Abstract
The aim of this study was to describe the characteristics and validate the use of a new, custom-built automatic smoking machine (ASM), primarily designed to study the effects of an environmental tobacco smoke surrogate (ETS surrogate) exposure in animals of various sizes, including large animals. The equipment includes a programmable ASM coupled to a vented whole body chamber, where animals can be exposed to both mainstream and sidestream smoke. The user-friendly interface allows for full programming of puff volume (1–60 mL), time interval between two puffs (1–60 s) and between two cigarettes (1–60 min). Eight newborn lambs were exposed to either 10 (4 lambs, C10 group) or 20 (4 lambs, C20 group) cigarettes, 8 h per day for 15 days. Four additional control, lambs were exposed to air (C0 group). Weight gain was identical in all three groups of lambs. Urinary cotinine/creatinine ratio increased with the number of cigarettes smoked (C0: 11 ± 7 ng/mg; C10: 961 ± 539 ng/mg; C20: 1821 ± 312 ng/mg), with levels in the C10 and C20 groups in keeping with values published in infants exposed to ETS. Overall, results show that our new ASM is especially well suited for ETS surrogate exposure in non-restrained, non-anaesthetized large animals such as sheep.
The early postnatal period is critical due to the immaturity of control centres involved in vital functions such as respiration, cardiovascular function, sleep–wake cycles, thermoregulation and swallowing function. Various innate or acquired factors can disrupt normal development and maturation of these functions, paving the way for frequent pathologies such as apparent life-threatening events of infancy or sudden infant death syndrome (SIDS). Following worldwide campaigns to prevent prone sleeping, perinatal passive exposure to tobacco smoke is now considered to be the single most important cause of preventable death by SIDS. 1–7 In fact, it has been calculated that one-third of reported SIDS deaths could have been prevented with avoidance of prenatal exposure to tobacco smoke. 8 Moreover, postnatal tobacco exposure has been reported to increase SIDS by 2- to 3-fold. 9 Finally, infants who died from SIDS tend to have higher concentrations of nicotine in their lungs than controls. 10
Most studies on the effect of environmental tobacco smoke (ETS) exposure in the perinatal period have focused on nicotine alone. However, of the at least 4000 different chemical compounds present in tobacco smoke, more than 100 are toxic, suggesting that animal studies focusing on nicotine should be complemented by studies on ETS exposure. In addition, studies on ETS must take into account that a burning cigarette produces a combination of mainstream smoke (MS) (inhaled, then exhaled into the environment by the smoker) and sidestream smoke (SS) (produced by a passively-burning cigarette). Indeed, it has been previously shown that the relative composition of both types of smoke is different. 11,12
Various smoking machines have been built for the tobacco industry over the years, with the primary aim of assessing and/or modulating the levels of various compounds in cigarette smoke. With time, smoking machines have refined from a manually operated system to fully automatic and programmable systems. However, a significant drawback of such systems designed for testing cigarettes by the industry resides in their complexity.
While several animal studies on the effects of ETS in the perinatal period have also used automatic smoking machines (ASMs), few bear significant relevance to SIDS pathogenesis. 13 Interestingly, some effects of postnatal exposure alone on brain cell development have been found to be identical to the effects of prenatal added to postnatal exposure in rats and monkeys. 13,14 In recent years, we became especially interested in assessing the effects of early postnatal ETS on cardiorespiratory control in our newborn ovine models. Such models have been used because of the unequalled amount of knowledge on fetal and neonatal cardiorespiratory physiology, the possibility to study preterm lambs and the ability to study numerous cardiorespiratory physiological signals during prolonged polysomnography in unrestrained, unanaesthetized, chronically instrumented animals. However, commercially available ASMs failed to meet our needs for a programmable, user-friendly and compact system allowing the assessment of the effects of both SS and (exhaled) MS smoke in freely moving, large developing animals during several days. We therefore designed and built a new system under close collaboration between the Departments of Physiology and Mechanical Engineering of the Université de Sherbrooke. The aim of the present study was thus to validate our custom-built system in newborn lambs exposed to cigarette smoke for 15 days.
Material and methods
Animals
Twelve mixed bred lambs (Dorset and Romanov species) were included in the study. All lambs were born at term by spontaneous vaginal delivery in a local farm, then transferred on the same day into our animal quarters. On their arrival, they were immediately examined for clinical normality and received an intramuscular injection of 0.75 mg selenium, 35 IU vitamin E, 1.25.105 IU vitamin A and 15.103 IU vitamin K. Daily cigarette smoke exposure was performed from day 1 to day 15, for a period of 8 h per day in a Plexiglas exposure chamber with a stainless-steel floor covered by a soft absorbent mattress. Between exposure periods, lambs were kept in pairs in an animal pen with wood shedding and hay. Daylight cycle was 06:00–18:00, humidity 50% and ambient temperature 24–26°C, as recommended by the Canadian Council on Animal Care. Lambs were bottle-fed with ewe milk throughout their stay in our animal quarters, but were not given colostrum. The study protocol was approved by the Animal Care and Use Committee of the University of Sherbrooke.
Equipment
A full system including an automatic, programmable cigarette smoking machine and a whole body exposure chamber was designed and built to expose freely moving lambs to both MS and SS. In brief, cigarette smoke is produced by an ASM set to mimic the action of a smoker. Both MS and SS are circulated in a whole body exposure chamber coupled with an in-line fan, which vents the smoky air out of the chamber via a filtration unit and into the main air evacuation system of the room.
