Abstract
Diabetes cell therapy by human islet transplantation can restore an endogenous insulin secretion and normal glycaemic control in type 1 diabetic patients for as long as 10 years post transplantation. Before transplantation, each clinical islet preparation undergoes extensive in-vitro and in-vivo quality controls. The in-vivo quality control assay consists of transplanting human islets under the kidney capsule of immunocompromised mice. Currently, it is considered the best predictive factor to qualify clinical transplant efficiency. This chimeric model offers a wide area of study since it combines the possibility of producing not only quantitative but also a maximum of qualitative data. Today’s technological advances allow us to obtain more accurate and stronger data from the animals used in research while ensuring their comfort and well-being throughout the protocol, including cage enrichment and pain treatment during and after surgery. As demonstrated in this valuable model, we are able to generate more usable results (Refine), while reducing the number of animals used (Reduce), by focusing on the development of ex-vivo analysis techniques (Replace), which clearly highlights the Burch and Russell 3Rs concept.
Introduction
Diabetes affects more than 420 million people around the world, and this rate has been increasing for several years.1,2 The most prevalent forms of diabetes are type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM). T1DM or insulin-dependent diabetes affects 10% of total diabetic patients. It is characterized by the loss of insulin-secreting Beta cells in the pancreatic islets of Langerhans, leading to an increase in blood glycemia. In the most severe cases, insulin-therapy is not sufficient to normalize the glycaemic range, and cell therapy is the only strategy to restore a glycaemic balance. This therapy shows long-term efficiency in re-establishing an endogenous insulin secretion in patients treated for more than 10 years.3,4 This approach consists of transplanting human islets from pancreases harvested from brain-dead patients. Islet isolation liberates these pancreatic islets via an enzymatic action followed by a ficoll-gradient purification. 5 After isolation, in-vitro explorations are done to verify the microbiological environment, the viability and islet functionality through a glucose-stimulated insulin response (GSIS) assessment. All these parameters permit the clinical-grade qualification of the final preparation, subsequently transplanted in T1DM patients.6,7
In-vitro studies are limited due to the unstable state of these cells; they dedifferentiate and lose their capacity to secrete insulin after 10 days in culture. 8 However, in-vivo transplantation of pancreatic islets in immunodeficient mice permits extended studies, from several months up to a year, opening the door to long-term studies and broader fields of research. 9 Thereby, the FDA (Federal Drug Administration) considers this model to be the best in predicting human islet functionality. 10 –12 Numerous studies were done to describe the best implantation site for pancreatic islets. From a simple subcutaneous site to something as sophisticated as the anterior chamber of the eye,13,14 almost all possible sites were tested in rodents (Table 1).15,16
Examples of islet transplantation sites and their characteristics (modified from Pepper et al. 201316 and 201515).
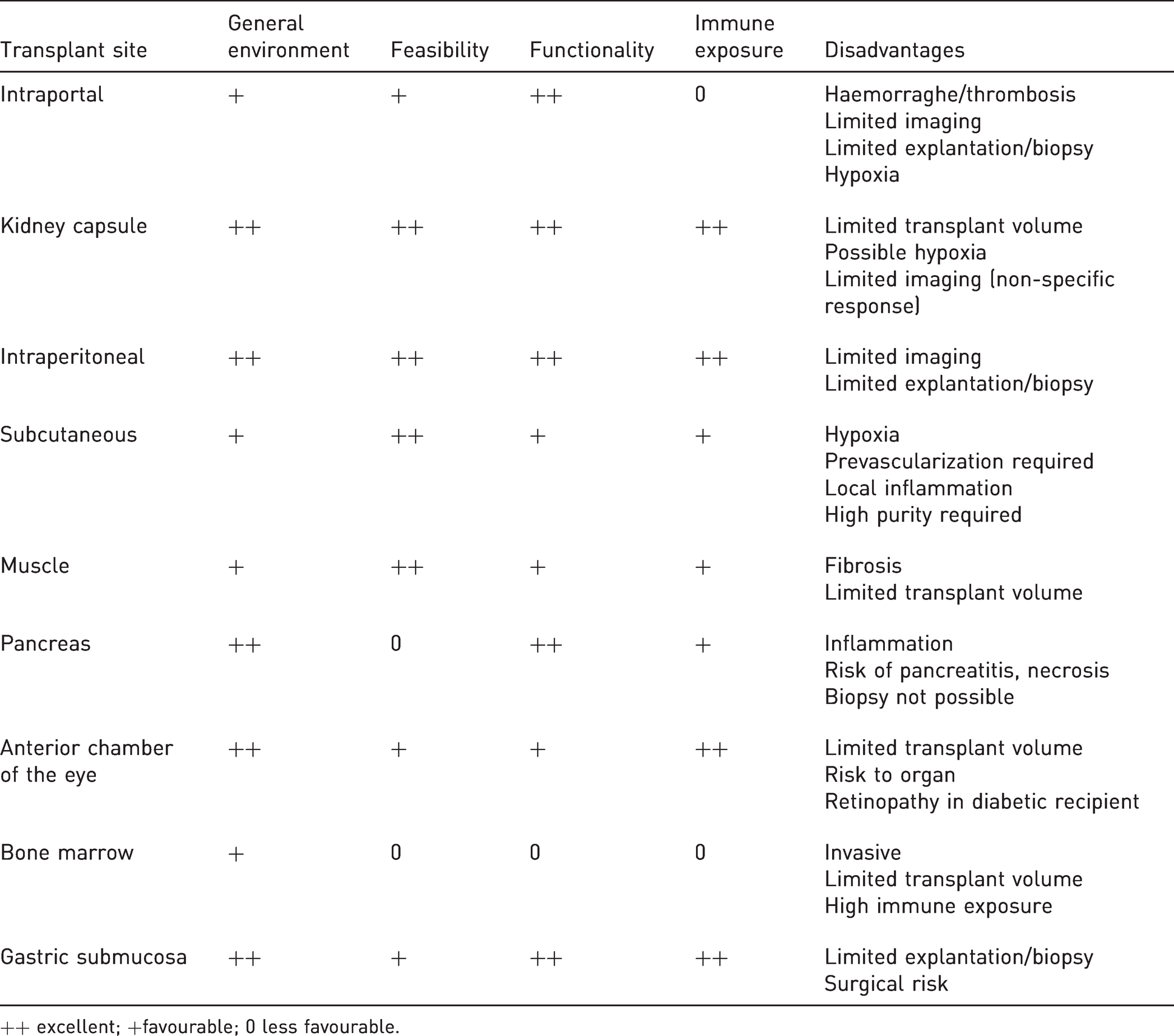
++ excellent; +favourable; 0 less favourable.
