Abstract
Intravenous (IV) administration in mice is predominantly performed via the lateral tail veins. The technique requires adequate training before it can be used safely and routinely. A novel anaesthesia induction chamber has been developed to simplify the treatment and to facilitate IV injection in mice, particularly for untrained personnel. We have assessed the benefits of the chamber in refining IV injection in isoflurane-anaesthetized mice in direct comparison with the common restrainer method on conscious animals. The body weight, nesting behaviour and concentrations of faecal corticosterone metabolites were taken as indicative of distress induced by the various procedures. The results suggest that both methods of tail-vein injection induce similar levels of momentary stress in the animals, revealed by a short-term increase in the levels of stress hormone metabolites in faeces. A temporary reduction of body weight was observed after IV injection under isoflurane anaesthesia but not for conscious mice injected in the common restrainer. We conclude that the severity of tail-vein injection in mice is ‘mild’ for both methods. There was no evidence that refining the procedure by using isoflurane anaesthesia in the induction chamber was associated with any benefit.
Introduction
Lateral tail-vein injection is the preferred method for single or repeated vascular administration of injectable substances in laboratory mice. The successful application of IV injection requires adequate practical skills, only attainable with sufficient practice, and specific equipment for proper immobilization. Currently, tail-vein injection in mice is commonly performed by using a restrainer. Several types of restrainer are on the market and have proven suitable to restrict the movement of the animal during the procedure, while leaving the tail freely accessible for injection. A correctly sized restrainer prevents the animals from turning around but allows normal respiration. Furthermore, rigid restraint devices should enable constant observation of the restrained animal to minimize the risk of injury and unnecessary stress. 1 To achieve dilatation of the vessels, tail-vein injection in mice is frequently carried out in combination with a heat treatment. Vasodilatation facilitates the procedure by rendering the tail vein more visible, which is especially helpful when dark-pigmented mice are to be injected. Local warming of the tail by infrared exposure or by immersing the tail in warm water, rather than heating the entire body, are the preferred techniques to minimize stress due to (over-) heating of the animal. 2
The procedure of tail-vein injection with a standard restrainer is routinely performed on conscious mice: no sedation or anaesthesia is given. Appropriate restraint of the animal is important to avoid injury to the animal or the handler. A sudden jerking motion by the animal is frequently observed and is a response to the acute pain caused by the vein puncture. It may complicate the procedure as abrupt movement of the animal often dislodges the inserted needle. Unsuccessful attempts are not unusual for an untrained person and might result in the repetition and prolongation of the procedure.
The severity of the intravenous administration of substances via superficial blood vessels is classified as ‘mild’, provided that the injected substance has no or only mild impact on the animal and the injection is performed in accordance with good veterinary practice (Directive 2010/63/EU, Annex VIII, Section III, 1.f). However, routine procedures such as restraint and injection are known to cause an acute stress response in laboratory mice.3,4 It is unknown whether restraining and IV injection together have a cumulative impact in terms of increased severity. The use of anaesthesia might have the advantage of avoiding discomfort for tightly restrained animals while simultaneously preventing the defence reaction of the mouse, which has no sensation of pain from the introduction of the needle. A novel anaesthesia induction chamber for tail-vein injection of mice (Rothacher Medical GmbH; Heitenried, Switzerland) has recently been recommended which claims to refine IV injections in anaesthetized mice. The manufacturer promises a simplified procedure without the need for pre-heating treatment to induce vasodilatation and to reduce pain for the animals.
For short-term procedures, inhalation anaesthesia represents the method of choice and provides a rapid onset of induction and recovery. Isoflurane is the most frequently used inhalation anaesthetic in mice because of its simple use and rapid induction of anaesthesia and of its comparatively slight cardiodepressive and hemodynamic effects, ensuring a low risk of complications.5,6 Mice generally recover immediately from inhalation anaesthesia, making additional post care procedures dispensable after minor interventions. However, anaesthesia has to be medically indicated due to the adverse physiological effects of anaesthetics, which can impair the animal’s well-being or alter experimental data.7–9
We have investigated the severity and duration of possible pain and distress after tail-vein injection carried out by the standard restrainer method on conscious mice, comparing the results with the pain and distress associated with the novel induction chamber in combination with isoflurane anaesthesia. Changes in body weight, concentrations of faecal corticosterone metabolites and nest-building behaviour were selected as reliable indicators to evaluate potential impact of the treatments.10–12 The study was conducted separately with experienced and inexperienced personnel. We expected that use of the novel induction chamber would reduce at least some of the adverse effects of tail-vein injection on the animals’ well-being, as well as it would simplify the injection procedure.
Animals, materials and methods
Animals
The study was approved by the institutional ethics committee of the University of Veterinary Medicine Vienna and an experimental license was granted under BMWFW-68.205/0042-WF/II/3b/2014 (Austrian Federal Ministry of Science, Research and Economy). The burden induced by the procedures was prospectively categorized as ‘mild’.
All mice were purchased from Janvier Labs (France) and the perpetuation of their specific pathogen free (SPF) quality according to FELASA recommendations was confirmed by a sentinel programme. 13 Animals were housed in a rodent facility (photoperiod 12L/12D, light on at 08:00 am, temperature: 21.3° ± 1.9℃, relative humidity: 60.3 ± 9.8%). Commercial mouse diet (V1126, Ssniff, Germany) and bottled tap water were available ad libitum. Cages were lined with bedding material (Lignocel®, heat treated, Rettenmaier KG, Austria) and enriched with nesting material (Pur-Zellin 4 × 5 cm; Paul Hartmann GmbH, Austria).
During the adaptation period of 2 wk the mice were housed in groups of five in transparent polycarbonate cages (Makrolon® Type III, Tecniplast, Italy).
