Abstract
Routine health assessment of laboratory rodents can be improved using automated home cage monitoring. Continuous, non-stressful, objective assessment of rodents unaware that they are being watched, including during their active dark period, reveals behavioural and physiological changes otherwise invisible to human caretakers. We developed an automated feeder that tracks feed intake, body weight, and physical appearance of individual radio frequency identification-tagged mice in social home cages. Here, we experimentally induce illness via lipopolysaccharide challenge and show that this automated tracking apparatus reveals sickness behaviour (reduced food intake) as early as 2–4 hours after lipopolysaccharide injection, whereas human observers conducting routine health checks fail to detect a significant difference between sick mice and saline-injected controls. Continuous automated monitoring additionally reveals pronounced circadian rhythms in both feed intake and body weight. Automated home cage monitoring is a non-invasive, reliable mode of health surveillance allowing caretakers to more efficiently detect and respond to early signs of illness in laboratory rodent populations.
Introduction
Accurate health monitoring of research animals is necessary to provide adequate care and avoid unnecessary suffering. In a recent survey of mice at a large research institution, “spontaneous mortality with unknown cause” was more common than all other adult morbidity types combined. 1 Simply put, most mouse illnesses were not detected until it was too late.
Common approaches to routine health surveillance include visual observation, body condition scoring, 2 and targeted assessment of experiment-specific outcomes.3–5 These labour-intensive methods become less practical as population size increases and have other important limitations. First, many diseases manifest in ways that are difficult for human observers to detect. The classic response to infectious disease – mirrored in several other disease states – is a suite of behavioural changes collectively known as “sickness behaviour.”6–9 Lethargy, reduced feeding and reduced grooming are virtually impossible to detect in brief health checks, particularly when conducted during the day (light phase), when mice are least active and often huddled together. 1 An additional concern is the oft-repeated (although not empirically proven) claim that mice hide signs of pain or disease to avoid being targeted by predators (e.g. humans) or aggressive conspecifics.1,3,9,10 Human observers often will not notice that mice are sick until their illness has conspicuous physical consequences, such as reduced body weight, low body condition score, or an unkempt coat. These subjective assessments also have the potential for inconsistency between observers.5,11
Second, handling mice for visual inspection, palpation or weighing 2 is typically aversive and provokes a physiological stress response,12,13 perhaps doubly so for animals who are sick or experiencing pain or distress. 14 Handling may also negatively affect health and experimental outcomes, for example by dampening mice’s subsequent immune responses to a challenge 15 or otherwise altering results of behavioural assays;16–18 this can be avoided by non-aversive handling techniques. 19
Therefore, routine health assessment should ideally rely upon non-stressful measurement – including at times when healthy mice are active – of parameters that are not prone to human error and are difficult for animals to hide. Various automated techniques allow continuous, objective, remote, handling-free observation of mice in their home cages, while they are unaware of being monitored.8,20–23
Automated home cage health assessment can detect condition-specific symptoms and changes consistent with sickness behaviour in various veterinary and experimental contexts. In neurodegenerative disease models, this approach distinguished sick from control mice weeks before standard assessments did, finding differences in rest, locomotion, grooming and their circadian rhythms, as well as in motor learning and performance.8,24–26 Automated monitoring revealed short-term decreases in locomotion and drinker visits in mice following traumatic brain injury 27 and decreased locomotion and social connectedness in commensal mice with experimentally induced sickness behaviour. 28 Automated video assessment readily detects reduced activity in post-surgical mice, where human observers instead rely on more obvious pain-specific behaviour (e.g. twitching or licking the affected area). 4
Here, we use automated home cage monitoring to detect changes in body weight and food intake, key signs of sickness behaviour that mice cannot voluntarily hide or alter and that can indicate deteriorating overall health. Sustained weight loss was predictive of imminent death in a rat cancer model, 29 and decreasing body weight and food intake are integral components of many humane endpoints for laboratory rodents.11,30,31 To our knowledge, automated body weight tracking has not previously been used for rodent health surveillance.
We developed an apparatus for automated, objective, accurate, continuous and non-invasive health monitoring of individual mice in group-housing. We hypothesized that a combination of body weight, feed intake and visual appearance could be used to monitor health. We predicted that automated assessment would show construct validity by successfully distinguishing mice with experimentally induced sickness from healthy controls. We predicted it would detect illness sooner than human observers conducting twice-daily assessments. Additionally, we opportunistically examined circadian fluctuations in feed intake and body weight.