In 1959, Burch and Russell emphasized the importance of the 3Rs in experimental protocols involving animals. 17 To do this, the ‘replacement’ of the animal must, in all cases, be favoured using in-vitro techniques, or in silico for example. Moreover, in the interest of ‘reduction’, the number of subjects must be reduced to a minimum without causing statistical analyses difficulties. Finally, in order to satisfy the principle of ‘refinement’, the entire project must be conducted taking into account the general well-being of the animal, focusing on the management of pain or discomfort, on the improvement of breeding and housing conditions and by introducing the notion of limit points. It is also a question of optimizing the methods used in order to obtain the most exploitable results while reducing distress to a minimum.
The goal of this review is to expose the most widely used model, the kidney capsule site, which offers a large array of possibilities.
Ethics
Commonly, ethical committees consider that this technique of transplantation under the kidney capsule inflicts a moderate level of pain. It is carried out with mice under general anaesthesia optimally with isoflurane gas (Isoflo, Zoetis, France) at 5% for induction and 2% to easily control the strength of anaesthesia. Pain treatment is undertaken with drugs that are administered locally on the skin and muscles such as xylocaine 2% (Astrazeneca, Sweden), lidocaine 0.5% (Aguettant, France) and morphinic substances (buprenorphine 0.05mg/kg; Bupaq, Virbac, France) injected subcutaneously or intraperitoneally. Our project entitled ‘Pancreatic islets transplantation in immunodeficient mice – #12758’ was approved by the local ethical committee (Comité Ethique en Expérimentation Animale 75 – North of France) and the National Instance (Ministère de l'enseignement supérieur, de la recherche et de l'innovation). In our centre, human islets use was approved by the institutional review board, and patients included in the clinical trial (NCT01148680 – Trial comparing metabolic efficiency of islet graft to intensive insulin therapy for Type-1 diabetes’ treatment) signed a consent form.
Advantages and disadvantages
The subcapsular space appears to be the optimal site for pancreatic islets. Indeed, it is well vascularized allowing enough oxygen to ensure the physiological dynamics of hormone secretion. Moreover, this site shows limited local inflammation after transplantation, promoting long-term survival. Also, the technique is minimally invasive so the graft can be easily removed for histological studies. Thus, the efficiency of the subcapsular site to study pancreatic islets is well established. 18 It combines advantages in being (a) a relatively easy technique for beginners, (b) well vascularized and (c) a technique that limits cells in a specific area, allowing for optimal histological studies. However, it is not without risk: the kidney capsule is thin and fragile, and even more so in immunocompromised nude mice. Possible cell leakages and fatal haemorrhages must be taken into account.
Technical approach – single or double graft
Animals used for transplantation are immunocompromised adult mice, housed in a specific opportunist- and pathogen-free animal facility, allowing xenografts. Both sexes can be used, depending on the study. Often, they are nude mice from various strains (CRL:NU(Ico)-Foxn1nu from Charles River Laboratories, Rj:NMRI)-Foxn1nu nude from Janvier Labs, or CD1-Foxn1/J from Jackson Laboratory). Other strains are used for more specific protocols such as CB17/lcr-Prkdcscid, B6.129S7Rag1tm1Mom, NOD.CB17/lcr-Prkdcscid or NSG mice (Jackson Laboratory, Charles River Laboratories, Taconic).
