Abstract
Blood collection in mice can be a challenge, in particular for samples used for coagulation analysis as initiation of coagulation during the procedures can influence the results. Blood collection from the retrobulbar venous plexus is commonly used but the method remains controversial. Several alternatives exist but not all are applicable to mice with a compromised coagulation system because of subsequently excessive bleeding. We therefore wanted to explore whether blood collection by puncture of the submandibular vein could replace blood collected from the retrobulbar venous plexus during pharmacokinetic and pharmacodynamic studies in mice lacking coagulation factor VIII (FVIII). The plasma concentrations of recombinant activated factor VII were independent of the blood collection method in a pharmacokinetic study. The same applied to the thromboelastographic profile of mice with normal coagulation in a pharmacodynamic study. However, excessive haemorrhages were observed in all FVIII knockout mice after a single puncture of the submandibular vein and 60% of the mice were euthanized 2–4 h after the blood collection. In contrast, no or only slight haemorrhage was observed in animals subjected to blood collection from the retrobulbar venous plexus. No signs of distress determined by blood glucose level or clinical abnormalities of the eye were observed after puncture of the retrobulbar venous plexus. In conclusion, blood collected by puncture of the submandibular vein and retrobulbar venous plexus has a quality which allows it to be used in coagulation assays. However, because of excessive bleedings, puncture of the submandibular vein is not recommended in mice lacking FVIII.
Mice are favoured laboratory animals due to their size, ease of handling and the existence of numerous genetically-modified models. However, blood collection poses a challenge because of the small blood volume and lack of easily accessible vessels. Approximately 300 µL (<20% of the total blood volume) can be drawn within 24 h 1 and blood collection tends to be a terminal procedure because of the large volume required for an analysis. With the improvement of analytical methods it is now possible to analyse a number of essential parameters on very small volumes. This improvement of the analytical methods allows for use of the same mice for repeated measurements over time and has increased the focus on humane blood collection techniques that allows for repeated collection of high-quality blood samples. 2 One of the most utilized techniques is puncture of the retrobulbar venous plexus. This method consistently yields a reasonable blood volume of high quality. 2 The technique is controversial, however, as poor performance can cause severe tissue damage and blindness which has led some institutions and countries to ban the method. 3
Alternative methods of blood sampling have been described including puncture of the tail vein, saphenous vein, sublingual vein or submandibular vein, laceration of the tail vein and amputation of the tip of the tail. 2–6 Even so, it is well documented that the choice of collection site has a deleterious impact on some of the haematological and biochemical parameters due to stress or other physiological changes such as consequences of restrain or anaesthesia, 7,8 which needs to be taken into consideration when designing a study.
During the research and development of new therapies for patients with coagulation disorders, i.e. lacking either coagulations factor VIII or IX (haemophilia A and B, respectively) factor VIII (FVIII) and IX (FIX) knockout mice have been a very valuable tool both for pharmacokinetic and pharmacodynamic studies. 9–12 For these two strains of mice prolonged bleeding after laceration of the tail vein 13 or amputation of the tip of the tail 14 is a well described phenotypic characteristic, and if left untreated the mice will bleed to death leaving these blood collection techniques unsuitable. Also, the sublingual technique appears unfeasible as it is frequently associated with haemorrhages even in mice with a normal coagulation system. 3 Thus, two possible alternatives to puncture of the retrobulbar venous plexus in FVIII and FIX knockout mice remains, that is puncture of the submandibular and the saphenous vein. In our search for an alternative to puncture of the retrobulbar venous plexus, we initially chose puncture of the submandibular vein solely because of the apparent ease of the handling procedure. The clinical biochemistry parameters after blood collection from the submandibular vein and retrobulbar venous plexus have recently been compared showing small but significant differences in the values, 8 but whether the same applies to coagulation parameters is unknown.
Functional coagulation tests are often used in pharmacodynamic studies to investigate the effect of new therapies for patients with coagulation disorders. 13,15 This also includes thromboelastography, which is a global coagulation test performed on whole blood. This analysis measures the onset of clot formation, kinetics of the clot formation and the resulting clot strength. 16 Thus, the clotting process is significantly impaired in blood from haemophilic patients (and animals) compared with individuals with a normal coagulation system. 17,18 However, because blood coagulation initiates as soon as the blood is drawn from the vessel or gets in contact with intravascular tissue, the result of the following analysis is very sensitive to the preanalytical conditions such as the blood collection method.
The studies described in the current paper were designed to investigate whether blood collected from the submandibular vein could replace blood collected from the retrobulbar venous plexus in pharmacokinetic and pharmacodynamic studies in FVIII knockout mice. The impact on animal welfare was investigated by characterization of bleeding from the puncture site and distress investigated by changes in blood glucose after the procedure.
