Abstract
Streptozotocin (STZ)-induced diabetes mellitus (DM) offers a very cost-effective and expeditious technique that can be used in most strains of rodents, opening the field of DM research to an array of genotypic and phenotypic options that would otherwise be inaccessible. Despite widespread use of STZ in small animal models, the data available concerning drug preparation, dosing and administration, time to onset and severity of DM, and any resulting moribundity and mortality are often limited and inconsistent. Because of this, investigators inexperienced with STZ-induced diabetes may find it difficult to precisely design new studies with this potentially toxic chemical and account for the severity of DM it is capable of inducing. Until a better option becomes available, attempts need to be made to address shortcomings with current STZ-induced DM models. In this paper we review the literature and provide data from our pancreatic islet transplantation experiments using single high-dose STZ-induced DM in NCr athymic nude mice with hopes of providing clarification for study design, suggesting refinements to the process, and developing a more humane process of chemical diabetes induction.
Investigators interested in performing small animal experiments involving diabetes mellitus (DM)-related research, including pancreatic islet transplantation studies, have a variety of methods to choose from. These methods include but are not limited to models of DM that are spontaneous or genetically derived, chemically induced, diet induced, surgically induced, and now transgenic or knockout derived. 1–5 Owing to the duration of time and substantial resources required for most of these techniques, chemically-induced DM offers the most rapid and cost-effective option. After the first report of its ability to induce diabetes in 1963, 6 investigators have used streptozotocin (STZ) extensively in animal models to study both the pathology of DM and complications related to the disease as well as possible interventions. STZ offers the additional benefit of being able to select specific traits of interest, since STZ can induce DM in most strains of rodents. This can be important for islet transplantation studies, since specific immune responses, or lack thereof, may be critical for experimental design.
For our experiments involving xenogeneic and allogeneic islet transplantation, we decided to use STZ-induced DM in a strain of mouse with a compromised immune system, NCr athymic nude (nu/nu), in order to avoid immune-mediated graft rejection. Since the athymic nude mouse lacks mature T lymphocytes, graft rejection does not occur, making these strains excellent models for transplantation studies. 7 Athymic nude mice have been extensively used to study the pathogenesis of DM for more than 30 years, 8 and successful STZ-induced DM in nude strains is well documented. 8–11 Since Ricordi et al. 9 reported their experience in 1988, the STZ-induced diabetes model in immunodeficient mice is being increasingly utilized as a bioassay to evaluate the potency of isolated human islets and test agents that may improve transplantation outcome. 11
Despite the large number of publications on the topic (more than 17,000 listings for STZ on PubMed as of 19 May 2010), investigators inexperienced with a model of STZ-induced diabetes may find it difficult to precisely design new studies. STZ-induced diabetes can be highly variable, and no standard protocol exists for the preparation, dose or administration of STZ. Most of the literature to date includes very little description of the technical details of diabetes induction, STZ-related moribundity and mortality rates, and severity of diabetes experienced. Additionally, if performing islet transplant studies little data have been reported concerning moribundity or mortality of recipients related to the islet transplantation and nephrectomy procedures in mice. These factors make it extremely difficult to assess optimal animal sample sizes for experiments and set realistic timelines without previous experience. In this paper we review these shortcomings in the literature and provide data from our experiments involving single high-dose STZ-induced DM with the hope of providing clarification for study design and suggesting refinements to the process.
STZ history and method of action
STZ is a broad-spectrum antibiotic that is toxic to the insulin-producing β cells of pancreatic islets. It is currently used clinically for the treatment of metastatic islet cell carcinoma of the pancreas 12 and has been used investigationally in a wide variety of large and small animal species. 2–5,13–17 The method of STZ action in β cell depletion has been studied extensively over the years. It is generally assumed that STZ is taken up via the cell membrane GLUT2 glucose transporter and causes DNA alkylation and eventual β cell death, 15,17 although STZ's actions as a protein alkylating agent 18 and nitric oxide donor may contribute to its cytotoxicity. 19 Because STZ enters the cell via GLUT2, the toxic action is not specific to β cells and can cause damage to other tissues including the liver and kidney. 5,15,17,19
Factors influencing variable STZ sensitivity
Strain
Unfortunately, STZ sensitivity is highly variable in rodents, a characteristic that can make development of a new animal model or switching strains very challenging. In mice, a general strain-dependent hierarchical response to STZ has been reported by Gurley et al. In their experience, the order of sensitivity to STZ measured by blood glucose level (BGL) is DBA/2 > C57BL/6 > MRL/MP > 129/SvEv > BALB/c. Furthermore, they were able to separate these strains into groups of low responders (MRL/Mp, BALB/c and 129SvEv) and high responders (C57BL/6 and DBA/2). 20 Hayashi et al. 21 reported that the smallest single dose of STZ required to induce diabetes in male C57BL/6, ICR, ddY and BALB/c mice is 100, 100, 125 and 150 mg/kg, respectively. It should be noted that although these trends in strain-dependent sensitivity exist, different subgroups of a strain may experience different sensitivity to STZ, 16 and it cannot be assumed that any strain originating from a specific background will react to STZ in the same manner. In our experience, even when using age- and gender-matched individuals from an inbred strain with little genetic variability between mice, the dose that causes severe hyperglycaemia in most animals may fail to do so for some or be fatal to others. Use of animals provided by different suppliers, animals from outbred strains or even animals of different generations within a colony may exacerbate this difference.