Cells dedicated to clinical transplantation are concentrated by centrifugation and aspirated in a blunt needle catheter (24–27 Gauges) with a syringe. Optimally, a syringe with a rotary plunger such as the model 1001 type C from Hamilton company (Nevada, USA) is used to (a) minimize the dead volume, (b) obtain high accuracy, precisely control the injection and (c) restrict to a minimum the air bubbles underneath the kidney capsule.
After anaesthesia, a lumbar laparotomy of the skin and muscle is performed to access the kidney (Figure 1(a)). The kidney is maintained outside the abdominal space and protected, to avoid drying of the capsule, so that it keeps its elasticity (Figure 1(b)). An incision (1–2 mm) is made to make way for the catheter in order to dispense the microvolume of cellular pellet to the opposite side of the kidney (Figure 1(c) and (d)). After injecting the graft, the catheter is removed, and the capsule is cauterised to avoid haemorrhage and cell leakage (Figure 1(e)). Suturing with 5/0 or 6/0 monofilament is done separately on muscular and skin layers. Intraperitoneal or subcutaneous morphine administration (buprenorphine, 0.05 mg/kg) helps to alleviate perioperative pain. The mice, identified with ear tags (National Band & Tag Company, Newport, USA), gain consciousness after a few minutes. The operative process lasts 10–15 min during which cardio respiratory function is monitored. Mice that have undergone surgery are housed, together, in a clean cage, maximum five per cage according to their weight; light/dark 12 h/12 h; food and water ad libitum; 22 ± 2°C. Individual ventilated cages are enriched with tunnels and nesting fibre (SAFE, France). Animals are checked daily.
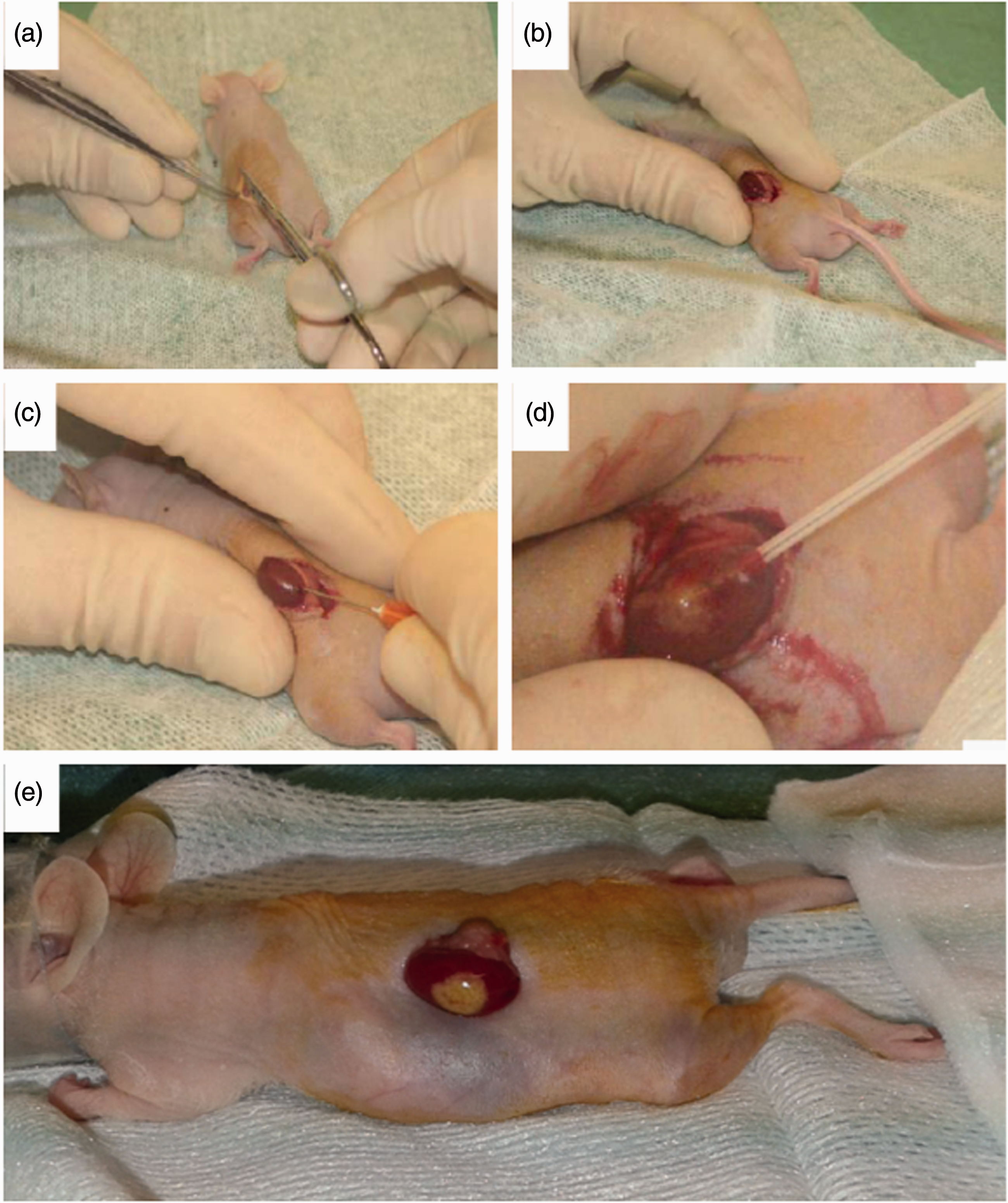
Main steps of transplantation under the kidney capsule. (a) Dorsal laparotomy is performed to expose the kidney. (b) Kidney is maintained outside the cavity and irrigated to avoid the capsule from drying. (c) and (d) Capsule incision is undertaken to allow access via a catheter for cell injection. (e) Transplanted islets are visible through the transparent capsule
Teams upgraded this model and used a bilateral subcapsular islet transplantation, which can be carried out in different ways depending on the purpose of the experimentation. On one hand, Jiao et al. performed double islet transplantations during the same surgery process to assess human and rodent cells under the same conditions of drug delivery. 19 On the other hand, others implemented a 2–4 weeks recovery period between the two surgical procedures. 20 In both cases, they minimized animal use by grafting cells under each of the two kidney capsules. This way each graft-bearing kidney can be used for different tests (see below).