In Part 1 of the study, we evaluated the discomfort induced by tail-vein injection in 10 females and 10 males of each of the two strains C57BL/6NRj and RjORL:SWISS (aged 8–10 wk). C57BL/6N was chosen because this inbred strain is frequently used as a genetic background of mutant mouse models. It is presumed that IV injection in dark pigmented mice is more difficult. In contrast, SWISS mice are larger albinotic outbred mice and their tail veins are more prominent. The results of both strains were pooled. For the investigation period of five days the animals were individually housed in Makrolon® type II cages. Because separation of mice will induce a transient increase of the faecal corticosterone metabolites (fCM) concentration, a five-day period for adaptation to the individual housing condition was included before starting the experiments. 14
The induction chamber is reported to facilitate and simplify IV injection. This aspect was separately tested in Part 2 of the study with 16 female C57BL/6NRj mice (aged 8–10 wk), which were housed in groups of eight in Makrolon® type III cages and only temporary separated for the collection of faeces. We choose C57BL/6 for this practicability test because this strain is predominantly used by researchers in our facility as genetic background for genetically engineered mutations.
Restrainer and induction chamber
A stainless steel restrainer (homemade, diameter 3 cm, length 12 cm) with a flexible stopper to secure the mouse was used to fix conscious mice (Figure 1). Before injection the tail was immersed in a 39–40℃ water bath for about a minute to cause sufficient dilatation of the lateral tail veins.
A conscious mouse is prepared for IV injection in the stainless steel restrainer.
The novel induction chamber for tail-vein injection of mice is made of a special polymethyl methacrylate (1 cm; 60 × 120 × 50 mm) with inlet and outlet adapters for fresh and waste anaesthetic gas. The cap has a small semilunar hole for fixation of the tail. The chamber was connected with the XGI-8 gas anaesthesia system (Xenogen, California, USA) and the Fluovac System (Harvard Apparatus, UK) to absorb anaesthetic vapours from the work area. To induce anaesthesia, the mouse was placed in the chamber and the isoflurane vaporizer was initially set at 3%. When the mouse was anaesthetized, which was first indicated by the cessation of body movement for a few seconds, the chamber cap was briefly opened to test the surgical tolerance of the animal by the hind limb withdrawal reflex and to place the tail in the correct position through the semilunar hole, a special feature of the novel device (Figure 2(a)). This hole in the chamber cap is padded with foamed plastic and after closing the chamber it will compress the tail to induce stasis of the tail veins. In comparison to the common restrainer, dilatation of the lateral tail veins should be induced by a contra pressure to the foamed plastic, making it unnecessary to warm the tail before injection (Figure 2(b)). After closing the chamber, the setting of the vaporizer was reduced to 2% until the end of the procedure. Oxygen (FiO2 = 1.0) was used as carrier gas at 0.5 l min−1. The chamber was placed in a filtration fume hood using activated carbon (A18B/AS Captair Flex M391 2C, Fisher Scientific, Austria) to avoid exposing personnel to anaesthetic vapours that leak from the chamber during opening.
(a) Induction chamber for anaesthesia. The tail of the anaesthetized mouse is placed in the correct position in the semilunar hole to induce stasis of the tail veins. (b) The anaesthetized mouse is prepared for IV injection.
Experimental design
Part 1. To compare the degree of discomfort induced by tail-vein injection with the novel induction chamber against that caused when the standard restraining method is used, animals were randomly assigned to four treatment groups and a control group (eight animals per group, two females and two males per mouse strain):
(0) Control group, without manipulation: mice were only handled for weighing and faeces collection. (1A) Restrainer group, without IV injection: mice were fixed in the restrainer and the tail was immersed in a 39–40℃ water bath for 1 min. (1B) Restrainer group, with IV injection: mice were fixed in the restrainer, the tail was immersed in a 39–40℃ water bath for 1 min and saline solution was administered. (2A) Chamber group with anaesthesia, without IV injection: mice were placed in the chamber and anaesthetized by isoflurane until surgical tolerance. (2B) Chamber group with anaesthesia, with IV injection: mice were placed in the chamber, anaesthetized by isoflurane until surgical tolerance and saline solution was administered.
Similarly, to the treatment groups, mice of the control group were observed for nest-building activity, weighed and transferred to a new cage for collection of faeces, but were not placed in any device. Assessment of nest building, measurement of body weight and faeces collection was performed for all mice in the animal room. The IV injections were performed in an adjacent procedure room. All mouse handling and IV injections were carried out by the same skilled (female) experimenter. Treatment of the experimental groups and the control animals was evenly distributed over five consecutive weeks.
Time schedule for experimental procedures before and after IV injection.