Automatic cigarette smoking machine
The apparatus is comprised of several components allowing for the automatic smoking of cigarettes, according to researcher-programmable parameters, and to produce both MS and SS (Figure 1).
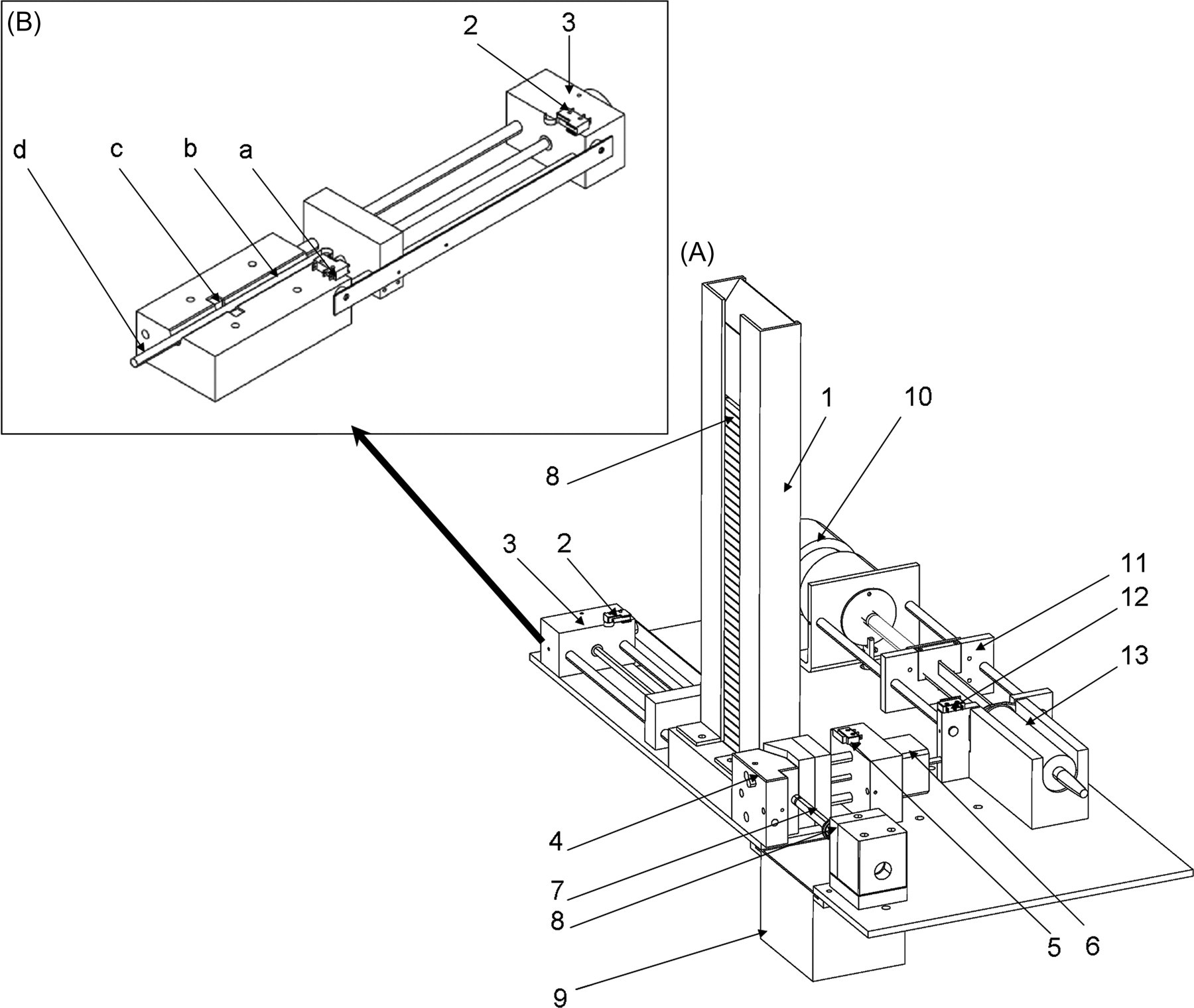
Technical schematics of the automatic smoking machine without tubing. A: 1. cigarette magazine; 2. limit switch sensor; 3. 24-volt motor; 4. holding unit; 5. limit switch sensor; 6. step-by-step motor; 7. cigarette; 8. car lighter; 9. ashtray; 10. 24-volt DC motor; 11. screw and sliding rods; 12. limit switch sensor; 13. 60 mL plastic syringe. B: Extracting unit: a. limit switch sensor, b. 13 mm slot, c. photomicrosensor, d. extracting rod
Extracting unit
The extracting unit consists of a cigarette magazine and an extracting system. The cigarette drops down by gravity from the cigarette magazine to a 13 mm slot where a photomicrosensor (EE-SPX303, Omron Canada Inc, Toronto, ON, Canada) detects its presence. Thereafter, a 24 V motor (S1054B, Colman Motor Products, Des Plaines, IL, USA) activates an extracting rod, which pushes the cigarette through the slot (from left to right, see Figure 1) to the holding unit. Two limit switch sensors (5A250V, Omron Electronics, Toronto, ON, Canada) and a photomicrosensor (EE-SX872, Omron Electronics) are responsible for the precise positioning of the extracting rod.
