Abstract
Hearing and balance receptors in the inner ear are highly susceptible to damage caused by a wide variety of toxic substances, including aminoglycosides. This class of antibiotics is commonly used in medicine, even though they may produce irreversible bilateral neurosensorial deafness. To identify potential ototoxic agents and novel therapeutic targets, it is necessary to generate standardized animal models of aminoglycoside ototoxicity, which will also serve to explore otic cell repair and regeneration. Although the mouse is the species most often used in biomedical research, due to the genetic information and genetically-modified strains available, there are few standard models of aminoglycoside ototoxicity in adult mice. Most protocols to produce ototoxicity in adult mice employ high doses of aminoglycosides for long periods of time, which causes systemic toxicity, side-effects and high mortality rates. Here, we compare the effects of systemic treatment with four different, yet common, aminoglycoside antibiotics in two mouse strains, evaluating their effects on mortality, cochlear morphology and auditory brainstem responses. Our data indicate that gentamicin and neomycin caused high mortality in the adult mouse without significantly changing the auditory threshold. Amikacin produced a tolerable rate of mortality but at doses that did not exhibit ototoxicity. Finally, intramuscular injection of kanamycin in C57BL/6JOlaHsd mice induced significant dose-dependent bilateral hearing loss with a moderate rate of mortality and less discomfort than following subcutaneous administration.
Otototoxicity refers to the damage generated by a chemical substance in the organ responsible for hearing and balance. 1 Although aminoglycoside antibiotics are commonly used to treat Gram-negative infections due to their effectiveness and low cost, their toxic effects on the inner ear are a major concern for the medical community. Certain aminoglycosides are primarily vestibulotoxic, whereas others are mainly cochleotoxic; however, the basis of this differential susceptibility has not been associated with any structural characteristic or tissue pharmacokinetics, and presents species variability. In humans, hearing loss due to aminoglycoside ototoxicity is typically neurosensorial, non-syndromic, bilateral and progressive, starting with loss at high frequencies. Vestibular toxicity usually develops in parallel and results in dizziness, ataxia and/or nystagmus. The severity of the symptoms depends on the drug, dose, route of administration and duration of the treatment, as well as the individual's susceptibility. 2
The combination of in vitro hair cell explant cultures and in vivo studies has contributed to our understanding of the basis of aminoglycoside ototoxicity. These compounds induce an increase in the formation of reactive oxygen species, nitric oxide levels and peroxynitrite radicals, which finally induce sensory cell death by apoptosis. 3
Aminoglycosides primarily damage the outer hair cells (OHC) of the organ of Corti, beginning at the basal region of the cochlea and then spreading towards the middle and apical zones. Inner hair cells (IHC) and support cells are usually less affected. Differential cell expression of nuclear factor kβ (NFkβ) and the levels of the antioxidant glutathione are among the factors thought to explain these differences. 4
Animal models have been widely used to study aminoglycoside ototoxicity and in drug screening for otoprotective therapies. The zebrafish lateral-line neuromasts display structural and functional similarities to cochlear hair cells in mammals. Ototoxicity and otoprotection studies in this species correlate with results in humans, supporting its use for preliminary drug screening. 5 The crista ampullaris of amphibians like Rana catesbeiana and Xenopus laevis has also been used to study vestibulotoxic effects after treatment. 6 Rodents like the chinchilla, rat and guineapig are good models for hearing research because their size facilitates surgical interventions at the cochlea. Additionally, the chinchilla has the same auditory frequency range as humans, unlike other animals that have higher frequency ranges. 7 However, the genetic background of these species is usually heterogeneous hampering the reproducibility of the data. By contrast, the genetic standardization of the mouse and its amenability to genetic engineering has made this specie the preferred model to explore the basis of cochlear pathologies and new therapeutic interventions. 8 Still there are not enough standard procedures to measure ototoxicity in adult mice because their cochlear hair cells are usually resistant to aminoglycosides and typically require very high, even close to lethal, doses before any ototoxicity is evident. 9 For this reason, a number of ex vivo organ and in vitro cell cultures have been established and used to screen for drug ototoxicity. 10
In this study, we have compared four systemic treatments with aminoglycoside antibiotics in adult mice evaluating auditory-evoked potentials and cochlear morphology. Accordingly, we found that gentamicin and neomycin cause high rates of mortality in adult mice without achieving any significant changes in the auditory threshold. While amikacin produced a tolerable mortality rate at high doses, it did not provoke ototoxicity. Finally, intramuscular injection of kanamycin induced significant dose-dependent bilateral hearing loss and changes in cochlear morphology, with a moderate rate of mortality and less discomfort than following subcutaneous administration. Our data indicate that this refined protocol could serve as a reference model for the in vivo study of ototoxicity in the adult mouse, as well as to develop cell repair and regeneration strategies.
Materials and methods
Mouse husbandry and handling
Six-week-old male mice supplied by Harlan Iberica (Barcelona, Spain) were used in this study and two strains were selected to test the possible strain-specific differences in terms of sensitivity to aminoglycosides: HsdOla:MF1, a robust outbred strain; and C57BL/6JOlaHsd, an inbred strain with genetic predisposition to age-related hearing loss 11 and high susceptibility to cochlear damage. Mice were housed in cages (Panlab Harvard Apparatus, Spain) in groups of six, with wood shaving litter (Harlan Iberica), under standard conditions: 12 h:12 h photoperiod, room temperature between 20 and 22°C, relative humidity of 45–55% and 20–22 air changes per hour. Standard mouse food (Teklad Global 2014 diet, Harlan Iberica) and water were available ad libitum. All mice were allowed to acclimatize to the animal facility for at least one week before the first test was performed. Health monitoring was performed following FELASA recommendations, 12 and all the experiments complied with local and European legislation regarding animal use and experimentation, and they were approved by the CSIC Animal Care and Use Committee.
