Abstract
Invasive lung function measurements are useful tools to describe respiratory disease models in mice but only result in one time-point measurements because of tracheostomy. We explored if intubation may overcome the need for tracheostomy thereby allowing invasive lung function monitoring of individual mice over time. Repeated invasive lung function measurements with Scireq© – FlexiVent or Buxco© – Forced Pulmonary Maneuvers® were performed three times in BALB/c mice with intervals of 10 days. Each lung function assessment following intubation was compared with a similar measurement in age-matched tracheostomized mice, the golden standard in lung function measurements. Tracheostomy and intubation gave similar results for resistance, elastance and compliance of the whole respiratory system as assessed by Flexivent. Likewise, Forced Pulmonary Maneuvers used to measure lung volumes such as total lung capacity, functional residual capacity, forced expiratory volume in 0.1 s and forced vital capacity, resulted in identical outcomes for both airway approaches. No interaction was found between the procedures for any of the pulmonary function variables. The observed changes over time were rather related to animal growth than to repetitive intubation. Eighty percent of the animals survived three consecutive intubations, which were hampered by transient breathing difficulties, weight loss and neutrophilic bronchoalveolar lavage immediately postextubation. Repetitive invasive lung function measurements by intubation are feasible and reproducible in healthy mice and results are comparable to the standard method. This may open new perspectives for longitudinal research in animal models of respiratory diseases.
Measuring pulmonary function in mice remains challenging, despite the major technical improvements of different commercial equipments. 1–5 Until now, no method is optimal in all regards and precision must be traded off against non-invasiveness. 3 Nowadays, non-invasive, unrestrained whole body plethysmography allows easy and repeatable measurements in mice without anaesthesia, but many have questioned the validity and accuracy of this method. 3,6,7 Invasive techniques, which directly measure pulmonary and airway mechanics, result in more precise and physiological variables but have the major drawback of tracheostomy and consequent sacrifice allowing no monitoring of mice over time.
Brown et al. 8 were the first to describe the measurement of respiratory resistance and compliance in intubated mice. However, these parameters were only assessed at one time point. Consequently, Glaab et al. 9 showed that repetitive measurements of respiratory resistance and compliance were feasible in intubated and spontaneously breathing mice. Still, many other pulmonary function variables assessed by Buxco© – Force Pulmonary Maneuvers® and Scireq© – FlexiVent® forced oscillations require ventilatory manoeuvres instead of spontaneous breathing. So far, the repetitive application of these manoeuvres, which generate high differential pressures in the airways, has not been explored in intubated mice. These ventilator-dependent variables such as airway resistance (Rn), tissue damping (G) and tissue elasticity (H), and more clinical variables such as static compliance (C st), forced expiratory volume in one second (FEV1), forced vital capacity (FVC), functional residual capacity (FRC) and total lung capacity (TLC) may though better describe obstructive and restrictive respiratory disease models. 10 As long as these variables are only available at the moment of sacrifice, their potential benefit for monitoring disease progression or therapeutic interventions remains questionable. Therefore, repeated measurements of the above-mentioned variables in the same animals would offer clear advantages for longitudinal respiratory research in mice.
The aim of this study was first to assess the feasibility of repeated intubation procedures and consequent lung function measurements with Buxco© – Force Pulmonary Maneuvers® or Scireq© – FlexiVent® forced oscillations methods. Secondly, we aimed to compare this new approach of repeated intubation and pulmonary function tests with the current and standard practice of endpoint measures in tracheostomized mice.
Methods
Animals
Male BALB/cJ mice (∼25 g, 10 weeks old) were obtained from Elevage Janvier (St Berthevin, France). All animals were housed in a conventional animal house and placed in sterilized type 2 plastic cages with filter top (267 mm × 207 mm × 140 mm, floor area 370 cm2) containing bedding (Lignocel® S8/15, J Rettenmaier & Söhne, Rosenberg, Germany). Tissues and toilet paper rolls were given as enrichment. The mice were housed in groups during the whole experiment. Mice were supplied with pelleted food (Ssniff® R/M-H, Soest, Germany) and filtered and UV treated water ad libitum. All experimental procedures were approved by the ethical committee of animal experiments of the University of Leuven.
Experimental design
For each device (Scireq© – FlexiVent and Buxco© – Forced Pulmonary Maneuvers) an intubation group and a tracheostomy group was included making a total of four groups. Animals were divided as followed: two intubation groups for repeated manoeuvres (Scireq© – FlexiVent or Buxco© – Forced Pulmonary Maneuvers) were compared with their respective group of age-matched tracheostomized mice. The study protocol is shown in Figure 1. In the intubation group (n = 14 for Scireq© – FlexiVent® and n = 11 for Buxco© – Forced Pulmonary Maneuvers®), mice were anaesthetized and intubated orotracheally on three consecutive time points with 10-day intervals. All mice were anaesthetized with a mixture of medetomidine (1 mg/kg, Domitor®, Pfizer Animal Health, Belgium) and ketamine (75 mg/kg, Ketalar®, Pfizer, Brussels, Belgium) intraperitoneally. Intubation was performed with an 18G, 30 mm catheter (BD Insyte, Madrid, Spain) through direct visualization of the vocal cords as described by Brown et al. 8 Lung function measurements in intubated mice were either performed with Scireq© – FlexiVent® (Montreal, QC, Canada) or with Buxco© – Forced Pulmonary Maneuvers® (Buxco Research Systems©, Wilmington, NC, USA) and compared with similar manoeuvres in tracheostomized, age-matched mice (n = 6) on each time point. After pulmonary function measurements, mice received an antidote (intubation) or were sacrificed through exsanguination (tracheostomy). Antidotation was performed with atipamezole (0.5 mg/kg Antisedan®, Pfizer Animal Health). Mice were weighed every other day and general health was checked daily. Bronchoalveolar lavage (BAL) and histology were performed in the tracheostomy group on days 0, 10 or 20 and directly after the last intubation (day 20) in the intubated group. In addition, in a subgroup of five mice, BAL and histology were performed seven days after the last intubation to see whether inflammatory cells present in BAL disappear over time.
