Abstract
Endotracheal intubation in mice is both a common and important technique. However, it is a difficult procedure because of the small orotracheal size and the success rate is variable. There have been many reports of refined techniques that facilitate intubation but only a few reports have proposed how to verify the proper placement of the endotracheal tube. We describe a novel, safe and reliable method to confirm endotracheal intubation in mice using an extension tube for intravenous infusion. One drop of water was instilled in the extension tube and connected to the end of the catheter used as an endotracheal tube. When the catheter was inserted correctly into the trachea, the water in the extension tube oscillated in synchrony with the movement of the mouse's thorax, indicating correct placement of the catheter. This method was simple, reliable and use materials that are routinely available. This method is helpful for experimental mouse models that require airway access.
Keywords
The trachea is the portal of entry to the lower airways, and the lung is the target of many infective agents and toxins. There is an increasing need for evaluating the efficacy of potential therapeutic agents delivered directly into the airway, and the majority of animal research is based on mouse models. Endotracheal intubation in mice is an essential technique for introducing drugs, toxins or infectious agents into the lower respiratory tract. However, the small anatomical size and the narrow glottic opening make the procedure difficult.
There are two commonly used methods to deliver the agents into the lung: tracheostomy and intranasal instillation. Although tracheostomy is a reliable method for airway control, it has many serious defects. Incision into the cervical skin, subcutaneous tissue and trachea leads to a high complication rate. Longer and deeper anaesthesia is also required. Nasal instillation is a simple method and has been widely employed, but the pulmonary delivery cannot be quantified.
Endotracheal intubation is an alternative method of accessing the lung without direct, surgical exposure of the trachea by cervical incision. Numerous procedures for intubation that vary in technique and equipment have been described. Several reports suggested direct visualization of the epiglottis and vocal cords during intubation by using a fibreoptic light guide, 1 a small bore arthroscope, 2 or a laryngoscopic blade. 3 Illumination of the larynx by an extraluminal light source (transillumination) has also been reported. 4
The use of these visualization techniques has made endotracheal intubation a major, indispensable tool in murine models of pulmonary disease. However, confirmation of the proper placement of endotracheal tubes has not been thoroughly discussed, and it still remains a major consideration. The tube may be inserted accidentally into the oesophagus at any time, and it is not always easy to distinguish between endotracheal and oesophageal intubation in mice. Confirmation of the correct insertion plays a key role in endotracheal intubation. The aim of our study was to develop a simple, safe and reliable method to confirm the correct placement of an endotracheal tube in mice.
We initially used 24 mice of four common strains (6 BALB/c, 6 ICR and 6 C57BL/6J, Charles River Japan, Yokohama, Japan; and 6 ddY, Tokyo Laboratory Animals Science, Tokyo, Japan) at 3–8 weeks of age. All mice used in our studies were cared for in accordance with the rules and regulations set out by the Prime Minister's Office of Japan. Animal protocols were approved by the Special Committee on Animal Welfare in our institution. Mice were housed in an air-conditioned room with a 12:12 h light–dark cycle and were fed with standard mouse chow and tap water ad libitum.
Mice were anaesthetized by intraperitoneal administration of a mixture of ketamine (65 mg/kg) and xylazine (13 mg/kg). The depth of anaesthesia was evaluated by the absence of both swallowing and righting reflexes. Once the mouse was unconscious, a standard, disposable intravenous catheter (BD Insyte-W, 22G × 1 in. or 24G × 0.75 in., Becton Dickinson Infusion Therapy Systems Inc, Sandy, UT, USA) was inserted into the trachea. Visualization of the trachea below the vocal cords was provided using a modified transillumination procedure. 4 Briefly, the mouse was suspended from a plastic box (85 mm × 85 mm × 65 mm) by the two front upper teeth with a suture thread. A 150 W halogen light source with flexible fibreoptic arms (MeCan Imaging Inc, Saitama, Japan) allowed transillumination of the trachea just below the vocal cords. The positions of the fibreoptic arms were adjusted in each mouse to provide the best visualization of the trachea.
After intubation with the catheter, we verified its proper placement. A drop of sterilized water (50–100 μL) was instilled into a standard extension tube that was originally designed for intravenous infusion (Figure 1) (TOP Extension Tube X2-50, TOP Corp, Tokyo, Japan). This tube was connected to the end of the intubated catheter cautiously so as not to cause extubation. When the catheter was properly inserted into the trachea, the water column in the tube moved simultaneously with the breathing pattern, and the column oscillated in the same phase as the movement of the mouse's thorax (Figure 2).