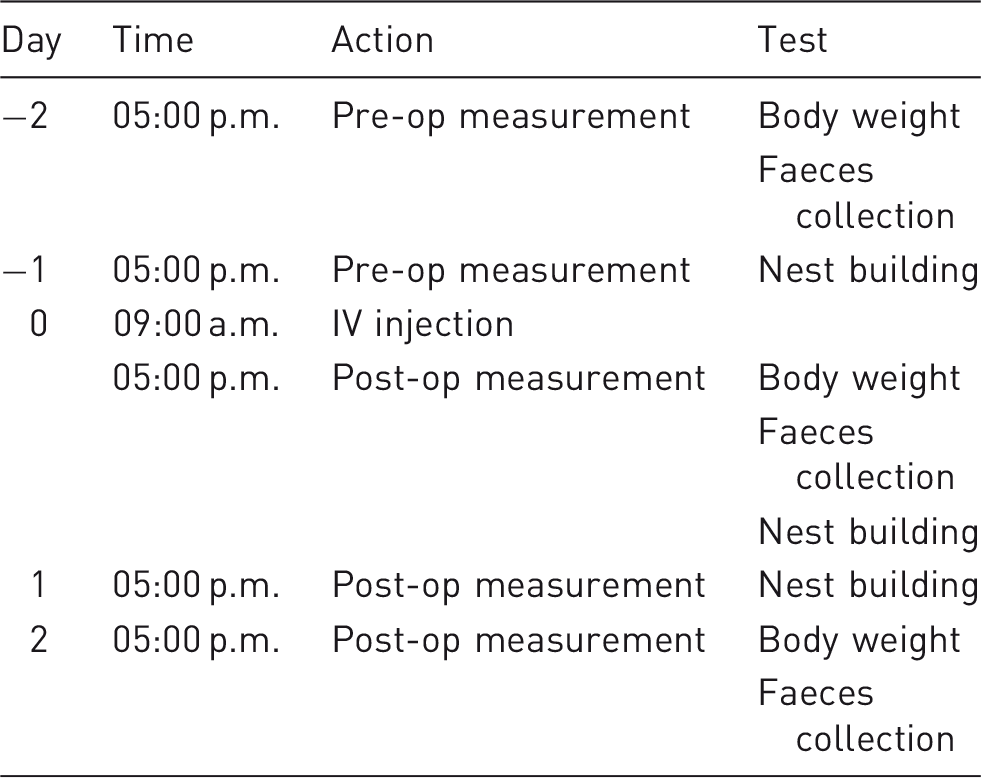
Part 2. As the novel chamber is reported to simplify and facilitate IV injection in mice, its applicability to the procedure was additionally tested by a group of eight untrained persons (students of veterinary medicine, tenth semester). The (female) students were given a short introduction in mouse handling but had no previous experience and had not previously performed IV injection in mice. Each participant injected a conscious mouse in the standard restrainer and an anaesthetized mouse in the novel chamber. The order of injections was randomly assigned either to start with the restrainer or with the chamber method. In the event of an unsuccessful injection, a second and a (maximum) third attempt per mouse was allowed. The study was conducted for all 16 mice in parallel on the same day according to the time schedule described in Table 1 for the restrainer group (1B) and chamber group (2B). To assess the effects on the animals, body weight and fCM were measured. Additionally, we counted the number of attempts per mouse and student until successful injection was performed and the number of terminations after the third unsuccessful attempt.
The IV injection was conducted in both parts of the study with 0.9% saline solution (Fresenius Kabi Austria GmbH) at a dosage of 5 ml kg−1 body weight with 30G x 1/2 in injection needles (Sterican®, B. Braun, Melsungen, Germany).
Body weight was measured in grams using an electronic balance (#440-47N, KERN & SOHN GmbH, Germany). To assess nest building we removed all old nesting material from the previous night in the morning (08:00–09:00 a.m.) and offered three fresh pads (Pur-Zellin®). Nest-building was evaluated at 05:00 p.m. for each single-housed mouse. Nest-building activity starts soon after nesting material is offered, which corresponds to the high values of nest scores normally observed at the end of the photoperiod. 11 A nest complexity scoring according to Jirkof et al. was used with scores from 0 to 5 for the assessment of such measures as the manipulation of nesting material and nest-building performance. 11 Briefly, 0 = pads not manipulated, 1 = most pads slightly manipulated, 2 = pads noticeably manipulated, shreds spread at a defined place of the cage, 3 = noticeable arrangement of manipulated nesting material, 4 = flat nest with incomplete encasing walls, 5 = complex nest with heightened walls.
Secretion of stress hormones was measured via excreted corticosterone metabolites in faeces. The level of circulating corticosterone and of excreted faecal metabolites follows a robust nycthemeral cycle.15,16 To control for the confounding effects of the 24-hour oscillations of stress hormones, faeces samples were collected at a fixed time of day. In the event of a stressful incident, the corresponding peak of fCM excretion in mice occurs 8–10 h after the initial impulse. 12 Therefore, 8 h after IV injection each mouse was placed for 2 h in a separate cage (lined with paper sheets instead of wood bedding), from which fresh faeces were collected and frozen in a tube at −20℃. After collection of faeces, the mice were returned to their home cages. For fCM analysis, clean faecal samples from each mouse were dried and homogenized and 0.05 g of the dry mass was mixed with 1 ml of 80% methanol. The concentrations of corticosterone metabolites were measured by an in-house 5α-pregnane-3β,11β,21-triol-20-one enzyme immunoassay, which has been validated for mice. For further details of the assay, see two studies by Touma et al. in 2003 and 2004.12,16
Statistical analysis
All statistical analysis and chart preparation was performed using IBM SPSS v24 (SPSS Inc. Chicago, IL, USA). Animals in both parts of the study were randomly allocated to the treatment groups. We analysed the effects of tail-vein injection on body weight, fCM and nest-building activity when the mice were immobilized by different procedures.
Body weight was analysed as a percentage of change, each relating to the measured individual value 48 h earlier (= 100%). Differences within each group were tested with a paired sample t-test, while between-group differences were tested with a t-test for unpaired samples. Concentrations of fCM were analysed separately for females and males by a General Linear Model (GLM) for repeated measurements, followed by post hoc tests using Bonferroni’s alpha-correction procedure. The comparison of fCM concentrations in the control group and the treatment groups was performed using simple a priori contrasts (least significant difference). Nest-building activity was assessed by scores from 0 to 5 and differences between repeated measurements within groups were tested by the Wilcoxon signed-rank test. All data are graphically depicted as box-and-whisker plots with medians (lines in boxes), IQR (25–75% interquartile range, boxes) and maximum range (whiskers), excluding extreme values. Two-tailed p values of p ≤ 0.05 were considered to be statistically significant.
Results
Part 1
Change in body weight
Because of sex- and strain-specific differences, changes in body weight are presented as percentage of the weight on day 0 relative to day −2 and day +2 relative to day 0. A significant change (increase) of body weight was measured for the control group (0) and the restrainer group without injection (1A) (group 0 p = 0.003; group 1A p = 0.005; Figure 3). Differences between treatment groups were not significant. It is worth noting that a (non-significant) decrease in body weight was observed for the chamber group with injection (2B) on day 0 relative to day −2, whereas all other groups showed an increase of body weight during this period.