Refinement and model use
Graft function: dynamic hormone secretion testing
Routine follow-up of immunodeficient mice with a human islet graft includes measuring weight and glycaemia. Weekly or bi-monthly blood samples according to the volume collected (7.5% total blood volume per week) 21 to measure pancreatic hormone levels and confirm graft function. Many teams use this model without hormone quantification and are interested in the islet transplant’s capacity to restore glucose levels to normal in hyperglycaemic mice that were made diabetic by streptozotocin or alloxan treatment.22,23
Insulin is well conserved between species. Due to a high rate of cross-reactivity, for years, it was hard to discriminate mouse insulin from human insulin. However, the c-peptide, equimolarly secreted with insulin by beta cells, has no cross-reactivity between rodents and humans. There are many specific kits (ELISA, EIA, etc.) that allow the specific measurement of the human c-peptide, secreted from transplanted human cells. Using this technology, we can evaluate the robustness of the model. Pasquetti et al. confirmed the high degree of reproducibility in this model between two mice, grafted with the same amount of islets (Figure 2(a)). 11 Moreover, fine-tuning of the method allows testing from minimal plasma or serum volumes (5–25 μl), which allows higher frequency of sampling and increases the precision of the follow-up of graft functionality.
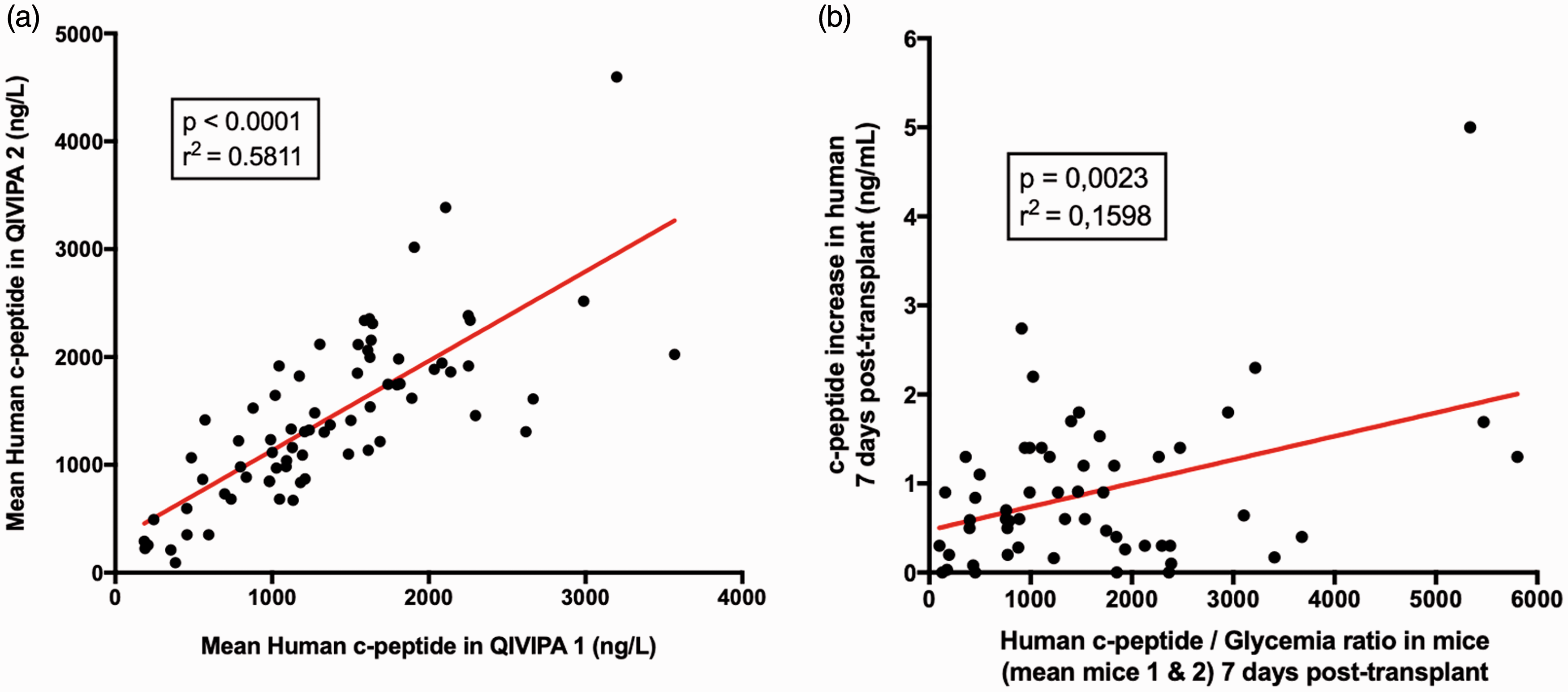
Human c-peptide dosage in plasma demonstrates the high reproducibility of the technique (r2 = 0.5811) between two mice grafted with the same number of islets (n = 146). (b) Correlation between c-peptide increase in transplanted patients and grafted islets in mice (c-peptide/glycaemia). Chimeric model is a good indicator of the clinical transplant functionality (n = 56 patients). (This figure was published in Transplantation, Bioengineering, and Regeneration of the Endocrine Pancreas, vol. 1, Pasquetti et al., In vivo quality control of human islets in the immunodeficient mouse to predict islet function in man: A retrospective study in 87 clinical transplants, 711–724, Copyright Elsevier, 2019.