Animals, material and methods
Animals
C57Bl/6J mice from Purdue University’s breeding colony were housed in same-sex sibling pairs from weaning. Six pairs (three male, three female) aged 17 weeks were used in a pilot to determine target doses of lipopolysaccharide (LPS). Five pairs (two male, three female) aged between 53 and 63 weeks then lived in the monitoring apparatus, with one member of each pair subjected to LPS challenge. In keeping with the 3Rs principles of replacement and reduction, 32 we used euthanasia-bound animals retired from previous experiments and strictly limited the overall number of animals exposed to LPS. Expecting pronounced treatment effects, we used just enough animals to reach adequate statistical power in a matched-pairs design. 33 Housing, care and experimental techniques were approved by Purdue University’s Institutional Animal Care and Use Committee (IACUC).
Husbandry
Mice were housed in standard shoebox cages (Alternative Design, Siloam Springs, AR; 18.3 cm W x 29.1 cm L x 12.6 cm H) with aspen bedding (Teklad, Harlan, Madison, WI, USA) and a wire lid. Room temperature, humidity and light-dark cycle were maintained at 20 ± 0.25℃, 55 ± 4%, and 14:10 (hr) (lights on: 0600 EDT) respectively. Tap water was available ad libitum. Food was available ad libitum before trials (Teklad Mouse Diet 2019, Harlan, Madison, WI, USA) and from the automated feeder during trials (0.025 g TUM, TestDiet, Richmond, IN, USA).
Animal identification
A member of Purdue’s veterinary staff surgically implanted all mice in the nape of the neck with a cylindrical, 2 mm x 12 mm, 96 mg radio frequency identification (RFID) transponder carrying a unique identification number (Trossen Robotics, Westchester, IL, USA). We allowed at least 1 week for recovery between surgery and experimental trials.
Health monitoring apparatus
The apparatus (Figure 1) consisted of a data collection tube attached to a standard shoebox cage. An RFID reader (PhidgetRFID 1023, Phidgets Inc., Alberta, Canada) identified mice as they entered the tube, triggering pellet distribution and recording of a picture. The reader’s small detection radius and position at the far end of the tube ensured animals would only be detected after having entered the tube completely. A custom computer program written in Access 2007 (Microsoft, Redmond, WA, USA) operated the apparatus and collected data.
Schematic representation of the mouse health monitoring apparatus. Mice lived in a standard shoebox home cage and regularly entered the transparent data collection tube on the scale to feed. Detection of a mouse at the distal end of the tube by the RFID reader triggered the hopper to dispense a single food pellet and (during the light period) the camera to take a picture.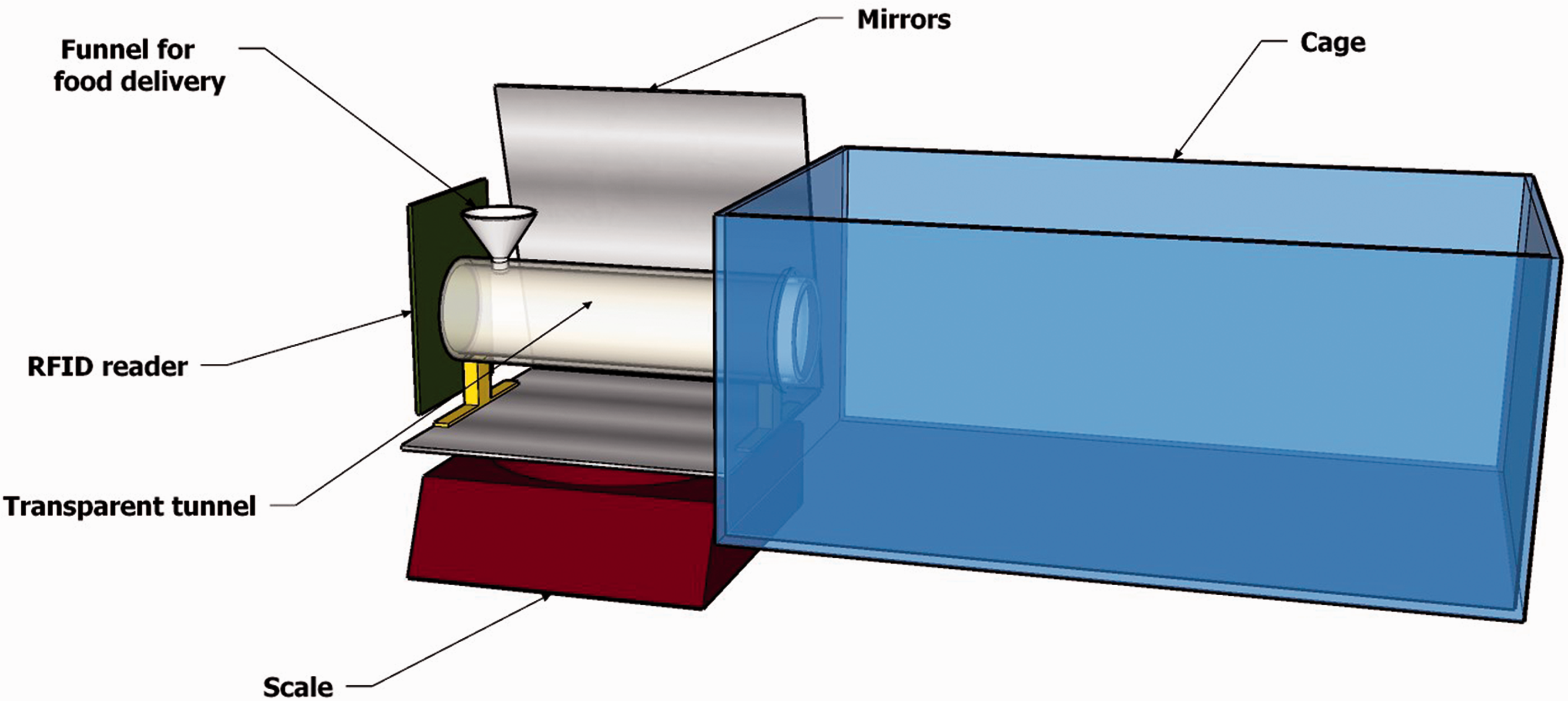
The photography unit consisted of a mirror and camera (UI-2230-C, IDS Imaging, Obersulm, Germany) positioned to capture the animal’s dorsal surface and reflected ventral surface in a single photograph. Pictures were recorded and timestamped at each visit to the data collection tube during the light period.
