance (ANOVA), with the Tukey–Kramer test to evaluate differences among the groups.
Results
Number of Syphacia muris individuals in the rats of the GI and GII groups.
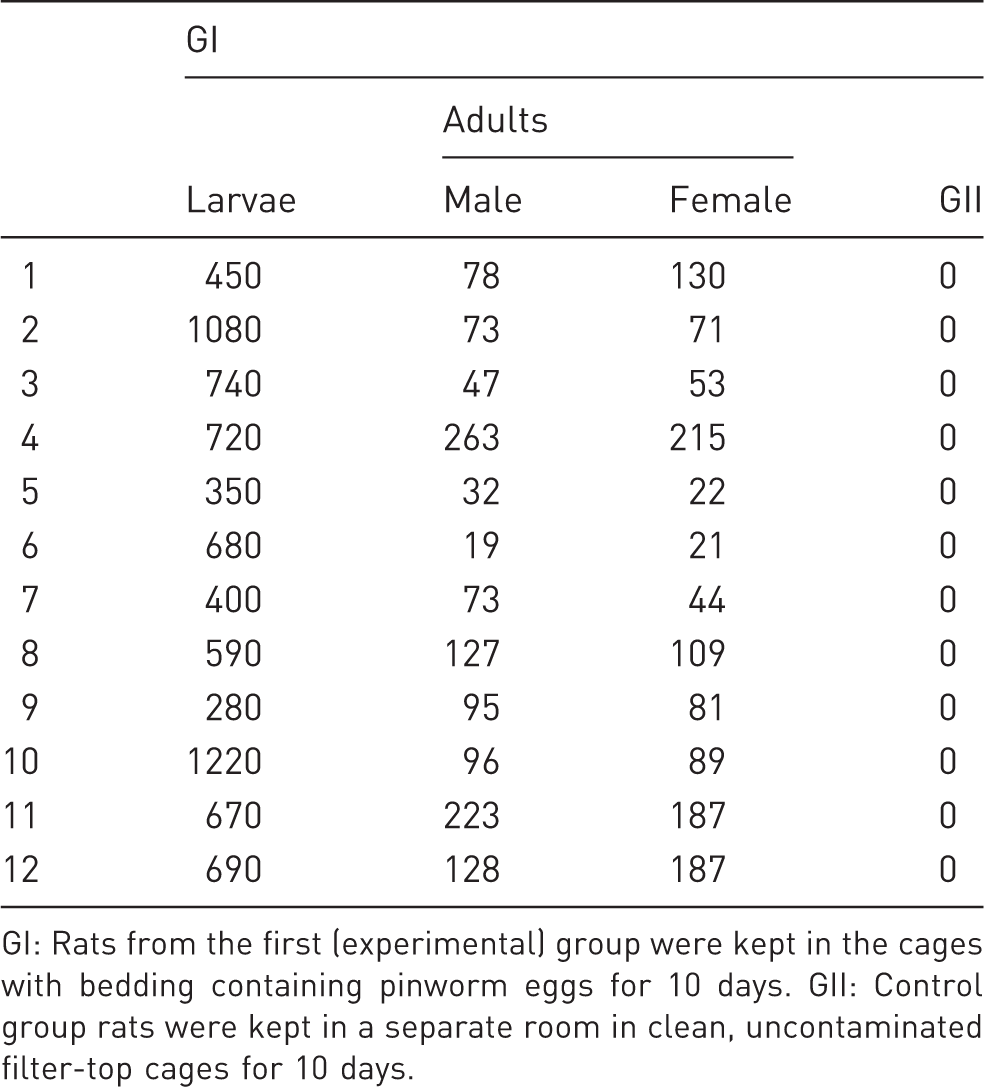
GI: Rats from the first (experimental) group were kept in the cages with bedding containing pinworm eggs for 10 days. GII: Control group rats were kept in a separate room in clean, uncontaminated filter-top cages for 10 days.
Average values of nutrient digestibility of monitored rat groups (%).