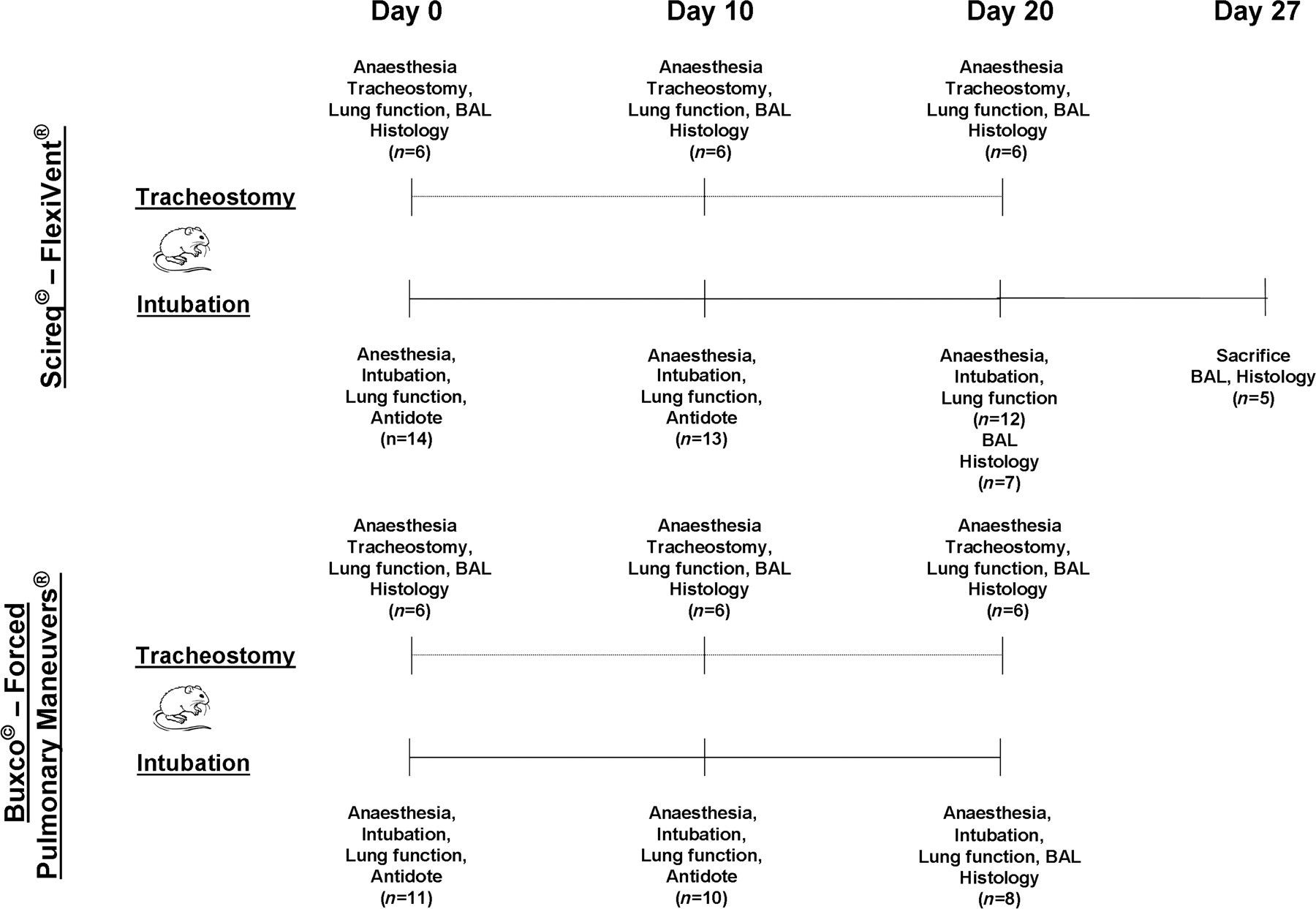
Study protocol. Two intubation groups for repeated manoeuvres (Scireq© – FlexiVent or Buxco© – Forced Pulmonary Maneuvers) were compared with their respective group of age-matched tracheostomized mice. Lung function measurements in intubated mice were performed on days 0, 10 or 20. n represents the number of surviving mice at a given point in time
Scireq© – FlexiVent®
Both intubation and tracheostomy were performed with an 18G intravenous catheter. To avoid spontaneous breathing, the animals were deeper anaesthetized with a low dose of pentobarbital (Nembutal®, CEVA Santé Animale, Brussels, Belgium). Next, mice were connected to the FlexiVent system and ventilated at a frequency of 150 breaths/min as previously described. 10 During the maximal vital capacity (MVC) perturbation, the lungs were inflated to a standard pressure of +30 cmH2O and then deflated to determine MVC. With the snapshot perturbation manoeuvre, a three-fold sinusoidal wave of inspiration and expiration was imposed, resulting in resistance (R), compliance (C) and elastance (E) of the whole respiratory system (airways, lungs and chest wall). Maximal pressure–volume-loops (PV-loops) between TLC (+30 cmH2O) and FRC were finally generated to obtain C st and static elastance (E st) of the respiratory system. In every animal, each manoeuvre was repeated until three acceptable measurements (coefficient of determination ≥0.95) were recorded. The average of three acceptable measurements was calculated.