The weight measurement unit was a 1000 g X 0.01 g scale (iBalance M01, MyWeigh, Phoenix, AZ, USA) that continuously monitored weights and recorded all changes. The data collection tube was attached to the scale platform with its open end inserted free-floating through a hole in the side of the cage.
When an RFID tag was detected, the computer program activated the food hopper at the far end of the tube, releasing a single pellet. To ensure repeated and frequent visits, the hopper only released a pellet if the mouse had left the tube and the scale had recorded a stable near-zero weight since the last pellet was dispensed.
Experimental trials
We tested whether the automated monitoring apparatus could detect the effects of an LPS challenge. LPS is a component of the outer membrane of many pathogenic bacteria that reliably elicits sickness behaviour with systemic administration in mice.7,34 We administered 1 mg/kg Escherichia coli 0111:B4 LPS (Sigma Aldrich St. Louis, MO, USA) in saline vehicle intraperitoneally to experimental animals after determining, in the pilot, that this sub-lethal dose would induce short bouts of illness.
The main experiment consisted of five trials in which a same-sex pair of mice lived in the apparatus and were monitored continuously. Each pair was allowed at least 2 days in the apparatus for acclimation and baseline data collection. Following this, at 1600 hours we injected one randomly assigned mouse per pair with LPS and the other with saline. We kept the pair in the apparatus for at least 1 full day following injection and then euthanized them using rising CO2. We kept some pairs in the apparatus for longer periods before or after injection to collect further data (not reported here). As a humane endpoint, we euthanized LPS-injected mice immediately if they did not resume feeding after 24 hours or if recommended by veterinarians. We thoroughly cleaned and disinfected the apparatus between trials.
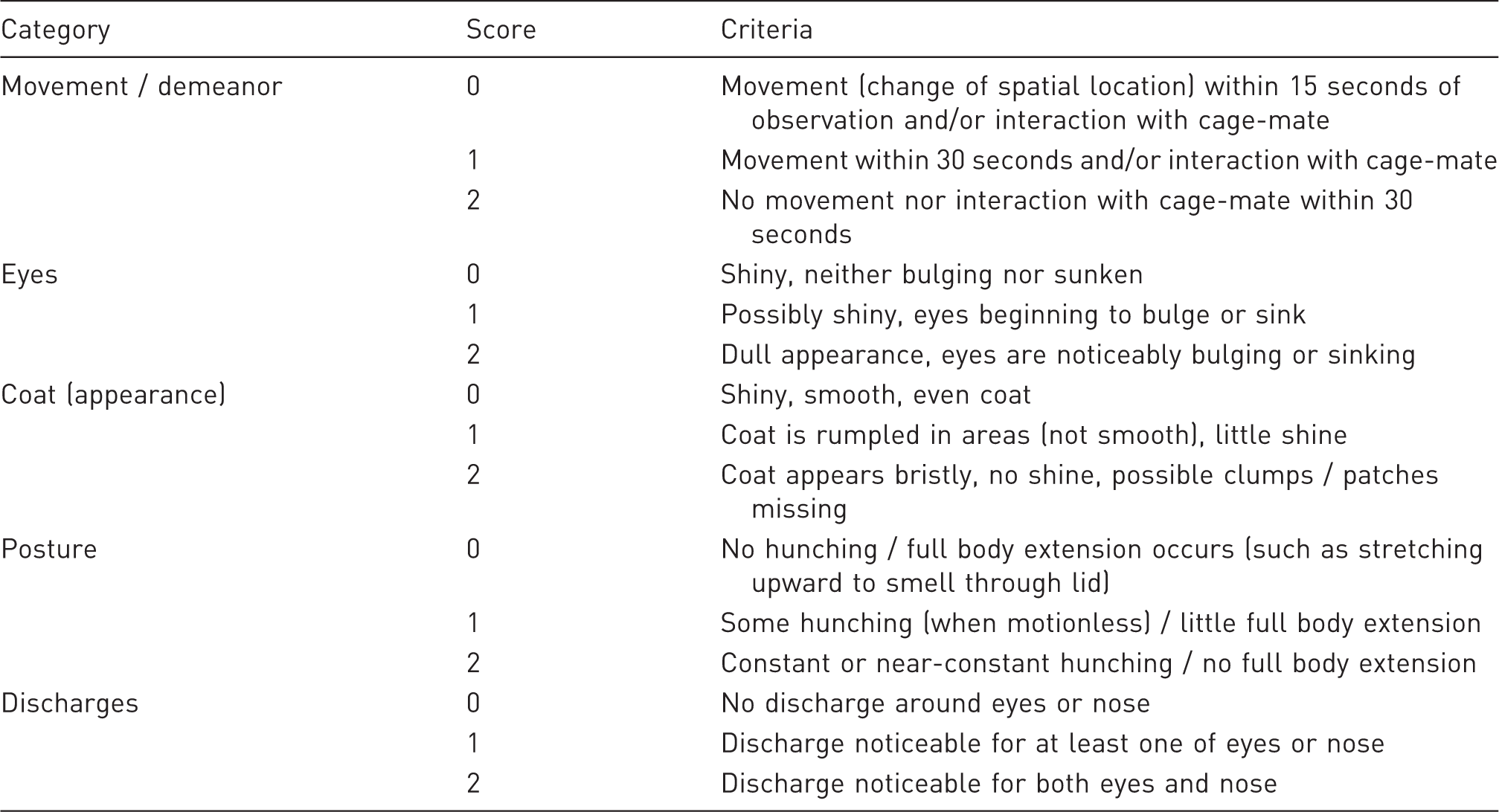
Illness Behaviour Score. Criteria were compiled from health assessment plans used in mouse and rat experiments monitoring illness and abdominal pain (LPS-induced and otherwise). Scores were summed across five criteria, with lower scores signifying better health.
Data management and statistical analysis
For each visit to the data collection tube (RFID detection), body weight was calculated as the average of stable scale readings recorded during the visit (if any), minus the last stable reading recorded without a mouse present. Baseline weights could shift over time due, for example, to bedding, droppings, or food in the tube.
