Abstract
Slightly acidic electrolysed (SAE) water is a sanitizer with strong bactericidal activity due to hypochlorous acid. We assessed the safety of SAE water as drinking water for mice at a 5 ppm total residual chlorine (TRC) concentration to examine the possibility of SAE water as a labour- and energy-saving alternative to sterile water. We provided SAE water or sterile water to mice for 12 weeks, during which time we recorded changes in body weight and weekly water and food intakes. At the end of the experiment, all of the subject animals were sacrificed to assess serum aspartate aminotransferase, alanine aminotransferase and creatinine levels and to examine the main organs histopathologically under a light microscope. In addition, we investigated the bacteria levels of both types of water. We found no difference in functional and morphological health condition indices between the groups. Compared with sterile water, SAE water had a relatively higher ability to suppress bacterial growth. We suggest that SAE water at 5 ppm TRC is a safe and useful alternative to sterile water for use as drinking water in laboratory animal facilities.
Slightly acidic electrolysed (SAE) water is produced by blending and diluting sodium hypochlorite with hydrochloric acid and adjusting the pH value to 5–6 (NDX-1500PLB; OSG Corp, Osaka, Japan). The main effective form of chlorine in SAE water is hypochlorous acid (HOC1), which has strong bactericidal activity. Earlier studies have demonstrated that SAE water inactivates many kinds of pathogens and has higher bactericidal efficiency than sodium hypochlorite solution, a commercial sanitizer.1–4 Hence, we use SAE water at a concentration of 50 ppm total residual chlorine (TRC) as an alternative sanitizer in our laboratory animal facility. In addition, in the future, we propose to provide laboratory animals with SAE water at a 5 ppm TRC concentration as drinking water, instead of sterile water, to save both labour and energy involved in sterilizing water. Although water with 5 ppm TRC is generally accepted to be as harmless as drinking water for laboratory animals5–7 and many facilities have used acidified hyperchlorinated water for mice for many years,8–11 few data show the precise effect of SAE water on the health of laboratory animals. In this study, we assessed the safety of administering SAE drinking water with 5 ppm TRC to laboratory mice.
We used 18 male and 20 female naïve five-week-old C57BL/6 mice. All of the animals were bred in our laboratory (originally purchased from Clea Japan, Tokyo, Japan) and were housed in groups of four or five in wire-topped, transparent cages (open top cages, 310 mm × 210 mm × 140 mm) with wood shavings for bedding and under standard environmental conditions (see below) from weaning to the beginning of the experiment. During that period, these mice were provided with sterile water. The animal experimentation protocols were previously approved by the Animal Research Committee, Akita University, Akita, Japan; all subsequent animal experiments adhered to the University's Regulations for Animal Experimentation. The mice were weighed at the beginning of the experiment and then housed individually in wire-topped, transparent cages (open top cages, 190 mm × 120 mm × 110 mm) with wood shavings for bedding, under a 12 h light–dark cycle, with lights off at 19:00 h. The vivarium was maintained at a constant temperature (23 ± 2°C) and humidity (50 ± 10%). All of the subjects were provided with water and food ad libitum; half were provided with SAE drinking water, and the other half received sterile water (males with SAE water: n = 9; males with sterile water: n = 9; females with SAE water: n = 10; females with sterile water: n = 10), using water bottles (200 mL). Both the bottle and the water were replaced and freshened every other week, at which time we recorded the body weight and weekly water and food intakes of each subject. At 17 weeks of age, the subjects were sacrificed to assess serum aspartate aminotransferase (AST), alanine aminotransferase (ALT) and creatinine levels using an automatic analyser (Fuji Dri-Chem 3000 V; Fujifilm, Tokyo, Japan). In addition, the liver, kidneys, heart, lungs, spleen, stomach and oesophagus of each subject were collected in 10% buffered formalin and stained with haematoxylin and eosin, following standard procedures for histopathological examination under a light microscope. At the same time, a 0.2 mL water sample was removed from each water bottle, and an aerobic plate count was conducted using brain heart infusion agar (Eiken Chemical, Tokyo, Japan). Analyses comparing the two types of drinking water considered male and female data separately. Statistical comparisons of body weight and water and food intakes were performed using two-way repeated measures analysis of variance (ANOVA). We used the Mann-Whitney U-test to statistically analyse serum AST, ALT and creatinine levels, and aerobic plate count data. All data are presented as means ± standard error. The criterion for statistical significance was P < 0.05 for all comparisons.
Regardless of the type of water provided, we could find no morphological and behavioural abnormalities in any subject animal, and no mice of either sex showed a significant difference in body weight (males: F1,16 = 0.024, P = 0.88; females: F1,18 = 0.028, P = 0.87; Table 1). The volume of SAE water consumed weekly by both males and females was almost equal to that of sterile water (males: F1,16 = 0.000016, P = 0.99; females: F1,18 = 0.51, P = 0.48; Table 1). Likewise, no significant difference was observed in the weekly food intake between the groups, regardless of sex (males: F1,16 = 1.2, P = 0.29; females: F1,18 = 1.3, P = 0.27; Table 1). Serum AST, ALT and creatinine levels showed no significant difference between groups, regardless of sex (AST males: P = 0.70, AST females: P = 0.66; ALT males: P = 0.39, ALT females: P = 0.79; creatinine males: P = 0.52, creatinine females: P = 0.63; Table 1). Histopathological examination revealed no significant lesion in any collected organ. The aerobic plate count for 0.2 mL of SAE water was zero, which was lower, but not significantly lower (males: P = 0.13, females: P = 0.07), than that of sterile water (males: 350 ± 170, females: 740 ± 710).