Buxco© – Forced Pulmonary Maneuvers®
Tracheostomy was performed with a standard catheter (CNS5002) provided by the Buxco equipment. Dead space was adjusted for the type of catheter: 0.85 mL for the standard catheter and 1 mL for the 18G intravenous catheter. After intubation or tracheostomy, mice were placed in a body plethysmograph and connected to a computer-controlled ventilator as described previously. 10 Briefly, a breathing frequency of 150 breaths/min was imposed by pressure control ventilation until a regular breathing pattern was obtained. Mice were then forced to breathe spontaneously against a closed valve at the mouth, by which FRC could be determined. During the quasistatic pressure–volume manoeuvre, the lungs were inflated to a standard pressure of +30 cmH2O (TLC) and then slowly exhaled until a negative pressure of −30 cmH2O was reached (residual volume or RV). Different lung volumes like TLC, RV and vital capacity (VC) were then determined. With the fast flow volume manoeuvre, lungs were first inflated to TLC and immediately afterwards connected to a highly negative pressure (−30 cmH2O) in order to enforce expiration until RV. This manoeuvre allowed the measurement of forced expiratory flows (peak expiratory flow or PEF) and volumes such as FVC and forced expiratory volume at 100 ms (FEV0.1). In every animal, each manoeuvre was repeated until three acceptable measurements were recorded, of which the average was then calculated.
BAL and histology
BAL was performed with three aliquots of 0.7 mL sterile saline. Total cell counting was performed using a Bürker haemocytometer and cytospins (Shandon) with May-Grünwald-Giemsa staining were made for differential cell counting. After BAL, the heart–lung block was excised and fixed in 4% paraformaldehyde at a constant hydrostatic pressure of 20–25 cm fluid column for at least 20 min. Paraffin sections were stained with haematoxylin–eosin and lung damage and inflammation were evaluated by a pathologist (EV) in a blinded manner.