The ‘human–mouse’ chimera predicts graft function in patients
Caiazzo et al. first showed in this QIVIPA model (Quantitative in-vivo Islet Potency Assay) that there was a high correlation between the c-peptide dosage in immunodeficient mice receiving 1% of human islet grafts and c-peptide increase in transplanted patients receiving 99% of the islets, suggesting that this in-vivo model is a good predictor of clinical transplantation in humans. 24 Recently, Pasquetti et al. confirmed this result with 87 clinical islet preparations transplanted in 165 mice (Figure 2(b)). 11
According to Burch and Russell and the application of the 3Rs, in-vitro methods are explored to predict in-vivo or clinical function. 17 Preliminary data suggests that dynamic insulin testing of islets by perifusion in vitro could predict transplant function in mice. If confirmed, this strategy could reduce and replace the use of animals in this research discipline. 25
Environmental impact on islet grafts: the high-fat diet model
After transplantation, drug treatments are routinely done to verify toxicity or beneficial effects on the phenotype of pancreatic cells. 26 It is also possible to feed transplanted mice with a specific diet. Thereby, Gargani et al. studied the effects of a high-fat diet on human cells and showed that pancreatic islets were able to adapt their mass and their function after several weeks on a high-fat diet. 27 Rescuing the human islets exposed to an obesogenic environment in vivo with drugs may make an ideal preclinical Phase 0 model prior to clinical translational in humans. 28
Environmental impact on islet grafts: the transgenic mouse
Different cell types such as stem cells are routinely transplanted under the kidney capsule of normoglycaemic immunodeficient mice, that can subsequently be rendered diabetic with streptozotocin, which only destroys mouse beta cells. 9