Drug administration
Mice from the different experimental groups (Table 1) were treated with the corresponding aminoglycoside or with physiological saline solution for two weeks. Commercial antibiotic solutions (gentamicin sulphate, B Braun Medical Rubi, Spain; amikacin, Bristol-Myers-Squibb, Madrid, Spain; neomycin and kanamycin, Sigma Aldrich Quimica, Tres Cantos, Spain) were diluted in 0.9% NaCl to obtain the desired concentration. The dosage administered was selected on the basis of previously published ototoxic protocols using gentamicin, 10 amikacin, 13,14 neomycin 15 and kanamycin, 10 and adapted to mice if necessary. Special interest was paid to the kanamycin treatment because this drug alone, or in combination with loop diuretics, presented the best bibliographic profile (summarized in Table 2). The injections were adjusted daily according to body weight and they were administered using a 1 mL syringe and 25-gauge needle by the same person at the same time each day to avoid stress and to maintain regular drug serum levels (08:00 h for once a day and 08:00–20:00 h for twice a day injections). The restraint and injection procedures were those recommended by FELASA. 16
Doses of the aminoglycoside antibiotics used
Mice were treated with aminoglycosides at the doses and times indicated. The dose selected was based on previously published ototoxic protocols in rodents with gentamicin, 9 amikacin, 13,14 neomycin 15 and kanamycin, 9 and adapted to mice where necessary. Dose: mg/kg BW, mg drug per kg of body weight. Route of administration: i.m. = intramuscular; s.c. = subcutaneous; i.p. = intraperitoneal
Reported ototoxic treatments with kanamycin in the mouse
Non-invasive evaluation of hearing
Hearing was evaluated by auditory brainstem responses (ABR) as previously described. 17 The ABR is a non-invasive technique commonly used for hearing evaluation in mice 11,17,18 that measures the electrical activity produced along the auditory pathway in response to sound. The data collected included the hearing threshold and the latency of four to five peaks that represent different steps along the auditory pathway. Mice were anaesthetized by intraperitoneal injection of 100 mg/kg of ketamine (Imalgene 500®, Merial Laboratorios SA, Barcelona, Spain) and 10 mg/kg of xylazine (Rompun®, Bayer Sanidad Animal, Barcelona, Spain). After loss of the withdrawal reflex, the animals were placed on a heated water-bed in a soundproof cage. Three electrodes were situated subcutaneously: the active electrode in the vertex zone, the reference electrode in the mastoid region, and the ground electrode in the lumbar region. Click and 8–40 kHz tone-burst stimuli were generated with SigGenRP™ software (Tucker-DavisTechnologies TDT, Gainesville, FL, USA) and calibrated using a 1/4 inch microphone (ACO Pacific Inc, Belmont, USA). The click-type sounds lasted for 0.1 ms and the tone-type sound for 5 ms (sinusoidal stimulation, 2.5 ms for the increase and decrease in frequency, with no plateau). Sounds were delivered from a free-field electrostatic speaker (ES1, TDT) placed 5 cm from the pinna. The electroencephalographic activity was amplified with a low impedance headstage (RA4-PA/RA4-LI, TDT) and then recorded with BioSigRP™ software (TDT). The average response from 1000 stimulations at different intensities was calculated, from a 90 dB sound pressure level (SPL) to 20 dB SPL in 5–10 dB steps. ABR was performed before starting the treatments, 24 h after the last injection and once weekly for a month thereafter. Auditory thresholds were determined as described. 17 In brief, the threshold value was defined as the lowest sound intensity capable of generating a recognizable evoked response where the average voltage of the peaks is greater than two standard deviations of the average of the baseline electrical activity. The audiogram was generated through the auditory thresholds in response to tone-burst stimuli. Aminoglycosides usually induce a bilateral deafness, but occasionally they could produce unilateral hearing loss. 27 Therefore, both ears were evaluated with this method.
Cochlear morphology
Five weeks after ending the treatment, the cochleae were dissected out of the mice as described previously. 28 Mice were anaesthetized with pentobarbiturate (Dolethal, Vetoquinol, Spain) and then they were cardiac perfused with 4% paraformaldehyde in 0.1 mmol/L phosphate-buffered saline (PBS), pH 7.5. The inner ear was dissected out, placed in a fresh fixative solution for 12 h and washed with PBS. The cochleae were then decalcified using a chemical fast bone decalcifier (RDO, Eurobio SA, Paris, France). Paraffin sections (5 µm thick) were obtained parallel to the modiolus with a microtome (RM2155, Leica Microsystems SA, Spain), and stained with haematoxylin–eosin or 1% cresyl violet (Fluka, Sigma Aldrich, Spain). The cochlear morphology and cytoarchitecture were studied in the sections using a Zeiss Diaplan microscope coupled to a digital camera (Leitz DFC300 FXC) to obtain light microphotographs. None of the alterations observed were associated with specimen manipulation or the histological technique used; morphology of cochlear structures was assessed as reported. 28
Statistical procedures
Statistical analyses were performed using SPSS 14.0 software (SPSS Inc, Chicago, IL, USA) applying analysis of variance and the Bonferroni post hoc test. All values are presented as the mean (SD) and P < 0.05 was considered statistically significant.