Boxplot for the percentage of change in body weight for female and male C57BL/6 and SWISS mice, which have received an IV injection from a skilled person in the restrainer, or under anaesthesia in the chamber. Body weight was measured 2 days before treatment, at the day of injection and 2 days post manipulation; changes were calculated for the body weight on day 0 relative to day −2 and on day +2 relative to day 0 (Part 1). Paired t-Test **p ≤ 0.01.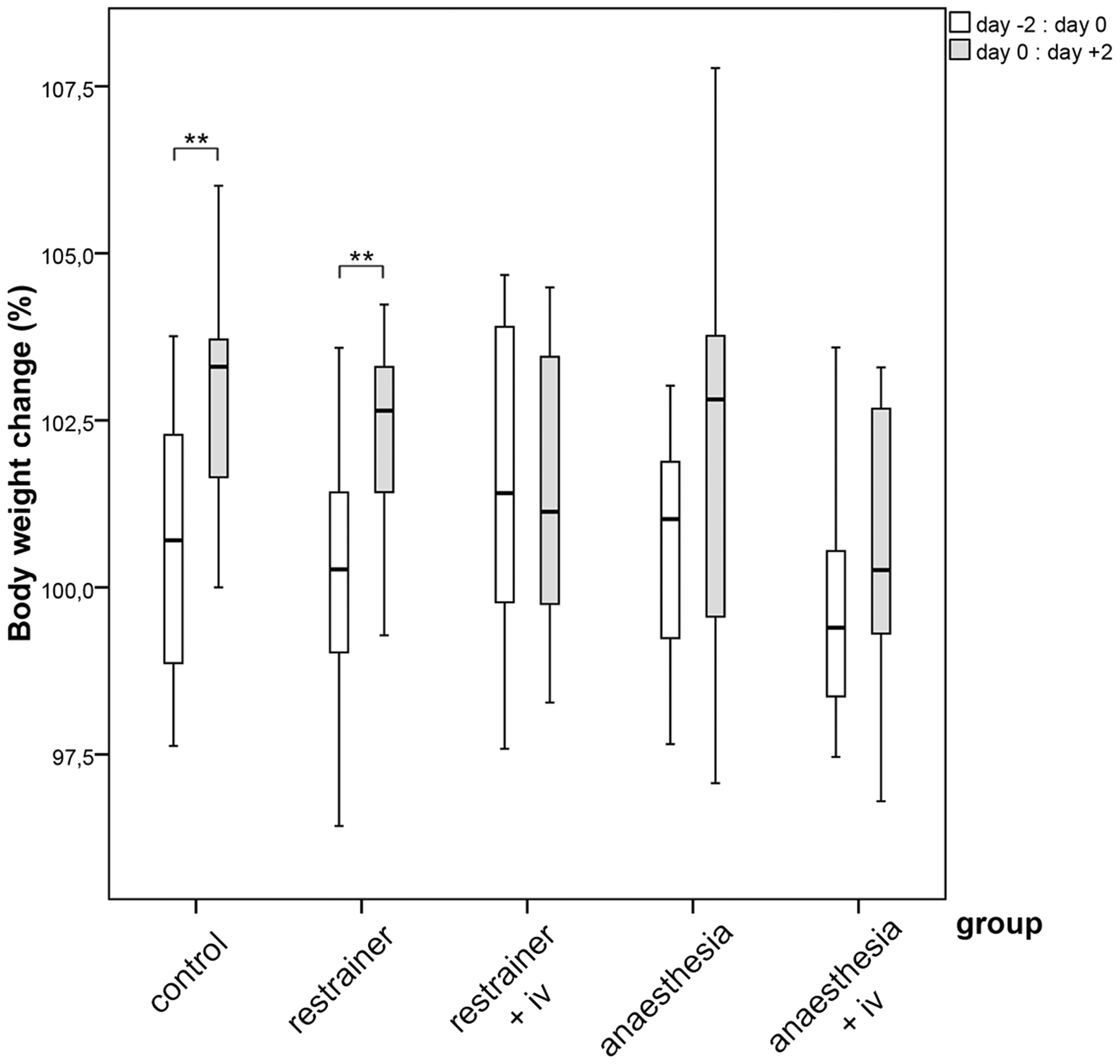
Stress hormone levels
Because there are sex-based differences in fCM concentrations in mice, females and males were evaluated separately (Figure 4(a) and (b)).
16
Our results confirmed the previously published data that females generally show about twice as high fCM values than males.
12
Differences in fCM levels were observed between females of both strains. However, the higher levels of SWISS females differed not significantly from those of C57BL/6 females (data not shown). Irrespective of the method applied, the impact of treatment was generally more distinct and consistent for males, reflecting a short but stressful event. A significant increase in fCM level was detected for males within the chamber group 2B with injection (day −2:day 0, p = 0.04). The fCM concentration immediately post injection (day 0) of males of the group 2B was also significantly higher in comparison to the control group at the same time (p = 0.014; Figure 4(b)). Interestingly, the increase of fCM values on day 0 (8–10 h post injection) was completely counterbalanced on day 2 (significant in group 2B, day 0:day 2, p = 0.01), suggesting that both immobilization by anaesthesia and IV injection are only stressful in the short term.
