Abstract
Diabetes mellitus (DM) is a major cause of morbidity and mortality worldwide, and its complications are prominent public health issues. Many experimental models of streptozotocin (STZ)-induced and high-fat diet (HF)-induced DM have been used to study this disease. Studies have indicated that unilateral nephrectomy (UN) accelerates the development of diabetic nephropathy. We hypothesized that UN stimulates HF and STZ combination-induced DM in mice. Seventy-two female C57BL/6J mice were divided into four treatment groups: HF; HF + STZ120 (HF and STZ, 120 mg/kg); UN + HF + STZ120 (UN, HF and STZ, 120 mg/kg); and HF + STZ200 (HF and STZ, 200 mg/kg). Onset of DM, survival rate, blood pressure, urine glucose level, and pancreatic histology were investigated. Additionally, renal function was evaluated in the UN + HF + STZ120 group after STZ injection. DM was induced in the UN + HF + STZ120 and HF + STZ200 groups within one week. The UN + HF + STZ120 group had lower mortality than the HF + STZ200 group and greater pancreatic destruction than the HF and HF + STZ120 groups. Two weeks after STZ injection, blood pressure was not significantly different among the groups. Nephrotoxicity associated with the combination of UN and STZ was not observed. In conclusion, the combination of these three techniques – UN, HF and STZ induced DM rapidly and effectively.
Extensive research of diabetes mellitus (DM) is warranted because this disease has reached epidemic proportions worldwide and is associated with significant health-care costs. 1 Many experimental models of DM are currently in use, but more efficient models are needed. 2 Experimental models of DM that use transgenic rodents or rodents fed streptozotocin (STZ) or alloxan are the most common. 3 STZ is an antimicrobial drug isolated from Streptomyces achromogenes that induces DM 4 and an analogue of glucose-containing methylnitrosourea, which alkylates DNA. 5 STZ is selectively toxic for pancreatic β-cells into which it is transported via glucose transporter 2. 6 The mechanism of action of STZ is not fully understood, but it is thought that its DNA alkylating activity causes β-cell necrosis and consequently induces diabetes. STZ has species specificity: mice, rats, hamsters, dogs, monkeys and pigs are sensitive to STZ, but rabbits and guinea pigs are not. 7 In beagles and rhesus monkeys, STZ injection induces the onset of diabetes and results in hyperglycemia, β-cell destruction and glucosuria. 3 Rodent models are used extensively to study diabetes owing to their ease of control. 8 Inbred mice have high genetic similarity to humans and have therefore been widely used for experimental models of diabetes. 9 C57BL/6J, C57BLKS, BALB/c, ICR, DBA2 and ROP mice are the main strains susceptible to STZ. 10
A high-fat diet (HF) is an effective method with which to induce type 2 DM. 11 HF results in obesity, hyperinsulinemia and insulin resistance. 11 C57BL/6J mice in particular have high susceptibility to HF and develop obesity, hyperglycemia and hyperinsulinemia in response. 10 Interestingly, results from studies using a diabetic nephropathy model have indicated that unilateral nephrectomy (UN) accelerates the development of diabetic nephropathy.8,12 However, it was also questioned that nephrectomy hastened the progress of DM. Based on these findings, we designed a new DM model combining UN, HF and STZ. The objective of this study was to investigate whether UN stimulated the induction of DM by combined treatment with HF and STZ.
Animals
All animal protocols were approved by the Institutional Animal Care and Use Committee of Kangwon National University (Chuncheon, ROK). Seventy-two healthy, 17-week-old female C57BL/6J mice (20–25 g) were purchased from Orient Bio (Seongnam, ROK). The mice were housed in groups of four in ventilated cages (Three-Shine, Daejeon, ROK) under standard laboratory conditions (12 h light/dark cycle, mean temperature of 22 ± 2℃ and 50 ± 10% relative humidity). Aspen shavings (Orient Bio) were provided as bedding in each cage, and food and water were available ad libitum. All mice were fed an HF (fat content >60 kcal%, D12491; Research Diets Inc, New Brunswick, NJ, USA).