Results
Comparison of hearing parameters after different aminoglycoside treatments in adult mice
Systemic administration of ototoxic doses of aminoglycosides was frequently associated with adverse effects and increased mortality rates. In our hands, the administration of neomycin at 300 mg/kg/24 h for 15 days showed the highest systemic toxicity with a mortality rate of 71%. By contrast, amikacin administration was better tolerated, presenting the lowest mortality rate of the treatments tested (Table 1). Twice daily treatment with 600 and 800 mg kanamycin/kg for 15 consecutive days produced a mortality rate of ∼30%. The animals treated with toxic doses of aminoglycosides also showed other side-effects during the study, such as weight loss, respiratory insufficiency and sudden death after injection. In addition, subcutaneous injection of amikacin frequently produced skin lesions at the site of injection.
Hearing loss associated with inner ear local toxicity was evaluated by measuring the ABR threshold shift after antibiotic treatment. At the beginning of the study, all mice showed similar hearing thresholds of between 40 and 45 dB SPL, as expected in young animals of the HsdOla:MF1 and C57BL/6JOlaHsd strains. Indeed, the ABR waves obtained displayed the five standard peaks (I–V).
Different doses of aminoglycosides were administered by the subcutaneous, intramuscular or intraperitoneal route, and their effects were compared. Intramuscular injections of gentamicin (300 mg/kg) for 15 days were poorly tolerated by HsdOla:MF1 mice (mortality rate 50%) and produced only small (0–5 dB SPL) and variable threshold shifts. Tolerance to amikacin and neomycin administration was also studied in the same strain and subcutaneous administration of amikacin (500 mg/kg/24 h) produced the lowest mortality rate (25%), although no significant auditory threshold shifts were evident. In contrast, intraperitoneal neomycin (300 mg/kg/24 h) caused extremely high mortality rates and a poor shift response (the auditory threshold shifts are summarized in Figure 1a).
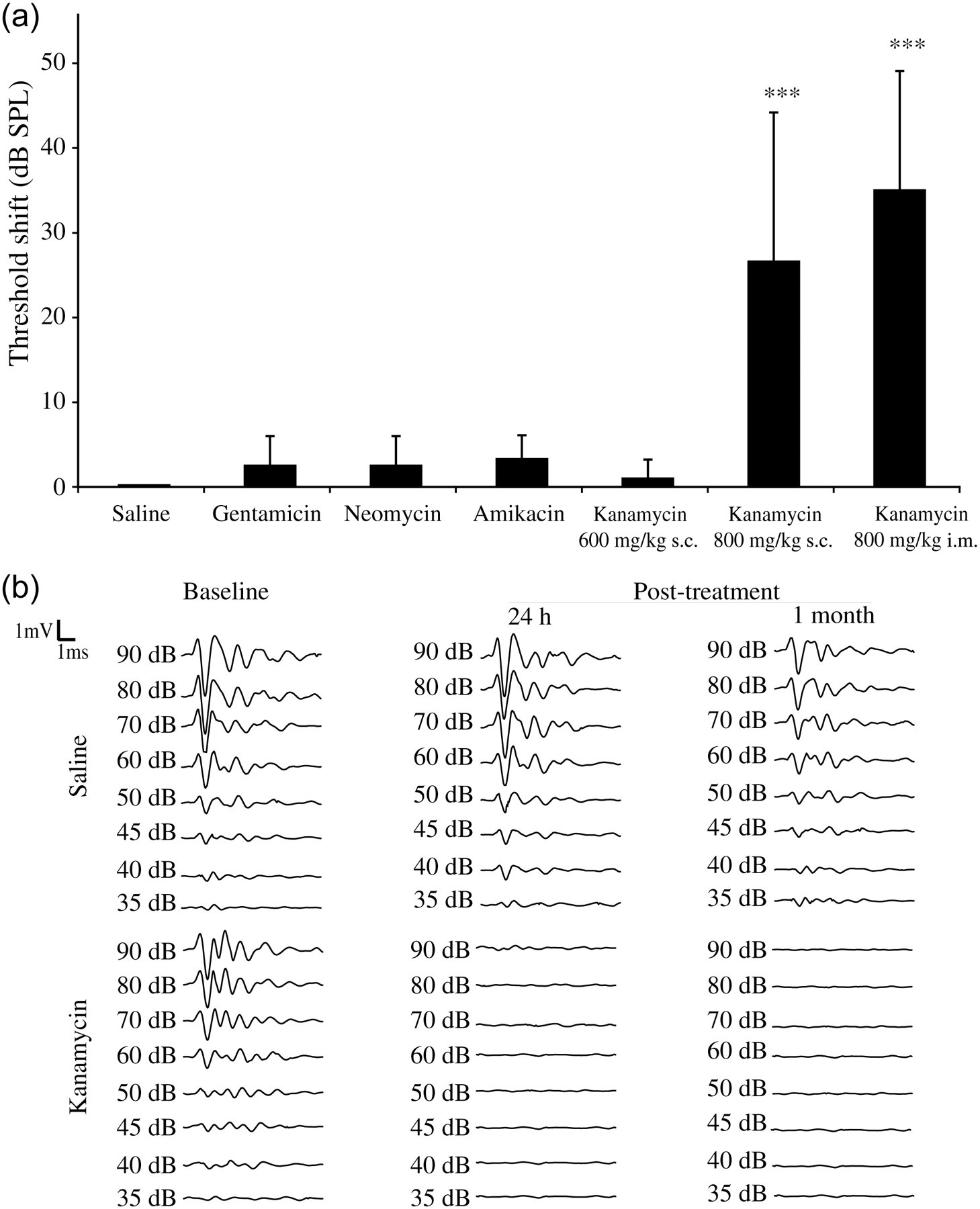
Kanamycin administration alters the auditory brainstem response (ABR) waves and increases hearing thresholds. (a) Threshold shifts in the experimental groups after aminoglycoside antibiotic administration for 15 days. The data are presented as the mean (SD) of groups of at least six animals at the beginning of treatment. After click stimulus, only kanamycin at a dose of 800 mg/kg body weight induced threshold shifts that differ significantly from those of the saline controls (P < 0.001). (b) ABR waves from control saline and kanamycin-treated mice at different time points. Baseline ABR waves exhibited normal morphology. At the end of the treatment and one month later, kanamycin-treated animals displayed an aberrant wave morphology when compared with control mice. SPL = sound pressure level; i.m. = intramuscular; s.c. = subcutaneous
Kanamycin has been documented to produce hair cell loss in mice (summarized in Table 2). Different treatment regimens were examined in C57BL/6JOlaHsd: an initial dose of subcutaneous kanamycin was tested (600 mg/kg/24 h), which did not display any effect on the ABR parameters studied (data not shown). As a result, two subcutaneous injections per day were administered in order to achieve sustained drug serum levels. Accordingly, while mice treated with 600 mg/kg/12 h did not experience significant changes in their hearing threshold in response to click stimulus, there was a noticeable threshold shift when kanamycin was administered at 800 mg/kg/12 h, confirming previous reports 10 (Figure 1a). Kanamycin frequently produced dermatitis at the site of the subcutaneous injection, a trait that was associated with increased mortality. In order to avoid this effect, the intramuscular route was evaluated. Kanamycin administered intramuscularly (800 mg/kg/12 h) provoked a consistent auditory threshold shift (TS = 35 dB SPL, P < 0.001) and a tolerable mortality rate. ABR wave alterations were comparable to those obtained following subcutaneous administration, but this protocol clearly improves the side-effects on the animals.