Gender
Rodents also show a substantial gender difference in STZ sensitivity. Male mice and rats tend to be more susceptible to STZ-induced diabetes. 10,15,16,22–25 This difference can be significant, with little or no response in female mice and severe hyperglycaemia present in male mice receiving identical doses. In experiments with male and female C57BL/KsJ mice, Leiter 25 reported a highly significant difference (P < 0.001) of mean blood glucose values at all time intervals after day 0, and a mean blood glucose>200 mg/dL higher in males compared with females 35 days after STZ injection. This decreased sensitivity experienced by females may be attributed to oestradiol's ability to protect pancreatic β cells from apoptosis induced by oxidative stress. 24
Diet
Changes in rodent diet can have a large influence on sensitivity to STZ, and have been used to create type 2 diabetes models when combined with STZ doses that do not cause diabetes in normal chow-fed mice. 1 Taking this even further, C57BL/6J mice can experience type 2 diabetes strictly with a high-fat diet. 2 Less drastic changes in diet may alter severity of STZ-induced diabetes, especially in strains that have already been found to be high responders to STZ.
Circadian rhythm and other
Additionally, the time of day that STZ is administered may alter diabetes induction. Candela et al. 26 injected STZ into groups of female C3H mice every 4 h throughout the day and found that the highest incidence of diabetes was seen in animals injected at 16:00 h, and the lowest incidence in animals injected at 08:00 h. This study did not alter the light cycle from a standard 12 h cycle exposing animals to light from 06:00 to 18:00 h; hence it is unclear as to how this may affect diabetes induction rates. The age and weight of animals may alter sensitivity to STZ, although reports on this topic have been conflicting. 5 Unidentified environmental conditions and stimuli may contribute to variability in STZ-induced diabetes outcomes, and steps should be taken to expose animals to uniform conditions to help assure consistent STZ-induced diabetes.
STZ dose
Multiple methods of STZ dosing exist in the literature depending on the type and severity of diabetes intended for the specific experimental protocol. 15,27,28 The dose can vary greatly between gender and strain (Table 1). Although variable, these methods tend to fall into one of three categories: multiple small doses of STZ over a period of several days, a single moderately sized dose or a single large dose of the drug. Multiple low doses are generally used in experiments attempting to mimic autoimmune insulitis and tend to produce mild insulin-dependent diabetes mellitus (T1DM). Previous reports suggested that hyperglycaemia from multiple low-dose STZ is related to insulitis 40 that is T-lymphocyte dependent. 32 However, more recent studies have refuted this claim and have shown that β cell apoptosis is responsible for the development of T1DM, 30 and that T1DM can be induced in this method with immune deficient animals. 28 A single moderately sized dose of STZ has been used to induce a slowly progressive DM. Ito et al. 36 reported that male ICR mice receiving a 100 mg/kg injection of STZ experienced a progressive hyperglycaemia with normal non-fasting serum insulin levels and preserved β cell mass in the pancreas, indicating a non-insulin-dependent diabetes mellitus (NIDDM) due to an increase of insulin resistance. A single large dose of STZ is used for experiments attempting to cause severe T1DM by direct toxicity to β cells. Unlike the moderate dose of STZ, large doses can cause near total destruction of β cells and little or no measurable insulin production. 21,36
Reported range of streptozotocin (STZ) dose for different strains of mice
Example doses reported in studies involving STZ-induced diabetes. Although variable between strains and studies, over the course of five days multiple low-dose studies administer a similar total dose of STZ to that used for single high-dose studies. Single moderate dose studies administer approximately 1/2–3/4 that used by the other dosing methods
For our islet transplantation experiments we have selected a single large dose of STZ capable of eliminating endogenous insulin production in order to rigorously evaluate islet graft function. Severity of STZ-induced diabetes is critical for islet transplantation studies, especially those attempting to mimic the human transplantation situation. Patients currently eligible for islet transplantation usually have no measurable C-peptide. C-peptide is produced when proinsulin is cleaved to form the mature form of insulin. 43 A lack of measurable C-peptide indicates a lack of endogenous insulin production, and ideally mice in a transplantation model would also fail to produce insulin. Additionally, in a mouse model DM must be severe enough to confidently identify both islet graft function and any changes in islet graft function over time due to experimental conditions. Pre-existing endogenous insulin secretion or recovery of endogenous islets can confound data obtained from mouse islet transplantation experiments. Although it does not indicate complete β cell destruction, blood glucose greater than 400 mg/dL provides a scenario close to the human situation with little interference from endogenous insulin while decreasing the likelihood of endogenous islet recovery.
STZ preparation
Although no set protocol for STZ preparation exists, most groups report using a method similar to those first described in the 1960s. Early publications on the diabetogenic action of STZ stressed that a low pH solution is required to maintain stability of STZ once reconstituted, 6 and most recently published literature continues to suspend STZ in a buffer solution with a pH of 4.0–4.5. 5,14,20,27,32–34,37,39,41,42,44,45 This does not appear to be a necessity though, as Axler 38 has shown that a solution with a pH of 7.2 is comparable to one with a pH of 4.5 when reconstituting STZ for the induction of DM. Additionally, Lenzen 17 reported that STZ is relatively stable at pH 7.4 and 37°C for at least up to one hour. Because of our previous experience using neutral pH solutions for STZ preparation, 46 we elected to use a single intravenous injection of STZ suspended in sterile Hank's buffered salt solution (HBSS) (pH 6.7–7.8) that was maintained on ice and used within 30 min of preparation for all of our islet transplantation studies. We found this method to be reproducible for DM induction in male NCr athymic nude mice. Other basic salt solutions such as saline should be acceptable diluents as well and reports from other groups support this. 38,47 Although we have not performed any degradation studies our experience suggests that STZ degradation at pH higher than 4.5 may not be as rapid as originally suspected. 6 It is also possible that STZ preparations used today do not have the same characteristics as those used in the first reports, since the purity of those samples was questionable. 48
Our ability to successfully induce diabetes using a STZ solution with a neutral pH may be due to the fact that the solution was used within 30 min of preparation and stored on ice. However, other studies also debate the need for low temperature and immediate use. Induction of diabetes has been performed in mice using solutions with a pH of 7.2 stored for up to 30 min at 37°C 38 and in hamsters using solutions with a pH of 4.4 stored for up to seven days at 6°C. 14 Additionally, the package insert for Zanozar®, the STZ preparation used for clinical treatment of metastatic islet cell carcinoma, reports that when reconstituted as directed (pH 3.5–4.5) the drug may be used for up to 12 h after being placed in solution with no requirement for storage temperature once opened. 12 Use of sterile, off-the-shelf reagents with a neutral pH reduces the effort and complexity of STZ preparation since no pH adjustment, filtration or sterilization of media is required prior to STZ suspension. Also, eliminating pH adjustment to acidic range removes the potential hazard of inducing significant acid–base disturbance and cardiovascular morbidity after intravenous injection, especially in a small animal like the mouse.