Boxplots for faecal corticosterone metabolite concentrations (fCM) in (a) females and (b) males in the different treatments. Faeces were collected 2 days before the experimental manipulation (day −2), at the day of manipulation (day 0) and 2 days post manipulation (day +2) (Part 1). Comparison using Bonferroni’s alpha correction *p ≤ 0.05, **p ≤ 0.01.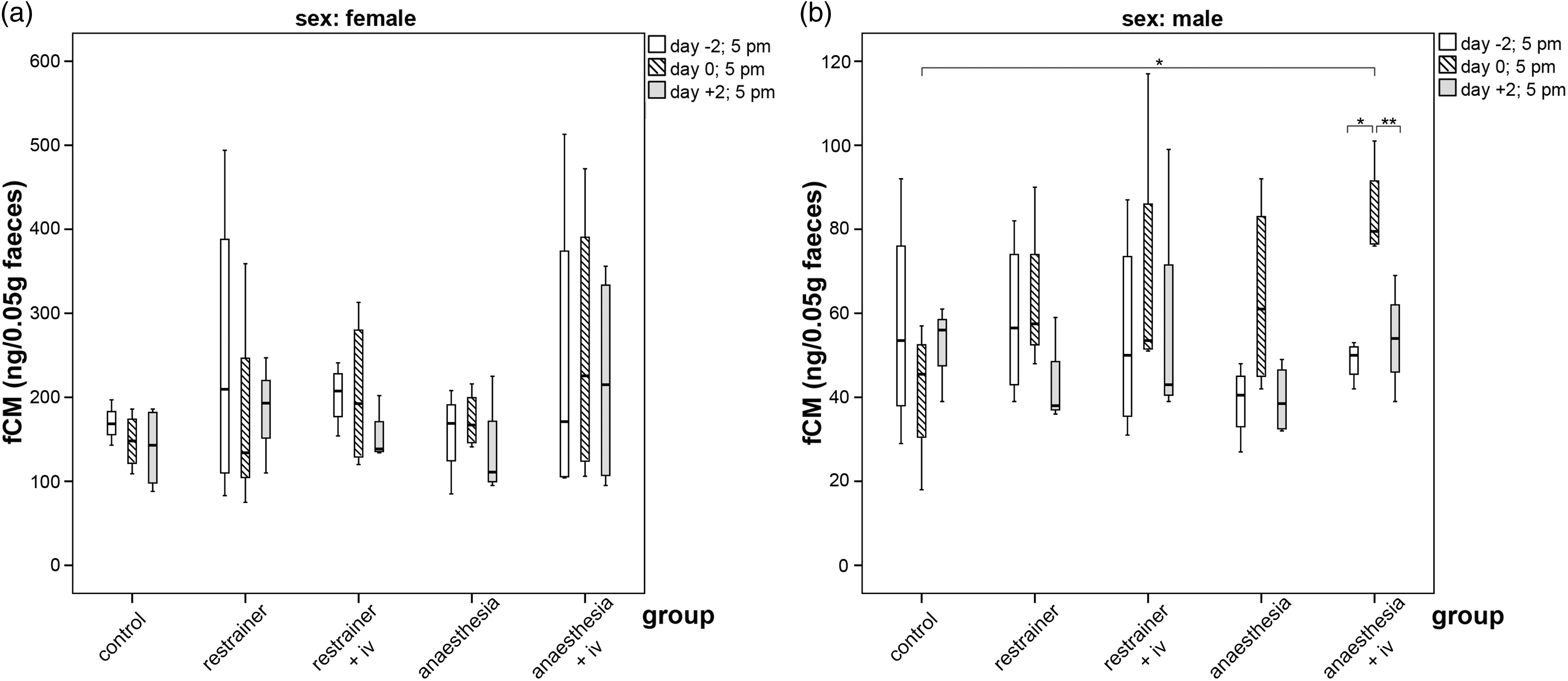
Nest building
Nesting behaviour was scored most frequently and alike (no significant differences detected) in both strains at level ‘3’ and ‘4’ prior to IV injection (day −1). Nest-building activity was slightly reduced in all groups, including the controls, on day 0 (after treatment). This general reduction might be the result of interference from the activities during the morning of day 0 in the animal room (transport of cages between animal room and procedure room for IV injection). An increase in nest-building activity was detected on day 1, which was significant in the chamber group 2A without injection (p = 0.04; Figure 5).
Boxplots for nest-building scores (0–5) in the different treatments. Nest building was recorded 1 day before the experimental manipulation (day −1), at the day of manipulation (day 0) and 1 day post manipulation (day +1) (Part 1). Extreme values more than ×1.5 IQR above Q3 or below Q1 (°). Wilcoxon Signed Ranks Test *p ≤ 0.05.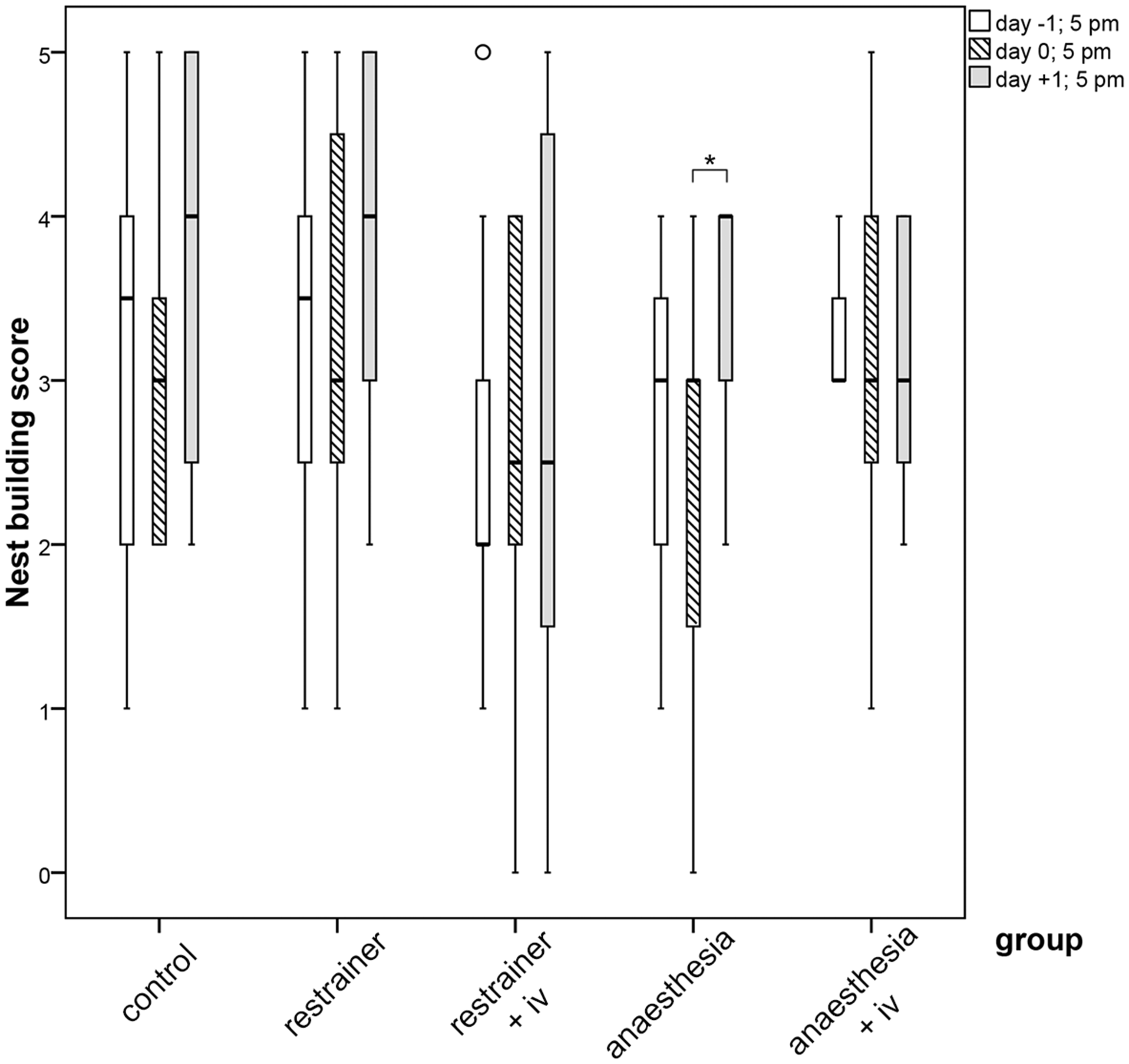
Part 2