To make the large quantity of data tractable for statistical analysis, we combined body weight and feeding data into temporal bins and restricted analysis to the 24-hour periods immediately preceding and following LPS or sham injections.
For each 1-hour bin, we calculated median body weight for each mouse, based on all visits with stable weight readings. We used the median to negate effects of occasional outlier recordings. For bins without stable weight recordings, wherever possible, we assigned body weights by a linear interpolation of the nearest hourly medians.
For feed intake, we calculated the sum of pellets delivered in 2-hour bins, to smooth out noise apparent in 1-hour bins.
We conducted statistical analyses using general linear mixed models in JMP 13.0.0 (SAS Institute, Cary, NC, USA). Models used to analyse circadian fluctuations in feed intake and body weight before LPS or sham injections included sex, time of day (1- or 2-hour bins as a categorical variable) and their interaction as fixed factors, and mouse nested in sex and the interaction of mouse with time of day as random factors.
Models used to analyse effects of treatment on feed intake and illness behaviour scores included treatment (LPS or sham), sex, time of day, experimental period (pre- or post-injection), and all their interactions as fixed factors, and mouse nested in treatment and sex and its first-order interactions with time and experimental period as random factors.
We verified general linear mixed model assumptions (homogeneity of variance and normality of residuals) graphically. Because these were violated in models of feed intake, we instead used the square root of the number of pellets obtained as a dependent variable.
We tested all fixed effects with an alpha = 0.05. We report tests of effects only where they are not included in significant higher-order interactions. We examined significant interactions using post hoc t-tests between factor levels of interest, with Bonferroni correction for multiple testing.
Results
During the 24 hours preceding LPS or sham injections, mice obtained an average of (+/− SD) 169.7 +/− 63.7 (range 90–283) pellets in 811.6 +/− 567.4 (range 381–2274) separate visits to the feeder, 227.5 +/− 103.7 (range 121–404) of which yielded stable body weight readings. The longest time without a stable reading for any individual mouse was just over 4.5 hours, during a period of typical inactivity (afternoon, light period).