Materials and methods
Mice were divided into four treatment groups: HF; HF + STZ120 (HF and STZ, 120 mg/kg); UN + HF + STZ120 (UN, HF and STZ, 120 mg/kg); and HF + STZ200 (HF and STZ, 200 mg/kg). Three weeks after UN, STZ (Sigma, St Louis, MO, USA) was administered to the mice via intraperitoneal injection after 4 h of fasting. For the nephrectomy, the mice were anesthetized via intraperitoneal injection of ketamine (80 mg/kg) and xylazine (5 mg/kg), and the left kidneys were removed after ligation of the renal artery, vein and ureter using 6-0 silk (Ailee, Busan, ROK).
Measurements
For measuring blood glucose level, all the mice were fasted for 6 h, and tail vein blood samples were collected at 13:00 h. Blood glucose concentration was assayed using an automated glucose analyzer (Beckman Instruments Inc, Fullerton, CA, USA). Mice with fasting blood glucose levels of 300 mg/dL or higher were considered to be diabetic. 13 Body weight was also checked weekly. Blood pressure was non-invasively measured using a CODA™ monitor (Kent Scientific, Torrington, CT, USA) according to the manufacturer’s protocols. Urine was collected in metabolic cages (Harvard Apparatus, Holliston, MA, USA) for 24 h and glucose levels were measured. Serum creatinine was measured using a QuantiChrom™ Creatinine Assay Kit (BioAssay Systems, Hayward, CA, USA) according to the manufacturer’s instruction.
Pancreatic histology
One month after STZ injection, the pancreas was collected, fixed in 10% phosphate-buffered formalin and embedded in paraffin. Sections were cut (4 um thickness) and stained with hematoxylin and eosin.
Kidney immunohistochemistry
Two days and one week after STZ injection, the mice were killed and their kidneys were collected. Frozen sections fixed in 4% paraformaldehyde were labeled with kidney injury molecule-1 (KIM-1) antibody (R&D Systems, Minneapolis, MN, USA) followed by Cy3-conjugated secondary antibody. Nuclei were visualized with 4′,6-diamidino-2-phenylindole (12.5 µg/mL; Invitrogen, Carlsbad, CA, USA).
Statistical analysis
Experimental data were analyzed statistically with analysis of variance (ANOVA). P value of 0.05 indicated significance.
Results
UN induced DM effectively with low mortality