Biological indices and statistical P values of mice provided with sterile water or slightly acidic electrolysed water
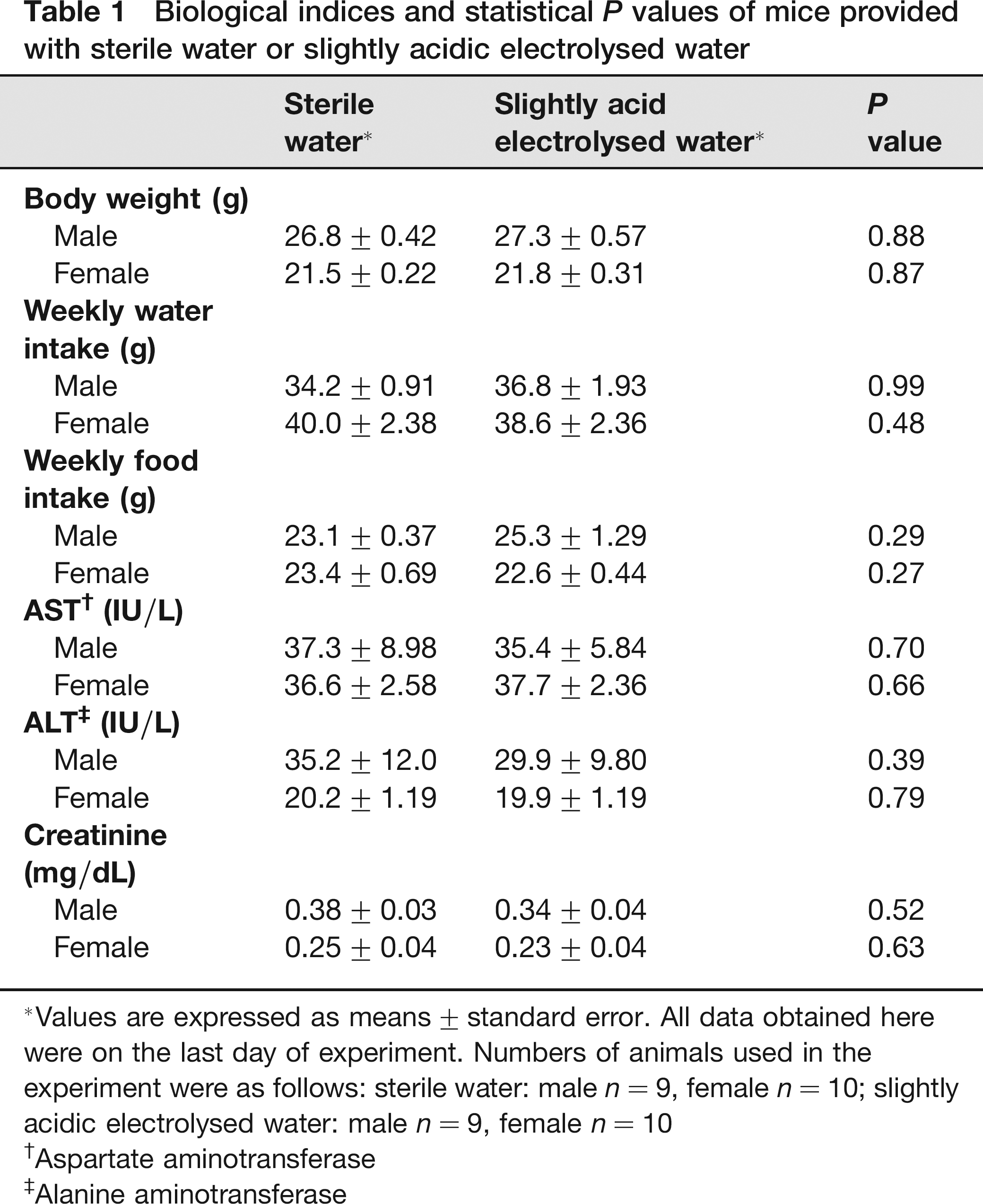
Values are expressed as means ± standard error. All data obtained here were on the last day of experiment. Numbers of animals used in the experiment were as follows: sterile water: male n = 9, female n = 10; slightly acidic electrolysed water: male n = 9, female n = 10
Aspartate aminotransferase
Alanine aminotransferase
Our findings indicate that SAE drinking water containing 5 ppm TRC has scant influence on several functional and morphological indices of laboratory mouse health. In addition, SAE water with 5 ppm TRC has a relatively higher ability to suppress bacterial growth compared with sterile water. Thus, we suggest that SAE water is a safe and useful alternative to sterile water as a drinking supply in laboratory animal facilities, even though the analyses on the effect of this water on mice are not complete. Future studies should further examine the appropriateness of SAE water for laboratory animal use by evaluating additional effects of SAE water on mouse health (e.g. reproduction, tooth enamel, gastrointestinal pH, bacterial flora of the ileum and enzyme activities of the intestinal mucosa), as well as its palatability compared with sterile water in a two-bottle choice test and its effects on the health of other laboratory animals (e.g. rats).5,12,13