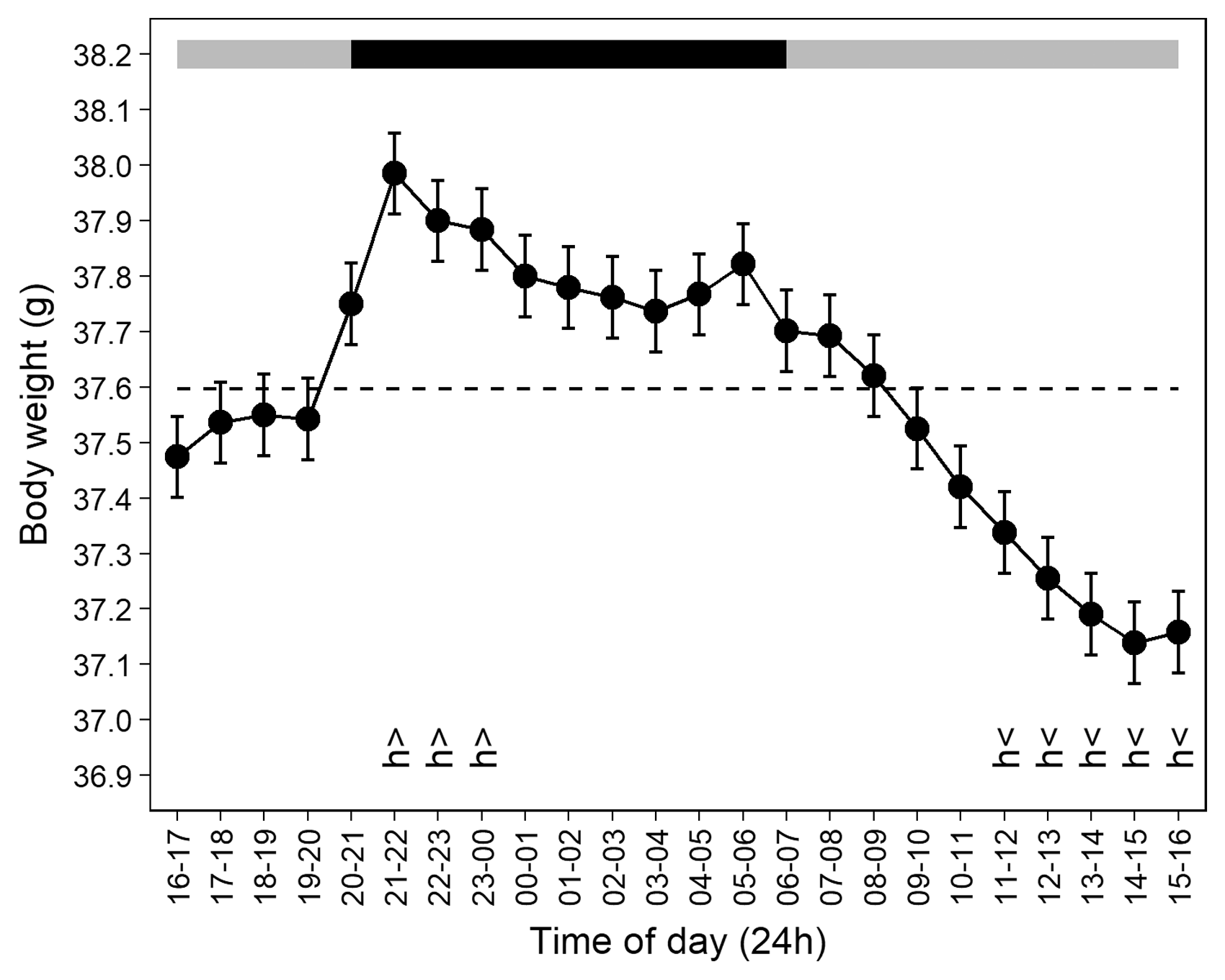
Prior to injection, feed intake and body weight showed marked circadian fluctuations, rising sharply at the start of the dark period, and being lowest during the light period. Time of day was the only significant factor affecting feed intake (F11,88 = 8.17, p < 0.0001; Figure 2) and body weight (F23,184 = 9.11, p < 0.0001; Figure 3).