Materials and methods
Animals and housing
Thirty-six FVIII knockout 14 mice (B6;129S4-F8 tm1Kaz /J) and 20 C57BL/6NTac mice were obtained from Taconic (Ry, Denmark). The animals were between 12 and 16 weeks old with an equal distribution of males and females. All animals (max 10 mice per cage) were housed in Tecniplast Makrolon type IV cages (Scanbur, Karlslunde, Denmark). The FVIII knockout mice were housed in cages with a normal lid, whereas the C57BL/6NTac mice had a 7 cm higher lid (Scanbur). All the mice had Tapvei aspen bedding (Tapvei, Kortteinen, Finland), Enviro-Dri® paper-based nesting material (Lillico, Surrey, UK), a Tapvei aspen wood biting stick (Tapvei) and a hide (Repsol, Brønderslev, Denmark) and were housed in a ScantainerNOVO rack (Scanbur). The mice were fed with Altromin 1320 diet (Brogaarden, Gentofte, Denmark) and a small amount of corn and wheat mixed up with the bedding material. Tap water was provided ad libitum through an automatic watering system (Edstrom Europe, Hereford, UK). Light was on from 07:00 to 19:00, temperature was 21 ± 3°C and air humidity was 50 ± 20%. The acclimatization period was at least one week before initiation of the experiment.
The four individual experiments described below were all performed under a licence issued by the National Animal Experimentation Board under the Danish Ministry of Justice. Danish animal experimentation legislation is based upon the EU Directive 86/609 and the Council of Europe Convention ETS 123 (2006).
Blood collection techniques
The main purpose of this study was to investigate whether blood collected from the submandibular vein could replace blood collected from the retrobulbar venous plexus. Thus, in order to avoid compromising the results because of lack of experience, both collection techniques were performed by competent technicians with more than two years of experience with the techniques and have used them routinely.
Submandibular blood collection
The blood was drawn from a small vascular bundle at the back of the jaw at the point where the orbital veins, the submandibular vein and other veins draining the facial region join to form the beginning of the jugular vein. 5 The conscious mouse was restrained with one hand, and the back of the jaw was localized – the puncture site is just caudal to the small cowlick. The vena fascialis was punctured by a Medipoint lancet (4 mm; Bie & Berntsen, Herlev, Denmark). The lancet was placed perpendicular to the skin when poking and quickly removed. The first two drops of blood was discharged and only free-floating blood was collected. Blood samples were stabilized in 130 mmol/L sodium-citrate (9:1 v/v). The blood was used immediately for thromboelastographic analysis or plasma was prepared and stored at −80°C before analysis. After blood collection, the stasis was released and the mouse replaced in its cage.
Retrobulbar blood collection
Anaesthesia was induced by placing the mice in a box (24.5 × 11.0 × 10.0 cm; l × w × h) saturated with isoflurane (5%, carried by 5 L/h O2 and 7 L/h N2O). The animals were removed from the box as soon as the palpebral reflex had disappeared. Blood was collected by inserting an uncoated 20 µL microhaematocrit tube (o.d. = 1.3 mm) into the medial cantus of the orbit while gently pushing and rotating it forward. The blood was stabilized and handled as described above. The mice showed no signs of consciousness during the blood collection and visual blood was removed by gently dabbing with a cotton swab. The mice were placed in their home cage after the procedure and recovered from the anaesthesia within a few minutes.
Comparison of the blood collection techniques for pharmacokinetic studies
FVIII knockout mice (n = 3) were administered a single intravenous dose of human recombinant activated factor VII (rFVIIa) (1 or 15 mg/kg, Novo Nordisk A/S, Maaloev, Denmark) in the tail vein. Blood was collected 5, 30 and 60 min after injection by puncture of either the submandibular or retrobulbar veins as described above. At each collection, six droplets of blood were obtained and stabilized in sodium citrate and diluted 5× in buffer (10 mmol/L sodium phosphate buffer, 145 mmol/L sodium chloride, 0.05% Tween 20 and 1% bovine serum albumin, pH 7.6) after which it was centrifuged at 4000
Comparison of blood collection techniques for coagulation analysis
The thromboelastographic profile was collected by ROTEM® delta (ROTEM, Munich, Germany). Citrate stabilized blood from FVIII knockout or C57BL/6NTac mice (n = 4) was collected using the two methods described above and 105 µL of the stabilized blood was added to a minicuvette containing 7 µL CaCl2 (final Ca2+ concentration 11 mmol/L) to initiate coagulation.