Alternatively, when interested in determining the direct effect of a gene on a specific cell type but only a total transgenic knockout (or knock-in) mouse model is available and not a cell-specific knockout, and the specific gene is expressed in multiple cell types or centrally and peripherally, a cell-specific transgenic mouse (knockout or knock-in) can experimentally and surgically be created from a total transgenic mouse. The specific cell type can be isolated from transgenic mice. The cells can then be transplanted in a wildtype or immunodeficient mouse 29 (Figure 3). Accordingly, Dror et al. specifically studied the phenotype of pancreatic cells from wild-type or interleukin-1 receptor-deficient mice (Il1r−/−), that were first isolated from the total knockout animals and subsequently transplanted in diabetic immunodeficient mice, creating surgically-induced islet-specific transgenic mice. This allowed the team to show the major role of IL1ß in post-prandial insulin secretion. 29
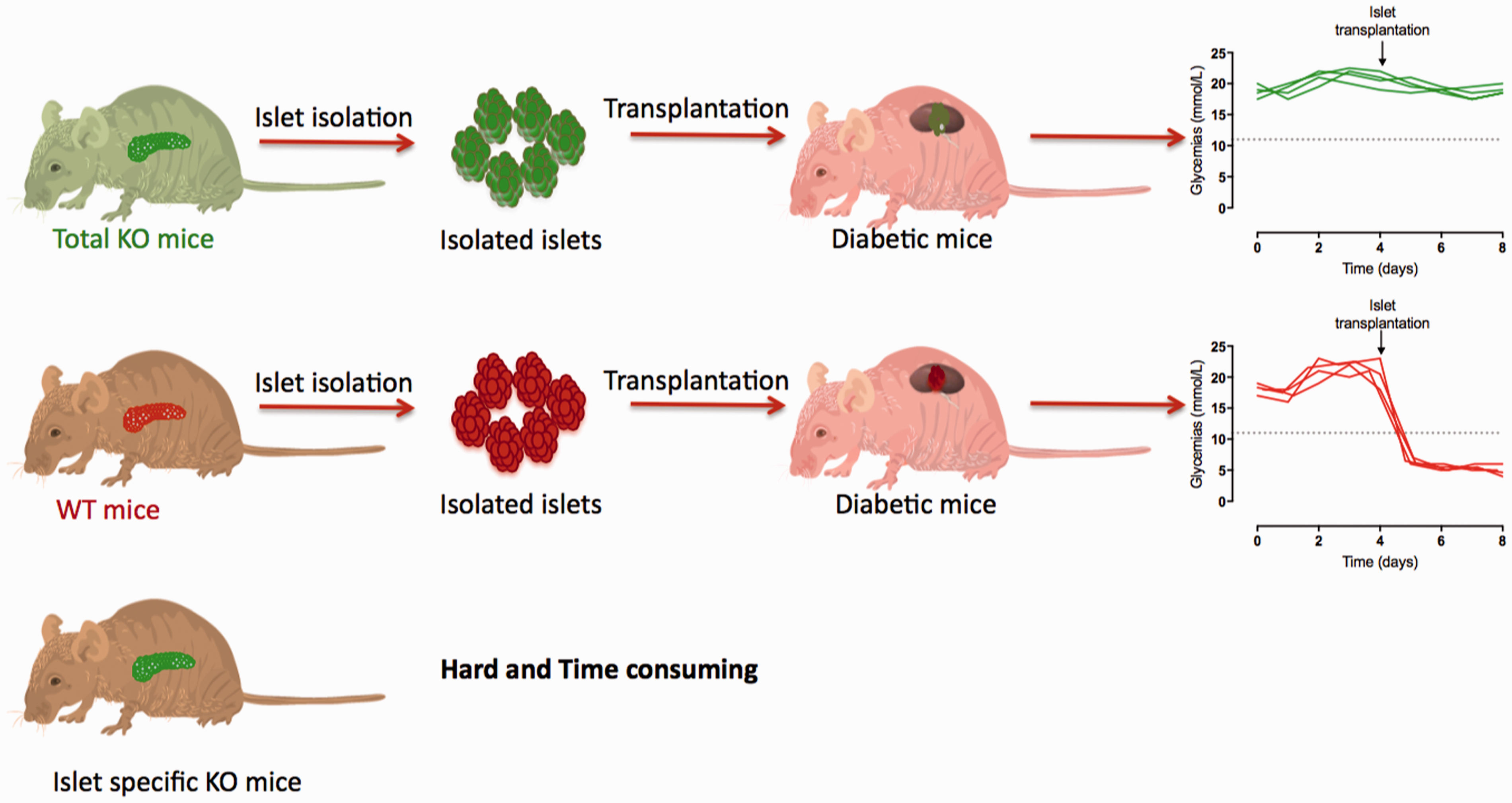
Cell-specific transgenic mouse model. Specific isolated cell type from wild type or mutant mice can be transplanted in another mouse to define their capacity to cure diabetes of the recipient host. Created and modified with picture from wikimedia commons (licensed under the Creative Commons Attribution 4.0 International License, DataBase Center for Life Science, http://togotv.dbcls.jp/ja/togopic.2014.63.html).
Refining the model, a step further for ex-vivo and molecular studies
Histology
At the end of the protocol, animals are euthanized by cervical dislocation, after anaesthesia, as recommended 30 and the graft-bearing kidney can be exploited in several ways. First of all, histology or immunohistochemistry is generally done. For that, the initial step is devoted to