Blood glucose levels in all groups were measured over 10 weeks to evaluate the effects of UN on DM induction. Within one week of STZ injection, the mice in the UN + HF + STZ120 and HF + STZ200 groups developed DM, whereas those in the HF and HF + STZ120 groups did not develop hyperglycemia for 10 weeks (Figure 1a). The mice in the UN + HF + STZ120 group had stable hyperglycemia; however, blood glucose levels in the mice in the HF + STZ200 group at two weeks after STZ injection were higher than those measured one week after injection. Unlike the mice in other groups, those in the HF + STZ200 group showed high mortality and died within three weeks (Figure 1b). Approximately 70% of UN + HF + STZ120 mice survived the experiment. These results showed that UN induced DM rapidly and effectively with STZ injection.
Unilateral nephrectomy (UN) stimulated the onset of diabetes mellitus (DM) with low mortality. (a) Changes in blood glucose level in the high-fat diet (HF), HF + streptozotocin (STZ) 120, UN + HF + STZ120 and HF + STZ200 groups over 10 weeks. (b) Survival rate was compared among groups. (c) A single high dose of STZ (120 mg/kg) or multiple low doses of STZ (30 mg/kg four times a day) were injected into UN mice. Significant differences in blood glucose levels were not found between protocols. (d) Changes in body weight in the HF, HF + STZ120, UN + HF + STZ120 and HF + STZ200 groups were compared. Mice in the HF + STZ200 group showed significant weight loss.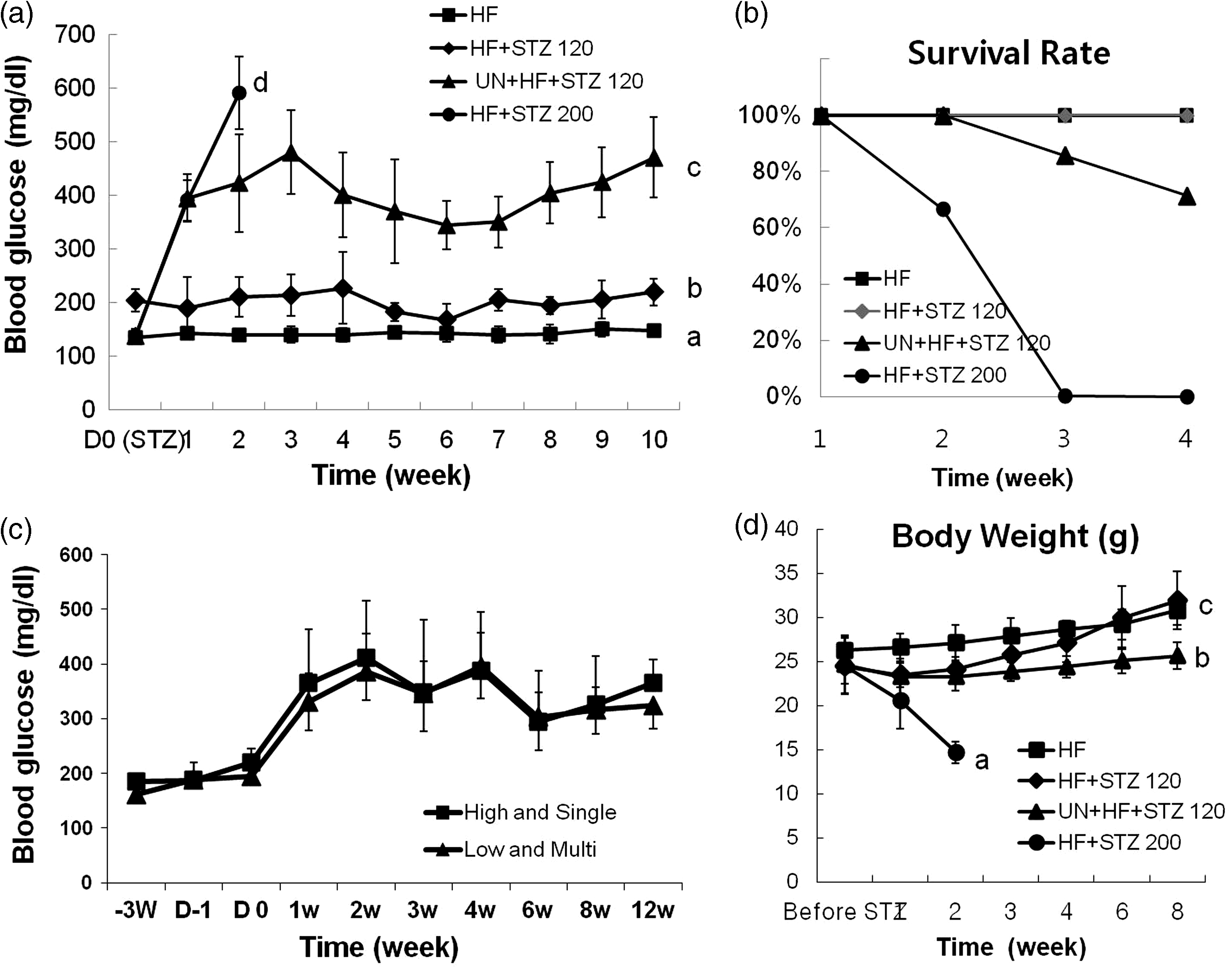
To determine whether the onset of DM is dependent on the number and dose of STZ injections, STZ was injected intraperitoneally into UN mice as a single high dose (120 mg/kg STZ in 0.1 mmol/L of citrate buffer, pH 4.3) or multiple low doses (30 mg/kg STZ, four times per day). Within one week of STZ injection, blood glucose levels in both groups rose above 300 mg/dL and induced DM (Figure 1c). The results demonstrated that the onset of DM was not dependent on the number of STZ injections.
To determine the effects of DM on body weight, the body weight of all mice was measured weekly. Body weight increased significantly in the HF and HF + STZ120 groups (Figure 1d). By contrast, the mice in the UN + HF + STZ120 group showed no significant weight increase through this experiment. Animals in the HF + STZ200 group showed significant weight loss after STZ injection. These results demonstrated that DM attenuated body weight increases.