Statistical analysis
All data are presented as mean (±standard deviation). Differences within and between intubated and tracheostomized mice were tested using two-way analysis of variance. Mean total and differential cell countings were compared by unpaired Student's t-test. All statistical analyses were performed with SAS 9.1 (SAS Institute, Cary, NC, USA) or Graphpad Prism 4 software (San Diego, CA, USA). A P value of <0.05 was considered significant.
Results
General health
Five of the 25 animals included in the intubation group died during one of the procedures (survival rate of 80%). Four of them did not wake up from the anaesthesia after the lung function measurement (2 on day 0 and 2 on day 10). One animal died during the intubation procedure on day 10. Most of the surviving animals experienced some difficulties of breathing during 1–2 days following intubation. All animals lost weight after intubation with a mean weight loss of 1.48 g after the first intubation and 1.06 g after the second intubation. Body weight was restored within one week after intubation and did not significantly differ from the weight of the tracheostomized group throughout the experiment (P = 0.087) (Figure 2).
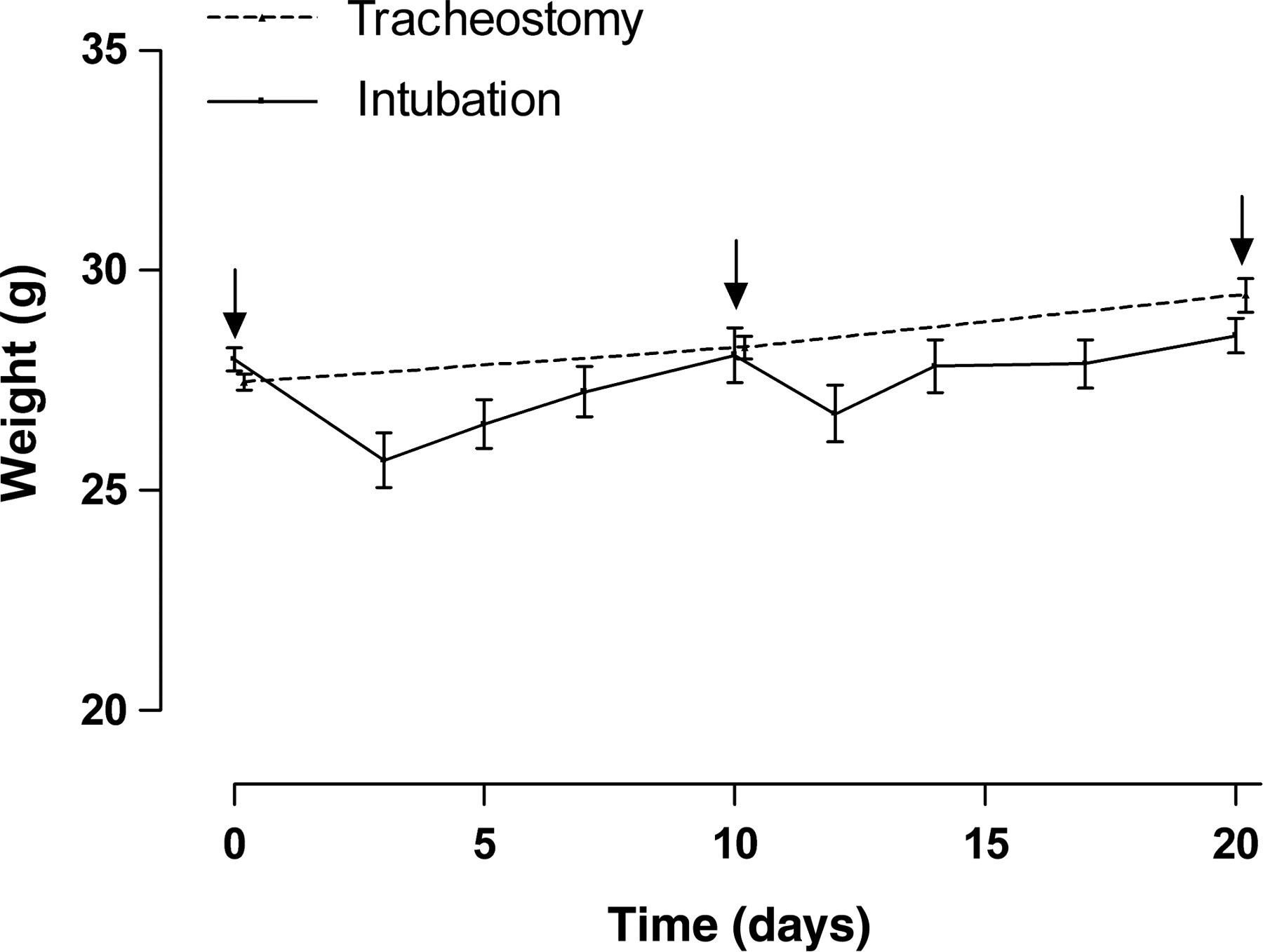
Weight evolution over time from the tracheostomy and intubation group. Repeated intubations were performed on days 0, 10 and 20. Arrow indicates day of lung function measurement. The transient weight loss in the intubation group was restored within one week and weight was never significantly different between both groups (two-way ANOVA test, P = 0.087). Data are shown as mean ± SD
Scireq© – FlexiVent®