Holding unit and lighting unit
When the cigarette pushed by the extracting rod reaches the lighter, a Mini-Beam sensor (SM312 FPH, Banner, Minneapolis, MN, USA) confirms the presence of the cigarette and activates the holder closure on the filter. Holder closure/opening is powered by a step-by-step motor (Z817G BKN-10-6, Eastern Air Devices, Dover, NH, USA). The open state is assured by a limit switch sensor (Omron Electronics), while the closed state is assured by an inductive sensor (DC 3-/4-Wire M8, Balluff Canada Inc, Mississauga, ON, Canada) and a step motor driver (2035, Applied Motion Products, Watsonville, CA, USA). As soon as the cigarette is firmly placed in position, a car lighter (212111, Casco Product Corporation, Bridgeport, CT, USA) is activated by a photomicrosensor for 2 s. The cigarette lighter unit is PVC isolated from the rest of the machine to prevent heat transfer and an electric transformer is connected to the lighter cable to prevent electrical transfer.
Smoking unit
The smoking unit includes a 60 mL plastic syringe plus tubing to collect the MS and SS from the burning cigarette as well as vent it out to the exposure chamber. The unit is powered by a 24 V DC motor (22VM51-020-5, Honeywell POMS, Herndon, VA, USA) connected to the piston of the 60 mL syringe via a screw and sliding rods. The syringe piston is pulled to aspirate the cigarette smoke from the holder unit to the syringe through a rubber tube (Fisherbrand diameter: 3/8”; wall thickness: 1/16”, Pure Natural Rubber Tubing, Fisher Scientific, Ottawa, Canada). The syringe piston is then pushed to vent the smoke out of the syringe to the exhaust hose of the smoking machine (tumble-dryer vent hose) through a second similar rubber tube. Both tubes are connected to the syringe using a Y connector. The inflow and outflow from the syringe is assured by a pinch valve activated by a solenoid (HS2506, Kuhnke Automation Inc, Wayne, NJ, USA). When the cigarette is detected as fully smoked (7 mm before the filter) by a Mini-Beam sensor (SM312 FPH, Banner) or when the preset time limit (5 min) is reached, the cigarette holder opens and the extracting rod subsequently pushes the cigarette into the ashtray below half-filled with water. The smoking machine is enclosed in an airtight box made of stainless steel and Plexiglas. The exhaust hose is located on the superior portion of the right side of the box and is connected to the exposure chamber. An in-line fan, located on the other side of the exposure chamber, continuously vents both the MS and SS from the box into the exposure chamber and then to the main air evacuation system of the room.
Control system
A Programmable Logic Controller (VersaMax Micro PLC, GEFanuc, Charlottesville, VA, USA) ensures the overall control of the smoking machine. The controller is connected to a graphical interface (Data panel 45, GEFanuc), which allows for easy control of a number of parameters to reproduce various smoking habits and hence various smoke exposures. The adjustable parameters include the number of cigarettes to be smoked for a given exposure (1–40 cigarettes), the time interval between two cigarettes (1–60 min), the volume of each puff (1–60 mL) and the time between two puffs (1–60 s). Duration of puffs is set at 2 s. Overall, this user-friendly interface allows for a high versatility of the control system.
In our laboratory, the ASM is operated in a room fully equipped with a continuous monitoring and alarm system (temperature, humidity, pressure, ventilation system, smoke detection). Hence, no monitoring and alarm system is included in the ASM itself, apart for an emergency stop button to prevent hazards during maintenance. The ASM is operated during daytime only, with at least hourly observation by a dedicated technician trained to check for good running of the equipment.
The exposure chamber
The whole body exposure chamber is composed of 1 cm thick Plexiglas. Dimensions are 1.2 m (length) × 1.2 m (width) × 1 m (height) with a floor surface of 1.44 m 2 , in accordance with Canadian Council on Animal Care standards for housing either one pregnant ewe or two newborn lambs at the same time. The chamber is airtight, easy to clean and simple to move using wheels. Air is vented from the smoking machine into the chamber through a tumble-dryer vent hose (diameter: 10.2 cm) using an in-line fan (PF100P Marbuco, Sherbrooke, QC, Canada). Air flows out of the chamber through an identical hose and an exhaust filtration unit attached to the room ventilation system. The exhaust filtration unit is comprised of a foam prefilter for big particles, a charcoal filter and a high efficiency particulate air (HEPA) filter. The in-line fan is permanently set to provide the level of ventilation required by the Canadian Council on Animal Care for one ewe or two lambs, i.e. 0.6–0.7 m 3 /min. Calibration and setting of the fan can be modified using a hand-held digital airflow/temperature meter (HHF92A, Omega Canada, Laval, QC, Canada), ultimately allowing the chamber to be adapted to different animal species.
Design of the validation study
For this study, the ASM was pre-set at 2 s puff duration, 35 mL puff volume (in accordance with ISO 3308 norms) and an interval of 30 s between two puffs. Measurements of carbon monoxide levels using the Q-trak plus 8554 system (TSI Inc, Shoreview, MN, USA) and particulate matter (including particulates <10 μm and respirable particulates <2.5 μm) using the TSI Dustrak 8520 (Ashtead Technology, Montreal, Canada) were performed in the exposure chamber in C10 and C20 conditions during a 4 h period to assess basal characteristics of our exposure conditions in the absence of the lambs.