A detailed analysis of hearing impairment was performed in all the surviving mice administered intramuscular injections of kanamycin (800 mg/kg/12 h). Of these animals, 85% developed profound deafness (hearing threshold ≥70 dB SPL: see the ABR pattern in Figure 1b) and 15% moderate deafness (50–60 dB SPL hearing threshold).
Indeed, at the end of the treatment the mice injected with kanamycin had a mean hearing threshold of 78 ± 18 dB SPL, whereas saline-treated mice preserved the baseline values (40 ± 4 dB SPL). The evolution of the hearing thresholds over one month did not indicate any functional recovery in kanamycin-treated mice (84 ± 13 dB SPL; n = 7 surviving mice), while the hearing thresholds of saline-injected animals remained stable (42 ± 5 dB SPL; n = 6). Thus, kanamycin caused irreversible bilateral deafness during the experimental period (Figure 2).
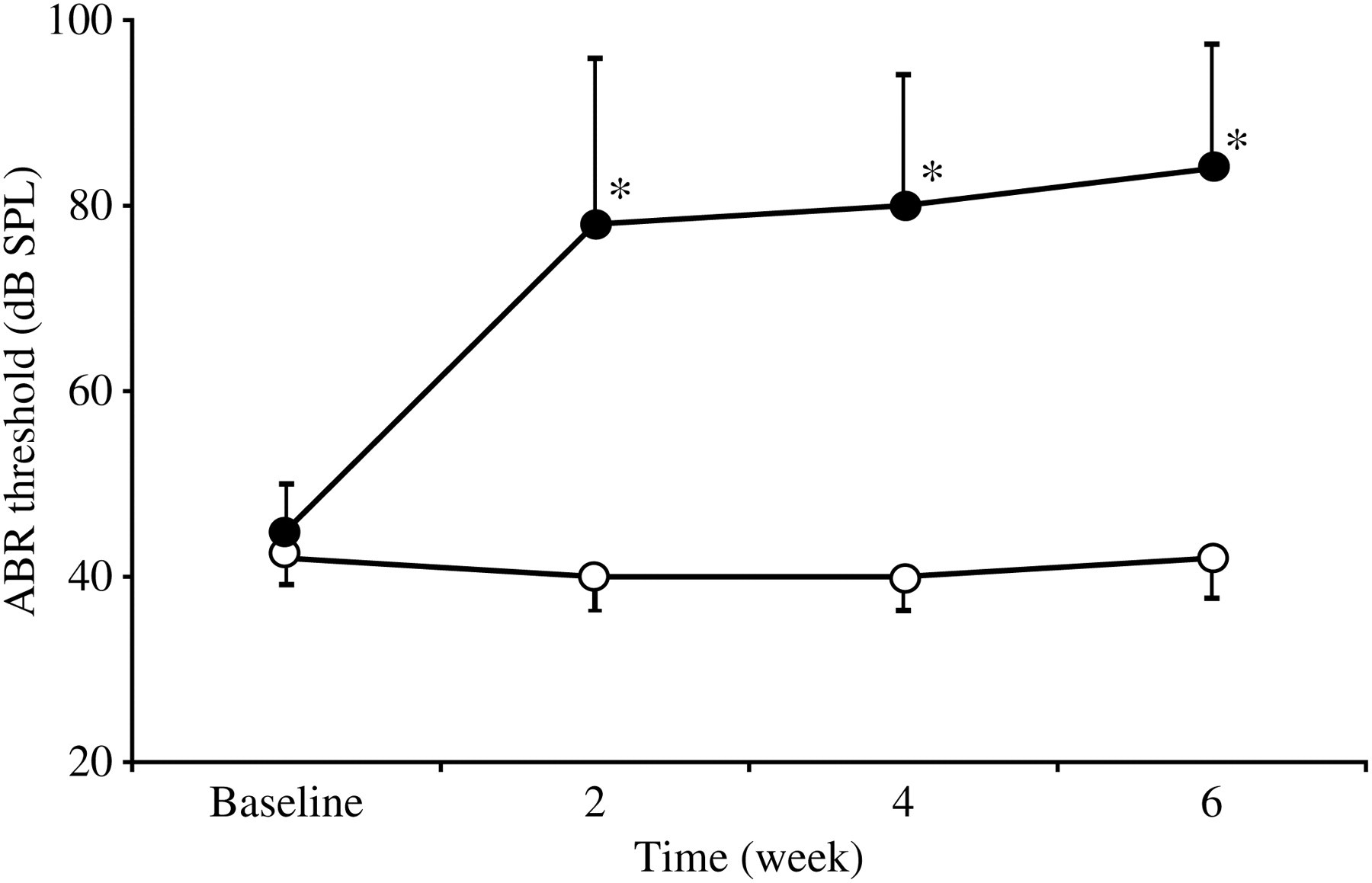
Evolution of hearing thresholds over time after kanamycin treatment. Mice treated with 800 mg/kg BW/12 h/i.m. kanamycin for 15 days showed severe and irreversible deafness (closed circles), unlike those treated with saline (open circles). Threshold shifts in kanamycin-treated mice differed significantly from those in saline controls (P < 0.05, n = 10) at all the times tested. SPL = sound pressure level; ABR = auditory brainstem response
To determine which frequencies were more strongly affected by kanamycin, the evoked response to 8, 20 and 40 kHz tone-burst stimuli was studied in mice with profound deafness (n = 6, Figures 3a and b). The threshold shift in mice treated with kanamycin was significant at all the frequencies analysed (P < 0.05), with values above 70 dB SPL.