Body weight
A significant decrease in the body weight was observed on day 0 relative to day −2 for animals of the chamber group in comparison to the restrainer group (p = 0.049; Figure 6). This weight loss for anaesthetized animals of the chamber group was completely compensated after the treatment, which was shown by the significant increase of body weight in the 48 h period after IV injection (p = 0.024).
Boxplots for the percentage of change in body weight in female C57BL/6 mice, which have received an IV injection from untrained persons in the restrainer, or under anaesthesia in the chamber. Body weight was measured 2 days before treatment, at the day of injection and 2 days post manipulation; changes were calculated for the body weight on day 0 relative to day −2 and on day +2 relative to day 0 (Part 2). Paired t-Test **p ≤ 0.05.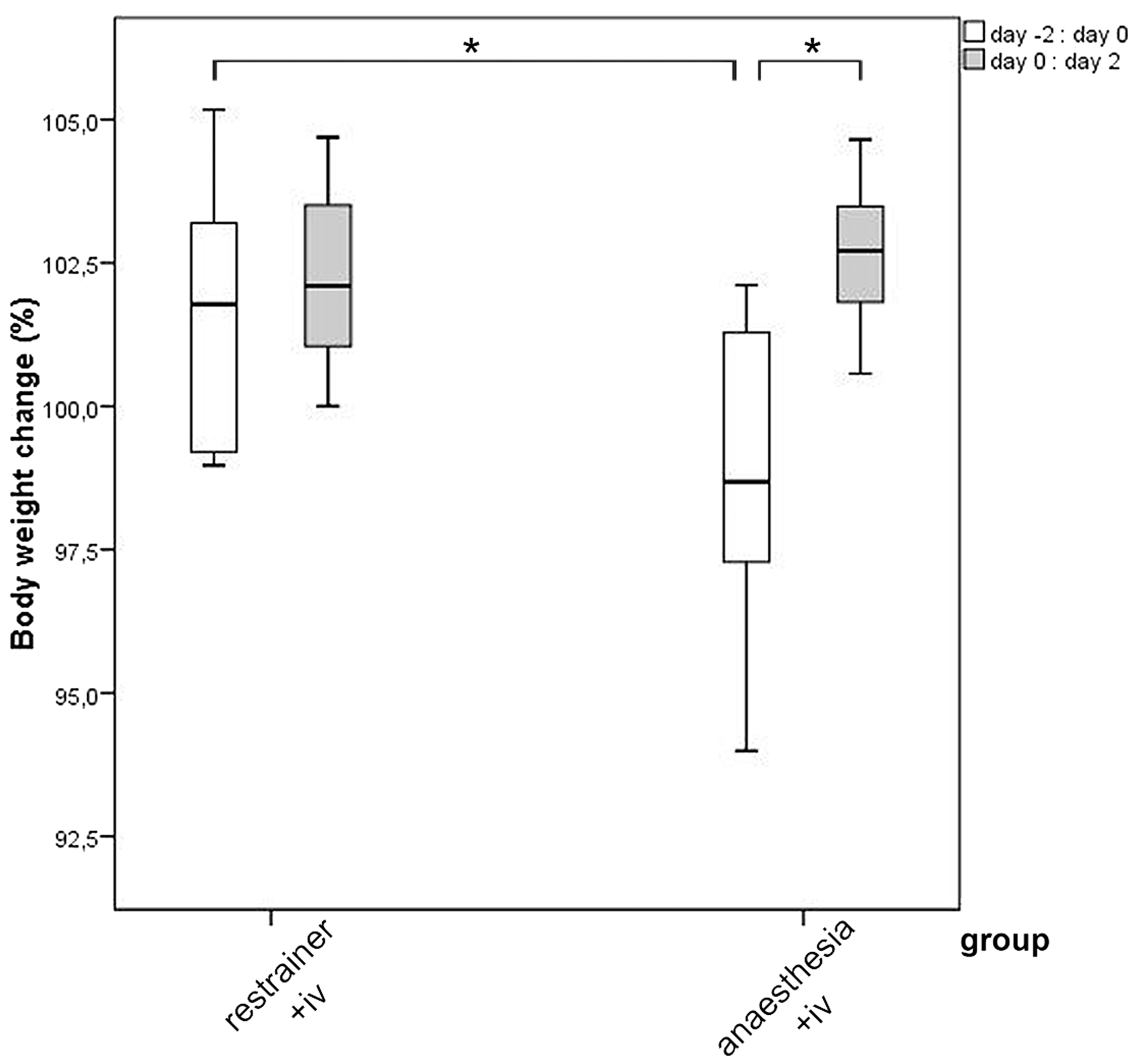
Stress hormone levels
Similarly, to Part 1, the analysis of fCM revealed an increase of concentrations on day 0 in both treatment groups. In the chamber group, the measured fCM concentration was significantly higher on day 0 than on day −2 and day 2 (p = 0.0004 and p = 0.011). An increased fCM level was also found for animals of the restrainer group on the day of treatment. The level on day 0 was significantly higher in comparison to the level measured on day 2 post injection (day 0:day 2, p = 0.047, Figure 7).