An extension tube with a water column. A droplet of sterilized water has been instilled into the tube. The water (arrows) is coloured with Alcian blue to improve its visualization
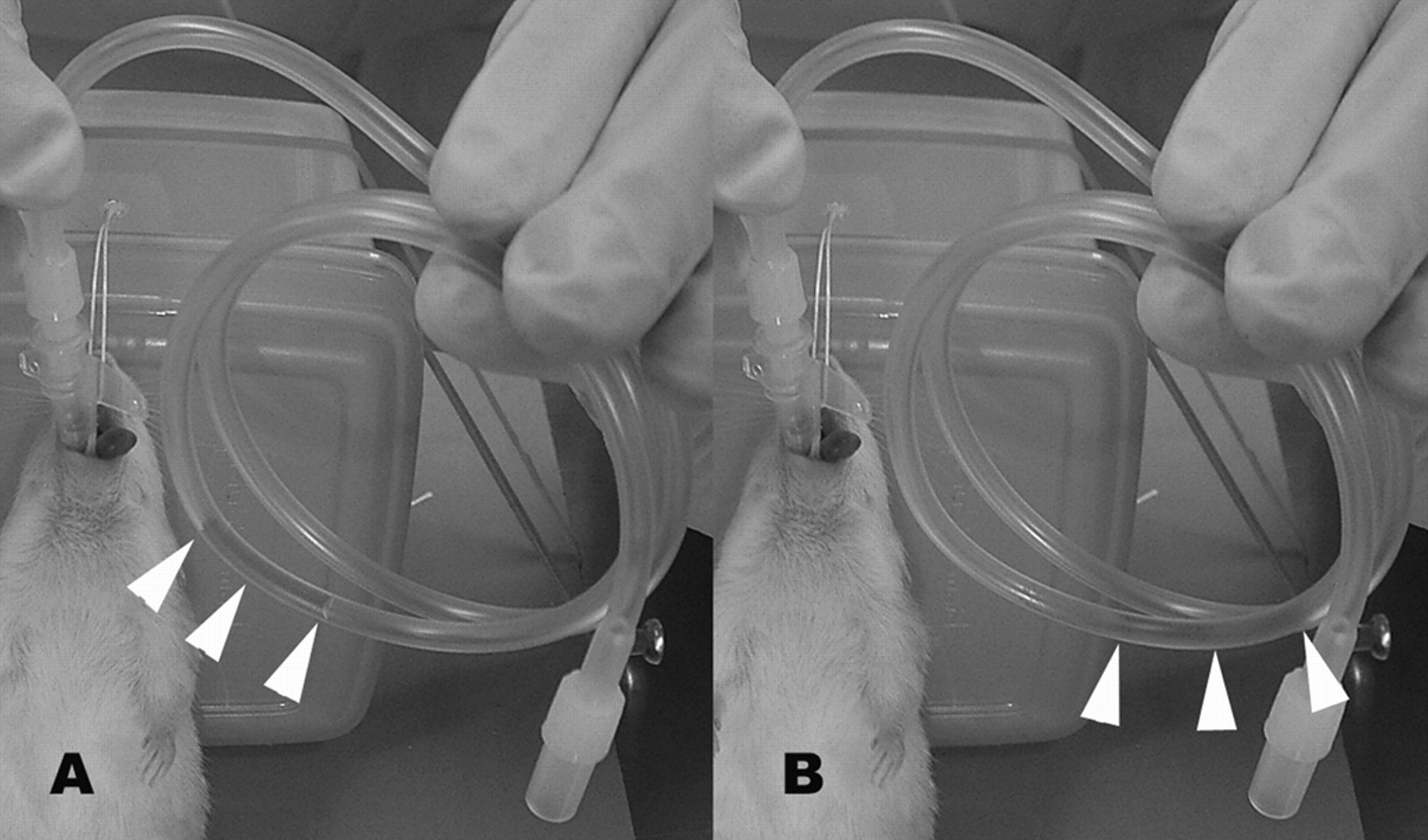
An extension tube connected to a catheter properly inserted into the trachea of a mouse. The water (white arrowheads) in the tube easily moves to and fro in synchrony with expiration (A) and inspiration (B). The water is coloured as in Figure 1
Even a slight airflow moves the water column, confirming the very high sensitivity of this system. When the catheter was accidentally intubated into the oesophagus, the water in the tube remained still. This method allowed us easy and secure confirmation of the proper endotracheal intubation. The mean length of time required for the verification was less than 10 s, but to test the tolerability of this time period, the connection of the extension tube to the catheter was continued for up to one minute in some mice. During these procedures, none of the mice showed any sign of respiratory distress. For ethical reasons, longer examinations were not carried out because of the risk of hypercapnia in the mice.
To determine the after-effects of endotracheal intubation, eight mice were observed for the following seven days for signs of respiratory impairment. An additional eight mice were intubated once and then the procedure was repeated after four or five days, and the mice were observed for the following seven days. A third group of eight mice was intubated for three consecutive days. No major complications resulted from this method, and the mice did not show any signs of respiratory impairment. Postmortem examination revealed no evidence of pharyngeal or laryngeal trauma.
Subsequently, in order to confirm correct intubation of the trachea, 30 μL of Gentian violet solution (Sigma, St Louis, MO, USA) was introduced into the catheter in each ICR mouse (n = 5). Immediately after extubation, these mice were killed humanely and their lungs were examined. In every mouse, the tissue of both lungs was diffusely stained violet with the dye, indicating the high reliability of this confirmation system (data not shown).
Here we describe a new, simple and reliable procedure for verification of the proper placement of an endotracheal tube. To date, few methods have been reported to verify the correct placement of an endotracheal tube, but each of these methods has some disadvantages. Balloy et al. 5 used a mirror placed in front of the external end of the endotracheal tube, checking the formation of mist on the mirror when the mouse exhaled. In this method, the formed mist is occasionally vague, making the confirmation unreliable. Rivera et al. 1 checked intubation in two ways, the first was to close the end of the tube by fingertip, and then to check if the mouse breathed abnormally. The second was to place a plastic transfer pipette onto the end of the tube and give squeezes to the bulb to deliver air into the lung. When the tube was placed correctly, the mouse's thorax would rise and fall with the air being pushed into the lung. However, both of these methods could lead to adverse effects on the animals. Vergari et al. 2 evaluated the success of the intubation by connection to a rodent pulmonary ventilator and visualizing the mouse's ventilation, a procedure that requires significant investments in equipment.
The method demonstrated here is simple, reliable and is easily mastered after brief instruction and demonstration. The devices are inexpensive and easily available. We performed intubation and extubation in mice without complication in all cases. Moreover, we were able to re-intubate the mice after several days 6 or even on the next day without difficulty. We believe that this method will be very helpful for longitudinal studies that require repeated intratracheal inoculation or drug administration.