Data of the TLC, snapshot and PV-loop with increasing pressure are summarized in Table 1. MVC was not significantly different between the two groups. Mild differences between intubated mice and tracheostomized animals were obtained for R, C and E of the respiratory system, which reached statistical significance for R (P = 0.042), and E (P = 0.049) and but not for C (P = 0.079) (Figure 3a). Time did not significantly affect these variables, but the small and consistent changes observed for both groups, likely resulted from the growth of the animals. For the PV-loops, important differences were observed with a statistically significant lower C st (P < 0.0001), lower K (P < 0.0001), lower hysteresis (P = 0.009) and higher E st (P < 0.0001) for the intubated mice when compared with tracheostomized animals (Figure 3b). It should be stressed however, that reliable PV-loops were almost impossible to obtain in the intubated mice as they required a deeper anaesthesia, which resulted in death after disconnection from the ventilator in pilot experiments. Indeed, as illustrated in Figure 4, PV-loops of the intubated mice were all disturbed by spontaneous breathing at the end of the manoeuvre. For similar reasons, forced oscillation variables were impossible to obtain in intubated animals (data not shown).
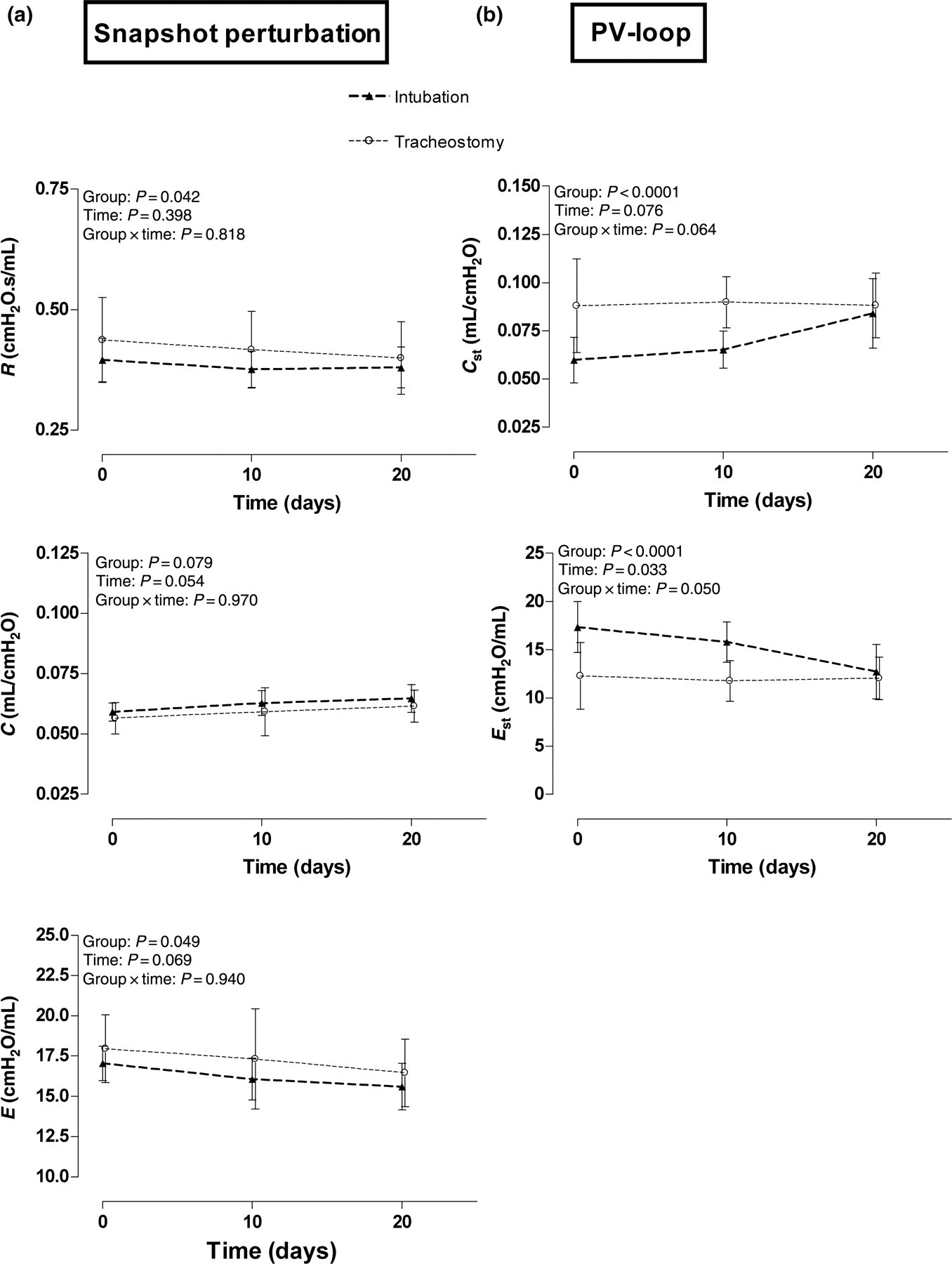
Results of Scireq© – FlexiVent. (a) The variables resistance (R), compliance (C) and elastance (E) of the respiratory system. (b) Static lung compliance (C st) and static lung elastance (E st). All variables were compared between and within intubation and tracheostomy groups. Data are presented as mean ± SD. P < 0.05 was considered as significant
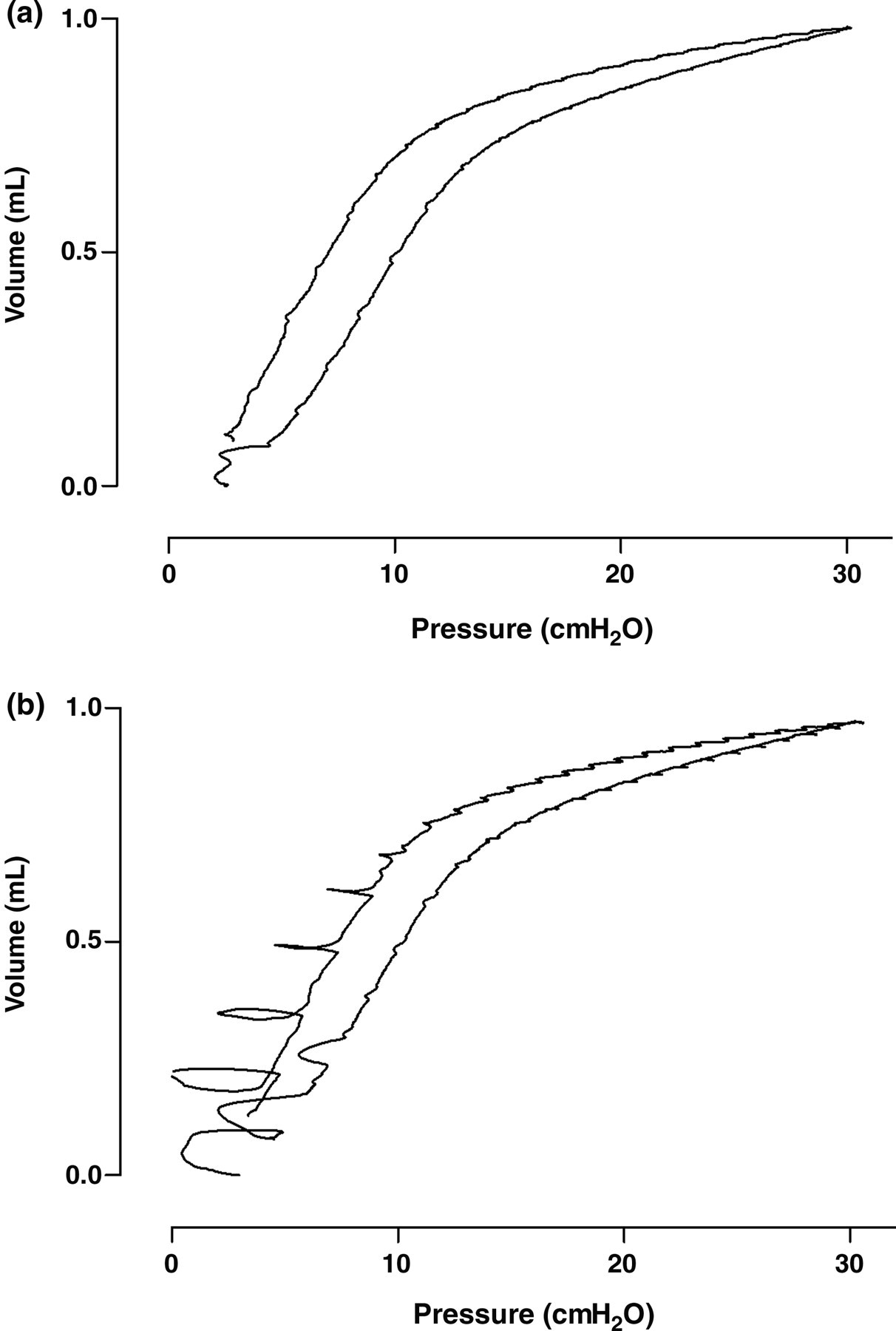
Illustrative figure of the pressure–volume (PV) loop assessed by Scireq© – FlexiVent. (a) PV-loop of tracheostomized mouse. (b) PV-loop of intubated animal with a different depth of anaesthesia