At their arrival in our animal quarters, all lambs underwent sterile surgery at day 1 of life under local anaesthesia (xylocain 2%) in order to introduce an arterial catheter into the brachial artery to collect blood samples for measuring pH, arterial PO2 and PCO2, HCO3 − concentration and haemoglobin oxygen saturation. The catheter was left in place for the entire duration of the study and flushed twice daily with heparin solution. Daily exposure to cigarette smoke (Peter Jackson King size, the most popular brand in Quebec at the time of the study) was performed from the first to 15th day of life from 08:00 to 12:00 and from 12:30 to 16:30. At 12:00, lambs were bottle-fed with ewe milk ad libitum and a urine sample was collected for cotinine and creatinine measurements (24 hour U-Bag for newborn, Libertyville, IL, USA). Before and after each daily exposure, lambs were also bottle-fed ad libitum with ewe milk. Body temperature and weight were measured daily at the beginning of the exposure and an arterial blood sample was collected at the beginning and at the end of the exposure. Three groups of randomly selected lambs were studied: four control lambs were housed in the exposure chamber throughout the 15-day period, but exposed to air only (C0); four other lambs were exposed to 10 cigarettes per day (C10); and four lambs were exposed to 20 cigarettes per day (C20). Lambs were systematically exposed in pairs in the Plexiglas chamber, at a temperature of 24–26°C, according to guidelines from the Canadian Council on Animal Care for newborn lambs. Wellbeing of the lambs was ensured throughout the exposure period by hourly observation by the technician specialized in animal care and assigned to good running, checking of the ASM. No recording was performed during exposure. Usual endpoints for lambs were included in the protocol accepted by our Institutional Animal Care and Use Committee.
At day 12 of life, aseptic surgery was performed under general anaesthesia (1–2% isoflurane; 30% NO2; 68% O2). Atropine sulphate (150 μg/kg intramuscularly) was given preoperatively with ketamine (10 mg/kg). Antibiotics (5 mg/kg gentamicin and 7500 IU/kg duplocillin) were administered intramuscularly before surgery and daily thereafter until the end of the experiment. One dose of ketoprofen (3 mg/kg intramuscularly) was systematically given immediately after induction of anaesthesia for analgesia; an identical dose of ketoprofen was repeated after surgery if needed. Two E2-12 platinum needle-electrodes (E2-12, Grass Instruments Company, Quincy, MA, USA) were glued on ribs at the level of the proximal forelegs for recording electrocardiogram. One E2-12 platinum needle electrode was also inserted under the scalp as a ground. Leads from these electrodes were subcutaneously tunnelled to exit on the back of the lamb. In addition, custom-made electrodes were inserted into a glottal adductor for recording electromyographic activity and two platinum needle electrodes were inserted into the parietal cortex directly through the skull for electrocorticogram recording, as part of another protocol aimed at studying the effect of ETS on swallowing–breathing coordination. At the end of the 15-day exposure period, i.e. three days after surgery, a polysomnographic recording was performed during 4 h in freely moving lambs while in the Plexiglas chamber, but after completion of smoke exposure. Just before the recording, two respiratory inductance plethysmography bands were placed on the thorax and the abdomen and a nasal thermocouple glued on the lateral aspect of the nostril for monitoring respiration. Heart and respiratory rates calculated from those recordings (Acknowledge 3.7.3 software, Biopac, Santa Barbara, CA, USA) were used in the present validation study. Following completion of the polysomnographic recordings, lambs were euthanized with an intravenous overdose of pentobarbital (90 mg/kg). The larynx and first 2 cm of the trachea were collected and fixed in 10% formaldehyde for histological assessment of local inflammation.
Data analysis
Weight and arterial blood gases were averaged daily for each group of lambs. Arterial blood gases were corrected for lamb temperature. 15 At day 15, heart rate (HR) and respiratory rate (RR) were calculated for each stable 60 s epoch and averaged in each lamb over the entire recording. Urinary cotinine was measured using an ELISA immunoassay kit (Bio-Quant Cotinine Direct ELISA, San Diego, CA, USA). The collected urine samples (3 mL) were stored at −20°C until measurement. Cotinine dosage was preferred to nicotine because of its longer half-life (15–20 h versus 30 min–2 h, respectively), its slow renal elimination and high urinary concentration (6- to 25-fold nicotine concentration). Creatinine was measured in the Department of Clinical Biochemistry at the Sherbrooke University Hospital using a Vitros 950 chemistry system (Ortho Clinical Diagnostics, Raritan, NJ, USA). Cotinine/creatinine ratio was calculated at days 14 and 15 and first averaged for each lamb and thereafter for each group. The collected laryngeal tissues were grossly sectioned and placed in a cassette for dehydration and fixation in paraffin. Paraffin blocs were cut in 3 μm slices using a microtome and stained with eosin–haematoxylin. Inflammation was then graded for epithelial and subepithelial changes at the level of the larynx and epiglottis. 16
Results
Functioning of the ASM
The ASM met all our requirements for studying ETS exposure (both SS and MS) in freely moving lambs for 15 days, while providing a versatile, user-friendly interface. Two resolvable problems were encountered during the validation period. The first was related to sleep disruption of the lambs by the too noisy ASM, which was solved by enclosing the ASM in a stainless-steel and Plexiglas box. The second problem was related to the cigarette magazine; gravity was not always sufficient for the cigarette to drop down. This was also rapidly solved by adding a small weight (copper ‘cigarette’) on top of the cigarette stack.
Behaviour, weight gain and cardiorespiratory function
All lambs except one (diarrhoea for 8 days) tolerated the 15-day exposure to cigarette smoke without any apparent problems. Indeed, no differences in sleep, respiration and feeding were clinically apparent between controls and exposed lambs. Figure 2 illustrates that mean weight at the onset of exposure and weight gain (C0: 126 ± 23 g/day; C10: 157 ± 49 g/day; C20: 141 ± 65 g/day) were identical in the three groups. However, although not quantified, an increase in spontaneous activity during wakefulness was noted in C10 and especially C20 lambs. Of note, lambs did not show any sign of distress while in the exposure chamber.