Boxplots for faecal corticosterone metabolite concentrations (fCM) in females, which have received an IV injection from untrained persons in the restrainer, or under anaesthesia in the chamber. Faeces were collected 2 days before the experimental manipulation (day −2), at the day of manipulation (day 0) and 2 days post manipulation (day+2) (Part 2). Comparison using Bonferroni’s alpha correction *p ≤ 0.05, ***p ≤ 0.001.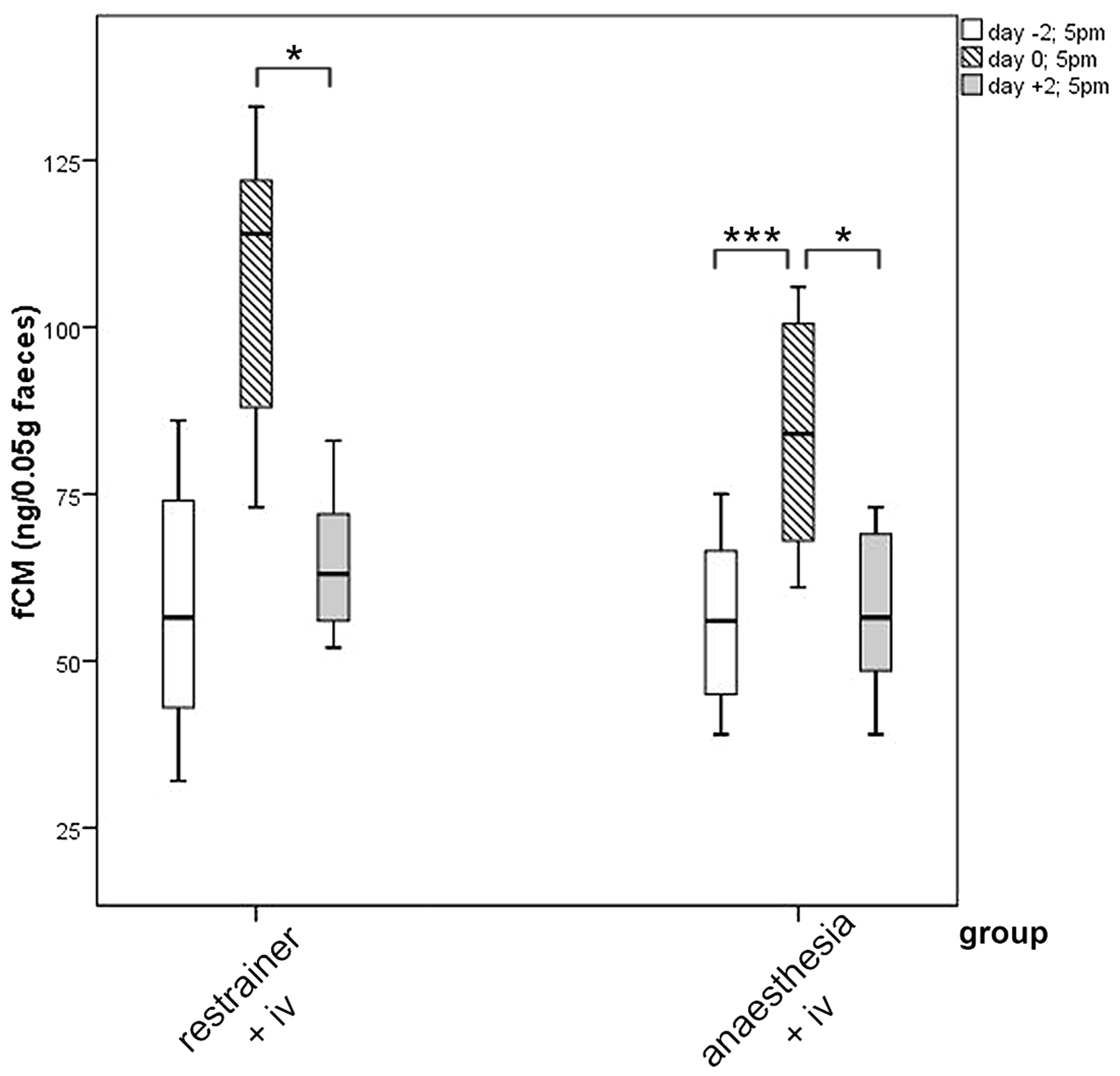
Practicability
Five of the eight students performed a successful tail-vein injection with conscious mice in the restrainer. On average 2.2 attempts were necessary to succeed. In contrast, only three of the eight students performed a successful tail-vein injection with anaesthetized mice in the induction chamber and all three needed a third (last possibility according to our protocol) attempt to succeed.
