Abstract
In a large number of animal experiments, blood collection is crucial for achieving the study aim. Requirements on sampling techniques used include their practicability, their effectiveness in terms of obtaining the desired blood volume, sample quality and low impact on animal's wellbeing. Numerous methods for blood collection from mice have been published. For large blood volumes, submandibular and sublingual bleeding was developed as alternatives to the retrobulbar bleeding method, which is considered controversial as it results in severe tissue damage. Only a few studies report the use of submandibular and sublingual techniques in mice. In particular, the degree of tissue damage or influence on animal's wellbeing due to submandibular bleeding in conscious mice has not yet been clearly assessed. This gap in the knowledge base has been filled by our exploratory study that revealed clear differences between both techniques. Defence movements during submandibular blood collection in conscious mice resulted in more complications and revealed insufficient blood sample quality due to the prolonged duration of blood collection. In addition, it is likely that these movements may have caused lesions to be more pronounced. Changes in red blood cell parameters (red blood cell count, haemoglobin and haematocrit), in glucose and in total protein concentrations observed in anaesthetized animals were most likely related to anaesthesia. Sublingually punctured mice gained significantly more body weight than submandibularly punctured mice, likely due to less severe tissue lesions and improved healing processes. Based on these results, we recommend the sublingual blood collection technique to be used in mice. However, if the submandibular bleeding technique is used, it should be performed in anaesthetized mice only.
In a large number of animal experiments, blood collection is crucial for achieving the study aim. Requirements on utilized sampling techniques are their practicality, their effectiveness in terms of desired blood volume, sample quality and low impact on animal's wellbeing. Numerous methods have been published for use in mice. 1–20
For larger blood volumes, retrobulbar bleeding (known as retro-orbital bleeding in some publications) is still one of the most commonly used blood collection techniques in mice and rats, although somewhat controversial due to resulting tissue damage. 1,21–32 Therefore, alternative methods were developed, i.e. the puncture of the sublingual vein. This method was first developed in rats 32 and extended to mice and hamsters. 31 This technique permits large-volume blood collection without a severe impact on the wellbeing of the animals used. Another alternative method is the puncture of the submandibular vein. 33 This technique could be criticized as some areas of venous structures in the facial region are covered by other tissues, i.e. relatively large glandular structures. 34,35 In addition, the submandibular method is described as being feasible in non-anaesthetized animals and this was highlighted as an advantage over other methods. Only one study on the use of this technique was available, 33 as this method is still under development. The degree of tissue damage or influence on animal's wellbeing due to abdication of anaesthesia has not yet been clearly assessed. Finally, the puncture is carried out on a vein that is not clearly visible, which may be disadvantageous due to the potential for increased missed vein punctures, especially with personnel with only limited experience of the technique.
The aim of our exploratory study was to compare direct and indirect tissue damage in mice after single blood collection from the submandibular vein and the sublingual vein. In addition, the necessity of using isoflurane anaesthesia during submandibular blood collection was evaluated.
Materials and methods
Animals and housing conditions
Fifty-four female CD1-mice (Crl: CD1 [CR] from Charles River Laboratories, L'Arbresle, Cedex, France) that were approximately 14 weeks old and weighed 29.6–45.1 g were randomized and divided into three groups of 18 animals each. The mice were group-housed (n = 4 or 5) in type III Makrolon boxes with autoclaved wood chip bedding and a mousehouse® (Tecniplast SA, Varese, Italy). Temperature was maintained at 19–25°C, with a relative humidity of 35–75%. A 12 h light–dark cycle was used (light on from 06:00 to 18:00, max. 100 lx light intensity), and background radio music was played. Animals received a pelleted diet ad libitum (Provimi Kliba SA, Kaiseraugst, Switzerland; mouse/rat husbandry ‘GLP’ NAFAG 890). Fresh water was available ad libitum from 300 mL Makrolon bottles. A sentinel program was used to monitor the health status during the experiment. All animals were microbiologically defined according to the Federation of European Laboratory Animal Science Associations (FELASA) guidelines. 36
Anaesthesia
Two of the groups of animals used for sublingual or submandibular blood collection (groups 2 and 3, see below) were exposed to 2.5% isoflurane inhalation anaesthesia (ForeneTM, Abbott Laboratories SA, Altishofen, Switzerland; oxygen flow rate of 4 L/min).
Materials used for blood collection methods and measurements of blood parameters
Submandibular blood collection was performed using 5 mm GoldenRod Animal Lancets (Medipoint Inc, Mineola, NY, USA). For sublingual blood collection, 24-gauge (24 G × 1 inch; 0.55 × 0.25 mm) hypodermic needles and cotton buds were used.
Blood was collected into micro blood collection tubes. Microtubes contained ethylenediaminetetraacetic acid to be used for haematological examinations. For measurements of clinical chemistry serum values, microtubes without anticoagulant were used.