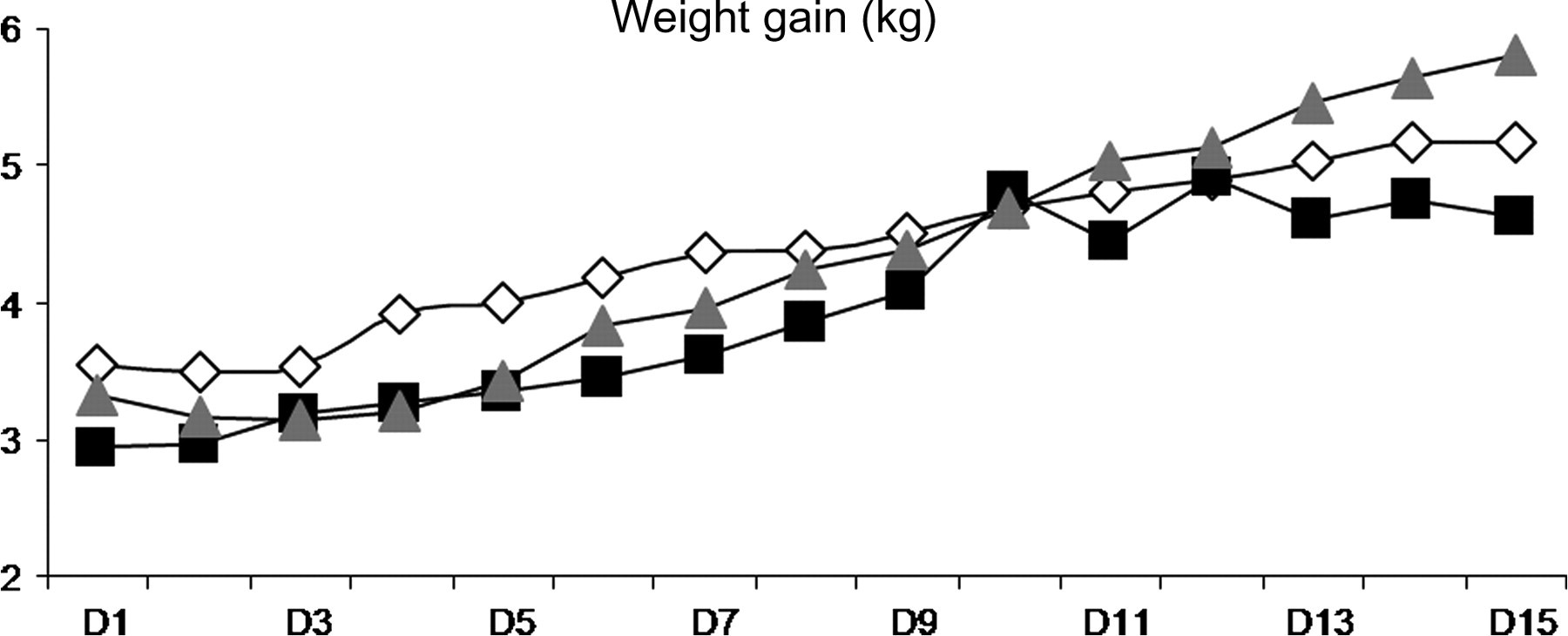
Mean weight throughout the 15 days of exposure. White diamonds are for control lambs (C0), black squares for daily exposure to 10 cigarettes (C10), grey triangles for daily exposure to 20 cigarettes (C20)
Results on resting RR, calculated from polysomnographic recordings performed at postnatal day 15, showed no differences between groups (C0: 41 ± 10 min−1; C10: 38 ± 9 min−1; C20: 37 ± 8 min−1), while C20 exposure seemed to increase HR (C0: 178 ± 26 min−1; C10: 176 ± 14 min−1; C20: 191 ± 15 min−1). Arterial blood gas values, obtained for control, C10 and C20 lambs, were, respectively, PaO2 = 85 ± 5 mmHg, 88 ± 6 mmHg, 92 ± 11 mmHg; PaCO2 = 44 ± 8 mmHg, 46 ± 4 mmHg, 42 ± 2 mmHg; pH = 7.36 ± 0.05, 7.40 ± 0.04, 7.41 ± 0.06 and [HCO3 −] = 23 ± 2 mmol/L, 27 ± 4 mmol/L, 25 ± 2 mmol/L; haemoglobin saturation in O2 = 95 ± 4%, 97 ± 1%, 97 ± 1%.
Urine cotinine measurement
Mean values of urinary cotinine/creatinine ratio at days 14 and 15 were 11 ± 7 ng/mg for C0 lambs, as compared with much higher values obtained in both the C10 group (961 ± 539 ng/mg) and C20 group (1821 ± 312 ng/mg).
Carbon monoxide and particulate matter measurement
Carbon monoxide was measured in the Plexiglas chamber in the absence of lambs, while temperature was 20.9 ± 0.1°C and relative humidity 43.2 ± 1.1%. Cigarette burning was consistently responsible for a peak in CO (from 9 to 10 ppm) during 10–15 min, and CO value was zero between peaks. CO peaks were twice as frequent in C20 lambs comparatively to C10 lambs, as shown in Figure 3.