UN did not induce significant hypertension
To determine whether UN induced hypertension, blood pressure was measured in the mice in all groups one and two weeks after STZ injection. No significant differences in systolic blood pressure values were found between mice in the UN + HF + STZ120 group and those in the other groups one week after STZ injection (Figure 2a). Two weeks after STZ injection, some mice in the UN + HF + STZ120 group had blood pressure values higher than those measured in previous experiments, but the difference was not significant (Figure 2b). These results demonstrated that UN did not induce significant hypertension in mice.
Unilateral nephrectomy (UN) did not induce significant hypertension. (a) One week after streptozotocin (STZ) injection, systolic blood pressure was not significantly different among groups. (b) Compared with other groups, the UN + high-fat diet (HF) + STZ120 group did not show significantly higher blood pressure two weeks after STZ injection.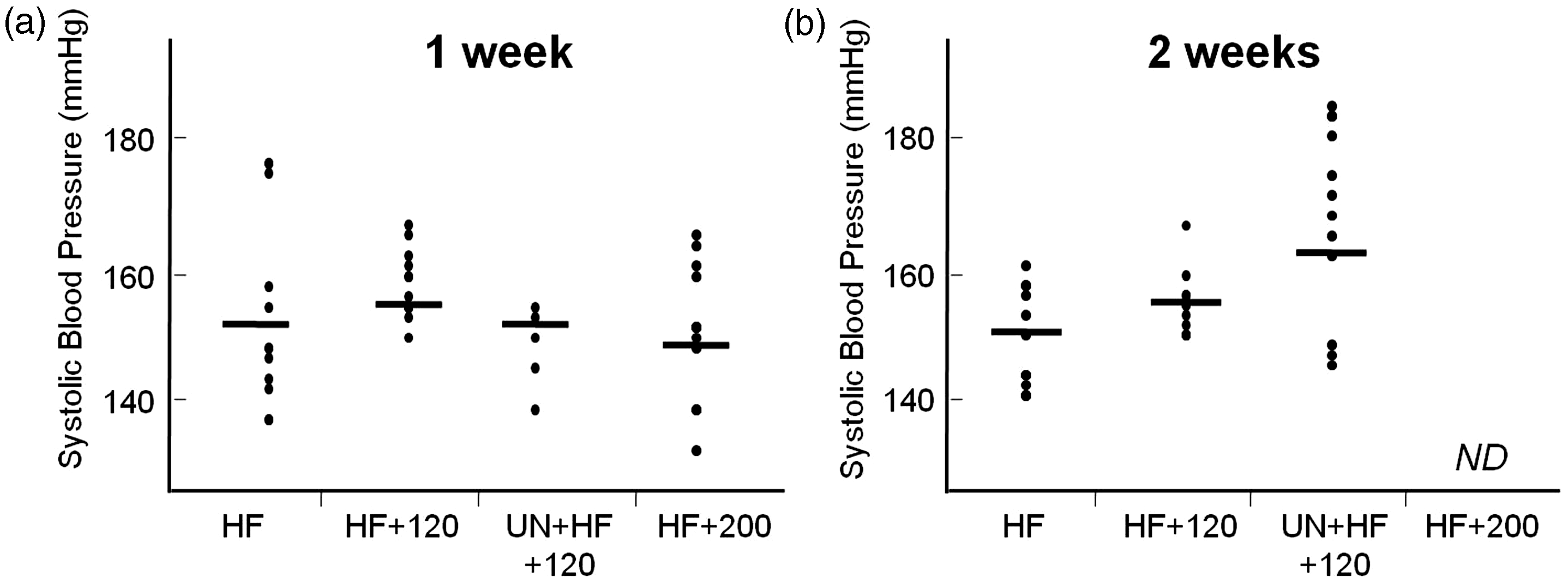
UN stimulates DM via impaired glucose regulation
To evaluate the cause of the higher blood glucose levels in the UN + HF + STZ120 mice compared with the HF + STZ120 mice, urine glucose excretion levels were measured. Compared with mice in the other groups, the mice in the UN + HF + STZ120 and HF + STZ200 groups showed significantly higher urine glucose levels, which demonstrated that UN did not result in less efficient glucose excretion (Figure 3a). Pancreatic destruction was compared next in each group four weeks after STZ injection. Contrary to mice in the HF and HF + STZ120 groups, those in the UN + HF + STZ120 group showed severe destruction and reduction of pancreatic islet cells (Figure 3b). These results demonstrated that the high blood glucose levels observed in the UN + HF + STZ120 mice were induced by impaired glucose regulation, not by insufficient glucose excretion.