Haematology parameters were determined with an ADVIA 120 (Siemens Medical Solutions Diagnostics, Eschborn, Germany). Total white blood cell count, differential white blood cell count, red blood cell count, haemoglobin, haematocrit, mean corpuscular volume, mean corpuscular haemoglobin, mean corpuscular haemoglobin concentration and platelet count were defined as parameter range. The clinical chemistry parameters alanine aminotransferase, alkaline phosphatase and aspartate aminotransferase activities, and albumin, creatine kinase, globulin, glucose and total protein concentrations were determined in the serum with a Synchron CX5 (Beckmann Coulter, International SA, Nyon, Switzerland).
Experimental design and procedures
Two groups that were used for submandibular bleeding (group 1 without anaesthesia and group 2 with anaesthesia) were compared with the third group that was used for sublingual blood collection (under anaesthesia). Animals were not fasted overnight before blood collection and had free access to water. Blood collection took place in the morning of day 1 and was performed over a period not exceeding 2.5 h. Blood was collected from each animal from either the right sublingual vein or the submandibular vein (right cheek). One technician performed the submandibular bleeding, whereas two technicians carried out the sublingual blood collection. For each animal, the time required to conduct blood sampling was measured using a laboratory stopwatch. The time to anaesthetize the animals was not measured as it was not considered relevant for the comparison of blood collection techniques.
To enable submandibular blood collection, the animal was restrained by the tail, neck and skin on the back, which insures venous congestion and immobilization of the animal's head and body. The lancet was used to make a puncture in an area approximately 3 mm caudal and 1 mm dorsal to the lateral tactile hair of the right cheek. Blood dripping from the punctured submandibular vein was collected into a microtube (using blood microtubes with volume scale). To stop the bleeding, the restraint was released. In anaesthetized animals, slight pressure on the punctured area was exerted with a cotton swab. The same was performed in the conscious animals, provided the animal did not struggle excessively.
For sublingual bleeding, the animal was grasped by the tail and the skin in the neck region in order to insure fixation of the animal automatically resulting in partial venous congestion. The mouse was placed in a supine position. A second person extended the tongue by holding it between their thumb and a cotton bud. The thick caudal part of the right vena sublingualis was punctured. Blood was collected into a micro blood collection tube holding the mouse in a horizontal position above the tube. To stop the bleeding, the hold on the animal's neck skin was released. The mouth of the mouse was cleaned with a dry cotton bud to remove any remaining blood.
From the first half of each group (n = 9), the blood samples were used for haematology determinations (differential blood count). Blood was immediately mixed by inverting the tube slowly at least 10 times in order to prevent clots and platelet clumps. A selected set of clinical chemistry parameters was determined in the serum from the second half of each group (n = 9). Microtubes containing blood samples were placed on ice prior to measurement of clinical pathology parameters, which took place as soon as possible after the last sample was taken.
Three different time points were scheduled for histopathological examination. Following the blood collection, six animals of each group were killed after 3 h, following two or five days carbon dioxide asphyxiation. From all animals, the following tissues/organs were processed histologically and examined microscopically: gl. lacrimalis supraorbitalis, gl. mandibularis, gl. parotis, mandibular lymph node, skin of mandibular region and the tongue. Tissues/organs were fixated in neutral phosphate-buffered formalin. After bone demineralization with formic acid, one 4–5 mm transverse section of the head including the site of the puncture for blood sampling and two transverse section of the tongue were trimmed and further processed histologically. Sections were embedded in paraffin wax, sectioned and stained with haematoxylin and eosin and Berliner blue for iron. In addition, for all animals of groups 1 and 2, a second transverse section of the block (head) was made and stained with haematoxylin and eosin.
The following investigations were performed daily in all animals: clinical signs including mortality check (once daily prior to blood collection, twice daily on the days following blood collection), body weight development (daily) and food consumption measurements (daily, mean consumption per animal was calculated from the total cage value).
Statistical analysis
For in-life data collected with our in-house computer system (body weight development and determination of food consumption), an analysis of variance test was used for overall assessment of parametric data followed, in case of a significant group effect, by Dunnett's test for group comparisons. For non-parametric data, a Kruskal–Wallis test was used for overall assessment followed, in case of a significant group effect, by Dunn's test for group comparisons. 37,38 For organ weight data, an automated program was used to decide whether parametric or non-parametric group comparisons should be made. This program used Kolmogorov's test to examine the normality of the data and Bartlett's test to examine the homogeneity of variances. Accordingly, either Dunnett's test or Student's t-test for parametric group comparisons or Dunn's test or Wilcoxon's test (U-test) for non-parametric group comparisons were used. No statistical analysis was performed for nominal or ordinal data, e.g. microscopic examination data. In these cases, the data were compared with data recorded in the reference groups or with historical control data.