Feed intake fluctuated with time of day over 24 hours pre-treatment. Feed intake was significantly above average (h>) during the 2000–2200 hour bin, immediately at the start of the dark period (2000–0600 hours, shown by the black horizontal bar), and below average (h<) during the 0800–1000 and 1200–1400 hour bins. The dashed line represents average feed intake. Data points represent least square means for each 2-hour bin (n = 10 mice), plus or minus standard errors of partial residuals (obtained by removing the estimated effects of sex and individual baseline feed intake). All are back-transformed following analysis of square-root-transformed data. Mouse body weight fluctuated with time of day over 24 hours pre-treatment. Weights increased at the start of the dark period (2000–0600 hours, shown by the black horizontal bar) and were significantly above average (h>) between 2100 and 0000 hours, and below average (h<) between 1100 and 1600 hours. The dashed line represents average body weight. Data points represent least square means for each hourly bin (n = 10 mice), plus or minus standard errors of partial residuals (obtained by removing the estimated effects of sex and individual baseline weight). Sexes are graphed together because their body weights did not differ significantly.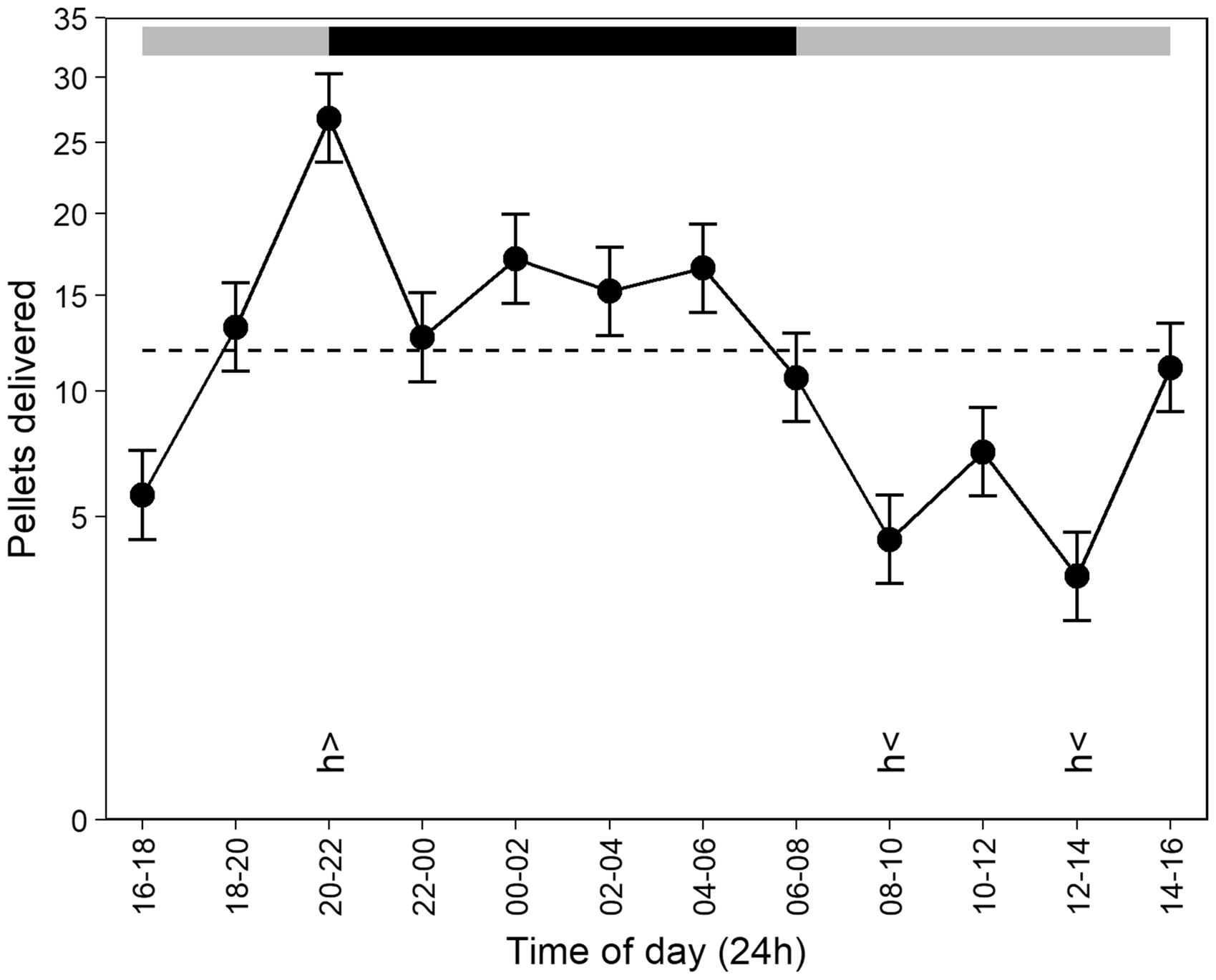
Feed intake declined precipitously after LPS injection, as shown by a significant three-way interaction between treatment (control vs treatment), experimental period (pre- vs post-injection) and time of day (F11,66 = 2.98, p = 0.0029; Figure 4). Treatment mice largely stopped feeding over 24 hours following LPS challenge. During the 10 hours of the dark phase following the injection, control mice consumed 70 to 138 pellets (1.75–3.45 g) while treatment mice consumed 0 to 3 (0–0.075 g). Because only two of five treatment mice visited the feeder after injection, we could not statistically analyse the effects of LPS on body weight.