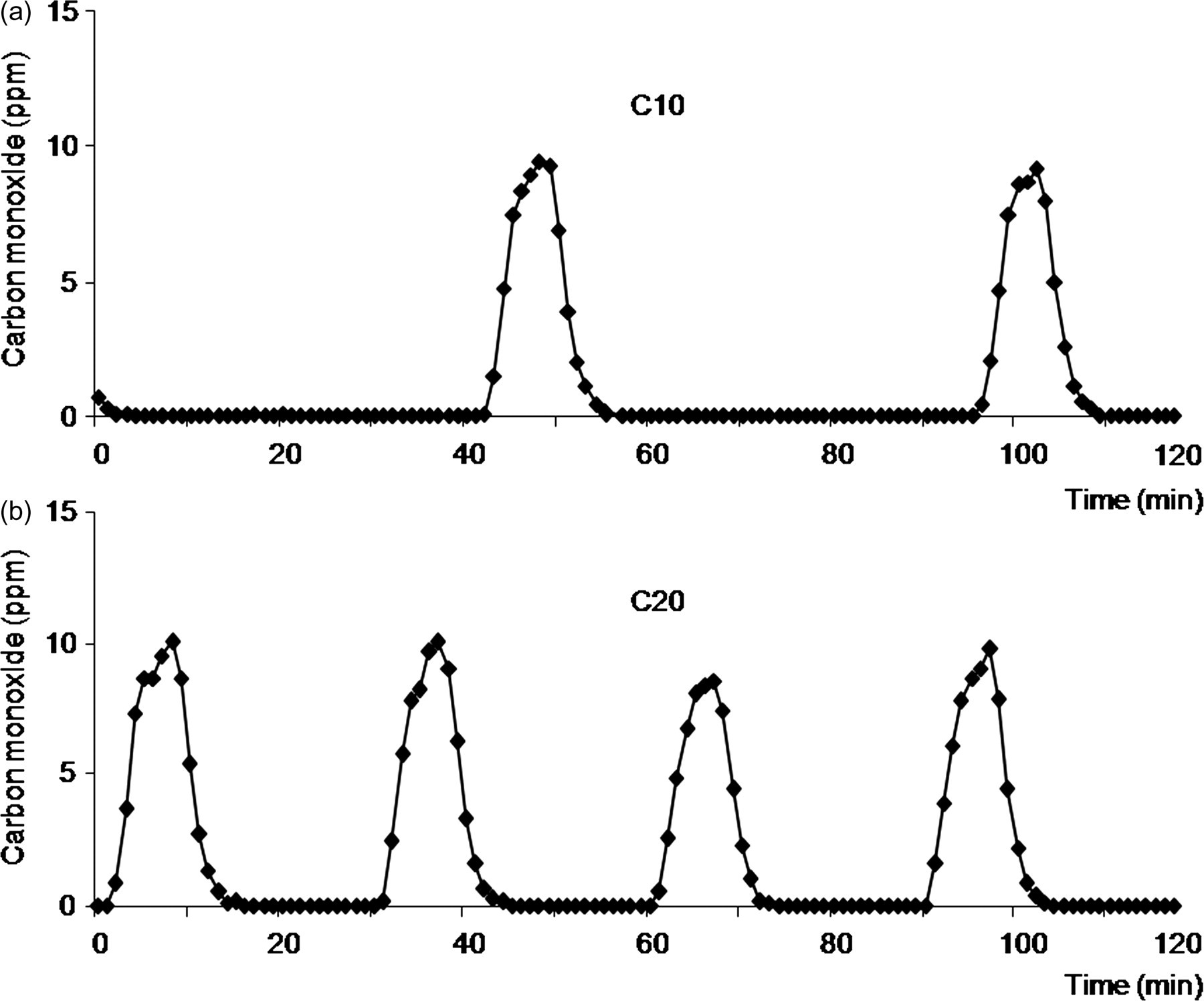
Carbon monoxide concentration in the ambient air during exposure to (a) 10 cigarettes (C10) and (b) 20 cigarettes (C20) daily
Similar variations of particulate matter concentration were measured, with peaks reaching 10–11 mg/m3. As expected, nearly all particulates had a mass median aerodynamic diameter inferior to 2.5 μm (Figure 4).