Impact on animal welfare
Characterization of excessive bleeding after blood collection
This study was designed to investigate the occurrences of rebleedings after puncture of either the retrobulbar or submandibular venous plexus in FVIII knockout mice (n = 10). All animals were frequently observed in their home cage for a period of 6 h after the actual blood sampling and the following day to ensure that the animals were euthanized for humane reasons, where appropriate. Humane reasons include, but were not limited to, cases where the animals show signs of permanent suffering, pain or distress, as described in the OECD guide: ‘Recognition, Assessment and Use of Clinical Signs as Humane Endpoints for Experimental Animals in Safety Studies (OECD, 2000)’. Twenty-four hours after the blood collection, the mice were euthanized with a mixture of CO2 (85%) and O2 (15%) and subjected to a postmortem examination, with emphasis on the cheek and eye regions. These examinations were performed by an experienced veterinarian pathologist.
Tissue changes after puncture of the retrobulbar puncture in rats with a normal coagulation system has previously been described extensively 21,22 and therefore we only included submandibular puncture in a group of mice with a normal coagulation system (C57BL/6NTac; n = 10).
Finally, the blood collected was used to confirm the thromboelastographic profile (n = 4). A platelet and red blood cell count was performed on EDTA-stabilized blood (Medonic Vet-620) from all mice.
Effect of retrobulbar blood collection on blood glucose
To determine the influence of the puncture of the retrobulbar venous plexus on blood glucose levels a blood sample was taken at baseline and 90 min later in mice with a normal coagulation system (C57BL/6NTac; n = 6). Five microlitres of blood were collected in a microcapillary (5 µL Modulohm A/S) and placed in an eppendorf tube with 250 µL Ebio buffer (Prahl Biolab A/S, Græsted, Denmark). The tube was thoroughly shaken, and the blood glucose was measured within 30 min in an EBIO® plus unit (Eppendorf AG, Hamburg, Germany).
Data analysis
Data are presented as mean ± SEM unless otherwise stated and were analysed using GraphPad Prism version 5 (La Jolla, CA, USA).
The data from the pharmacokinetic study were analysed by a two-way analysis of variance (ANOVA) for repeated measurements. The thromboelastographic data were analysed by a two-way ANOVA. The comparison of the blood glucose level over time was done with a paired t-test. All other analyses were performed with an unpaired t-test. P < 0.05 was considered significant.