STZ administration
STZ is most commonly delivered by one of two routes, intraperitoneal or intravenous, 15,47 although other methods including subcutaneous, intracardiac, and intramuscular delivery have been used in rodents. 5 Although the intraperitoneal route offers a quick and easy method of administration, especially for studies involving multiple doses of the drug, accidental delivery into the bowel or subdermal space may result in increased moribundity or decrease in diabetogenic effect. Additionally, other studies have reported that intravenous administration of STZ produces a more stable and reproducible model of diabetes than intraperitoneal administration. 47 Therefore, we decided to employ intravenous injection for our studies. The tail vein is commonly used for intravenous injection in the mouse, but this method can be a difficult procedure which requires experience and patience to be performed well, 49 especially in mice with dark pigmentation that can make the vein harder to identify. Blebs are common if the vein is missed initially, which not only requires additional puncture(s) in a more proximal location but also delivers a portion of the dose into the soft tissue of the tail. An incomplete intravenous dose such as this may cause variability in onset or severity of diabetes, especially when injecting highly concentrated solutions. Once mastered, tail vein injection can be performed effectively and consistently, eliminating concern of variable intravenous administration. However, for those inexperienced with the procedure other methods may be considered. For studies using male mice, injection into the penile vein is another option and the technique is easy to learn and perform due to the size and accessibility of the vessel, and has been used in the past for STZ delivery in rodents. 50 Although this method requires all recipients of STZ to be male and sedated during the injection, the procedure takes advantage of the increased sensitivity to STZ in male mice. We have adopted this method and report consistent and complete vascular delivery of STZ to all animals in our study with the only complication observed being rare transient paraphimosis that subsided within a few hours (<2% of animals).
Onset and severity of diabetes
For accurate study design one must consider the percentage of mice that will reach the required severity of diabetes, the timeframe in which they do so and how long they can be maintained with untreated severe DM. We designated a non-fasting blood glucose of ≥400 mg/dL as our criteria for islet transplantation because, as stated earlier, we feel that it provides a scenario close to the human situation with little interference from endogenous insulin while decreasing the likelihood of endogenous islet recovery. Other groups have adopted similar criteria for their studies as well. 37 Although it may not be the case with other dosing strategies, mice receiving a single large dose of STZ develop severe hyperglycaemia very quickly. Lu et al. 28 reported that high-dose STZ caused 12/12 C57BL/6 mice and 10/12 C.B17-SCID mice to develop hyperglycaemia with weight loss within 48 h.
In our experience, NCr athymic nude mice injected with STZ had a mean random blood glucose of 142.8 ± 19.7 mg/dL (n = 150) prior to STZ administration. Following STZ injection, animals developed DM rapidly with mean blood glucose of 459.4 ± 90.7 mg/dL within two days of STZ injection and 516.6 ± 95.2 mg/dL by day 7 (Figure 1). Although mice were considered diabetic when blood glucose exceeded 250 mg/dL for three consecutive readings, as our experience grew mice were not considered eligible to receive islet grafts until they were determined to be severely diabetic (>400 mg/dL). Table 2 shows that 82.5% of animals experienced their first blood glucose reading ≥400 mg/dL within five days of STZ injection. Over the course of our studies, more than 90% of mice developed diabetes severe enough to qualify for islet transplantation according to the blood glucose criteria for our experiments; however, blood glucose levels were usually far above the ≥400 mg/dL threshold. Peak blood glucose level experienced at any time before transplantation or removal from the study was between 400 and 499 mg/dL in 12.5% of mice, between 500 and 599 mg/dL in an additional 38.75% and ≥600 mg/dL in a remaining 41.25% (Figure 2). Insulin secreting ability was almost completely eliminated in the islets of these animals, a state that was confirmed by the changes in islet integrity and insulin staining observed in pancreas tissues removed two days after STZ injection (Figure 3).