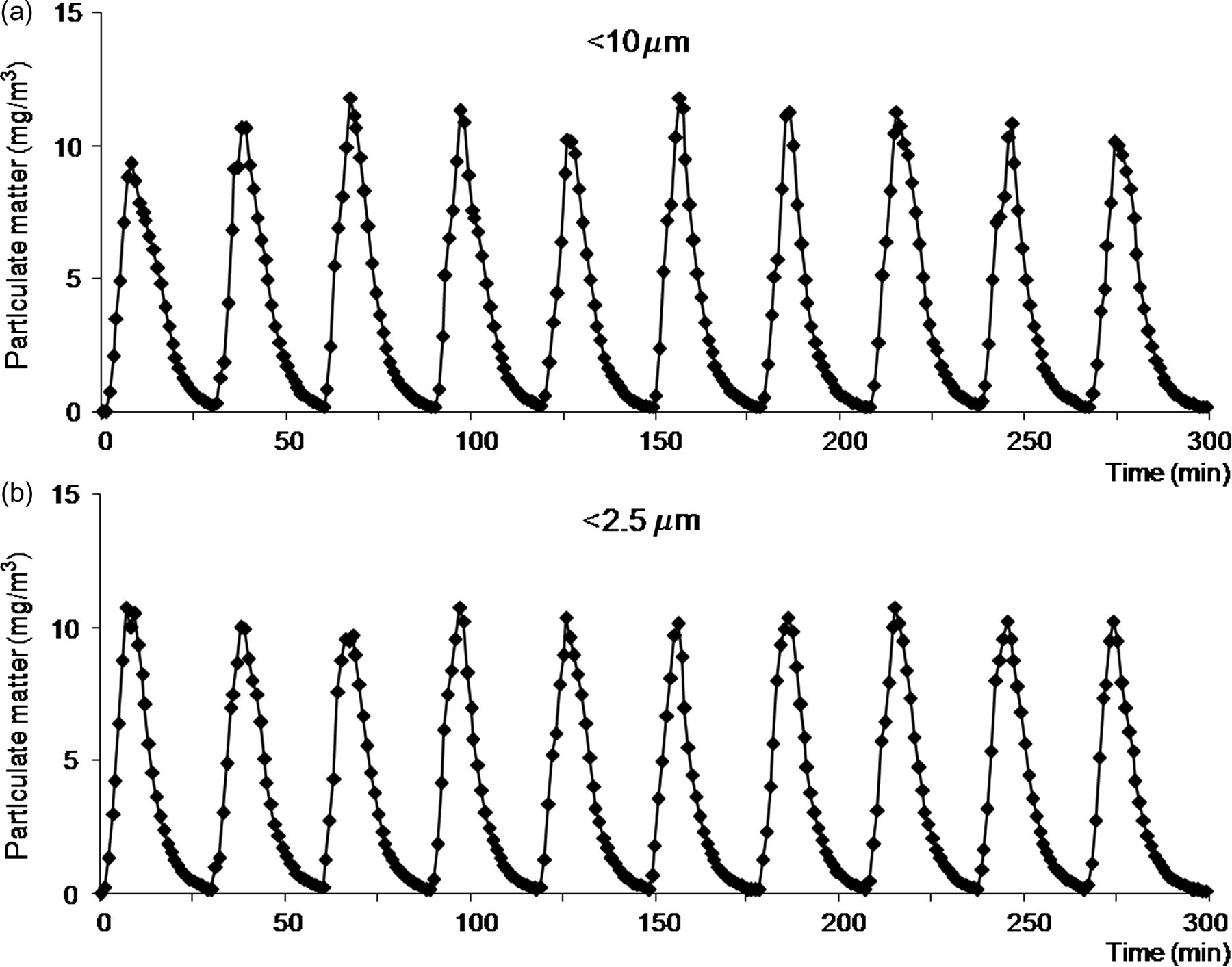
Time variation of particulate matter concentration in the Plexiglas chamber during exposure to 20 cigarettes (C20) daily. (a) Particulates <10 μm; (b) particulates <2.5 μm. Results show intermittent exposure with particulate concentration increasing transiently with each smoked cigarette. As expected, median aerodynamic diameter is almost entirely in the ‘respirable’ (<2.5 μm) particulate range
Histological examination of larynx and epiglottis
No significant epithelial or subepithelial inflammation was observed in the larynx of either C10 (mean score 1.8/15) or C20 (mean score 0.5/15) lambs using the inflammation scoring system of Koufman 16 when compared with control lambs (mean score 0/15).
Discussion
In the present study, we were able to validate a new custom-built ASM primarily designed to be versatile, user-friendly, and which can be used and set to different conditions by non-specialized personnel to study the effects of ETS surrogate in non-restrained, developing lambs. While our preliminary experience with the use of the machine allowed us to readily correct the very few initial problems that arose, such as the noise associated with the running of the machine and the malfunctioning of the cigarette magazine, overall, our ASM has proven to be ideally suited to our needs.
Various smoking machines have been built for the tobacco industry throughout the years, with the primary aim of assessing and modulating the levels of various constituents in cigarette smoke. The first smoking machines were manually operated and able to burn only one cigarette at a time; in addition, only MS could be studied. Subsequent smoking machines (e.g. Filtrona ASM) were automatic, able to burn several cigarettes and a number of parameters could be set. Currently available ASMs for the tobacco industry, such as the Borgwaldt or Cerulean ASM, can burn up to 20 cigarettes with four different smoking regimes (puff duration and volume, time interval between 2 cigarettes) at the same time. The exact concentration of several smoke constituents, including nicotine, carbon monoxide and total/respirable suspended particulate matter, can be automatically analysed in SS and/or MS. While some of those ASMs have been used in animal inhalation studies, they are primarily made for the tobacco industry, to provide precise chemical analysis of MS and/or SS under standardized regimens (FTC/ISO standards), which is mandatory in many countries. 12
Various systems have been used since the 1950s in numerous animal inhalation studies to assess the effect of MS or ETS (see Coggins 19 for a review). Most often, ETS surrogates used in previous studies were diluted and aged SS 17–19 or room-aged 20,21 SS, with no exhaled MS, due to the technical difficulty to produce the latter. However, chemical composition of MS and SS is known to be different, especially due to the lower temperatures, which generate SS, as compared with MS. 11 We have taken a somewhat different approach. In our system, the ETS surrogate, to which each lamb is exposed, is not simply the smoke generated by the ASM (fresh SS and MS). It is rather a mixture of SS, MS and exhaled MS (from the other lamb), which is diluted by the system ventilation and somewhat aged in the exposure chamber. Also, continuous measurement of CO (Figure 3) and particulate matter concentration (Figure 4) shows that exposure level follows important variations with time. We believe that such an ETS surrogate exposure is at least as relevant as continuous exposure to aged and diluted SS alone with a fixed composition for our studies attempting to infer the effects of ETS on infants. Indeed, infants are often nursed in the immediate vicinity of the smoker (in their arms), hence the levels of SS and exhaled MS, to which they are exposed, inevitably vary with time. Finally, it must be recognized that, while no ETS surrogate perfectly reproduces real life ETS, composition of the latter is highly variable with the cigarette brand, the smoker and from one moment to another. 12
The user-friendly interface, which enables the programming of the various parameters to be changed independently from one another, is a unique characteristic of our ASM. In the present validation study, the ASM parameters (time interval between 2 cigarettes, volume of each puff) were set in accordance with the ISO 3308 norms established in 1977, except for the time between two puffs, which has since been shown to be, on average, 30 s instead of 60 s. 22 While our ASM is not currently designed to deliver exact levels of smoke constituents, the latter can be easily modulated by varying the number or pattern cigarettes are burnt, e.g. frequency, duration and/or volume of the puffs (see ISO 3308), and/or by modifying exposure chamber venting. In addition, rather than burning several cigarettes at the same time, the exposure level can be increased by decreasing the time duration between two cigarettes from one hour to one minute. The level of exposure can then be readily assessed by measuring urinary cotinine, whose knowledge again may be more relevant to animal exposure studies than that of constituent levels in smoke. Indeed, intermittent repeated exposure to cigarette smoke constituents, such as gases or suspended particulates, may bear different physiological effects than constant exposure to the same chemicals. An important result in our validation study concerns urinary cotinine/creatinine measurements in C10 and C20 lambs, which are in keeping with findings in infants exposed to ETS. 23–26