Results
About 300 μL of blood was collected from every animal (using blood microtubes with volume scale). The duration of submandibular blood collection in conscious animals was between 12 and 79 s (mean duration: 44 s, Table 1), whereas it was clearly shorter in anaesthetized animals (duration between 7 and 51 s/mean duration: 23 s). In some animals from both groups, the puncture needed to be repeated as the ideal site was not always found on the first attempt. Strong defence movements, i.e. pedalling or struggling motions of hindlimbs and/or forelimbs and/or of the head partly prevented a successful puncture and/or interfered with the blood flow in conscious animals. Therefore, repetition was necessary more often in conscious animals. In addition, excessive bleeding occurred in three animals, most likely due to puncture of the collateral artery.
Mean duration of blood collection and observed complications
During blood collection, anaesthesia with 2.5% isoflurane (Forene™, Abbott Laboratories SA, Switzerland; oxygen flow rate of 4 L/min) was applied
Sublingual blood collection was as quick as the submandibular in anaesthetized animals and was performed in 15–45 s (mean duration: 25 s; Table 1).
Sublingual blood collection was repeated in some animals since blood flow was limited or the animals recovered from the anaesthesia too quickly. The duration of blood collection in this group was comparable to that of anaesthetized animals of the submandibular.
Unlike the other groups, the sublingually punctured animals showed a slight body weight gain on the first day following blood collection (+1.7%). This difference achieved statistical significance as the submandibularly punctured animals showed a slight body weight loss (−2.2% for conscious animals and −1.7% for anaesthetized animals, respectively). Between days 2 and 3, this difference in body weight gain persisted without statistical significance and correlated to a slight increase in food consumption (without statistical significance) in sublingually punctured mice within the first three days (in comparison with the other groups). Over the entire observation period following blood collection, sublingually punctured animals gained body weight (+1.2%), whereas submandibularly punctured animals lost body weight (−1.7% for conscious animals and −1.8% for anaesthetized animals, summary data shown in Figures 1 and 2).