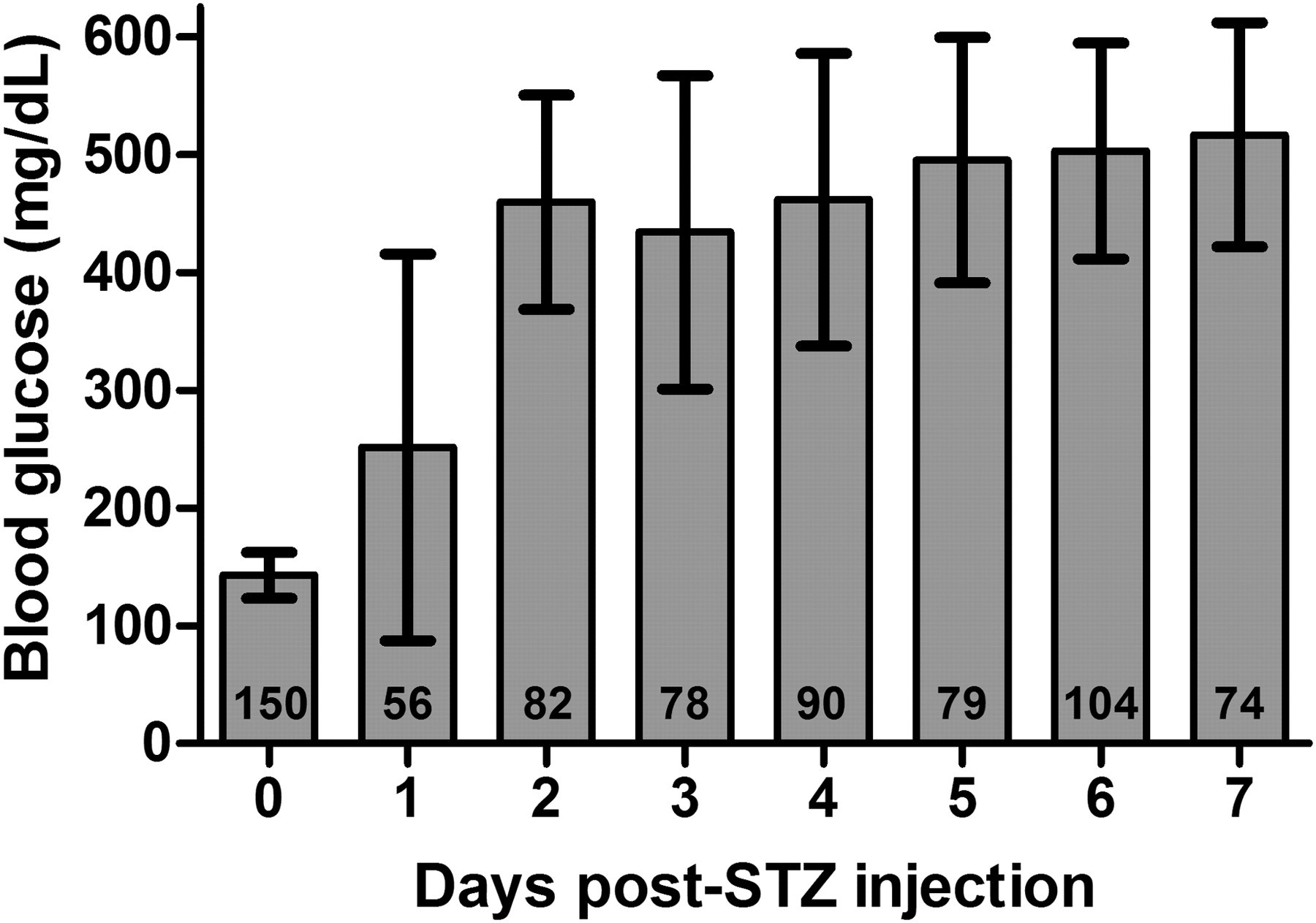
Mean blood glucose measurements of mice receiving streptozotocin (STZ) with standard deviation. Day 0 measurements were taken immediately before STZ injection. Blood glucose was not measured in every animal on every day. The number of animals for which blood glucose was measured each day is displayed at the base of each bar
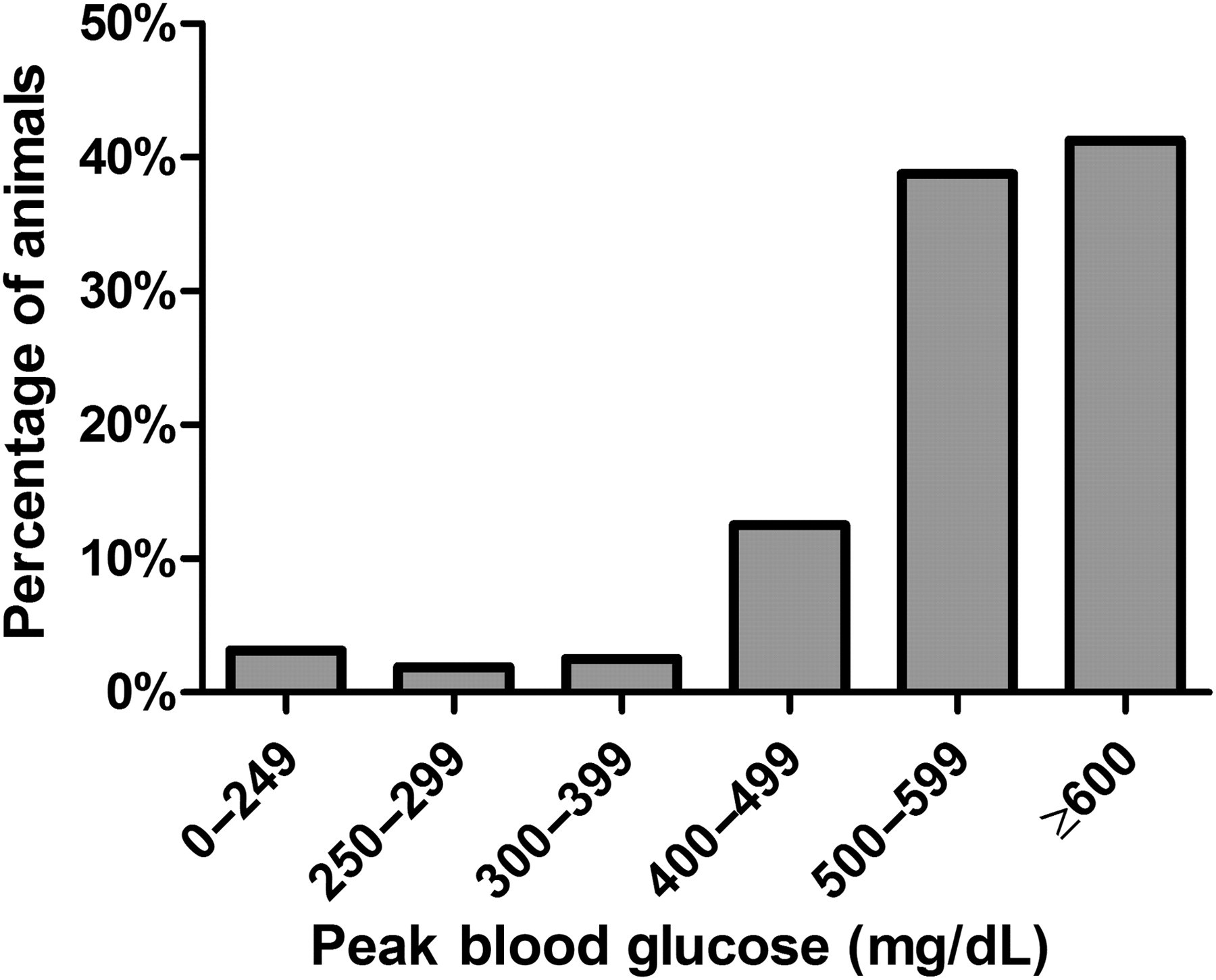
Peak blood glucose recorded from each animal after streptozotocin (STZ) injection but prior to islet transplantation (n = 160). Peak blood glucose exceeded 400 mg/dL in 92.5% of animals
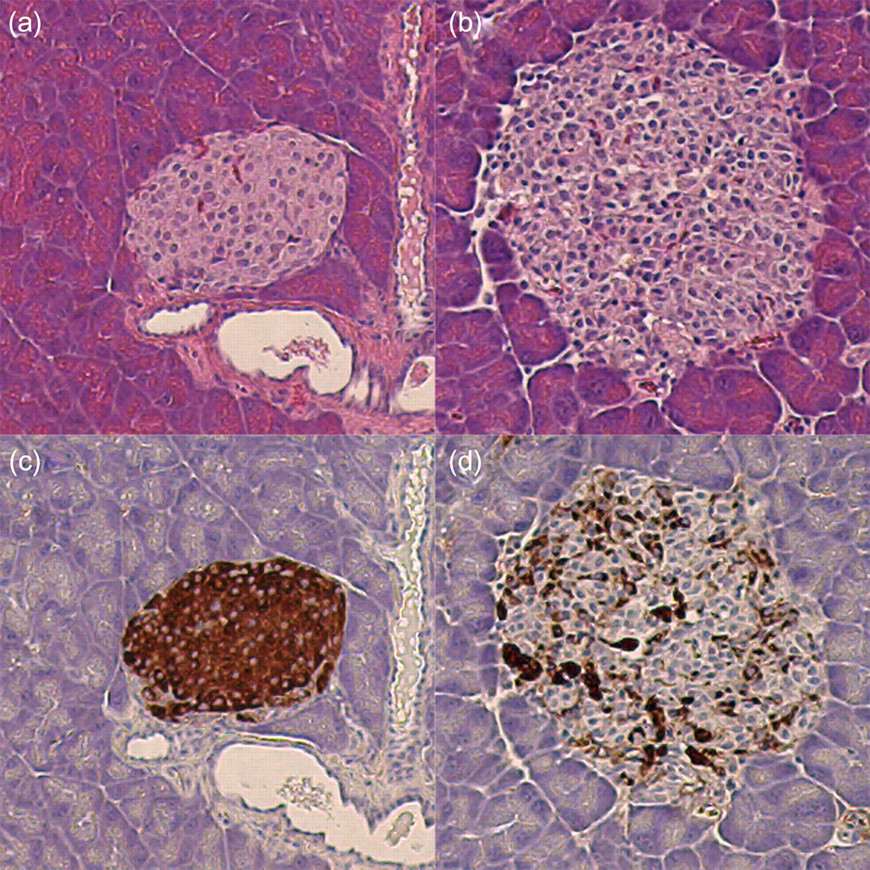
Haematoxylin and eosin and insulin staining of pancreases two days after animals that received either an injection of Hank's buffered salt solution (HBSS) (a, c) or HBSS with streptozotocin (STZ) (b, d). Panels a and c show healthy islets with normal morphology and substantial dark brown insulin staining. Panels b and d show damaged islets with abnormal morphology and minimal dark brown insulin staining
Cumulative incidence of severe DM after STZ injection
Number of animals that developed severe diabetes mellitus each day after STZ injection in 160 mice. Numbers reported are for the first day that a blood glucose reading ≥400 mg/dL was recorded
STZ: streptozotocin; DM: diabetes mellitus
This five-day timeframe for the onset of severe DM should not be prohibitive for transplantation studies in which islet availability can be predicted; however, it does not lend itself well to the use of islets freshly isolated from deceased organ donors. Owing to the unknown availability of fresh human islets, groups wanting to use this model for a biological assay of fresh islet products will need to keep a supply of severely diabetic recipients ready for when donor tissue becomes available.