Results
Comparison of blood collection techniques for pharmacokinetic studies
After intravenous injection of 1 and 15 mg/kg of rFVIIa in FVIII knockout mice, a dose-dependent increase in the plasma level of FVIIa activity was observed in both blood collection techniques (Figure 1). Two additional blood samples were successfully collected over a period of 60 min. During this period, the FVIIa activity in plasma gradually declined. No statistic significant differences in the FVIIa activity were determined between the two different blood collection techniques.
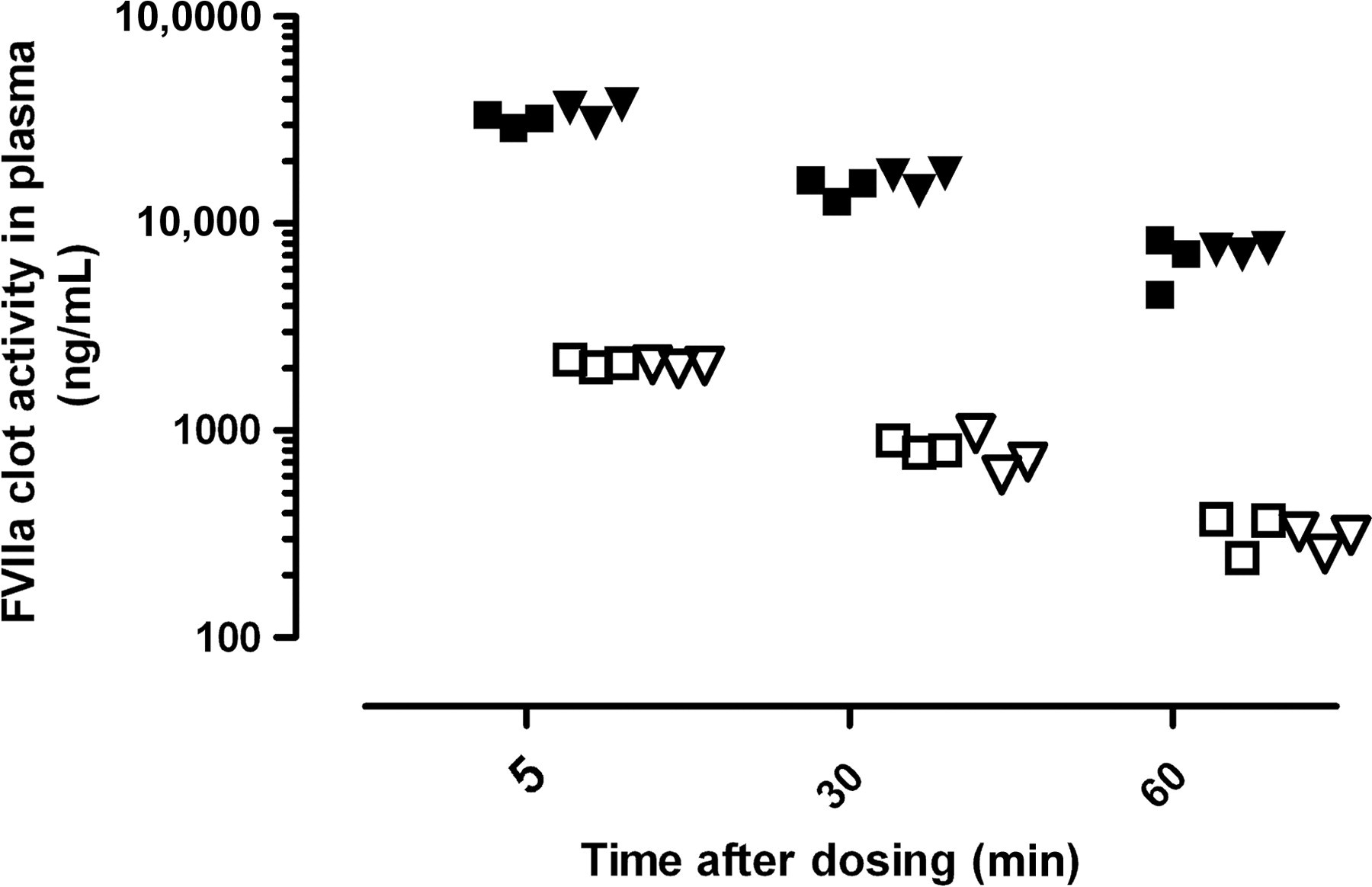
Pharmacokinetic profile of rFVIIa (1 [open symbols] and 15 [closed symbols] mg/kg, iv) after collection of the submandibular vein (square) and retrobulbar venous plexus (triangle) in FVIII knockout mice. FVIIa levels in plasma were determined by FVIIa clot activity (n = 3). No difference was observed in the FVIIa clot activity between the two blood collection methods
Comparison of blood collection techniques for coagulation analysis
Blood from FVIII knockout mice coagulates significantly later than the blood from a mouse with a normal coagulation system (C57BL/6NTac) (clot time: 1804 ± 146 versus 304 ± 15 seconds; n = 4; Figure 2). Also, when coagulation was initiated, the propagation of the clot formation progresses significantly slower (angle: 23 ± 2° versus 61 ± 5°; n = 4). No statistic significant differences were observed between the data obtained from the two different blood collection techniques.
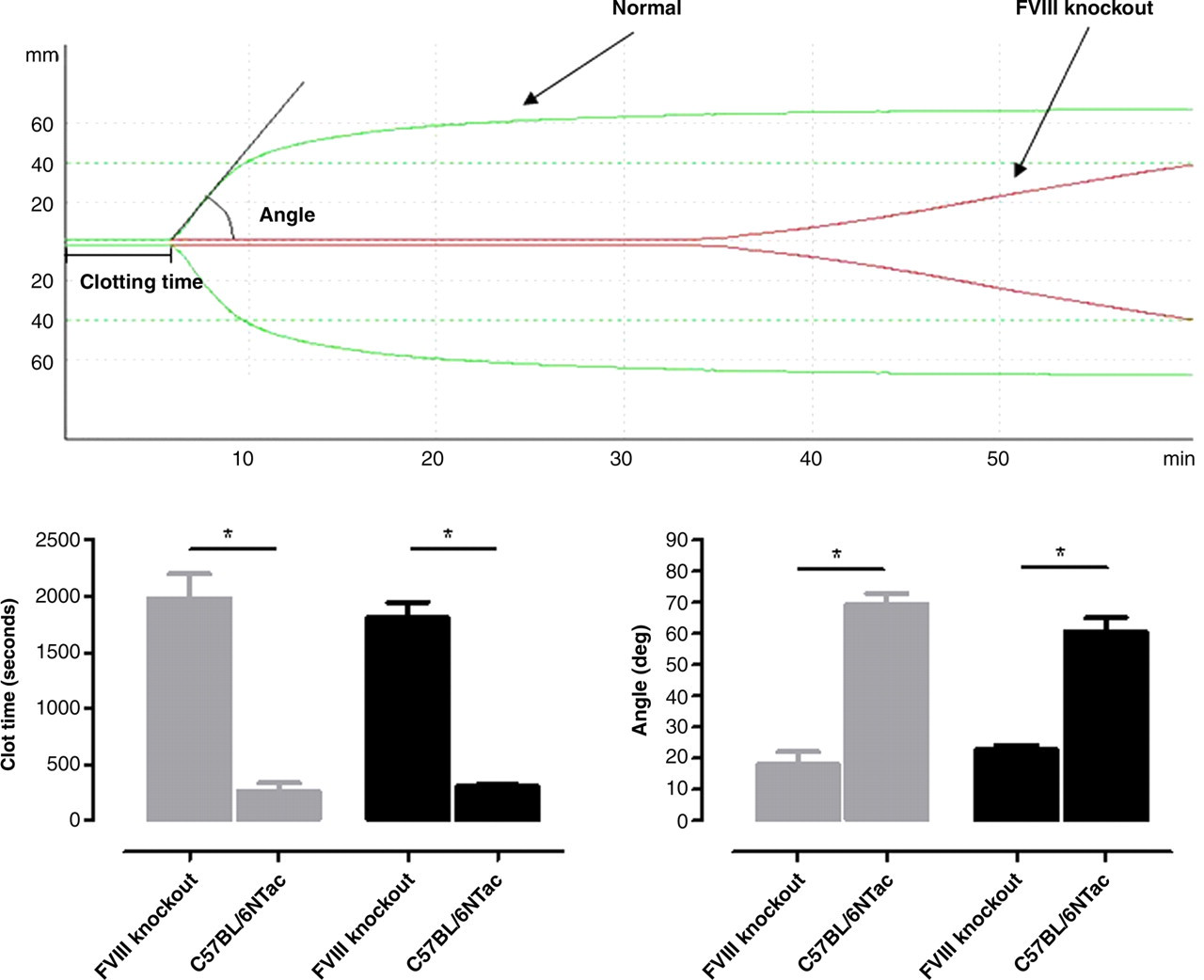
Top panel: Thromboelastographic profile of blood from a mouse lacking factor VIII and a mouse with a normal coagulation system (C57BL/6NTac) collected by puncture of the retrobulbar venous plexus. The time to initiation of the clot formation is measured from the initiation of the tracking to the point where the curve is 1 mm wide. The propagation phase is quantified by the angle. The angle is measured between the midline of the tracking line and the line drawn between the time to clot formation tangential to the curve. Bottom panel: Comparison of the clot initiation (left panel) and propagation phase (right panel) from the thromboelastographic profile obtained in whole blood collected from FVIII knockout and mice with a normal coagulation system after blood collection from either the submandibular vein (grey) or retrobulbar venous plexus (black) (n = 4). *Indicated significant (P < 0.05) differences between strains. No difference was observed between the blood collection methods