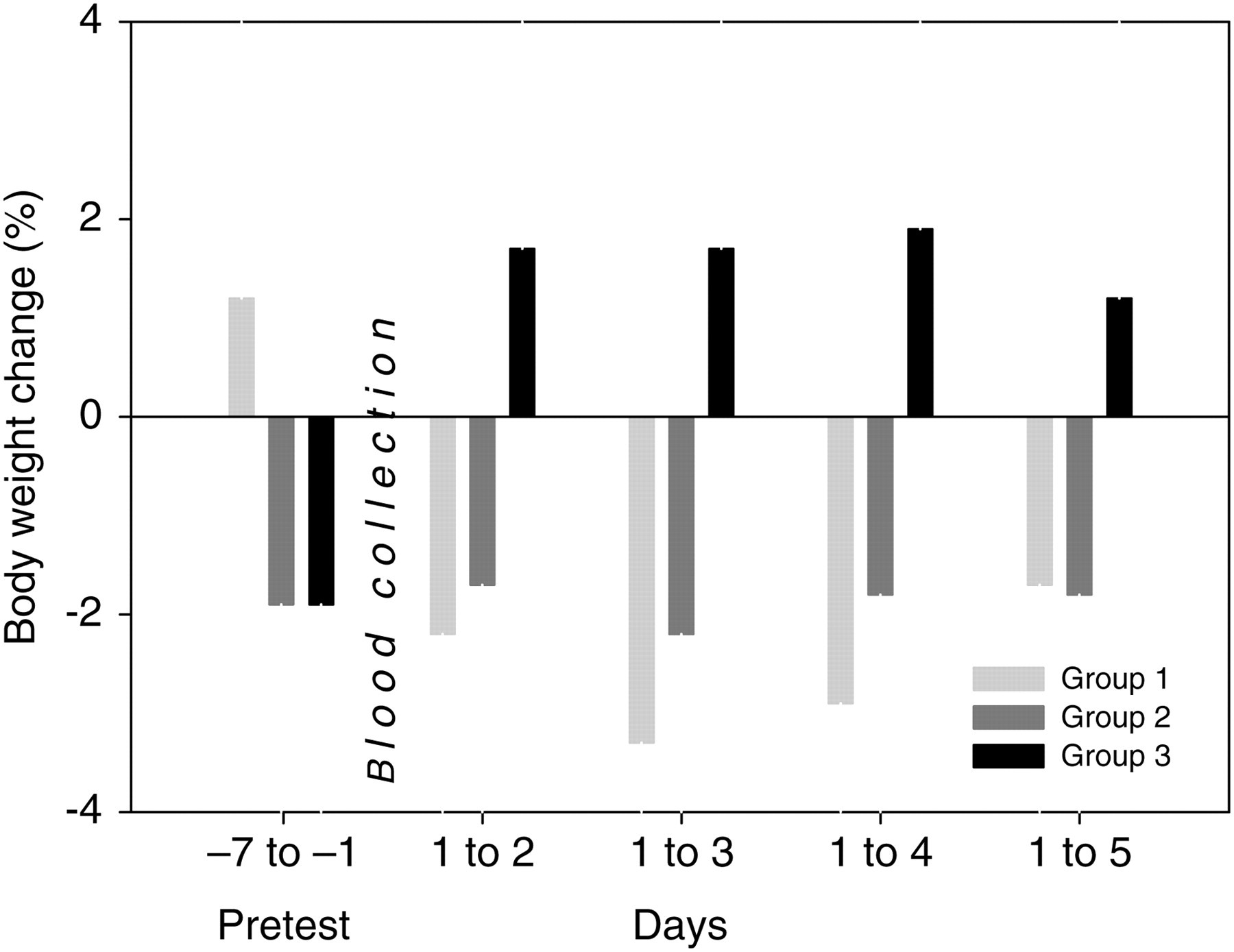
Mean body weight development per group is given in percent. Values of the observation period (days after blood collection) were related to the first day of blood collection, whereas preblood collection period (pretest period) was completely covered. Group 1: submandibular blood collection technique, no anaesthesia; group 2: submandibular blood collection technique, isoflurane anaesthesia applied; and group 3: sublingual blood collection technique, isoflurane anaesthesia applied
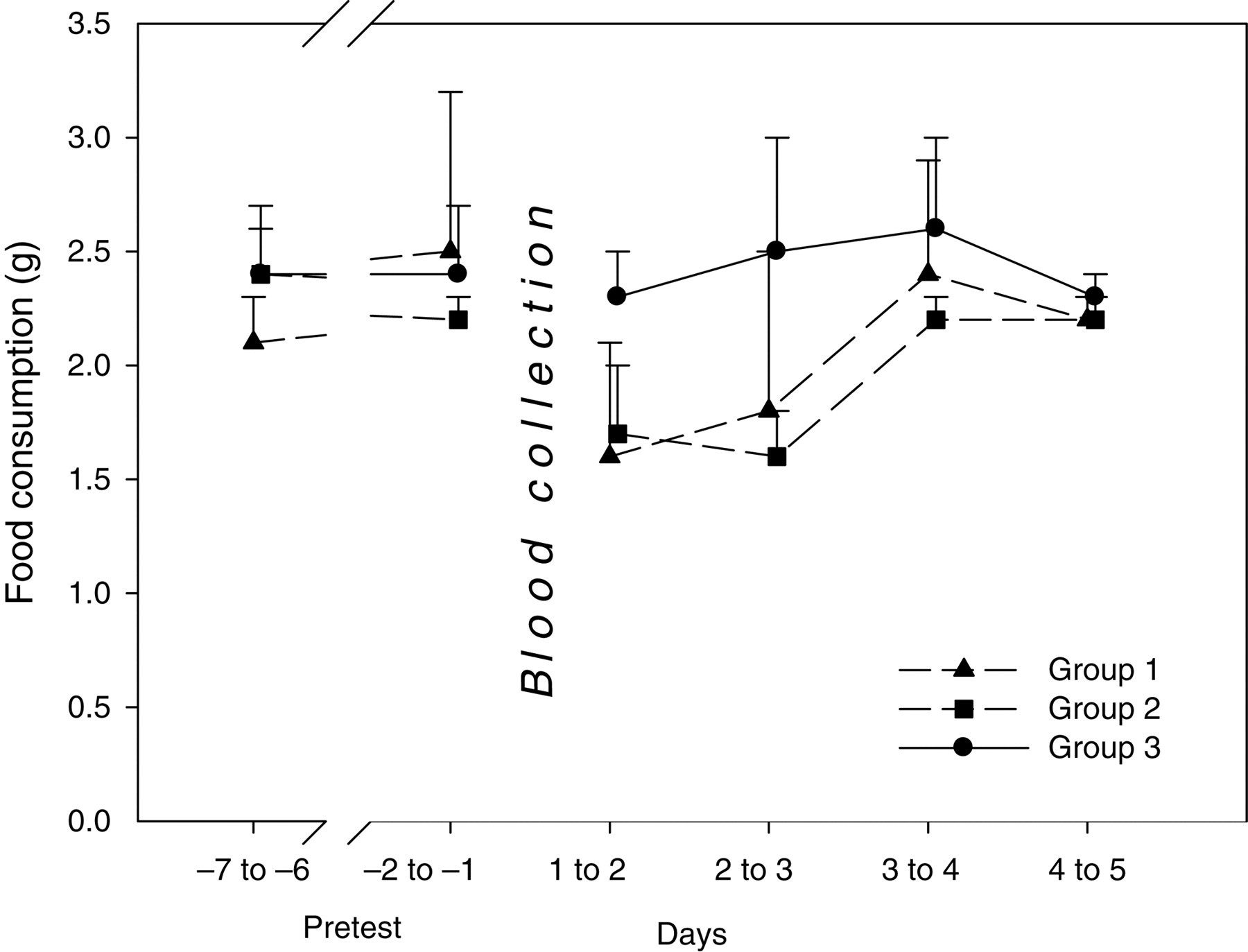
Mean values for food consumption are shown in gram per animal and day. For the pretest period, means of selected days are reported. Group 1: submandibular blood collection technique, no anaesthesia; group 2: submandibular blood collection technique, isoflurane anaesthesia applied; and group 3: sublingual blood collection technique, isoflurane anaesthesia applied
Platelet count could not be assessed in most of the conscious animals due to the quality of the samples (7/8, clumped platelets detected by the analyser), whereas the samples collected from anaesthetized animals showed clumped platelets less often (3/9/submandibular technique and 2/9/sublingual technique, respectively). Moderate decreases in red blood cell count, haemoglobin, haematocrit and total protein (mainly due to a decrease in albumin) and slight to moderate increases in glucose were observed in anaesthetized animals used for submandibular or sublingual technique when compared with conscious animals used for submandibular bleeding. In addition, slight to moderate increases in creatine kinase were observed in individual animals of the submandibular groups when compared with the sublingually punctured group (selected relevant data shown in Table 2).