Different mechanical parameters obtained with the FlexiVent system
A two-way analysis of variance (ANOVA) test was performed for the statistical results. P < 0.05 was considered as significant. Group: differences between groups without taking time into account; time: evolution in time, groups pooled; group × time: evolution in time compared between groups. Data are presented as mean ± SD. Significant P values are indicated in bold
MVC: maximal vital capacity; C st: static compliance; E st: static elastance; K: shape parameter of deflation pressure–volume (PV) curve; hysteresis: difference in the pressure–volume relationship during the inflation and deflation curve of a PV-loop
Buxco© – Forced Pulmonary Maneuvers®
Data from the different manoeuvres are summarized in Table 2. No significant differences between intubated and tracheostomized mice were seen for the different lung volumes such as FRC, VC, TLC, RV, FVC and FEV0.1. When looking at time, lung volumes such as VC (P = 0.007), TLC (P = 0.018), FVC (P = 0.003) and FEV0.1 (P = 0.007) significantly increased over the course of the experiment in both groups due to growth of the animals. PEF and static lung volumes such as RV and FRC did not significantly change during the experiment (Figure 5). Finally, a representative example of the fast flow–volume curve is given in Figure 6. This figure explains the statistically significant difference in PEF between both groups due to a difference in diameter and consequently resistance between both catheters.
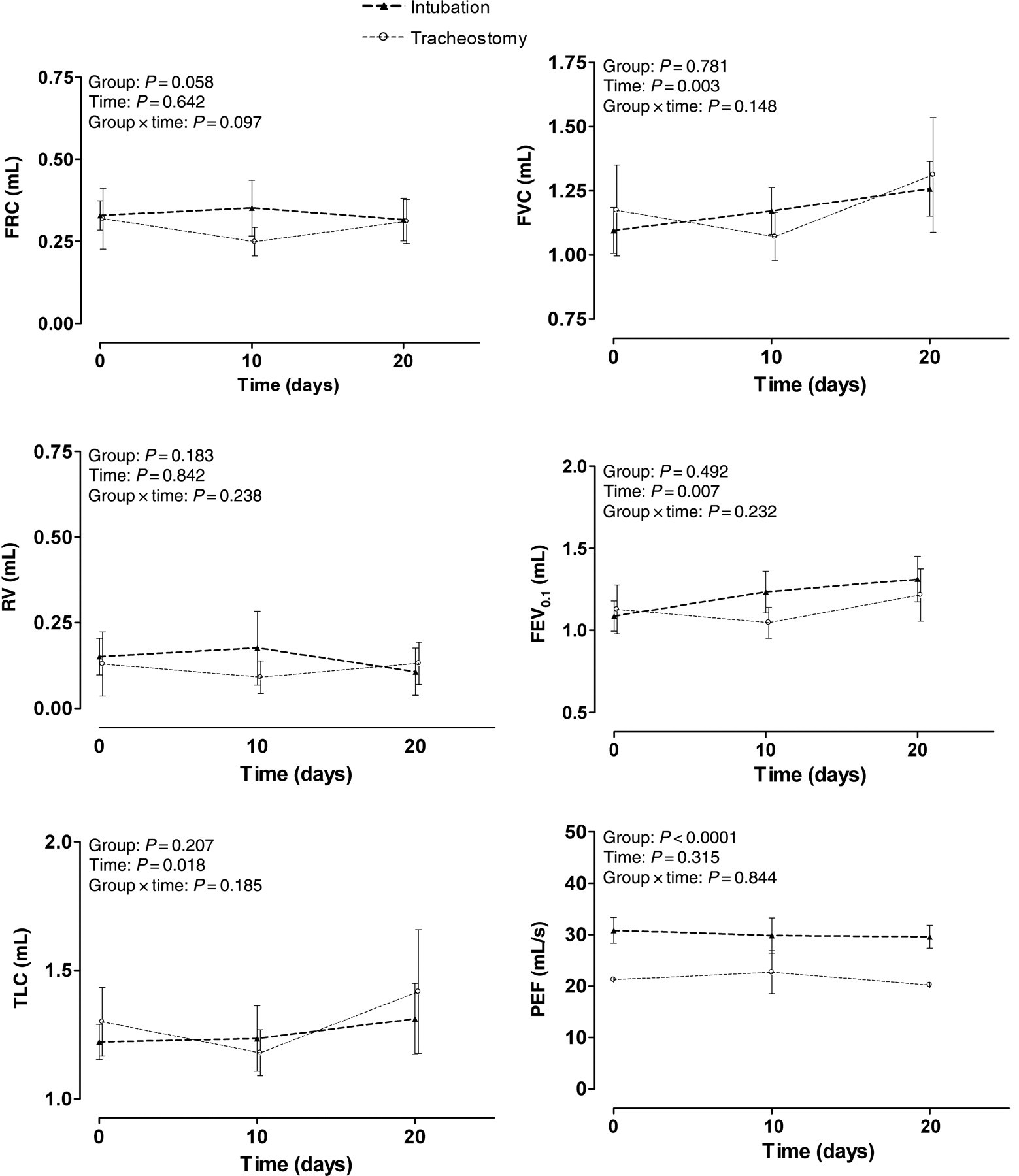
Results of Buxco© – Forced Pulmonary Maneuvers. The variables functional residual capacity (FRC), residual volume (RV), total lung capacity (TLC), forced vital capacity (FVC), forced expiratory volume at 100 ms (FEV0.1) and peak expiratory flow (PEF) were compared between and within intubation and tracheostomy groups. Data are presented as mean ± SD. P < 0.05 was considered as significant