STZ toxicity and moribundity of severe DM
Investigators must account for the loss of animals from STZ toxicity and complications of severe DM if left untreated over time. Unfortunately, most reports fail to mention such data, and even the more comprehensive articles make general statements of mortality without reporting specific data. 21,51 Bloch et al. 52 reported a survival of 70 ± 7% over their 27-day study in diabetic control ICR mice that had received a single dose of 200 mg/kg STZ and had blood glucose values >400 mg/dL. In our experiments, after injection of a single large dose of STZ, mice experienced three distinct periods of disease progression and mortality. In the first five days after STZ injection blood glucose increased significantly, mice lost weight and some experienced diarrhoea. During this period 4.38% of animals were found dead (Table 3). STZ has a documented acute toxic effect on multiple organs 53 and it is possible that STZ toxicity rather than complications of DM contributed to their deaths. In days 6–10 after STZ injection many mice developed sustained severe hyperglycaemia, polyuria, torpor, hunched appearance and continued weight loss, though no mortality was observed. When animals were maintained more than 10 days after STZ injection, the symptoms previously described persisted and a 20% mortality rate was observed. Because of the long duration of time after STZ injection and the observed polyuria and polydypsia, these mice may not have died due to STZ toxicity but instead due to complications of severe hyperglycaemia. The observed window of survival from days 6–10 supports the concept that the mice that expired within five days of injection did so due to acute STZ toxicity while the animals that died more than 10 days after STZ injection did so due to complications of severe hyperglycaemia.
Mortality involved with each procedure
Mortality of animals associated with STZ toxicity (≤5 days post STZ), diabetic complications (>10 days post STZ), and the islet transplantation and nephrectomy procedures. Total number of animals is reduced at each time point due to death, islet transplantation or removal from the study due to moribundity or lack of available islet tissue
*Death was observed within 24 h of the surgical procedure
STZ: streptozotocin
Although a total of only 10% of 160 mice in our experiments died following STZ injection, an additional 26.25% of animals had to be removed from the studies due to lack of available islet tissue or STZ treatment-related moribundity. The high number of mice removed from the study is complicated by experimental design because animals were used for several different protocols. Some protocols required mice to be maintained with severe untreated DM for extended periods of time while waiting for human islets to become available, resulting in higher rates of STZ treatment-related moribundity and culling of animals due to humane study endpoints. Other studies did not require this long waiting period and mice were injected with the knowledge that tissue may be made available on a certain day. These studies accounted for lower rates of moribundity but higher rates of sacrifice due to lack of available tissue in the event of failed islet isolations. In our experience it is reasonable to perform islet transplantation in mice up to 10 days after STZ injection with the assumption that approximately 5% of STZ-treated mice will die and 5% of mice will need to be removed from the study due to humane endpoints. However, this loss of animals increases substantially when attempting to maintain a group of mice with severe DM for extended periods of time and alternate methods of DM induction may need to be developed.
Mortality related to islet transplant and graft resection
Additionally, when designing studies involving islet transplantation under the kidney capsule, attrition of animals needs to be considered with the islet transplantation and nephrectomy procedures. These data are not often reported in the literature, although some studies have made an attempt to do so when comparing islet transplantation sites. Kim et al. 54 reported that in C57BL/6 mice that had received a single dose of 200 mg/kg STZ with preoperative blood glucose values >350 mg/dL, an operative mortality rate of 6.7% (1/15) was observed when performing islet transplantation under the kidney capsule. At the time of transplantation, mice are in a weakened state due to severe hyperglycaemia and a relatively high mortality can be expected with the surgical procedure. A total of 12.75% of our transplanted animals died within 24 h of the procedure, which was performed 7.81 ± 3.59 days after STZ injection. When comparing our operative mortality rate to that of Kim et al., it appears that our rate may be elevated. Kim et al. do not report pretransplantation blood glucose numbers for all recipients, but of the eight mice that received functional grafts the blood glucose exceeded 400 mg/dL in only one animal on the day of transplantation. By comparison, in our studies the mean peak blood glucose value that 102 mice receiving an islet graft experienced prior to transplantation was 562.5 ± 54.8 mg/dL. This more severe state of DM for our animals may account for our higher operative mortality rate.
If the islet graft proves to be functional, mice recover well, diabetic symptoms are alleviated and the animals gain weight (5.55 ± 2.82 g during a period of 23.69 ± 19.05 days). Generally, mice that underwent a nephrectomy in our study had functional islet grafts and had recovered from severe DM. These animals tended to be robust and handled the procedure well with only 1/59 animals (1.69%) dying within 24 h of the procedure. The large difference observed between the survival of transplantation recipients and nephrectomy recipients may be attributed to the more fragile state of transplantation recipients due to stress from severe hyperglycaemia. It should be noted that our surgical staff has extensive experience with both islet transplantation and nephrectomy in mice. Groups inexperienced with these procedures can expect a steep learning curve, and may need to adjust their expectations of mortality associated with the procedure accordingly.
STZ-related weight loss
Mice with severe DM tend to lose a large percentage of their weight after STZ treatment, potentially requiring a large number of animals to be culled from a study before islet transplantation can be performed. Dekel et al. 37 reported weight loss of up to 40% within 14 days of STZ injection in their high-dose model, and consequently had to reduce the amount of STZ administered. We recorded day of STZ administration and day of transplantation weight in 92 animals. These mice weighed 26.88 ± 3.11 g on the day of STZ administration and 22.54 ± 3.27 g on the day of transplantation. Although the mean weight loss was 16.15%, weight loss approaching 20% of body weight within 10 days of STZ injection was not uncommon. Despite this, mice tend to survive islet transplantation well and thrive with functional grafts. It should be determined with each Institutional Animal Care and Use Committee as to whether this is a concern and how to treat such animals. To decrease the percent weight loss, STZ dose can be reduced, or in certain models only larger mice may be used. Dekel et al. 37 reported a reduced STZ-related weight loss in heavier ICR mice; however these animals experienced corresponding lower BGLs. We did not observe a difference in percent weight loss or peak blood glucose between heavier and lighter recipients in our experiments, but our mice were substantially smaller than those used in the studies of Dekel et al.