Selected mean clinical pathology
Anaesthetized groups (submandibular or sublingual method) were compared with the conscious group (submandibular method) independent of the puncture technique. For statistical analysis, analysis of variance/Dunnett test (*P < 0.01; **P < 0.05) was used. Mean values are shown with standard deviation within parentheses. No data are shown for total white blood cells, differential white blood cells, mean corpuscular volume, mean corpuscular haemoglobin, mean corpuscular haemoglobin concentration, platelet counts, alanine aminotransferase, albumin, alkaline phosphatase and aspartate aminotransferase, since values were not considered to show relevant or significant changes
No relevant external macroscopic observations in the region of the head (submandibular method) or the tongue (sublingual method) were recorded. The extent and severity of traumatic lesions decreased with time in all groups (see Table 3). The incidence of effects and their severity were significantly higher in submandibularly punctured animals when compared with the other technique. In submandibularly punctured animals, macroscopic observations were mainly characterized by subcutaneous haemorrhage and oedema with an acute inflammatory response on day 1. In addition, acute focal head muscle necrosis was observed. Over time, the oedema and haemorrhage were resorbed and the inflammatory response became more chronic. Tissue damage such as muscle necrosis showed myophagocytosis and regeneration. Signs of traumatic lesions were still present after five days. On day 1, the traumatic lesions recorded in conscious submandibularly punctured animals were slightly more pronounced than those noted in anaesthetized animals used for the same blood collection method. They were mainly characterized by oedema, haemorrhage and focal to diffuse acute neutrophilic/fibrinoid inflammation. In addition, muscle necrosis and presence of thrombi were recorded only in individual conscious submandibularly punctured animals (Figure 3). On day 2, the extent and severity of traumatic lesions of both submandibular groups were comparable. However, the nature of the inflammation noted in conscious animals was similar to day 1, namely diffuse acute neutrophilic/fibrinoid, whereas the inflammatory response was more chronic in anaesthetized animals. The underlying causes of this different inflammatory response in anaesthetized animals were related to the presence of numerous hair fragments deep within the puncture site seen in four animals of this