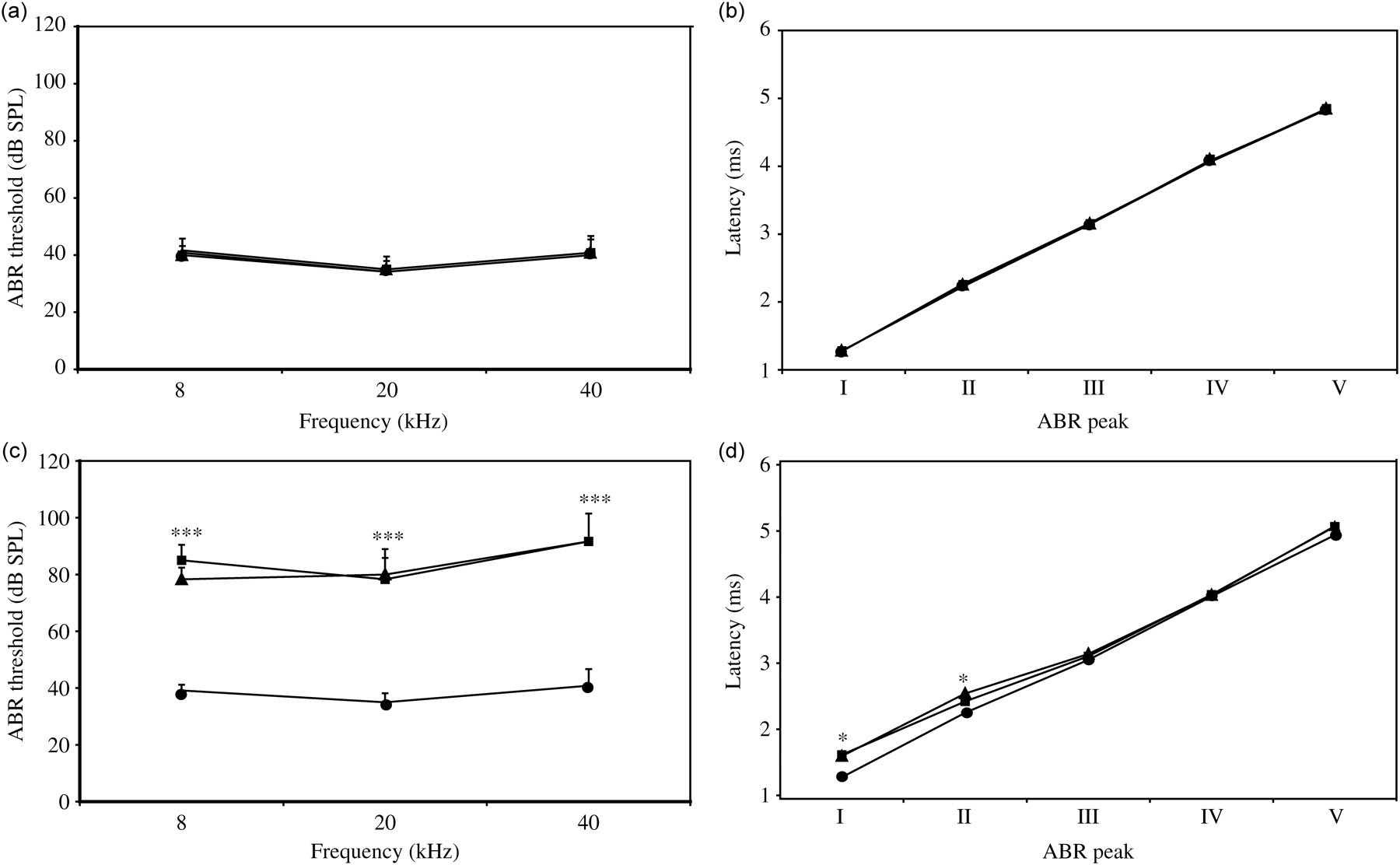
Evolution of the audiogram and I–V peak latencies of kanamycin-treated mice. Audiograms and I–V peak latencies of mice treated with saline (n = 6) and kanamycin (800 mg/kg BW/12 h/i.m.) for 15 days (n = 6) at three different moments: baseline (circles), end of treatment (triangles) and one month later (diamonds). (a, b) Saline-treated animals maintained a similar audiogram profile (a) and ABR peak latencies (b) at all the times tested. (c) Mice treated with kanamycin presented a bilateral increase in hearing thresholds at all frequencies, with significant differences between the baseline and post-treatment values (P < 0.001). (d) When compared with the baseline values, kanamycin-treated mice also showed an increase in the latency of peaks I and II after treatment and one month later (P < 0.05). SPL = sound pressure level; ABR = additory brainstem response
Specifically, kanamycin induced an auditory threshold shift of 50 ± 7 dB SPL at high frequencies (40 kHz), of 43 ± 8 dB SPL for middle frequencies (20 kHz) and of 39 ± 5 dB SPL at low frequencies (8 kHz), indicating that the ototoxic damage affected all the cochlear turns, although it produced more damage in the basal one.