finding the graft on the paraffin-fixed kidney, which is completely sectioned and stained with Masson’s trichrome (Figure 4(a) and (b)). Sections can also be stained with specific antibodies to discriminate the different hormonal markers and identification of proliferation, apoptosis (Figure 4(c)). Graft volume can be quantified with planimetry. Alternatively, kidneys can be rendered transparent with the iDISCO clearing method 31 which has recently been applied to determine human islet graft volume under the kidney capsule. 32
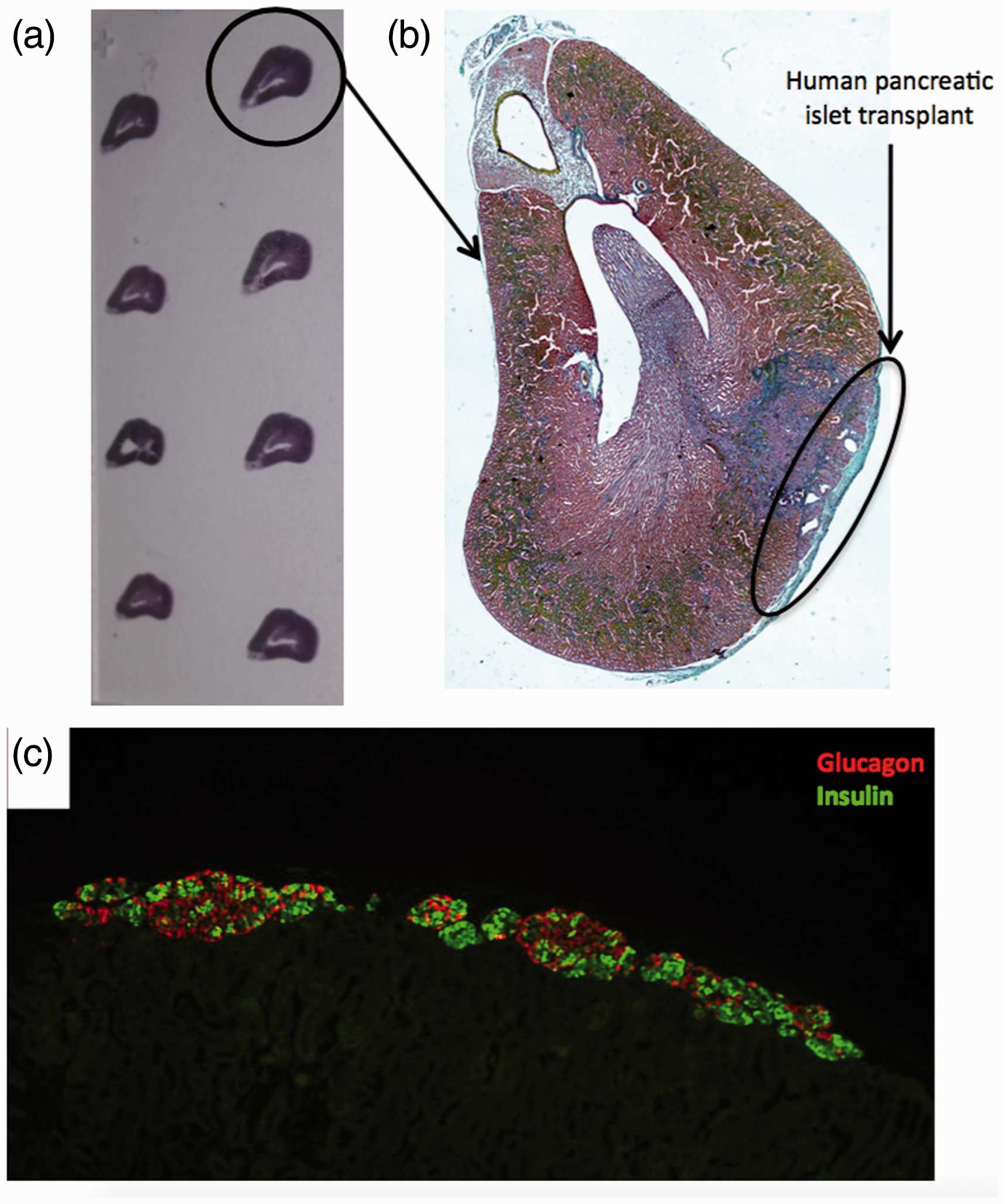
Histological sections of graft bearing-kidney. (a) The entire kidney is sectioned and one section is mounted every 50 microns. (b) Masson’s trichrome staining (c) Immunofluorescence of transplanted human islets revealing pancreatic alpha (anti-glucagon antibody in red) and beta (anti-insulin antibody in green) cells.
Graft-bearing kidney dissection for molecular biology and perifusion
Intracellular insulin can also be determined. However, a more meticulous approach allows the dissection of the cellular transplant from the kidney (Figure 5(a) and (b)). First of all, it allows the extraction of proteins, RNA or DNA, of very good quality, making it easy to look for specific factors 33 (Figure 5(c)). Explanted human transplants containing beta, alpha and delta pancreatic cells could be studied more functionally by the technique of perifusion 34 (Figure 5(d) and (e)). Cellular grafts are placed in a thermostatic chamber (37°C) and are submitted to a constant feed of glucose medium. Effluents are collected for quantitative assays by a fraction collector. Multiple hormones can be simultaneously tested (somatostatin glucagon, insulin). Preliminary data showed that several months after transplantation, transplants remained functional and glucose sensitive (Figure 5(f)).