group. Hair fragments were surrounded by a mixed-cell population of inflammatory cells (neutrophils, lymphocytes, monocytes; Figure 4). The presence of hair fragments in the subcutis likely negatively influenced the healing of the tissue damage. On day 5, the traumatic lesions of both submandibular groups were comparable. The extent and severity of subcutaneous haemorrhage were significantly reduced and more focalized. Traumatic lesions and necrosis were noted in the tongue tissue in one animal sampled by the submandibular technique. No sublingual blood collection was performed in this animal. Therefore, this finding was considered to be secondary to a handling injury during submandibular blood collection.
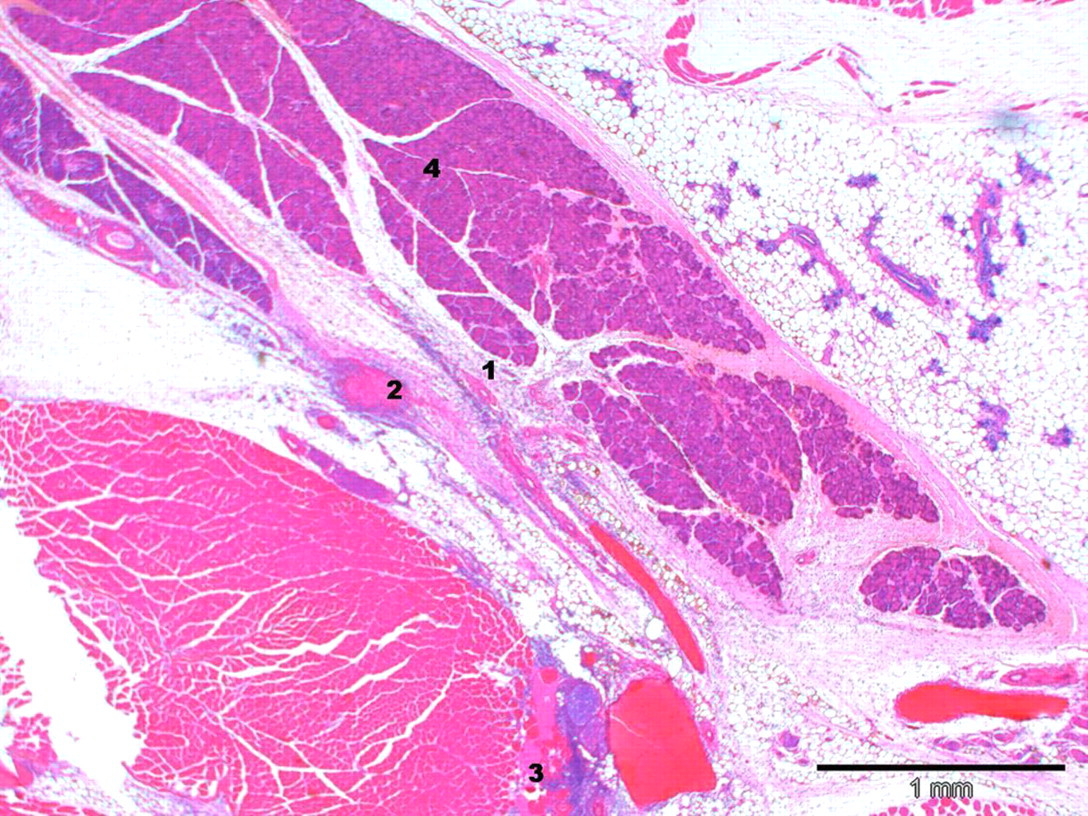
Lesions in a conscious submandibular punctured mouse 3 h after blood collection. Diffuse acute neutrophilic/fibrinoid inflammation (1) with moderate haemorrhage (2), slight muscle necrosis (3) and oedema of right puncture site. Salivary glands (4) are shown in the middle of this figure (staining: haematoxylin–eosin; magnification: ×4)
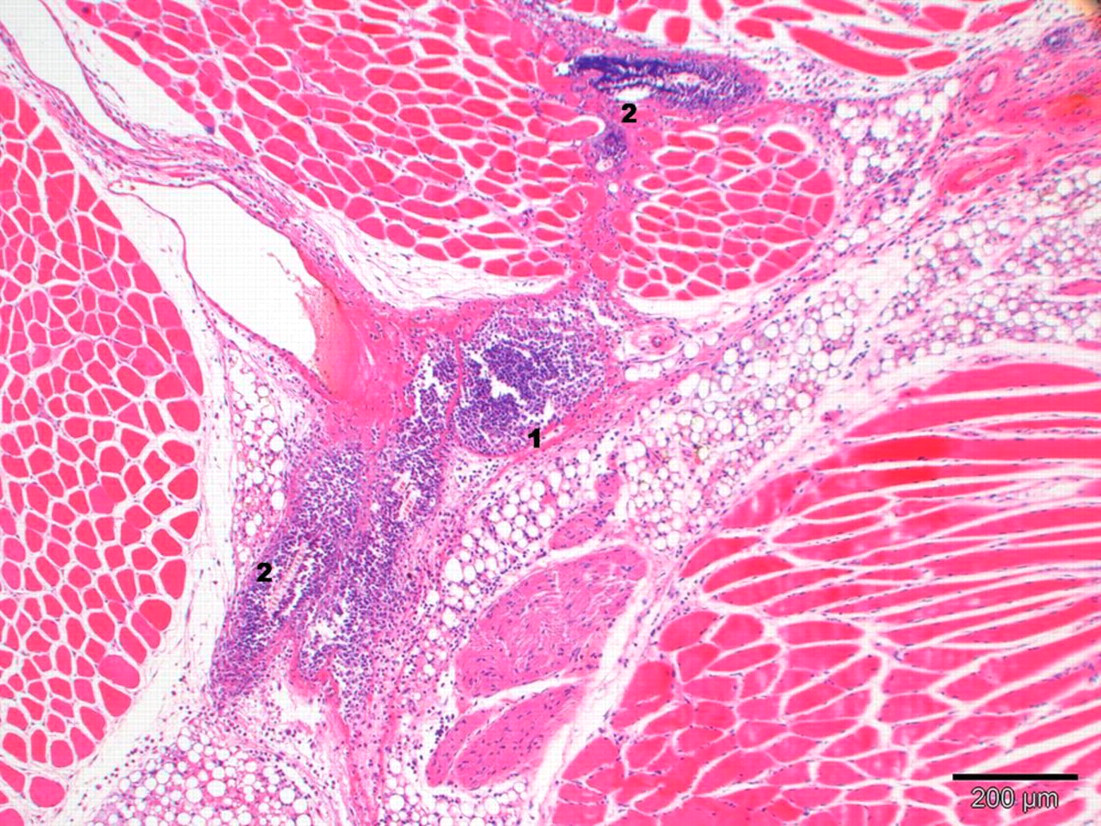
Lesions in an anaesthetized submandibular punctured mouse two days after blood collection. Multifocal mixed-cell inflammation (1), focal acute muscle necrosis, slight haemorrhage and moderate oedema and congestion of right puncture site. Hair fragments (2) are found deeply in the puncture site and are surrounded by a mixed-cell population of inflammatory cells (staining: haematoxylin–eosin; magnification: ×10)
Summary table of relevant microscopic findings after submandibular and sublingual blood collection
Relevant microscopic findings after submandibular or sublingual blood collection are shown (number of affected animals, average grading within parentheses)
Finding level for average grading: –: not found; grade 1: minimal; grade 2: slight; grade 3: moderate. Grading may relate to severity, incidence and/or size of an effect
The incidence of traumatic lesions was lower in sublingually punctured mice (when compared with the other technique). Traumatic lesions in this group were mainly characterized by minimal to slight haemorrhage on day 1 (with minimal acute inflammation in two female mice), resulting from tissue damage/vascular lesions (Figure 5). In two animals, minimal to slight haemorrhage was still present on day 2, with an inflammatory response and/or tongue muscle necrosis. Repairing processes (scar formation/granulation tissue) were observed after five days in these two animals.