Another option for minimizing the degree of STZ weight loss may be to use female animals. The findings of Cortright et al. 23 show that male Lewis rats are more susceptible than females to weight loss after STZ treatment. Rats were treated with variable amounts of STZ to equalize the severity of diabetes between groups and after the nine-week observation period male STZ-treated animals experienced a 45% reduction in weight when compared with controls, females only experienced a 13% reduction. 23 Bell et al. 22 reported a difference in weight loss between male and female Wistar Furth rats receiving STZ. In their study, male rats experienced significant weight loss over the course of the study, but female rats did not. At the end of the six-week study, STZ-treated control males weighed 221 ± 20 g compared with 276 ± 20 g for untreated male controls (19.9% decrease). On the other hand, STZ-treated female controls weighed 179 ± 6 g compared with 195 ± 11 g for untreated female controls (8.2% decrease). Unfortunately, despite overt hyperglycaemia in all female rats, the mean plasma glucose level in this study was significantly lower than that in males. 22 Interestingly, the study of Bell et al. 22 showed that these female rats experience an enhanced efficacy of islet grafts when compared with males, an effect that may be related to gender but may also be explained by the significant weight and blood glucose differences between genders. Additionally, islets were transplanted into the portal system and no histology of the pancreas was reported, hence potential recovery of endogenous islets cannot be ruled out. For transplantation studies where near complete β cell destruction is desired to mimic the human condition, STZ dose reduction, the use of heavier animals or the use of females may not be desirable due to the risk of less severe hyperglycaemia or endogenous islet recovery after transplantation.
DM treatment
Maintenance of mice with severe STZ-induced DM for an extended period of time is challenging. If left untreated, large numbers of mice will be lost due to DM-related mortality and moribundity. Some investigators have opted for insulin treatment in an effort to prolong the time that mice can be maintained and to improve the overall health of animals. Unfortunately, insulin administration may drastically alter endogenous islet function or recovery, confounding the transplantation model experimental outcomes. Grossman et al. 31 reported that C57BL/6 mice with STZ-induced DM experienced substantially decreased BGLs after 60 days of insulin therapy (280.9 ± 35.8 versus >400 mg/dL) and β cell regeneration and normoglycaemia after 120 days of insulin treatment. Additionally, Merino et al. 29 showed improved islet transplantation outcome when C57BL/6 mice received insulin treatment from 10 to 14 days before islet transplantation until 14 days after islet transplantation. Whether insulin therapy is used to treat severe STZ-induced DM for extended periods of time, or used in combination with islet transplantation, it appears that its presence may alter the study of transplanted islet engraftment and function. Because of this, alternate methods of severe hyperglycaemia treatment need to be developed. Islet transplantation studies, especially any utilizing insulin therapy, should incorporate graft explant to observe the return of hyperglycaemia as well as examination of the native pancreas to verify a lack of endogenous islet function.
Conclusion
Despite more than 40 years of widespread use of STZ in small animal models, standardized techniques and consistency in the resulting DM have not been achieved in the scientific community. This is due in part to the very complicated nature of the disease that investigators are attempting to mimic. Although not perfect, STZ-induced diabetes offers a very cost-effective and expeditious technique that can be used in most strains of rodents, opening the field of diabetes research to an array of genotypic and phenotypic options that would otherwise be inaccessible. Unfortunately, due to the complexity of DM and the huge number of possible animal models, investigators inexperienced with STZ-induced diabetes may find it difficult to precisely design new studies. To further challenge investigators, most of the literature to date is highly variable and often incomplete when it comes to detailing STZ preparation and administration, time to onset and severity of DM, moribundity and mortality experienced, etc. Until a better option becomes available, attempts need to be made to address these shortcomings with current STZ-induced DM models.
For studies attempting to mimic T1DM in which patients have little or no measurable C-peptide, such as pancreatic islet transplant studies, the dose of STZ should be large enough to cause near-total destruction of β cells. Use of a low pH solution may not be necessary for STZ preparation, and use of neutral pH off the shelf reagents simplifies the procedure and reduces the risk of potential acid–base disturbances in the recipient. The need for immediate injection and cold storage of STZ is also questionable, although no study combines all three scenarios of neutral pH, delayed injection and ambient temperature. If possible, STZ should be administered via intravenous injection because it allows for a more stable and reproducible model of diabetes than intraperitoneal administration. 47 Both tail vein and penile vein injection offer viable options. The resulting hyperglycaemia should be severe to eliminate as much interference from endogenous islet function and/or recovery as possible, and in our opinion non-fasting random blood glucose should be ≥400 mg/dL before animals should be used for islet transplantation studies. Weight loss, moribundity and mortality can be issues when maintaining mice with severe STZ-induced DM, and although regulatory standards may differ between countries, humane endpoints must be adopted and animals should not be maintained for extended periods of time without DM treatment. Unfortunately, insulin therapy has been shown to both reverse STZ-induced DM 31 and alter islet transplantation outcomes, 29 and its use should be reconsidered or accounted for when designing studies. New strategies for severe hyperglycaemia treatment that do not stimulate endogenous islet recovery and thus confound islet transplantation outcomes need to be developed before long-term maintenance of these animals becomes a humane option.