Impact on animal welfare
Comparison of haemorrhage 24 h after collection
Two to four hours after puncture of the submandibular vein, six out of 10 FVIII knockout mice were euthanized for humane reasons. Though responsive, these animals were pale and appeared non-alert. In four of the mice the postmortem examination showed large diffuse haematomas in the area of the masseter muscle defined by the triangle between the parotid gland, mandible and ear. In addition, two of these mice had blood in the cranial cavity possibly caused by an ooze through the stylomastoid foramen. The remaining mice had a moderate haematoma (5–7 mm diameter) or a slight haematoma (1–3 mm diameter) on the masseter muscle. The four mice that completed the 24 h observation period all had a slight haematoma on the masseter muscle at the site of the puncture (Table 1).
Overview of the haemorrhages observed 24 h after blood collection by different techniques in FVIII knockout mice and mice with a normal coagulation system (C57BL/6NTac) at the postmortem examination
Definition of haemorrhage:
Slight: Haematoma/bleeding (1–3 mm diameter) at the puncture site
Moderate: Defined haematoma at the masseter muscle (5–7 mm diameter)
Severe: Large diffuse haematomas in the area of the masseter muscle defined by the triangle between the parotid gland, mandible and ear
*After puncture of submandibular vein, six FVIII knockout mice were euthanized 2–4 h after the puncture for humane reasons. These animals were pale and appeared non-alert though responsive (see Results for further details)
All the mice with a normal coagulation system completed the 24 h observation period without signs of distress. Nevertheless, upon the postmortem examination, all mice had signs of bleedings on the puncture site and one mouse had a moderate haematoma on the masseter muscle (Table 1).
Puncture of the retrobulbar venous plexus in FVIII knockout mice only caused a small bleeding (1–2 mm) in two out of 10 animals (Table 1). No clinical signs of damage to the eye or surrounding tissues were observed in the current study after a single sample.