Each of the five peaks in the ABR wave is characterized by a latency (measured in milliseconds) and amplitude (measured in volt units). At the beginning of the study, peak latencies were similar in all mice (Figure 3c). Mice administered kanamycin then displayed an increase in the latency of the ABR peaks, particularly in peak I (from 1.27 ± 0.09 to 1.61 ± 0.06 ms, P = 0.001) and II (from 2.24 ± 0.11 to 2.42 ± 0.06 ms, P < 0.05), which represents activity in the cochlear–auditory nerve and the cochlear nucleus, respectively (Figure 3d). The delayed latencies decreased gradually in the following steps of the auditory pathway from the cochlea (peak I) to the lemniscus (peak IV). This pattern is illustrated by the interpeak latencies (IPL), whereby IPL I–II represents the time delay in the peripheral auditory pathway (from the cochlea to the cochlear nucleus), and IPL II–IV that in the central pathway (from the cochlear nucleus to the lemniscus). There were no significant differences between IPL I–II before and after treatment with intramuscular kanamycin (from 0.97 ± 0.09 to 0.93 ± 0.11 ms, P = 0.35), or for IPL I–IV (from 1.76 ± 0.20 to 1.69 ± 0.11 ms, P = 0.31). Hence, the delay appeared to start in the peripheral region of the pathway while central transmission speed was not affected. Concomitantly, there was a reduction in the amplitudes of the peaks I and IV in mice that received kanamycin at all stimulation intensities in a range from 90 to 60 dB. Furthermore, the downward slopes of the amplitude reduction versus stimulation intensity were similar (data not shown).
Since strain differences in the susceptibility to ototoxins have been reported, the same protocol was evaluated in HsdOla:MF1 mice. No significant differences were found at the functional level between the two strains tested after kanamycin treatment (data not shown), possibly since young animals were evaluated. However, there were differences between the cochlear cytoarchitecture of the strains as discussed below.
Cochlear histopathology of kanamycin-treated adult mice
The gross morphology of the inner ear was evaluated in cresyl violet (Figures 4A–C) or haematoxylin/eosin (Figures 4D–I) stained sections from mice that exhibited functional alterations after intramuscular kanamycin administration. A normal cytoarchitecture was evident in the organ of Corti from animals administered saline, with three rows of OHC and one row of IHC (Figure 4B). In addition, the spiral ganglion had a normal neuronal packing density with no acellular patches (Figures 4C and E). By contrast, intramuscular kanamycin (800 mg/kg/12 h/15 days) injection provoked structural damage, which was in accordance with the functional deficits, including the progressive loss of both nerve fibres at the spiral osseous lamina and of neurons in the spiral ganglia (Figures 4F and G). Indeed, IHC and OHC degenerated in the cochlea and along the entire organ of Corti, and they were even lost at basal turns (Figure 4H). The upper turns of the cochlea displayed a collapsed organ of Corti, associated with the loss of OHC or both hair cell types (Figure 4I). Spiral ganglion cell packing density diminished rapidly following the appearance of deafness in parallel with IHC loss. After comparative evaluation of cell density and strial thickness on these sections, no obvious alterations were observed in the stria vascularis or in the spiral ligament (not shown).
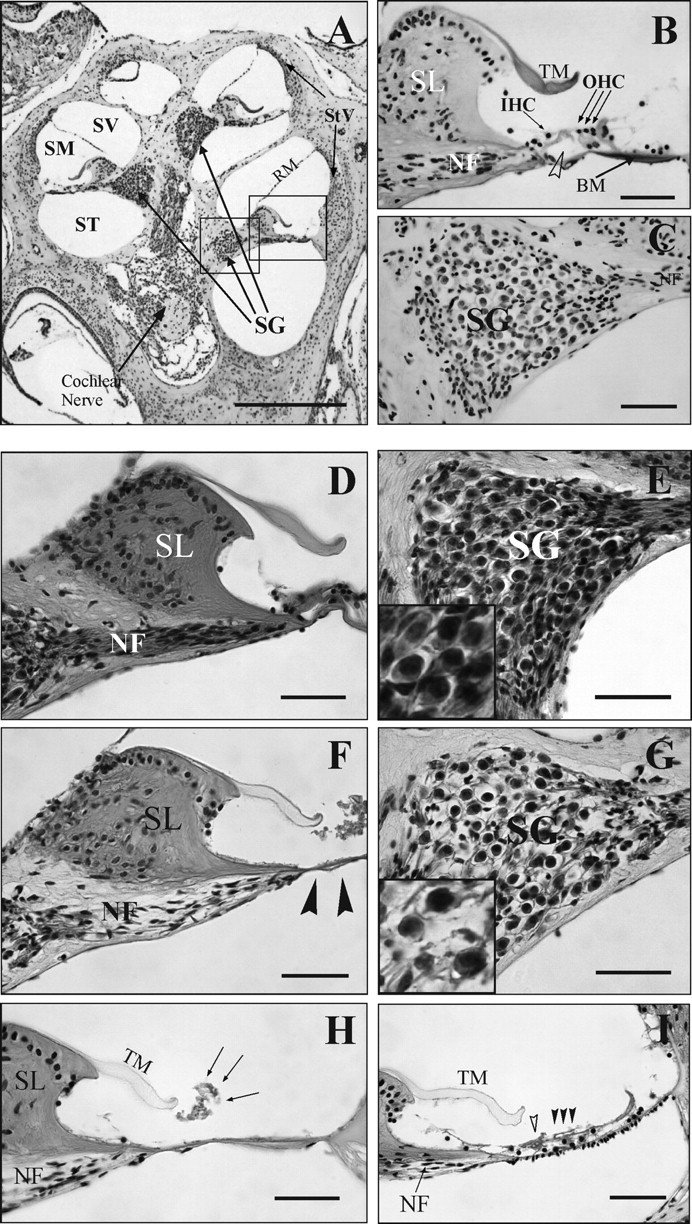