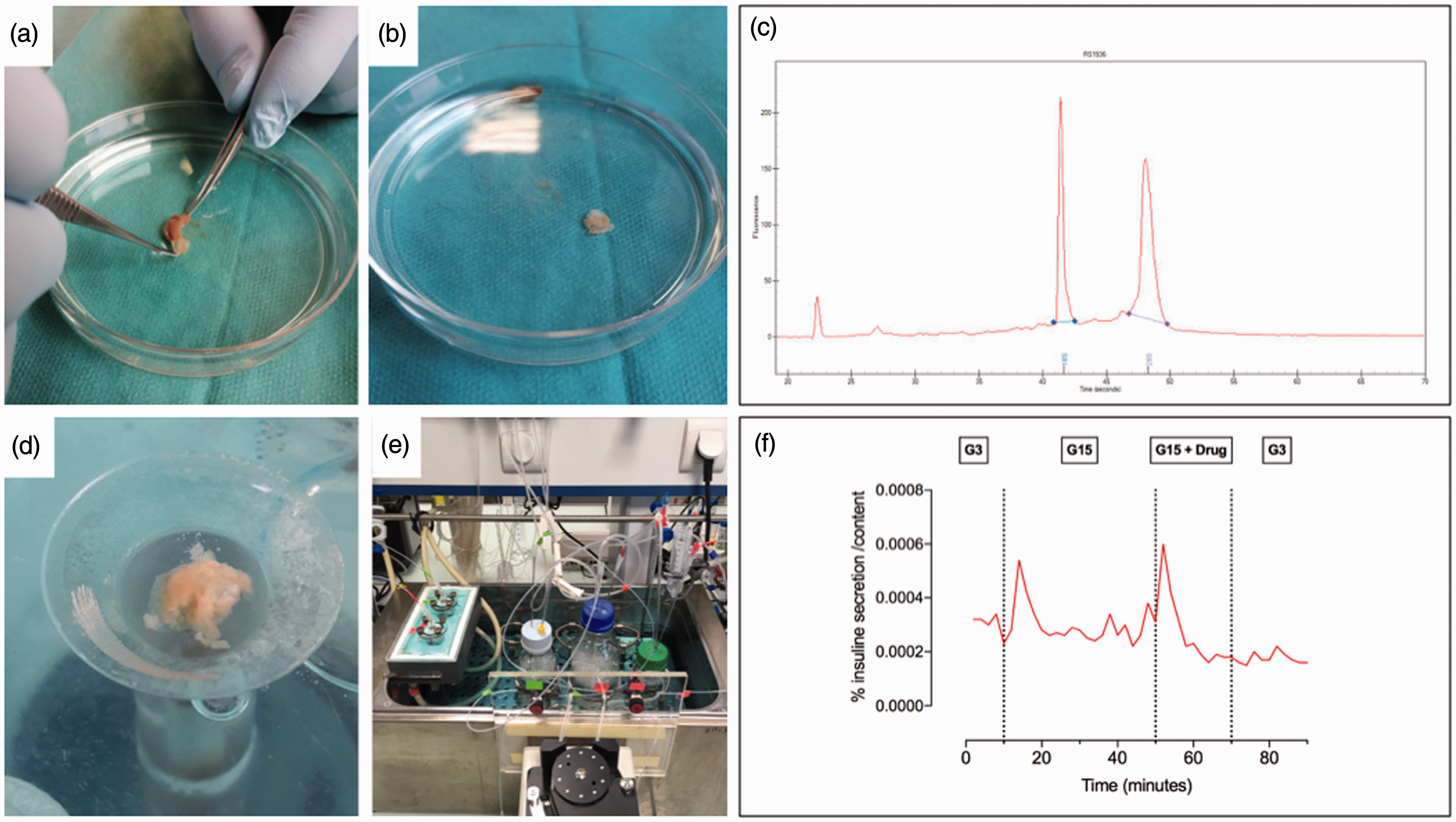
Graft-bearing kidney dissection for RNA extraction or perifusion. (a) and (b) Islet graft is removed from the kidney in a liquid phase to avoid desiccation. (c) RNA quality of extracted transplant. 33 (d) Human transplants are placed in a thermostatic chamber. (e) Effluents are collected by fraction for hormone assays. (f) Cells are studied according to several phases: first phase at 3 mM glucose (G3), followed by a stimulation with glucose at 15 mM ± drug, finished by a final basal phase at 3 mM glucose.
Conclusion
Pancreatic cell transplantation under the kidney capsule is an effective and reproducible technique. It is considered as the gold-standard for human islet in-vivo evaluation. The kidney capsule has all the advantages of an optimal cell transplantation site for evaluating the functionality of the transplant. This model could be used to test toxicity or the beneficial effects of different compounds over a longer period than in-vitro studies. 26 It has become an essential model in pancreatic research. 29 Furthermore, cellular transplant studies are no longer limited to histology; modern techniques allow access to much more information. Generating more information with less animals is the goal. Hence, this model is part of a concrete process of refinement and reduction in the number of animals used in research.
Footnotes
Acknowledgements
We sincerely thank Audrey Quenon, Thomas Hubert and the animal facility team (Julien Devassine, Kim Letten, Cyrille Degraeve, Caroline Declerck) for their help. We also thank the coordination teams and surgeons for pancreas procurement and we thank Nathalie Delalleau, Anaïs Coddeville, Isanga Aluka, Amanda Elledge, Pauline Petit for islet isolation, and Ericka Moerman and Gianni Pasquetti for evaluation. Caroline Bonner, Chiara Saponaro, Ana Acosta, Alix Vaissié, Rofigua Hakem and Alexandre Thevenot are also thanked, and Marie-Hélène Gevaert is thanked for histology and Masson’s trichrome. We would like to thank Amanda Elledge for preparation of the manuscript, Markus Muehlemann and Lorea Zubiaga for abstract translation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from Agence de BioMédecine (2014), Association pour la Recherche sur le Diabète (2015), Agence Nationale de la Recherche (European Genomic Institute for Diabetes – Grant number ANR-10-LABX-46) and JDRF-ECIT (Juvenile Diabetes Research Foundation – European Consortium for Islet Transplantation – Grant number #2RSC-2019-726-I-X).