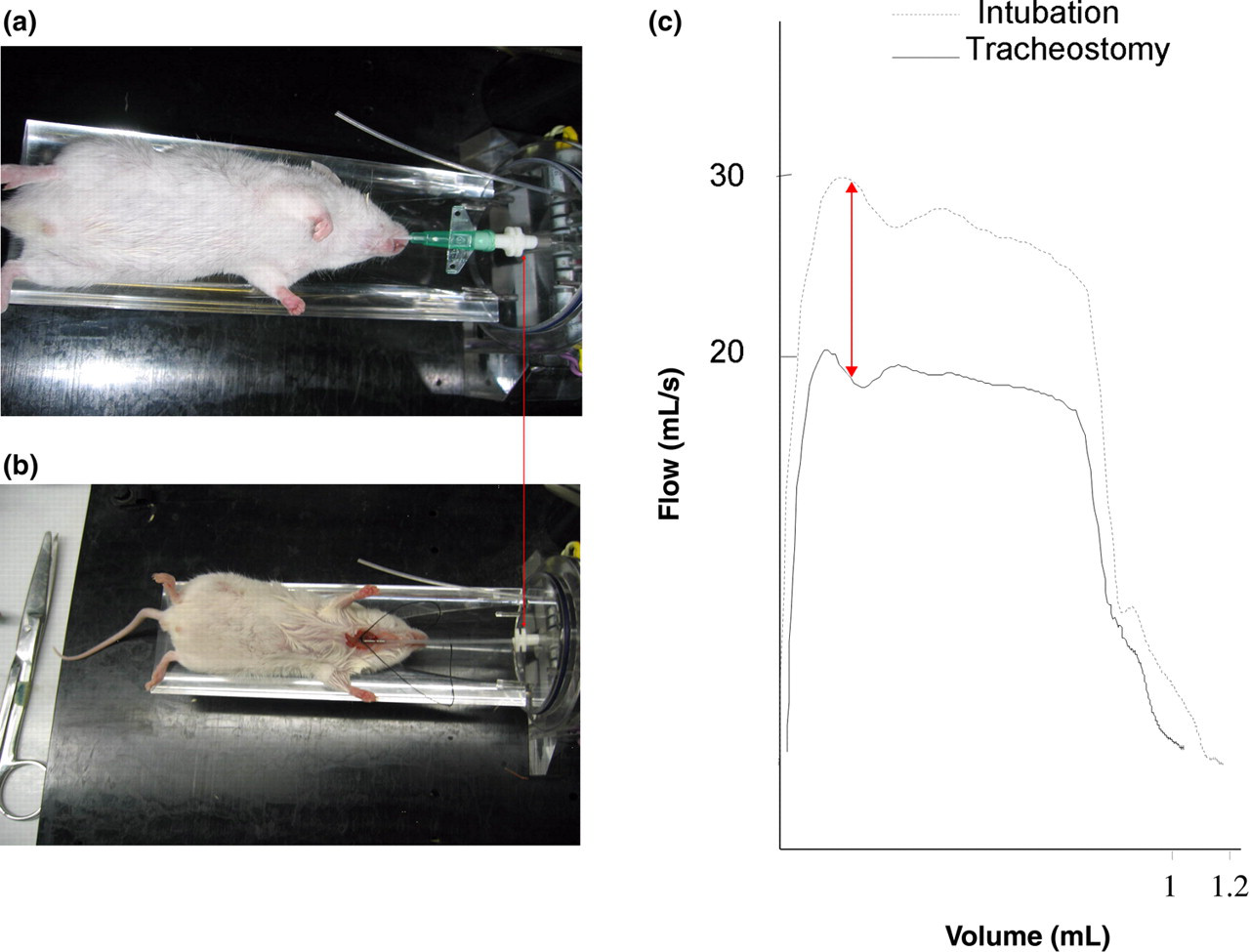
Buxco© – Forced Pulmonary Maneuvers. Left panel: (a) Intubated mouse, (b) tracheostomized mouse. Right panel: illustrative figure of fast flow–volume curve for an intubated and tracheostomized mouse. Arrow indicates difference in peak expiratory flow because of difference in diameter between two catheters
Different function parameters obtained with the Forced Buxco Maneuver system
A two-way analysis of variance (ANOVA) test was performed for the statistical results. P < 0.05 was considered as significant. Group: differences between groups without taking time into account; time: evolution in time, groups pooled; group × time: evolution in time compared between groups. Data are presented as mean ± SD. Significant P values are indicated in bold
FRC: functional residual capacity; VC: vital capacity; RV: residual volume; TLC: total lung capacity; FVC: forced vital capacity; FEV0.1: forced expiratory volume in 0.1 s; PEF: peak expiratory flow
BAL cell counts and histology
Total cell counts were not different between tracheostomized (n = 36) and intubated mice (n = 15) (P = 0.237). Differential cell counting showed a statistically significant shift towards an increased percentage of neutrophils in intubated mice versus tracheostomized mice (5% versus 1%, P < 0.0001). When looking at the BAL fluid obtained from animals that were sacrificed seven days after the last intubation (n = 5), the differential cell count was restored to normal values (Table 3). Despite these temporary changes in the BAL cellular profile, histological sections of all mice lungs were normal with no signs of inflammation at any time during the experiment.