Cochlear morphology after kanamycin administration. (A–C) Paraffin sections from saline-treated mice stained with cresyl violet. Mid-modiolar section (A) illustrating the cochlear anatomy. The organ of Corti (B) shows a normal cytoarchitecture with ordered rows of hair cells (black rows) and an open tunnel of Corti (open arrowhead). The spiral ganglion has a normal neuron packing density (C). (D–G) Comparison of haematoxylin–eosin stained cochlear sections. Sections from saline (D and E) and kanamycin-treated mice (F and G) are shown. Note the normal tracts of nerve fibres in the spiral osseus lamina (D) and the normal appearance of the spiral ganglion (E, inset). By contrast, cells are absent at the organ of Corti (F, arrowheads), nerve fibres staining is diminished and the neuronal packing density is decreased in the spiral ganglion (G, inset) of the mice that received kanamycin. (H and I) Severe degeneration of the organ of Corti in C57BL/6JOlaHsd mice stained with haematoxylin–eosin. Note the total absence of auditory receptor cells in the basal turn: the arrows indicate the area where there are no support cells, pillar elements or hair cells (H). In the apical turn there is a loss of hair cells in the auditory receptor (I). IHC, open arrowhead and OHC, arrowheads. SV = scala vestibuli; SM = scala media; ST = scala tympani; RM = Reisner's membrane; StV = stria vascularis; Spl = spiral ligament; SL = spiral limbus; NF = nerve fibres; TM = tectorial membrane; BM = basilar membrane; SG = spiral ganglion; OHC = outer hair cells; IHC = inner hair cells. Scale bars: 0.5 mm (A) and 50 μm (B–I)
As already mentioned, there were differences in the cochlear cytoarchitecture between strains, and the cochlea of C57BL/6JOlaHsd mice exposed to kanamycin was seriously affected with cell damage evident in the basal turn at the organ of Corti. In addition, sensory cells were absent in areas of the spiral ganglion that also showed extensive neuronal loss (Figures 4G–I). The OHC were lost at the middle and apical turns, although the IHC and pillar cells of the tunnel of Corti maintained their gross structure. Similarly, the neurons of the spiral ganglion were less affected. By contrast, light microscopy images indicated that sensory cell damage was less severe in HsdOla:MF1 mice treated with kanamycin and it was apparently restricted to the basal turn. In this strain, although the tunnel of Corti collapsed, the OHC and IHC survived the treatment. Nevertheless, spiral ganglion cell density rapidly decreased following ototoxic damage (data not shown), although the middle and apical turns were practically unaffected. Therefore, the two strains studied suffered different changes to their cochlear cytoarchitecture, with a more severe pattern of lesions in the inbred C57BL/6JOlaHsd mice when compared with the outbred HsdOla:MF1 strain.
Discussion
Drug-induced hearing loss is an important cause of deafness worldwide and a concern for the medical community. More than 130 different chemicals are known to be potentially ototoxic and many of them are common drugs, such as aminoglycoside and macrolide antibiotics, loop diuretics, non-steroid anti-inflammatory drugs, anti-neoplastic or antimalarial drugs. 1 Hearing loss is therefore a central aspect of drug-induced toxicity but, surprisingly, it is not routinely evaluated in preclinical studies, at least in part because there are no standard reference models.
In vitro cell-based assays have been used to screen for ototoxicity and to explore factors that contribute to otic cell maintenance and repair. 10 However, they cannot assess the complex cellular interactions and metabolic responses that determine drug efficacy or cause toxicity in vivo. Therefore, many drugs that appear to be effective in cell-based assays fail in animal testing, and vice versa. Several species can be used to establish animal models of aminoglycoside-induced hair cell loss. Since the genetic standardization and relative ease of genetic engineering in the mouse makes it the preferred species in hearing research, 8 it would be valuable to establish standard procedures to study ototoxicity, as well as reliable in vivo mouse models for drug screening and otoprotection studies.
