Abstract
Endotracheal intubation in rats is challenging due to difficulties visualizing the epiglottis and vocal cords. No visualization of these structures results in repeated intubation attempts which can cause trauma to the oral cavity and/or oesophagus, and death of the animal due to respiratory failure. Here, we describe a simple blind oral tracheal intubation technique in the rat that decreases the frequency of repeated intubations using an intubation device that comprises a 16 G intravenous catheter and a modified 18 G epidural needle, and a rodent ventilator. The epidural needle is bent in such a way that it curves in conformity with the rat's oral airway in order to direct the catheter into the larynx, and the rodent ventilator is used to verify its correct placement. The first attempt success rate of endotracheal intubation using the blind oral tracheal intubation technique with a rodent ventilator was greater than the first attempt success rate using the blind oral tracheal intubation technique without using a rodent ventilator. Although this method is a simple modification of a previously described method of blind oral endotracheal intubation, our method is easy to learn, inexpensive and does not require specialized equipment.
Keywords
Provision of a patent airway is a refinement for many experimental procedures in laboratory animals, and endotracheal intubation is usually necessary for artificial pulmonary ventilation during open-chest thoracic procedures in rats. Many techniques for endotracheal intubation in rats have been described in the literature, and include blind oral tracheal intubation 1 or direct visualization or transillumination of the trachea2-5 with or without the use of an intubation wedge, 6 an otoscope on an inclined metal plate 7 or a straight-tip guide wire for a vein catheter and a small infant laryngoscope. 8 Although commercially available equipment for rodent intubation is available, such as the rat intubation pack (Hallowell Engineering and Manufacturing Corporation, Pittsfield, MA, USA) and the BioLITE Intubation Illumination System (Bioseb, Chaville, France), endotracheal intubation in the rat is technically difficult because of the small size of the animal and its oral cavity. Rivard et al. 9 reported that repeated attempts on the same animal can increase the frequency of complications, such as laryngospasm, mucus plugging, laryngo-epiglottic oedema and glottic and local tissue trauma. The authors also noted that, if the rat was not successfully intubated after the second attempt, the animal would regain glottic reflexes, which made subsequent intubation attempts difficult. In this report, we describe a modification of the blind oral tracheal intubation technique of Stark et al. 1 in which a hollow intubation stylette is used as an aid to correctly place the endotracheal tube, and a rodent ventilator (Model 683; Harvard Apparatus, Holliston, MA, USA) is used to verify correct placement of the tube.
This method was evaluated in 120 male Sprague-Dawley rats (150–350 g) during the course of studies on myocardial infarction and bleomycin-induced lung injury. The rats were maintained in the Animal Care Facility in our institute, and all experimental procedures were approved by the Institutional Animal Care Committee. For anaesthesia, each rat was anaesthetized by placing it in a 5 L anaesthesia chamber with 5% enflurane-O2 (Alyrane, Baxter, Guayama, Puerto Rico, USA) (1 L/min) until the righting and palpebral reflexes were lost. Ketamine (50–150 mg/kg) (Huons, Korea) was injected intraperitoneally in order to supplement the inhalation anaesthesia. For intubation, the endotracheal tube was a 45 mm long 16 G intravenous catheter (01.77 mm; BD Angiocath Plus, Seoul, Korea), and an 18 G epidural needle (BD Perisafe, Basel, Switzerland), which was used as an intubation stylette in order to direct the catheter into the larynx (Figure 1). The epidural needle was bent in such a way that it curved in conformity with the rat's oral airway in order to direct the catheter into the larynx, and the needle tip was blunted and sandpapered smooth (Figure 1c).
Equipment and assembly required for blind oral tracheal intubation. The equipment comprises an 18 G epidural needle (A), and a 45 mm long 16 G intravenous catheter (B). The epidural needle was bent in such a way that it curves in conformity with the rat's oral airway, and the tip of the epidural needle is blunted and sandpapered smooth (C). The intubation device, which consisted of a 16 G intravenous catheter and the modified epidural needle (D), is connected to the Y connector (E) of the rodent ventilator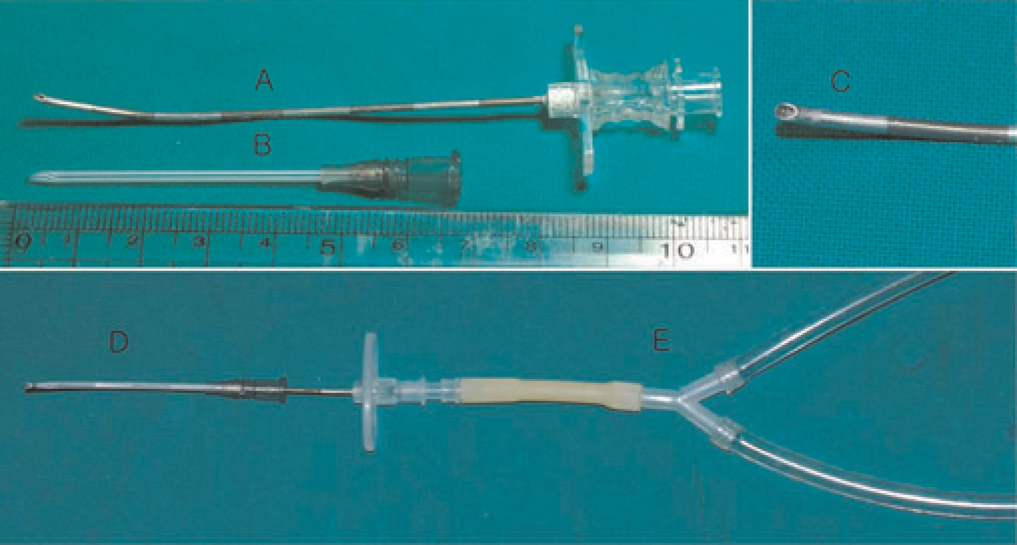
The 120 rats were randomly assigned to one of two groups for blind oral endotracheal intubation: group V in which the intubation device (i.e. the endotracheal tube and intubation stylette) was placed in the trachea while it was connected to a rodent ventilator (new method group), and group C in which the intubation device was placed while it was not connected to the ventilator (control group). The settings of the rodent ventilator were 10 mL/kg (tidal volume) and 60 cycles/min. All intubations were carried out by one person. Before intubation, the operator checked that there were no air leaks between the intubation device and ventilator when the ventilator was not used to ventilate the rat. For endotracheal intubation, the mouth of the anaesthetized animal was opened, and the tongue was then pulled cranially and ventrally using the