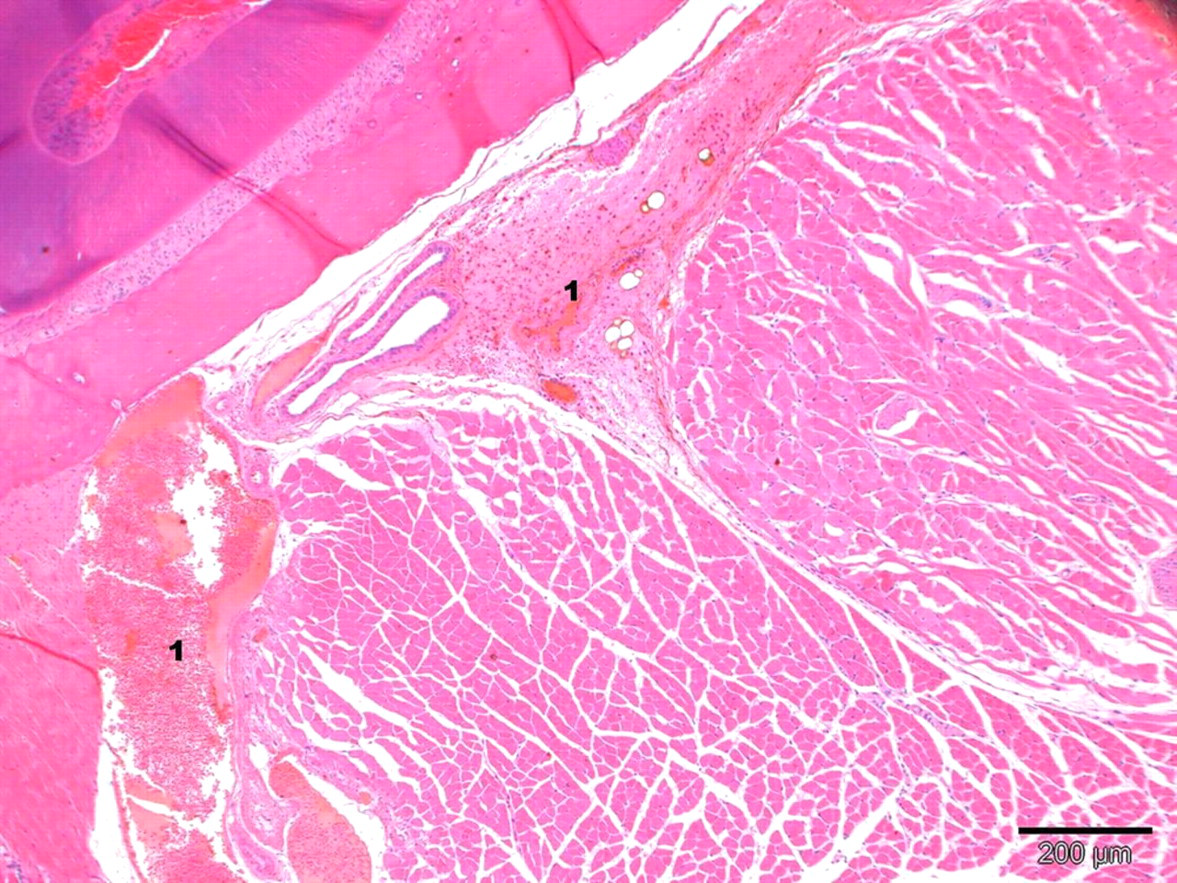
Lesions of the sublingual region in an anaesthetized sublingual punctured mouse 3 h after blood collection. Slight traumatic lesions with multifocal haemorrhage (1) and congestion are shown. In the left corner tooth tissue is displayed (staining: haematoxylin–eosin; magnification: ×4)
No relevant alterations related to the sampling methods, especially the submandibular method, were noted in the lacrimal gland, salivary glands and mandibular lymph nodes.