Several studies have previously defined aminoglycoside doses that produce hair cell loss in mice and other rodents. 10,13–15 Together with the results presented here, these studies show that adult mice are highly resistant to aminoglycoside ototoxicity and that treated-mice often die due to systemic toxicity before any appreciable alterations in auditory function or cochlear morphology occur. Indeed, high doses of aminoglycosides, often close to lethal, are usually required to damage hair cells, while lower doses decrease the mortality rate but they do not produce noticeable alterations in the cochlear sensory epithelia. On the other hand, functional follow-up in ototoxicity studies in mammals have been based on classical behavioural testing, which frequently generates false-negative data since the mouse and other rodents can lose most of their high-frequency hearing and still respond to ambient sound. In this context, electrophysiological techniques like ABR provide precise and quantitative information on the functional alterations induced by aminoglycosides. Therefore, to establish a reliable aminoglycoside ototoxicity mouse model, it is necessary to carefully choose the drug, the dose and the route of administration, as well as to assess the alterations in cochlear function and morphology using objective methods. It is also important to consider the characteristics of the mouse strain. For example, the C57BL/6JOlaHsd strain carries a mutation in the Ahl gene which may contribute to otic insult susceptibility. 9,11
In our hands, intramuscular administration of gentamicin (300 mg/kg/24 h for 15 days) did not produce a noticeable threshold shift. Moreover, gentamicin was not well tolerated and caused systemic toxicity in the adult mouse, which seems to tolerate high gentamicin doses worse than other rodents like the guineapig. 29 These results are in accordance with a previous report on C57BL/6JOlaHsd mice 9 and indicate that gentamicin produces very variable hearing loss and an unacceptable rate of mortality in adult mice at high doses.
Mouse ototoxicity caused by treatment with either neomycin or amikacin alone has not been previously reported; therefore, the doses used were adapted to mice from studies in other species. 13–15 Since they did not produce observable ototoxicity in adult mice at these doses, these antibiotics do not appear to be suitable for this purpose. In addition, intraperitoneal neomycin produced the strongest systemic toxicity, which was inappropriate for in vivo experimental procedures. When administered subcutaneously, amikacin produced limited systemic toxicity but a high incidence of severe skin lesions, particularly swelling and irritation at the injection site.
According to previous ototoxicity protocols with kanamycin in mice, we used a starting dose of 600 mg/kg BW/day for 15 days. Our results confirmed that in adult C57BL/6JOlaHsd mice, this protocol had no effect on any of the parameters studied. By contrast, a lower dose of 400 mg/kg/day for 10 days induced a noticeable threshold shift in 10-day-old mice. 24 This suggests that pre-weaning mice have a higher susceptibility to kanamycin ototoxicity. Once daily injection regimens in adult mice require very high doses of kanamycin (up to 1000 mg/kg) to cause a significant threshold shift in one mouse in five. 9 Therefore, a protocol of twice daily subcutaneous injection of 600 or 800 mg/kg BW of kanamycin during 15 days was tested. 9 While the lower dose did not produce functional impairment in adult mice, the twice daily dose of 800 mg/kg BW produced a significant auditory threshold shift in response to click and tone-burst stimuli. Following the general base-to-apex pattern of cochlear damage, 2 there was a higher threshold shift at high frequencies (40 kHz) than at 8 or 20 kHz. These results confirmed that administering high doses of subcutaneous kanamycin twice daily produces ototoxic damage in adult mice, extending previous experimental data by providing more detailed information on the ABR of mice treated in this way.
To avoid the skin lesions observed in some animals after subcutaneous injection, the intramuscular route was evaluated. Intramuscular administration of kanamycin at 800 mg/kg every 12 h over 15 days also caused significant, consistent and reproducible otic damage with slightly lower mortality rates than when administered subcutaneously. However, since repeated intramuscular injections produced less discomfort to the animals than repeated subcutaneous injections, this alternative administration route refines the protocol and improves animal welfare.
Kanamycin treatment not only increased the auditory threshold but it also produced an increase in peak I latency, and consequently in the following peaks II to V. Both alterations are caused by the damage at the organ of Corti and reflect the malfunction in the mechano-transduction process of the hair cells or in the activity of the auditory neural pathways. However, no significant differences were found in the IPL, indicating that the damage is restricted to the cochlea.
Morphological changes in the ABR waves of kanamycin-treated mice were correlated with alterations to the cochlear cytoarchitecture. Kanamycin administration caused progressive alterations from the basal turn of the cochlea to the apex, and it primarily affected the OHC and to a lesser extent the IHC. This suggests a pronounced differential sensitivity to ototoxic damage between IHC and OHC in the mouse, which may have implications for the use of the mouse as models for human deafness. Nevertheless, there was also IHC loss that was well correlated with the alterations to the spiral ganglion. The neuronal packing density decreased after ototoxic treatment in parallel to the number of surviving IHC, but not those of OHC, confirming earlier data from the guineapig. 30 At the times studied, the qualitative morphological assessment did not identify obvious alterations in the stria vascularis of kanamycin-treated animals. However, light microscopic assessment could not judge more subtle types of damage and, therefore, a loss of marginal cells or changes in strial thickness could not be ruled out. In fact, accelerated strial thinning has recently been reported in CBA/Ca mice after treatment with a single dose of kanamycin plus bumetanide. 26
Despite the lesions observed, there were also regions in the organ of Corti and spiral ganglion that maintained their structure after kanamycin treatment. Therefore, the procedure reported here produced clear but still limited local damage that makes this model highly suitable for otoprotection studies in the adult mouse.
In conclusion, systemic treatment with gentamicin, amikacin and neomycin are not suitable to provoke ototoxicity in adult mice, whereas high doses of intramuscular kanamycin cause reproducible and consistent ototoxic damage, and when compared with the subcutaneous route, they improve animal wellbeing. Therefore, this protocol could be used as a standard model for ototoxicity screening and cell repair studies.
Footnotes
Acknowledgements
This work was supported in part by grants from DIGNA Biotech SA, the Spanish Ministries of Health and Education (BFU-200500084 and SAF 2008-00470), the Community of Madrid (CAM-PRICIT0530) and Mutua Madrileña (MM 2007). SM held contracts from MM and CIBERER.
We appreciate the help of Laura Barrios (CTI CSIC, Madrid) with the statistical analysis of the data and the helpful comments on the manuscript of Dr Jose M Zubeldia (Hospital General Universitario Gregorio Marañón, Madrid).