Discussion
The procedure for lateral tail-vein injection consists of different steps depending on the method used. The type of restraint and the techniques to immobilize an animal significantly influence the well-being of the animal, both during as well as following treatment. We have compared the widely-used restrainer method for IV injection in conscious mice with a novel induction chamber for tail-vein injection of mice using isoflurane anaesthesia. The outcome of this study generally supports the classification of lateral tail-vein injection in mice as severity degree ‘mild’ for both methods. All tested characteristics revealed either no significant impairment of or only a short-term mild impact on the well-being of the animals. In direct comparison to the common restrainer method we found no benefit in using the novel induction chamber, neither as refinement to improve animal welfare nor as a simplification of the procedure.
For direct comparison of both devices we choose C57BL/6 and SWISS mice. C57BL/6 is the most often used genetic background for genetically engineered mice. However, mice of this inbred strain are small, dark pigmented and their tail veins are less distinctly visible. In order to test both methods with a second strain, we selected SWISS albinotic mice. These outbred mice are bigger than C57BL/6 and the larger diameter of their tails could additionally improve the stasis of the tail veins after compression in the cap hole of the closed chamber.
To assess the degree of severity of tail-vein injection we measured three key characteristics: changes in body weight, the level of faecal metabolites of corticosterone, the major glucocorticoid in mice, 17 and nest-building behaviour.
The rate of change in body weight is a recommended indicator for previous and prolonged pain or distress, which is usually manifested in weight loss.10,18 It is frequently used in scoring systems for the assessment of animal welfare. 19 Mice have a very high motivation for nest building and deficits in this activity are often associated with a reduction in well-being. 20 The analysis of corticosterone metabolites in faecal samples is a non-invasive method to monitor adrenocortical activity in mice and is a well-established technique to evaluate pain-induced distress. 16 In contrast to a stressful event, simple handling of mice will not have an impact on the changing levels of fCM. 21
According to the measured fCM levels in males of Part 1 and females of Part 2 of the study, IV injection in the chamber with isoflurane anaesthesia appears to be more stressful than it is for conscious mice in a common restrainer. This finding is in accordance with a previous study that showed that isoflurane anaesthesia alone induces a short rise in serum corticosterone.22,23 Although the elevated fCM level on day 0 in Part 1 of males in the chamber group without injection (group 2A) indicate a previously stressful event, fCM increase reached significance only for anesthetized mice with IV injection (group 2B). This result suggests that anaesthetizing and IV injection together could have a cumulative impact in terms of increased severity. Interestingly, the significantly increased fCM levels for the chamber group in Part 2 are paralleled by a reduction in body weight of these animals which was not observed in the restrainer group. This is important because an effect on body weight suggests a stronger or more prolonged impact of anaesthesia on an animal’s well-being, possibly as a consequence of a timely restricted reduction of food intake.24,25
On average we measured lower and less variable fCM concentrations in Part 2 of the study, in which exclusively C57BL/6 females were used. The difference can be mainly explained by higher but not significantly different fCM levels for female SWISS outbred mice in the merged groups of Part 1. However, an additional impact of the housing system (single housing in Part 1 and group housing in Part 2) cannot be excluded. Nevertheless, the average level of measured fCM values in Part 2 is similar to previously reported data of C57BL/6 females.12,14
It has been shown that spontaneous nest-building behaviour of mice is affected by painful treatments and anaesthesia. 11 In this study nest-building behaviour was not evidently affected by the treatments, suggesting only a mild impact, even if short-term anaesthesia was applied. One exception was seen in mice of the chamber group with anaesthesia (group 2A), in which a significantly lowered nest-building activity was observed on day 0 (in the afternoon of the treatment day) in comparison to day 1 post treatment.
The restrainer was used as it is frequently applied in our routines and the novel induction chamber strictly according to the product description of the manufacturer. In Part 1 of the study, mice of both strains were successfully injected independent of the device used by the skilled experimenter. In Part 2 we were predominantly interested in the practicability of both methods when applied by inexperienced personnel because the novel device is explicitly advertised for a simplified injection procedure and a considerably lower number of unsuccessful attempts. Although the rate of successful injection attempts of untrained persons did not significantly differ between the two methods, the results tend to be more favourable for injecting conscious mice in the common restrainer than when anaesthetized mice were injected in the induction chamber. The reason may be the improved vasodilatation of lateral tail veins after warming of the tail in the restrainer method. Isoflurane anaesthesia is known to cause low blood pressure, 6 which may reduce the effect of stasis in the tail when pinched in the hole of the chamber cap, reducing the visibility and accessibility of the vein in anaesthetized mice. Whether additional tail warming would have improved the success rate of injections in anaesthetized mice or not, we did not test, as this would have necessitated two additional experimental groups: (a) tail warming in the chamber groups and (b) anaesthesia in the restrainer groups. However, from a practical point of view, tail warming of anaesthetized mice in the chamber would have abolished the accentuated and beneficial simplification of the procedure with the novel device. Inhalation anaesthesia for mice in a standard restrainer is not applicable. In order to isolate the effect of anaesthesia on its own in the chamber group, IV injection on conscious mice would have been necessary. However, this would have harboured a high risk of injury to the not adequately immobilized mouse and therefore, in our opinion, it was not a practically feasible option.
Anaesthesia with inhalants requires appropriate equipment (which may turn out to be costly), including an effective scavenging system to protect personnel, as well needing specific clinical experience. At worst, prolonged exposure to significantly higher doses of anaesthetic may result in mortality of the animals via respiratory and cardiovascular depression. Even in professional clinical practice, experimental results can be significantly influenced by side-effects of anaesthesia. We found no refinement of the injection procedure with the induction chamber and isoflurane anaesthesia. Although both methods are categorized at the lowest degree of severity, we assess IV injection with the common restrainer as the more suitable method, although the novel chamber might have advantages for the induction of anaesthesia and for short-time transport of anaesthetized mice for use in other experimental procedures.
Footnotes
Acknowledgements
We acknowledge Sebastian Gehrig for providing the induction chamber, Edith Klobetz-Rassam for fCM analysis and Nina Hornacek, Dalma Batkay and Graham Tebb for editing the figures and text of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