thumb and index finger or a mosquito clamp until resistance was felt. The middle finger of the other hand was placed on the skin overlying the larynx in order to locate the position of the intubation device and for feeling its entry into the larynx or trachea. The intubation device was then introduced into the mouth with the tip pointing ventrally. The device was slowly and carefully advanced along the sagittal plane following the curve of the oral cavity towards the vault of the hard palate. A slight resistance was typically encountered as the intubation device passed across the vocal cords. For group C animals, the intubation device was then advanced approximately 2–3 mm before advancing the intravenous catheter off the stylette and into the trachea. The intravenous catheter was then connected to the ventilator. The operator judged the endotracheal intubation to be successful when movements of the chest wall coincided with the pre-set cycling rate of the ventilator or by pulmonary auscultation. If endotracheal intubation failed, the operator removed the intubation device, and then attempted to re-intubate the animal using the identical method. For group V animals, the intubation device was advanced approximately 2–3 mm, and the operator then observed movements of the chest and abdominal walls for 5–10 s without further advancement. Due to the fact that the epidural needle was connected to the ventilator and the ejection phase of the ventilator coincided with the inspiratory phase of the rat's respiratory cycle, synchronous movements of the chest wall was observed when the tip of the intubation device had been placed beyond the vocal cords. When such movements were observed, the chest was auscultated using a paediatric stethoscope, and the intravenous catheter was then advanced off the stylette into the trachea. If the tip of the intubation device had been placed in the oesophagus, synchronous movements of the abdominal wall and abdominal distension would be observed. The chi-squared test was used to compare the number of animals in each group that were successfully intubated at the first attempt and underwent repeated intubation attempts.
The number of successful endotracheal intubations at the first attempt in group V rats was more than twice as many as that in group C rats (Table 1; P < 0.0001), and more repeated intubation attempts were necessary in group C rats than group V rats (Table 1; P < 0.0001). Two group C rats died after repeated intubation. One rat died after five intubation attempts, and the second rat died after six intubation attempts, and both rats died within 10 min after the last intubation attempt. Although we found oesophageal injuries in the two dead rats at necropsy, we presume that the cause of death in each rat was hypoxia due to ventilatory failure that resulted from unsuccessful endotracheal intubations.
Frequency chart of repeat attempts for successful endotracheal intubation

The numbers, 0, 1, 2, 3, 4 and 5 refer to the number of repeated intubation attempts for successful endotracheal intubation. The letters ‘C and ‘V refer to the two experimental groups (see text for definitions)
In the original description of their method of blind endotracheal intubation, Stark and others 1 recommended that the catheter be fully advanced after it enters the larynx. This ‘blind’ step is risky when the oesophagus has been intubated because the intubation device may puncture the oesophageal wall if it is advanced too far. In our modified method, the operator can observe chest wall expansion if the tip of the needle has been placed just beyond the vocal cords or abdominal distension if the tip of the needle has been placed in the upper part of the oesophagus. In addition, oesophageal damage will not occur by not fully advancing the needle if the needle has been placed in the oesophagus. Delaying full advancement of the needle in order to observe synchronous chest wall movements and ventilator operation can also prevent a potential cause of death if the needle has been placed in the oesophagus. In addition to puncturing the oesphageal wall following full advancement of the needle, it is possible that ventilation, while the tip of the catheter is presumably in the mediastinum, can result in a pneumomediastinum and lung collapse. Recently, Watanabe et al. 10 described a simple method for verifying successful endotracheal intubation in mice. In their method, one drop of water was instilled in the extension tube which was connected to the end of the catheter that was used as an endotracheal tube. If the catheter is successfully placed into the trachea, the water in the extension tube will oscillate in synchrony with the movement of the mouse's thorax. Although our method is simple and reliable, the rat should be anaesthetized for endotracheal intubation to such a depth that the animal is not spontaneously breathing and stimulation of the vocal cords will not result in reflex responses and/or epiglottic and tracheal spasms. If the depth of anaesthesia is not adequate to cause suppression of these reflexes and the rat is spontaneously breathing, the procedure becomes more difficult using our method.
In conclusion, we found that our facilitated blind intubation technique increases the success rate of endotracheal intubation at the first attempt. Although this method is a simple modification of a previously described method of blind oral endotracheal intubation, our method is easy to learn, inexpensive and does not require specialized equipment.