The thromboelastographic analysis of the collected blood confirmed the data shown in Figure 2. No differences in the red blood cell count were observed (Figure 3), whereas the platelet counts were significantly less in blood collected from the submandibular vein (374 ± 28 × 109/L) compared with blood collected from the retrobulbar venous plexus (485 ± 18 × 109/L) (Figure 3).
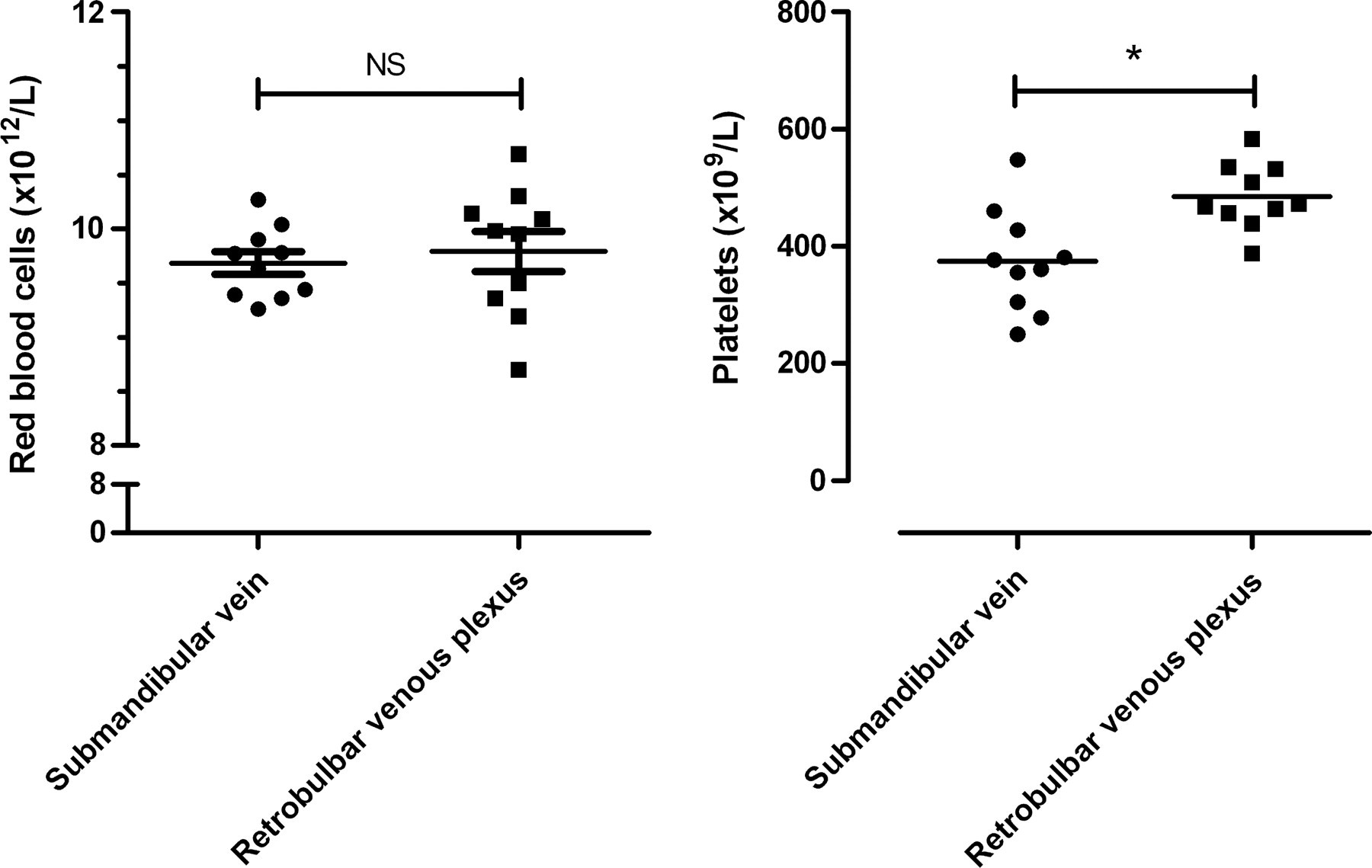
Comparison of red blood cell and platelet count after collection from either the submandibular vein or retrobulbar venous plexus in FVIII knockout mice (n = 10). *P < 0.05, unpaired Student's t-test. NS: not significant
Effect of retrobulbar blood collection on blood glucose
Under the conditions applied in the current study, no changes in blood glucose were observed 90 min after the first measurement. Thus, the mean blood glucose was 8.1 ± 0.3 mmol/L at baseline and 7.7 ± 0.3 mmol/L 90 min later (Figure 4).