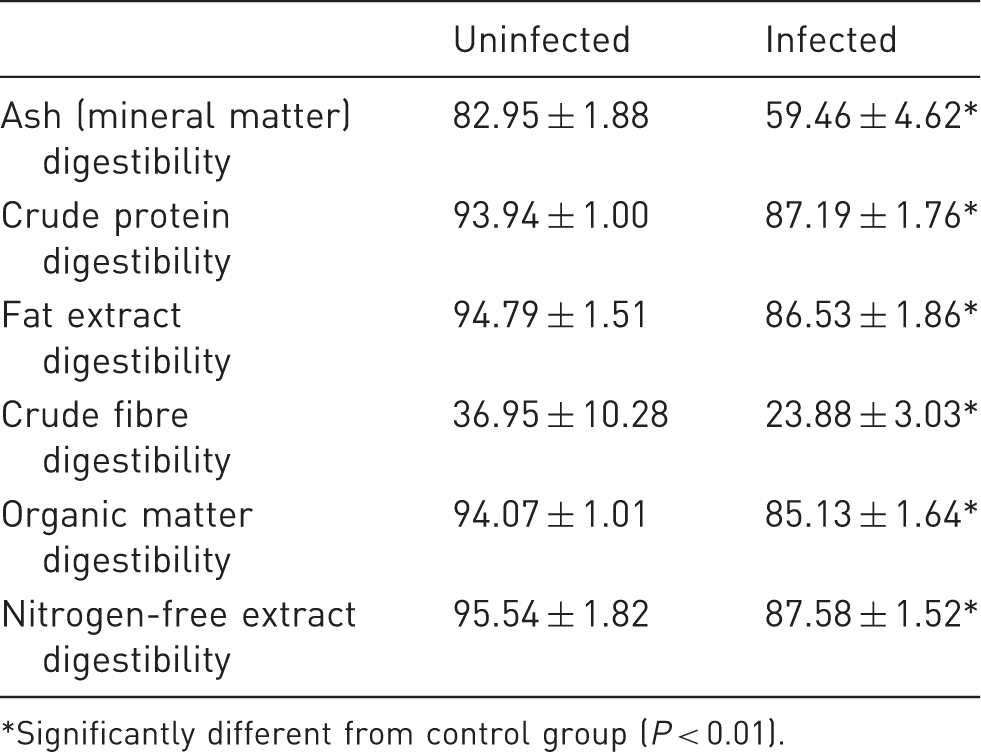
Significantly different from control group (P < 0.01).
Growth parameters of monitored rat groups.
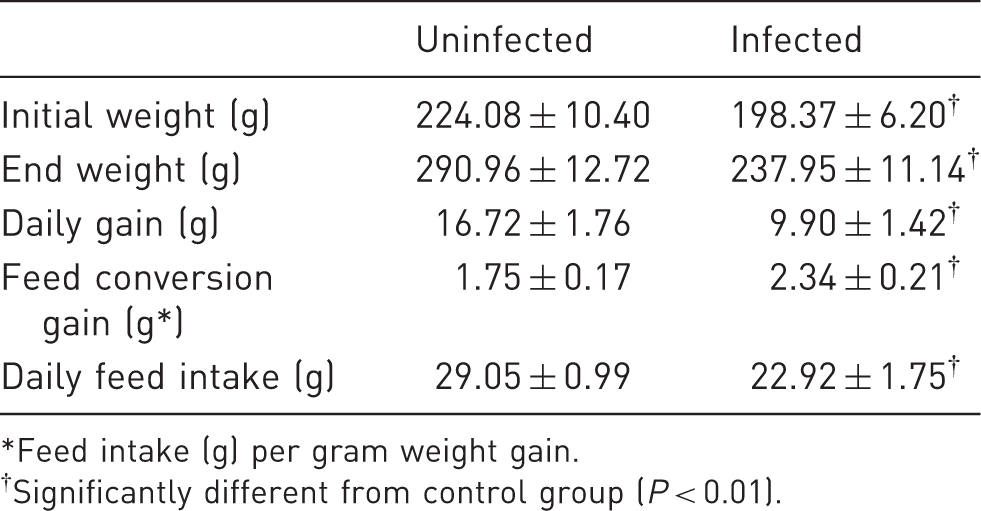
Feed intake (g) per gram weight gain. †Significantly different from control group (P < 0.01).
The most significant differences in digestibility were observed in the cases of crude fibre and mineral matter (P < 0.01). Crude fibre digestibility in the uninfected rats was very high (36.95 ± 10.28%), whereas crude fibre digestibility values in the animals with pinworms averaged 23.88 ± 3.03%. Similarly, mineral matter retention was significantly lower in the infected animals (59.46 ± 4.62%) than in the control rats (82.95 ± 1.88%). The values of nutrient digestibility are shown in Table 3 and growth parameters are presented in Table 4.
Discussion
Rats are among the most important laboratory animals. They are used for a variety of experiments, including metabolic trials and food testing. Unfortunately, pinworm infection is not taken into account in many of these assays. Although these parasites are considered non-pathogenic, pinworms definitely affect their hosts in a variety of ways. Wagner 9 reported that uninfected rats grew faster and attained weights (at 6 weeks) that were, on average, 12% higher than those of their infected counterparts. The competition for and utilization of the host nutrients are likely explanations for growth depression. 10 Pinworms cause changes in haemopoiesis, 11 affect lymphocyte proliferation 12 and reduce water and electrolyte transport in the intestine. 13
The results of this study show that pinworms also negatively affect the digestibility of all evaluated nutrients. We compared parasitized rats to non-parasitized rats given access to the same amounts of food, so that the consequences of parasitism on food utilization could be directly quantified and evaluated. Statistically significant differences were observed between groups GI and GII with respect to digestibility of all tested nutrients.
The effects were most marked for crude fibre and mineral ash. This is a consequence of the fact that pinworms are located primarily in the caecum, where fermentable polysaccharides act as an energy source for microorganisms, and where mineral absorption takes place. Similar results have been noted in pigs infected with the nematode Oesophagostomum dentatum, which is localized in the colon; and all of these digestibility coefficients measured were affected by this parasite. However, the effects were most marked for crude fibre and mineral ash. 14 Similar changes were also reported in studies by Kaarma. 15 Munger and Slichter 16 reported significantly low dry matter digestibility in kangaroo rats (Dipodomys microps) with Trichuris dipodomys infection.