Unilateral nephrectomy (UN) stimulates diabetes mellitus via efficient pancreatic destruction. (a) Urine glucose was higher in the UN + high-fat diet (HF) + streptozotocin (STZ) 120 and HF + STZ200 groups than in the HF and HF + STZ120 groups. Asterisks indicate statistical significance (*p < 0.05). (b) Hematoxylin and eosin staining of pancreatic tissue demonstrated that compared with mice in the HF (a) and HF + STZ120 (b) groups, mice in the UN + HF + STZ120 (c, c′) group had severe pancreatic islet destruction.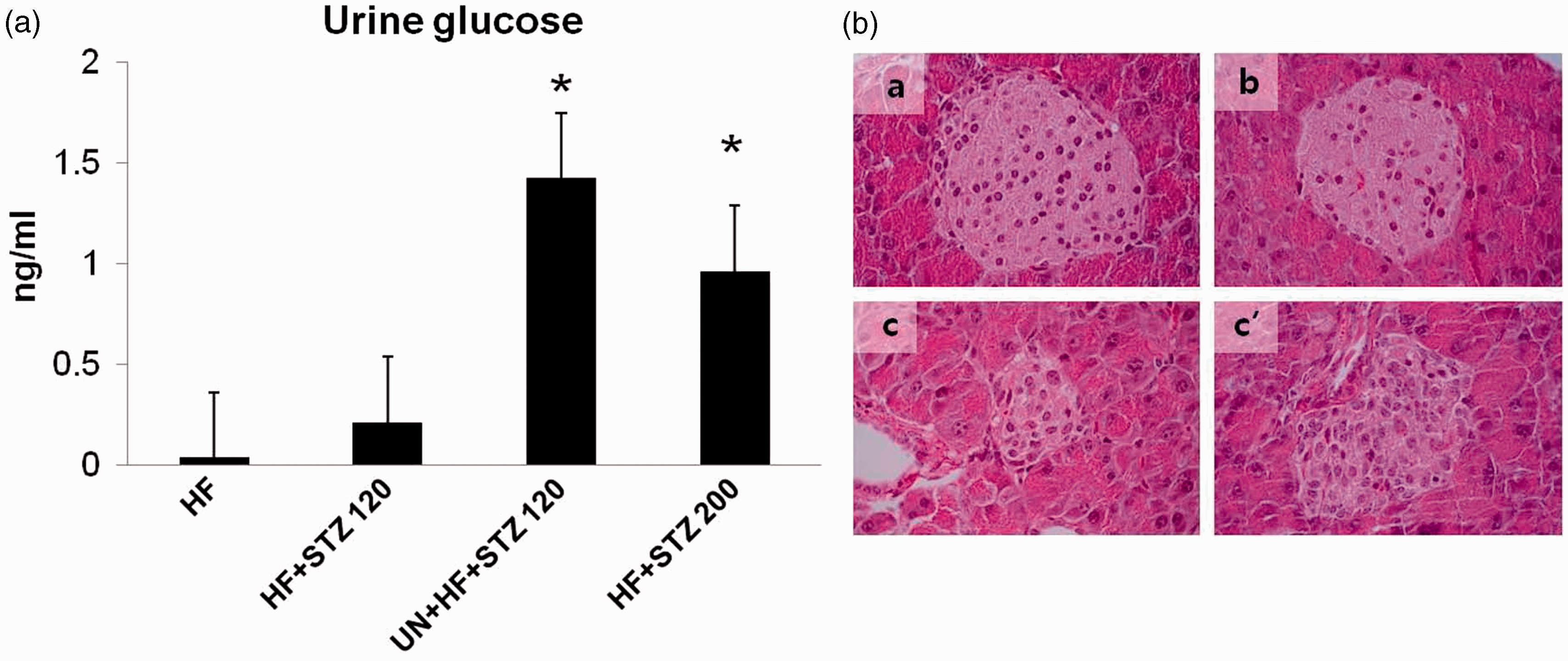
UN and STZ did not induce nephrotoxicity
To determine whether STZ induced renal toxicity in the UN mice, serum creatinine levels and KIM-1 expression were measured in the kidney tubular epithelium. Significant increases in serum creatinine did not occur during the three-week observation period (Figure 4a). Furthermore, nephrotoxicity associated with high-dose STZ was low, as demonstrated by low levels of KIM-1 expression (Figure 4b and c). These results showed that UN and STZ injection did not alter renal function.