Feed intake patterns differ between control and treatment mice following LPS injection. Treatment mice fed significantly less than controls (asterisk) 2–4 hours after injection (1800–2000 hour bin) and in all 11 remaining 2-hour post-injection bins. Control mice maintained normal circadian feeding patterns following saline injection (light and dark periods indicated by grey and black bars, respectively). Data points represent least square means for each 2-hour bin (n = 5 mice per treatment), plus or minus standard errors. All are back-transformed following analysis of square-root-transformed data.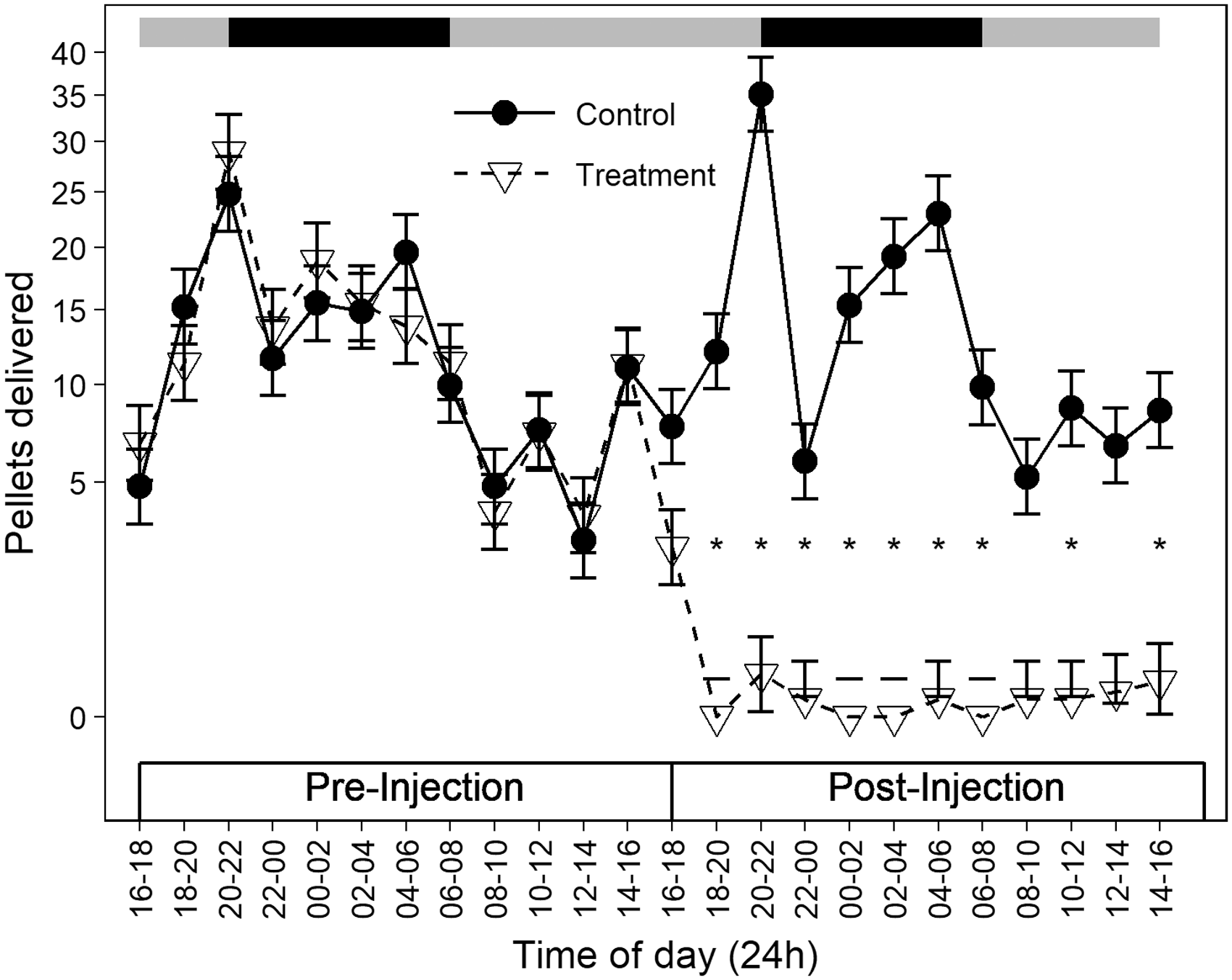
Human observers failed to detect a difference between treatment and control mice. Whereas IBS was affected by a significant interaction between treatment, experimental period and time of day (F1,8 = 7.58, p = 0.0249), post hoc tests were not significant: at no time did the scores of LPS-injected mice differ significantly from those of controls or from their own pre-injection scores. We could not statistically analyse the effects on BCS because only two mice, both LPS treated, showed any change on the day after injection: a one-point increase and a one-point decrease.