The role of the large intestine in digestion has not been fully documented. In rats this is mainly where bacteria degrade the fermentable part of fibre to short-chain fatty acids, which are sources of energy and are precursors in the synthesis of glucose and body fat, and which also play an important role in various physiological functions. 17 The main site of fibre degradation in rats is the caecum, and in this study, 98% of all pinworm populations were found in this region. Although pinworms did not cause visible clinical symptoms or changes in the mucosa, caecum function was likely to have been affected. At the end of the experiment, we found 404 to 1405 individual S. muris nematodes in one rat!
Most studies dealing with nutrient digestibility in relation to parasitic infection have been carried out on ruminants infected with nematodes, which are located in the abomasum and small intestine.18–23 The interaction between intestinal helminth infection and nutrition has been reviewed by Poppi et al., 24 Van Houtert and Sykes, 25 Coop and Holmes, 26 Coop and Kyriazakis, 27 Petkevičius, 28 and Holmes et al. 29 In general, gastrointestinal nematodes reduce nutrient availability in the host through reductions in both voluntary feed intake and/or absorbed nutrient efficiency. The degree to which these two mechanisims impair production is, to some extent, dependent on the species of parasite and its location in the gastrointestinal tract.27,30
The key feature of gastrointestinal nematode infection is an increased loss of endogenous protein to the gastrointestinal tract,31–34 partly as a result of plasma protein leakage, increased mucoprotein production and sloughing of epithelial cells in the alimentary tract. 32 Poppi et al. 31 and Kimambo et al. 35 have suggested that the amount of non-reabsorbed endogenous nitrogen that leaves the terminal ileum may be considerable (up to 4–5 g of nitrogen per day). In this study, high protein digestibility was observed in both rat groups. The main reason for this was the feed composition, which contained large proportions of fish; as a result, both groups of rats grew very quickly. However, there was also a statistically significant difference in CP digestibility in the control group. This may have been due to limited caecum function as well as to reduced absorption or utilization of ammonium nitrogen in other forms, which also takes place in the caecum. 36
The most significant effect that gastrointestinal parasitism has on its host is voluntary feed intake depression.22,23,37,38 Large acute infections result in significant decreases in the feed intake of parasitized animals; 39 however, a degree of inappetence is present even in subclinical infections. 28 Several hypotheses have been postulated for this reduction in voluntary feed intake, such as alterations in amino acid availability, changes in flow rates and pH of digests, alterations in gut peptides and hormones, and direct neural effects on the central nervous system.28,40 Even in this current study, the feed intake of healthy animals was higher than that of infected animals, and the difference was statistically significant.
Recently, the influence of nutrition on gastrointestinal parasites has been examined from different perspectives. Some studies show that reductions in voluntary feed intake, as measured by a depression of dry matter intake, are observed only during primary infection.22,23 Other studies have shown that adding protein to the animal diet improves resistance and resilience to several nematode infections.21,28,41 In recent times, the influence of host nutrition on helminth populations has also received attention, and much information is available.28,42–47 Gastrointestinal helminths have very specific physicochemical requirements of their host gut environment, and nutritionally mediated changes may have a direct influence on parasite populations. 48
The possible effects that varying levels of dietary fibre intake have on the occurrence of endoparasitic infection in pigs was reported in the study by Pearce, 49 in which the use of grower diets high in non-starch polysaccharides was associated with an increased risk of Trichuris infection.
In our study, S. muris infection caused no visible signs; no visible changes in the mucosa, no alteration in faecal structure, and no diarrhoea. However, pinworms definitely affect their hosts in different ways. Pinworms alter the gastrointestinal tract environment as well as nutrient digestibility, and they most likely also affect the bacterial community structure, including possible secondary bacterial infections.A secondary bacterial infection would likely lead to impaired absorption and an increase in the flow of materials. 50 Pattison et al. 14 have reported that a high flow rate of digesta in the large intestine would reduce the duration of bacterial action on dietary fibre, thus accounting for a reduction in digestibility.
In conclusion, rats infected with S. muris are not suitable for experiments designed to test nutrient digestibility in various feeds, because these parasites significantly reduce nutrient digestibility of individual nutrients (P < 0.01). It is difficult to understand the mechanisms involved in these reductions, but it has been suggested that subclinical parasitism may affect the nutrition of the host, particularly through reduced absorption, an increased flow rate of digesta, and reduced enzyme activity.14,28 Further detailed investigations would be beneficial.
Footnotes
Acknowledgement
The authors gratefully acknowledge Brian Kavalir (Ontario, Canada) for his proofreading services.
Declaration of conflicting interests
On behalf of all authors the corresponding author states that there is no conflict of interest.
Funding
This study was supported by the University-wide internal grant agency of the Czech University of Life Sciences Prague (CIGA) project No 20142053.
Compliance with ethical standards
All experiments with laboratory animals were conducted in compliance with the current laws of the Czech Republic Act No. 246/1992 coll. on the protection of animals against cruelty and EC Directive 86/609/EEC.