The NCr athymic nude mice used in our studies developed DM quickly and required no pretransplantation DM treatment. Approximately 80% of animals experienced severe DM and were healthy enough to receive islets 10 days after STZ injection despite routinely having blood glucose values in excess of 500 mg/dL for the majority of that time period. Mice with severe DM handle islet transplantation well and animals can be used for studies of human islet potency as well as xenoislet and alloislet experiments. When designing future studies we can confidently estimate that 65% of mice receiving STZ will experience severe diabetes (10% with blood glucose < 400 mg/dL), survive STZ treatment for at least 10 days (5% mortality), be healthy enough to receive an islet graft (5% moribundity) and survive both the islet transplantation and nephrectomy procedures (15% mortality).
Materials and methods
Animals
Animals were handled in accordance with the regulations of the Institutional Animal Care and Use Committee of the Mayo Foundation (Rochester, MN, USA). NCr athymic nude homozygotes (nu/nu) were purchased from Taconic (Hudson, NY, USA). Male mice aged 5–6 weeks were maintained in conventional housing with unrestricted access to food and water.
Husbandry during experiments
Animals were maintained in an open system and housed in polycarbonate static microisolator cages (Allentown Inc, Allentown, NJ, USA). Cages provided 451 cm2 of floor area and up to five non-diabetic mice were housed in each cage. Bedding consisted of heat-treated Aspen Sani-chips® (PJ Murphy Forest Products Corp, Montville, NJ, USA). Environmental temperature was regulated within a range of 69–71°F. Environmental humidity was not regulated, but was monitored weekly with a range of 22–60%. Animals were on a 12 h light cycle with artificial light provided between 06:00 and 18:00 h, and the facility's ventilation system performed 10–14 air changes per hour. Mice were allowed unrestricted access to food and water. Food consisted of irradiated PicoLab® Rodent Diet 20, catalog number 5053 (Lab Diet, Richmond, IN, USA) and tap water was provided with no additional treatment in our facility.
STZ preparation and administration
STZ was obtained from Sigma-Aldrich (St Louis, MO, USA) and USB Corporation (Cleveland, OH, USA). Immediately before administration, STZ was suspended in cold HBSS (Invitrogen, Carlsbad, CA, USA) with no adjustment of pH at a concentration of 40 mg/mL. STZ solution was stored on ice for less than 30 min before injections were performed. Animals were given an intraperitoneal injection containing 90–120 mg/kg ketamine and 5–10 mg/kg xylazine. Once sedated, the prepuce was pushed back and the penis was gently pulled taut using forceps. A total of 220 mg/kg of STZ was injected into the penile vein using a 27g needle. A cotton-tipped applicator was used to apply pressure as the needle was withdrawn. Once bleeding stopped the penis was pushed back into the prepuce and the animal was allowed to recover before being returned to the housing facility. If necessary, mice were placed on a heating pad or received intermittent exposure to a heat lamp during the recovery period to prevent heat loss and potential hypothermia-related anaesthetic death.
Blood glucose determination
Prior to diabetes induction BGLs were measured in tail vein blood using an AccuChek Compact glucometer (Roche Diagnostics, Indianapolis, IN, USA). The range for the AccuChek Compact is 10–600 mg/dL, and any value >600 mg/dL registers as ‘HI’. Readings of ‘HI’ were recorded as ≥600 mg/dL. After STZ injection, blood glucose and animal health were monitored three times per week. DM was diagnosed when blood sugars exceeded 250 mg/dL for three consecutive readings, but transplantation was not performed unless blood sugars were substantially higher, preferably in excess of 400 mg/dL. All blood glucose measurements were taken in the fed state early in the afternoon to eliminate variability in BGLs caused by feeding patterns of the mice.
Maintenance of diabetic animals
Cages were changed multiple times per week after STZ injection to provide dry bedding for polyuric animals. Mice with DM were often housed in groups of two or three to reduce the amount of urine in bedding and were maintained without antihyperglycaemic treatment until islet tissue became available for transplantation. This period of time was variable due to the logistics of islet availability. Experiments involving the transplantation of dog, pig or mouse islets could be planned in advance and tissue could be collected once animals were eligible for transplantation. However, in experiments involving human islets, mice were maintained in the diabetic state for up to 39 days while waiting for pancreases to become available from deceased organ donors. Humane endpoints outlined in our Institutional Animal Care and Use Committee approved protocols were adhered to for all studies.
Histology
To verify the destruction of β cells within islets two mice were sacrificed two days after STZ injection. The pancreas was placed in formalin and embedded in paraffin. Sections were cut and haematoxylin and eosin (H&E) staining was performed as well as insulin staining using polyclonal guineapig anti-insulin, catalog number A0564 (DAKO, Carpinteria, CA, USA).
Statistical analysis
Statistical analysis, including paired t-test and Wilcoxon signed-rank test, was performed using GraphPad Prism 5 (GraphPad Software, La Jolla, CA, USA).
Footnotes
ACKNOWLEDGEMENTS
This publication was made possible by Mayo Clinic; Grant Number DK56890 from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), a component of the National Institutes of Health (NIH); Grant Number 1 UL1 RR024150 from the National Center for Research Resources (NCRR), a component of the NIH and the NIH Roadmap for Medical Research; and the Richard F Emslander Career Development Award in Endocrinology and Nutrition. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of the NIDDK, NCRR or NIH.
We wish to thank the Department of Comparative Medicine, especially Michael Blanco, DVM and Wayne Kingsbury.