Most studies on the effects of cigarette smoke in adult animals have been performed in rodents. 19 The few studies on the effects of cigarette smoke in adult, non-rodent species were performed either acutely in anaesthetized ewes through a tracheal tube, 27 or chronically in tracheotomized sheep 28,29 and dogs, 19,30 in intact dogs using a mask 31,32 or in baboons taught to inhale through the mouth. 33 Studies in large newborn mammals were also initially performed in lambs as a model of bronchitis, using a tracheostomy tube 29 or an ASM custom-made from a Bird ventilator. 29,34 More recently, studies on the cerebral effects of chronic cigarette smoke exposure (up to 13 months) were performed in non-sedated newborn rhesus monkeys. 14,35 In these latter studies, the Teague ASM originally built for rodent or cell exposure 17 was used in association with a 3.5 m 3 exposure chamber similar to the Plexiglas chamber used in the present study. Whole body exposure was preferred in the present study, both for ethical considerations (no contention) and to better mimic real life exposure in infants.
To the best of our knowledge, our ASM is the first specifically designed and validated device for large newborn mammals. An advantage of our equipment, both from a physiological and ethical standpoint, is the possibility of housing two newborn lambs at the same time in the exposure chamber. Moreover, dimensions of our exposure chamber allow the housing of one ewe during gestation. Furthermore, our chamber could readily accommodate various animal species such as piglets, dogs, cats, monkeys or encaged rodents. Versatile programming of the various parameters of our ASM via the user-friendly interface allows for easy adaptation of ETS surrogate to every experimental condition and animal, up to the size of an adult sheep.
Although results from previous studies suggest that some brain effects can be directly ascribed to nicotine exposure alone in the perinatal period, 36 ETS studies clearly remain important. Indeed, while we did not observe upper airway inflammation, which may be a significant risk factor for SIDS or apparent life-threatening events in infants via alteration of upper airway sensitivity, 37 comparing the effects of nicotine alone to the effects of ETS surrogate using our ASM in the same study would allow recognizing the direct effect of nicotine more readily. Of note, the increased activity observed in some lambs during ETS surrogate exposure in the present study is remindful of the behavioural problems reported in children following ETS exposure, 38 such as attention-deficit hyperactivity disorder. 39
The choice to perform our validation exposure using postnatal instead of prenatal (or prenatal added to postnatal) exposure was not solely based on the cost or easiness of caring for lambs, comparatively to a ewe. Previous studies on the effects of cigarette smoke exposure on brain cell damage in monkeys suggest that postnatal exposure has the same consequences as prenatal and prenatal added to postnatal exposure, probably due to adaptive changes in defence mechanisms. 14 Accordingly, part of our forthcoming research programme will focus on postnatal exposure to cigarette smoke.
In conclusion, the ASM designed herein is able to mimic mainstream and sidestream cigarette smoke exposure of variable intensity. Validation of the machine has shown that our initial aim to build a versatile, user-friendly device for use in newborn lambs has been reached. Our newborn ovine models will be used to better ascertain the effect of cigarette smoke exposure on laryngeal chemoreflexes, swallowing–breathing coordination, control of heart rhythm variability, all of which are involved in apparent life-threatening events of infancy and SIDS. In addition, our versatile equipment, which can easily be built by other research teams using the information provided herein, can be readily used in large as well as small animal species to assess the biological effects of cigarette smoke exposure, especially in the perinatal period.
Footnotes
Acknowledgements
The authors gratefully acknowledge the technical assistance of Jean-Philippe Gagné and Nathalie Samson and Dr Alexandre Doueik for histological analyses. The study was supported by the Canadian Institutes of Health Research (Grant MOP 15558) and the Foundation of Stars. J-P Praud is the holder of the Canada Research Chair in Neonatal Respiratory Physiology and a member of the FRSQ-funded Centre de recherche clinique Etienne-Le Bel du Centre Hospitalier Universitaire de Sherbrooke.