Discussion
Blood collection (either from the submandibular or sublingual vein) caused no mortality or noteworthy clinical signs during the five-day observation period after the puncture. About 300 μL of blood was collected from each animal. The mean duration for blood collection was comparable in anaesthetized mice irrespective of the technique used. In contrast, the mean duration of submandibular blood sampling in conscious animals was significantly longer due to struggling and defence movements, which resulted in a higher incidence of clumped platelets in the blood samples.
Another complication with the submandibular blood collection technique was the ideal puncture site at the right cheek was not always found on the first attempt in some mice (occurring at a higher incidence in conscious mice), since veins are covered by skin and are not visible during blood collection. Additionally, defensive movements in conscious animals interfered with the blood collection. Finally, excessive bleeding was noticed as a third complication in three submandibularly punctured mice, most likely due to a puncture of the collateral artery. This complication may result in the maximal permitted blood volume being inadvertently exceeded. Sublingual blood collection was performed with fewer complications, although it was necessary to repeat blood collection in some animals due to a limited blood flow or early recovery from the anaesthesia.
Over the entire observation period following blood collection, sublingually punctured animals showed a slight body weight gain, whereas submandibularly punctured animals slightly lost body weight (with statistical significance on the first day following the blood collection). Body weight gain in sublingually punctured animals correlated with increased food consumption during the first three days following the blood collection (without statistical significance) when compared with the other groups. More favourable body weight development correlating with increased food consumption indicates that the sublingual puncture technique has a lower impact on the wellbeing of the animal.
Decreases in red blood cell count, haemoglobin, haematocrit and total protein (mainly due to a decrease in albumin) were observed in anaesthetized groups (independent of the puncture technique) when compared with the conscious group. These findings were likely related to anaesthesia. Increases in glucose seen in sublingually punctured mice were not considered to be effected by salivary contamination based on a comparative study using sublingual and retrobulbar blood collection in rats. 28 This study revealed no significant differences in amylase activities in blood taken from the sublingual vein or the retrobulbar plexus. Glucose values were significantly higher in sublingually collected blood, indicating no negative impact of salivary enzymes on this parameter. In addition, one should consider a technical aspect that might prevent the samples from salivary contamination: the use of cotton tip during sublingual blood collection. The cotton tip enables the optimal fixation of the tongue before the sublingual vein is punctured, and it may also absorb the majority of the saliva. Based on these considerations and since glucose values of anaesthetized submandibular and sublingual groups differed minimally, the increase of glucose was considered to be related to anaesthesia. Finally, slight to moderate increases in creatine kinase observed in individual animals in groups 1 and 2 (submandibular technique) were consistent with muscle injury and correlated with the microscopic findings.
The extent and severity of lesions after submandibular and sublingual blood collection decreased with time. On day 1, tissue damage was noted in all animals. However, in sublingually punctured animals, the findings were limited to minimal traumatic lesion, haemorrhage and acute inflammation. These findings resulted from tissue damage (vascular lesions) during blood puncture and their severity was significantly lower compared with submandibularly punctured groups. In two animals of the sublingual group, minimal haemorrhage was still present on day 2, with a minimal acute inflammatory response and/or tongue muscle necrosis. Repairing processes (scar formation/granulation tissue, absence of inflammation and signs of inflammation) were observed in two out of six sublingually punctured animals after five days, whereas slight chronic inflammation (with slight oedema and slight haemorrhage) was still observed in most submandibular animals five days after puncture.
After submandibular puncture, the traumatic lesions were mainly characterized by subcutaneous haemorrhage and oedema that were slightly more pronounced in conscious animals as compared with anaesthetized animals. In addition, muscle necrosis (including myophagocytosis and regeneration) and presence of thrombi were recorded only in individual conscious animals. These effects (lesions that are more pronounced, muscle necrosis and thrombi) may relate to defence movements of the conscious animals during blood collection.
From days 1 to 5, the extent and severity of traumatic lesions in both groups sampled for the submandibular technique were comparable. However, the nature of the inflammation noted in the group of conscious animals on days 1 and 2 was diffuse acute neutrophilic/fibrinoid, whereas the inflammatory response in anaesthetized animals was minimally more pronounced and changed to a more subacute response, likely related to the presence of numerous hair fragments deeply within the injection site that negatively impacted the healing of the tissue damage.
In conclusion, about 300 μL of blood was collected from anaesthetized or conscious mice using either the sublingual or submandibular blood collection technique. Due to defence movements, submandibular blood collection in conscious mice took more time, induced more complications and in several animals a poor quality blood sample was obtained due to the prolonged duration of blood collection. In addition, defence movements of the conscious animals during blood collection may have caused lesions that were more pronounced as compared with anaesthetized animals. Sublingually punctured mice consumed more food and gained more body weight than submandibularly punctured mice, most likely due to less severe tissue lesions and improved healing processes. Based on these results, we recommend the sublingual blood collection technique to be used in mice. However, in cases where submandibular bleeding is to be used, this technique should be performed in anaesthetized mice, for animal welfare reasons and an improved quality of blood sample.