Mean (±SD) of total and differential cell counts in bronchoalveolar lavages from the control and intubation group
An unpaired Student's t-test was performed for the statistical results. P < 0.05 was considered as significant. Significant P values are indicated in bold
Discussion
The present study investigates the possibility of repeated invasive lung function measurements in mice with two of the most commonly used equipment, namely Forced Pulmonary Maneuvers (Buxco©) and FlexiVent forced oscillations (Scireq©). We found that most of the pulmonary function variables obtained by standard methodology (tracheostomy and sacrifice) can be correctly reproduced on a repetitive basis with intubation.
The FlexiVent forced oscillations (Scireq©) showed very reproducible results for the snapshot perturbation manoeuvre. R, E and C of the whole respiratory system in intubated mice were slightly different from tracheostomy but followed exactly the same evolution over time. As these measures are often used in asthma models to investigate bronchial hyper-reactivity following methacholine challenge, 11,12 our technique of repetitive measures in single animals may offer great advantages in the long-term follow-up of these models. When considering PV-loops and forced oscillation variables however, the data obtained in tracheostomized mice could not be reproduced in intubated animals. For these manoeuvres a more profound anaesthesia was required, which resulted in a high mortality after extubation in pilot experiments. When anaesthesia was less profound, forced oscillation manoeuvres were no longer possible as PV-loops were severely disturbed by spontaneous breathing at the end of the expiration. At present, we are not able to overcome this problem but it is anticipated that a better tuned anaesthesia may resolve this in the near future. Low doses of muscle relaxants, central or peripheral, could be added to the anaesthesia protocol or could be given instead of the barbiturates. Of course, such animals should only be disconnected from the ventilator when spontaneous breathing has fully returned.
The Forced Pulmonary Maneuvers method is designed to measure static and dynamic lung volumes and flows. Although some mild variation was present in tracheostomized animals, variables such as FRC, RV, TLC, FVC and FEV0.1 were consistently reproduced with the intubation technique. The significant increase in PEF in intubated animals might be explained by the larger diameter and lower resistance of the intubation catheter, which obviously did not result in larger expiratory volumes because of the shorter time of expiration. At present, lung volumes and flows are not routinely used in animal models of respiratory diseases. Their promising potential in the long-term follow-up of animal models of emphysematous and interstitial lung disease 10 may certainly increase with the current validation of repeated measures by intubation.
The strength of our study design is that each lung function assessment following intubation was compared with a similar measurement in age-matched but tracheostomized mice. Although we found some mild but statistically significant differences between the intubated and tracheostomized mice, no statistical interaction was found between the procedure (intubation) and the change of lung function measures over time for any of the assessed variables. Indeed, the observed changes over time, which relate to the expected growth of animals, were not influenced by the intubation procedure itself as they followed the same course in intubated and tracheostomized animals. However, when considering the differences in lung function variables obtained by intubation or tracheostomy at a given point in time, it was clear that in the case of the Flexivent forced oscillations (Scireq©) method the depth of anaesthesia explained the observed variation, whereas for the Forced Pulmonary Maneuvers (Buxco©) a different catheter size was responsible. It should be stressed that the statistically significant differences between both approaches were still very mild, certainly when opposed against anticipated differences between health and disease. Together, our data demonstrate that variables obtained with intubation are reproducible and reliable over the course of a long-term experiment but that caution should be taken when absolute values of the intubation procedure are compared with those of a standard procedure.
The technique of repeated lung function measures by intubation has some shortcomings. We were forced to use 18G catheters instead of 20G to avoid leakage and pressure drops during ventilation and respiratory manoeuvres. These larger catheters complicate intubation and may induce more difficulty in breathing (stridor) afterwards. The complexity of intubation depends not only on the catheter size but also on the experience of the manipulator and the anaesthesia. In our experiments, the overall survival was 80% after three consecutive intubations, which is acceptable but at the same time also limiting for the number of assessments in a long-term study design. All surviving animals lost weight after the procedure, which was restored within one week postintubation. Similarly, a transient shift towards neutrophils was observed in the BAL fluid. From these side-effects, we suggest that after intubation and invasive measurements a recovery phase of at least one week should be respected when using our approach to monitor chronic experiments. As the same technique and anaesthesia are now integrated in our standard laboratory measurements of pulmonary function, overall mortality in these long-term experiments with repeated measurements has dropped down to less than 10%. It indicates that insufficient experience in the intubation technique is likely the most limiting factor for a broad application.
In conclusion, the present study is the first to demonstrate that repetitive invasive pulmonary function assessments in mice are feasible and reproducible over time. Our data warrant the validation of this new approach in the long-term monitoring of different chronic respiratory disease models.
Footnotes
ACKNOWLEDGEMENTS
The authors would like to thank Hans Scheers for the help in the statistical analysis. The project was supported by a grant from the ‘Fonds voor Wetenschappelijk Onderzoek Vlaanderen’ (FWO) (FWO G.0643.08, G.0723.10, G.0687.09). GMV is supported by FWO. BV, JV and WJ are postdoctoral research fellows of the FWO, RV is a PhD fellow of the FWO.