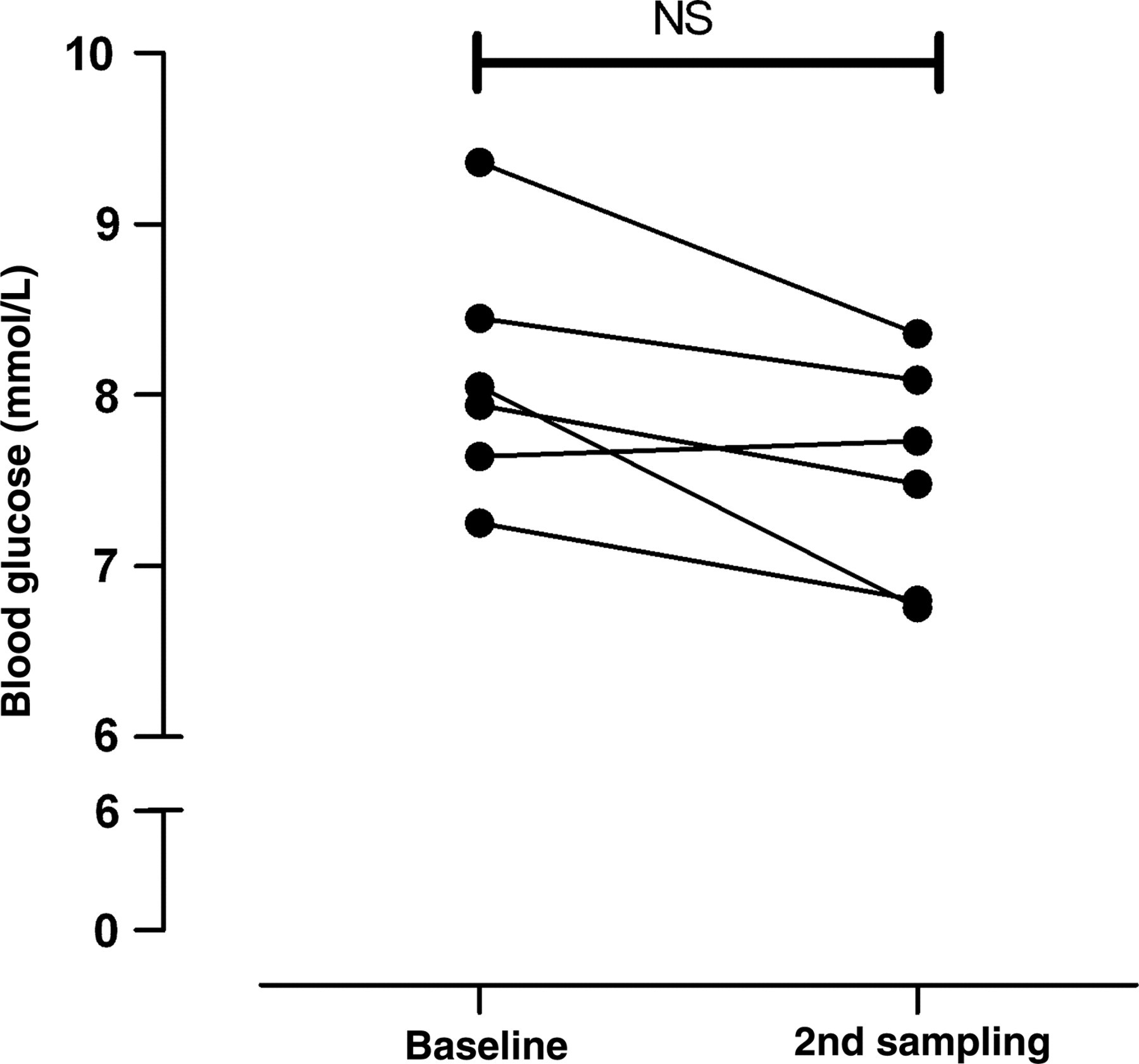
Comparison of the blood glucose level at baseline and 90 min after collection of a blood sample by puncture of the retrobulbar venous plexus under light isoflurane anaesthesia in C57BL/6NTac mice (n = 6). NS: no statistic significant differences between time points (paired Student's t-test)
Discussion
The data generated in this study show that blood collection by puncture of the submandibular vein provides blood of a high quality that is suitable for coagulation analysis. Unfortunately, this method cannot replace blood collection by retrobulbar puncture in FVIII knockout mice due to excessive and life-threatening bleedings from the puncture site in these mice.