Discussion
As hypothesized, the automated health-monitoring apparatus successfully detected experimentally induced illness in laboratory mice. Decreased feeding became apparent 2 to 4 hours after LPS challenge, persisted throughout the 24-hour period, and allowed perfect discrimination between treatment and control mice. In contrast, human observers failed to detect any significant difference between sick mice and controls. Automated round-the-clock home cage behavioural monitoring thus allows sensitive and rapid detection of illness: flagging subtle cues that human observers may miss, and doing so well before they are scheduled to conduct daily health checks. Configuring software to alert caretakers when signs of illness are detected would allow them to respond promptly by increasing observation, administering treatment or euthanizing sick animals.
The apparatus enables minimally invasive health monitoring for any number of group-living mice. Sub-cutaneous RFID transponder implantation is itself invasive, but it is a one-time procedure from which mice typically recover readily and resume normal activity. The tag’s small size and location in the nape of the neck make it unlikely to interfere with behaviour or physiology. Monitoring is otherwise completely “hands-off.” The absence of a human observer and lack of handling may both reduce stress12,13 and possibly decrease the likelihood that mice will suppress behaviours that indicate pain or illness.3,9 Note that automated behavioural monitoring is intended only as a complement to brief daily visual checks by caretakers, as it would be unlikely to catch certain problems, such as cages flooded by leaky water bottles.
Because several animals completely stopped visiting the data collection tube (feeder) after LPS challenge, we could not test whether the apparatus can detect illness via other measured outcomes. With a less severe challenge, we may have observed progressive declines in body weight 29 or changes in physical appearance (e.g. Table 1). Photographs of the dorsal and ventral surfaces recorded at each feeder visit could also aid in detecting and retrospectively examining progression of some of the most common mouse morbidities, such as fight wounds or ulcerative dermatitis, both of which are visually salient. 1 Changes in behaviour and physical appearance could potentially aid in other applications, such as detecting pregnancy, parturition, and even estrous in breeding mice. 36
Our apparatus adds to many existing efforts in automated tracking of mouse home cage behaviour.8,20–23 RFID reader arrays have been used to track location and activity of individual mice in large enriched enclosures.37–41 Video and machine vision have been used to track location and quantify social and other behaviour in mice.39,42–46 Feeding bouts have been tracked using RFID-equipped feeders,47,48 feeders with infrared presence detectors, 49 and feeders with built-in scales that additionally measure feed consumption.50,51 Fluid intake can be regulated and monitored using RFID-based systems,26,41,52,53 including one developed in our lab (publication forthcoming). Scales at drinking stations or other locations can additionally monitor body weight.41,54 Intraperitoneal telemetry implants can track locomotor activity and body temperature,55–57 as can much smaller sub-cutaneous implants (similar in size to the RFID transponders we used).58,59 Respiratory rate and various behaviours have also been tracked non-invasively using electric field sensors. 60 Many of these approaches could readily be combined or integrated with our apparatus to extend monitoring capabilities.
Feed intake and body weight showed circadian fluctuations, rising markedly during the dark phase. This is consistent with previous findings of nocturnal (or crepuscular) circadian rhythms in laboratory mouse feeding,48–51 drinking,49,51–53 body temperature,56,57 and general activity.22,49,51,56,61 Mice nonetheless obtained nearly 40% of their feed during the light period. Feeding patterns are sensitive to the energetic or macronutrient content of feed 50 and mice in semi-naturalistic enclosures may feed without a clear circadian pattern or even primarily diurnally at certain times of year. 47 Diurnal feeding did not seem to be a result of competitive displacement, as mice who shared a cage showed similar feeding patterns – it is unknown if mice in larger groups would attempt to monopolize the collection tube. 62 Circadian rhythms in body weight, meanwhile, have not to our knowledge been examined as closely as we have here. Weights declined by nearly 1 g on average from their peak early in the dark period to late afternoon. Frequent automated weighing thus provides a much more accurate picture than even the most diligent daily weighing schedule. In an analogous case, automated monitoring via intraperitoneal implants revealed circadian rhythms in mouse core body temperature that would have remained hidden with manual measurement, particularly as handling affects body temperature.56,63
This experiment provides proof-of-concept that automated monitoring of health and sickness behaviour in laboratory mice is more sensitive than assessment by humans. Deployment for large-scale routine health surveillance will require adjustments to minimize cost54,64 and space requirements, and/or large-scale modifications to conventional caging systems.38,39 Overcoming these hurdles could drastically improve laboratory mouse health, ending the sad situation in which most sick mice “fall through the cracks” and die before caretakers realize there is a problem. 1
Footnotes
Acknowledgments
The monitoring apparatus is described in patent US9497928 B2 granted to JPG and colleagues.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This publication was made possible in part by grant number R21NS088841 from the National Institute of Neurological Disorders and Stroke (NINDS) at the National Institutes of Health. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the NINDS.