Unilateral nephrectomy (UN) did not impair kidney function. (a) Serum creatinine levels in mice in the UN + high-fat diet (HF) + streptozotocin (STZ) 120 group. No evidence of renal failure was found. Few cells expressed kidney injury molecule-1 (KIM-1) at two days (b) or one week (C) after STZ injection in the UN + HF + STZ120 group.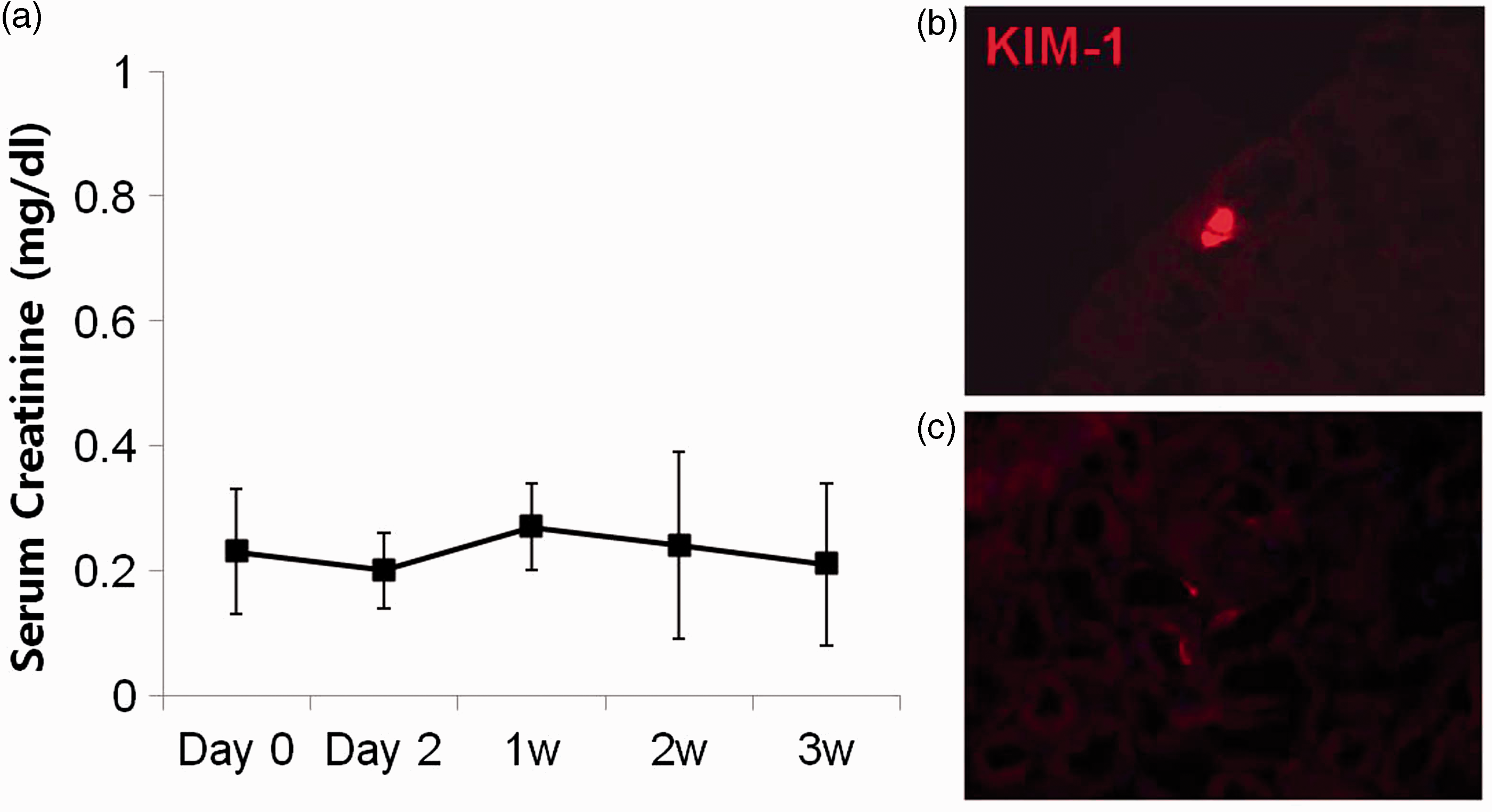
Discussion
In this study, we developed a modified mouse model of DM using UN, HF and STZ and demonstrated that the combination of these treatments effectively stimulates the induction of DM in C57BL/6J mice. STZ has a toxicity specific to β-cells because of its uptake via glucose transporter 2. 14 However, it also has non-specific toxicity to other tissues such as the liver and kidney. An injection of 150–200 mg/kg STZ is recommended for inducing stable hyperglycemia in mice. 10 However, Tay et al. have reported that acute tubular necrosis appeared in C57BL/6J that received a single intravenous injection of STZ (200 mg/kg) two weeks after they developed diabetes. 15 Our protocol used mice that underwent UN, which required the exclusion of the possibility that renal injury affected the results. Our results showed that serum creatinine was unaltered. Also, a biomarker of renal injury, KIM-1, was only slightly expressed. Therefore, the STZ dose of 120 mg/kg used in this protocol is also the optimal dose.