It is crucial for an experiment to obtain blood of consistent high quality to decrease variation in the results and thereby minimizing the number of animals necessary to reach a conclusion on the hypothesis tested. Blood for coagulation assay is very sensitive to the collection method. Most coagulation assays are functional assays based on the time for the clot formation and this process is initiated as soon as the blood is drawn from the vessel or gets in contact with intravascular tissue. Thus, the blood collection method for coagulation assays requires that tissue contamination is eliminated and that only free-floating blood is collected. Thromboelastography is a global coagulation test performed on whole blood measuring not only the onset of clot formation but also the kinetics of the forming clot and the resulting clot strength. 16 In blood from humans the method can differentiate not only normal blood from a patient with haemophilia but also the severities of the disease. 23 A similar picture is observed in murine blood. Thus, blood from FVIII knockout mice coagulates later than blood from normal mice 18 and after coagulation is initiated, the propagation of the clot formation is slower. When comparing the data from the mice with a normal coagulation system and FVIII knockout mice the thromboelastographic analysis revealed the expected difference between the two mouse strains. Furthermore, there were no differences in the data obtained from the two collection sites. The same applies for the pharmacokinetic evaluation of rFVIIa. This suggests that blood collected from the submandibular vein consistently has the same high quality as blood collected from the retrobulbar venous plexus. Finally, the study also demonstrates that despite the high quality of the blood samples obtained from both collection methods, haematological values depend on the collection site as differences in the platelet count between the two collection sites were observed. This has previously been described for other blood collection methods 7,24 and recently a comparison of the biochemical values obtained after puncture of either the submandibular vein or retrobulbar venous plexus showed significant differences in some clinical biochemistry parameters. 8 This emphasizes that it is only possible to compare data from blood collected by identical methods.
Collecting blood inevitably damages the punctured tissues and cause bleeding. In animals with a normal coagulation system the bleeding quickly ceases, though all animals exhibited small signs of bleeding 24 h after puncture of the submandibular vein in the current study. In contrast, the mice lacking coagulation factor VIII all had signs of haemorrhage at the puncture site 24 h after the submandibular vein puncture and six animals had to be euthanized due to excessive haemorrhages. The initial clot formation is initiated in these mice however because of the lack of factor VIII the amplification and propagation of the clot formation does not occur. This makes the formed clot very fragile and leaves the mice susceptible to repeated bleeding from the site of puncture. This is a very pronounced phenomena for this strain and two other blood collection techniques employing the tail are actually used to describe the phenotype of this and other strains with an impaired coagulation. 13,14 Unlike the puncture of the submandibular vein, puncture of the retrobulbar venous plexus only caused a small haemorrhage in two out of 10 animals. It is likely that the pressure on the surrounding tissue together with the small mechanical stress in the area provide sufficient protection of the formed clot to prevent rebleeding. Based on these observations we find it unlikely that collection of blood from the saphenous or sublingual vein will provide a better result leaving the puncture of the retrobulbar venous plexus the only method for repeated collection of blood samples from FVIII knockout mice. No clinical signs of damage to the eye or surrounding tissues were observed in the current study after a single sample. The blood collection was performed by a very skilled technician which probably contributed to this result as it has been described that severe tissue damages is avoided when the procedure is performed by experienced and skilful technicians. 22
Finally, puncture of the retrobulbar venous puncture has been argued to cause pain and distress. Only a few studies have addressed this issue in mice; one paper, 25 however, concludes that the method is non-stressful when performed under light halothane anaesthesia based on measurement of plasma catecholamines. Similar conclusions were drawn in the current study based on blood glucose levels. Increase in blood glucose is a well-known stress response in rodents 26,27 but the blood glucose level in the current study was unchanged 90 min after puncture of the retrobulbar venous plexus when performed under light isoflurane anaesthesia. This is in contrast to puncture of the retrobulbar venous plexus in non-anaesthetized animals where a significant increase in the blood glucose has been observed. 2 Several studies have been performed in rats to investigate behavioural and physiological changes after retrobulbar blood collection with different conclusions. 28–32 Unfortunately, in the majority of these studies, retrobulbar puncture was performed on animals under ether anaesthesia, which in itself causes stress. 33
In conclusion, blood collected by puncture of the submandibular vein has a quality that allows it to be used in coagulation assays. However, because of excessive bleedings puncture of the submandibular vein is not recommended in mice lacking FVIII and most likely in any mice with a severely compromised coagulation system. In the hands of an experienced and skilful person, collection of blood by puncture of the retrobulbar venous plexus under light isoflurane anaesthesia has a quality that allows it to be used for coagulation analysis and the procedure does not cause excessive bleeding at the site of puncture nor does it seem to cause distress to the animal as demonstrated by no changes in the blood glucose level.