The control group in our study showed no hyperglycemia. However, mice treated with UN, HF and STZ showed stable hyperglycemia within one week of STZ injection, which was a significant decrease in DM induction time. In a previous study, female C57BL/KsJ mice did not show onset of DM until 35 days after STZ injection. 11 Therefore, our combination method is more efficient than current methods.
Conventionally, two STZ injection methods are used: multiple low-dose injection and single high-dose injection. The former requires repetitive injection, whereas the latter may cause non-specific STZ toxicity. 10 We tried to determine which method induced DM more efficiently. However, we found no significant differences between the two methods. In the absence of non-specific cytotoxicity, single high-dose STZ injection was more convenient.
In this study, an HF was fed to experimental animals because it increases sensitivity to the onset of DM.11,16 A previous study has shown that body weight increased significantly in female mice after only one week of consuming an HF. 16 By contrast, weight gain was usually inhibited after the onset of DM in mice. 17 Our study also showed that the DM mice in the UN + HF + STZ120 and HF + STZ 200 groups presented relatively low body weights compared with the non-DM mice in the HF and HF + STZ120 groups. Therefore, this result confirmed that DM occurred readily in the UN + HF + STZ120 group.
In a previous study, a combination of STZ and nephrectomy was used in a rat model of combined hypertension and diabetes. 18 However, in our study, UN mice did not show significant hypertension. Similarly, a previous report has demonstrated that, unlike in rats, UN in mice did not induce hypertension without the use of deoxycorticosterone salt. 19
Various animal studies have shown that STZ accumulates in the kidneys, which is the main site of STZ excretion. Previously, 10–20% of STZ was detected in the kidney.20,21 UN affects drug secretion by the kidney, and although compensatory renal change occurs, renal function does not fully return to normal. 21 As in STZ metabolism, kidney excretion is the main pathway of excretion for atenolol, a β-adrenoreceptor-blocking drug. 22 Atenolol elimination is impaired one year and one month after UN, which increases serum concentration. Because the kidney is the primary site for STZ excretion, UN also affects STZ levels. STZ remains in animals longer and causes more pancreatic damage. After intravenous injection of STZ (200 mg/kg), the serum half-life is 5 min, and STZ is not detected in the serum within 2 h. 23 This explains why delayed metabolism of STZ resulted in more extensive pancreatic damage in our study. To compensate fully for impaired renal function, the remaining kidney must increase its function to 100%. However, after UN, the glomerular filtration rate increased by only 52.3%. 24 UN is therefore an efficient method for inducing pancreatic damage and hastening the onset of STZ-induced DM.
The combination of these three techniques – UN, HF and STZ – shortened the time required to achieve a DM model. Furthermore, non-specific STZ toxicity was negligible in this protocol, although long-term observation is required. Therefore, this modified model is potentially helpful in the study of DM and the development of methods to reduce the progression of DM.
Footnotes
Acknowledgement
The authors thank Dr. JV Bonventre and Dr. T Ichimura, Harvard medical school, USA, for supporting this experiment.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the 2013 Research Grant from Kangwon National University (No. C1009825-01-01) in Republic of Korea.
