Abstract
Catheterization of the carotid artery and the jugular vein is one of the most commonly applied techniques used to gain intravascular access in pharmacology studies on rodents. We catheterized 10 rats by conventional clean techniques, 10 rats by aseptic techniques and 10 rats by conventional clean techniques using a heparin-coated catheter rather than an ordinary non-coated polyvinyl chloride catheter. In all groups, approximately 80% of the rats developed kidney infection and 10-30% of the rats were septicaemic. Clinical chemistry did not indicate severe kidney damage, but serum haptoglobin and body temperature rises indicated an inflammatory response in rats independent of the surgical method. Heparin coating did not seem to improve the usability of the catheter. It is concluded that this commonly used method for catheterization has an impact on animals that may very well render them unsuitable for the purpose, e.g. pharmacological research, and therefore an alternative method would be preferable.
Catheterization of the carotid artery and the jugular vein is one of the most commonly applied techniques used to get intravascular access in pharmacology studies on rodents. Various versions of the technique described by Wixson et al. 1 are commonly used. Although aseptic surgery is recommended by experts in the field,2,3 best practice in aseptic techniques is not necessarily followed in all facilities as there is no universal acceptance that adverse effects are a direct consequence and as infection is not lethal in rats. The catheters being used are generally simple, such as uncoated Tygon® polyvinyl chloride (PVC) catheters. However, this imposes a risk that bacteria and/or emboli from an infectious nidus at the site of the catheter tip may induce septicaemia or be carried with the blood to form an embolus in the kidneys and eventually develop a secondary nidus of infection. This may lead to physiological and immunological changes and thereby uncontrolled variation 2 and alteration of parameters.4–8 With regard to animal welfare, survival alone is not a satisfactory criterion as significant physiological and behavioural changes such as less open field activity, elevated plasma fibrinogen, altered serum glucose levels and elevated white blood cell counts are observed even in the absence of obvious clinical signs after non-aseptic surgery. 9 Furthermore, the studies may be invalidated by thrombotic complications in catheterized veins due to catheter sepsis. 10
Foley et al. 11 found Carmeda BioActive Surface (CBAS®) heparin-coated catheters (Instech Solomon, Plymouth, PA, USA) after aseptic insertion into the jugular vein to be efficient in a closed vascular access port. Other techniques to prevent blood clotting and catheter obstruction include continuous infusion of small quantities of saline or using a viscous lock solution of heparin-polyvinylpyrrolidone (PVP),12,13 which has also proven to reduce bacterial colonization. 14 However, although regarded as biocompatible, PVP has been shown to induce inflammation,15–18 and it may just relocate the bacteria to the pericatheter macrophages. 19 Using dexamethasone in the lock solution, smaller diameter, softer materials and deep insertion into a vessel with high blood flow may increase catheter patency. 20 Chlorhexidine, silver, copper and antibiotic agents have been reported to resist bacterial colonization. 21–27 Silicone catheters are more easily infected by bacteria than catheters made from polyurethane (PU), teflon or PVC.28,29 Silicone and PU are less thrombogenic than PVC, 30 and bacteria adhere better to PVC surfaces than to teflon. 31
From awelfare point of view there seem to be good reasons to avoid catheter infections and from a scientific point of view the use of infected animals can be severely questioned. The aim of this study was to evaluate (a) whether vascular catheterization of rats by commonly applied techniques induces infection and inflammation that can invalidate research; and (b) the extentwhich this is related to surgical hygiene oranticoagulant coating of the catheters.
Materials and Methods
Animals and treatments
The experiment was licensed under the Danish Animal Experimentation Act according to the EU directive 86/609. Male Crl:SD rats (Charles River, Sulzfeld, Germany) weighing 200-300 g provided with ad libitum Altromin 1324 rodent chow (Altromin mbH, Lage, Germany), bottled municipal tap water, and aspen wood bricks (Tapvei, Kortteinen, Finland) and paper towels as enrichment, were pair-housed in Makrolon type IV cages (SCANBUR Ltd, Karlslunde, Denmark) on aspen bedding (Tapvei), and after surgery singly-housed in rat microdialysis swivel systems (Instech Solomon, Plymouth Meeting, PA, USA) at a room temperature of 19-24°C, a relative humidity of 30-70% and lights on 06:00-18:00. After four days of acclimatization they were randomly assigned to one of three groups of 10 animals. The day prior to surgery they were moved to the operating room, baseline blood samples were taken by tail vein puncture and an IPTT 30 thermochip (Plexx, Elst, The Netherlands) was implanted subcutaneously in the right lower back area under anaesthesia using fentanyl/fluanison (VetaPharma, Leeds, UK) and midazolam (Roche, Basel, Switzerland) and isoflurane (Forene, Abott, Copenhagen, Denmark). In total, 10 mg/kg of enrofloxacin (Baytril, Bayer Health Care, Kgs. Lyngby, Denmark), 5 mg/kg of carprofene (Rimadyl, Orion Pharma, Espoo, Finland) and 0.05 mg/kg buprenorphine (Temgesic, Schering-Plough, Brussels, Belgium) were given either by the subcutaneous or intramuscular route on the day of surgery and enrofloxacin and carprofen were continued for the first three days after surgery. During the postsurgical recovery period the animals were kept warm using a heating lamp.
Experimental design
All rats were subjected to cannulation of the left carotid artery under anaesthesia. Instruments were washed, sonicated for 10 min in a 2% deconex® (Borer Chemie AG, Zuchwil, Switzerland) solution, washed in demineralized water and dried using normal tissue paper. An approximately 4 × 4 cm2 field on the dorsum centred between the scapulae and an approximately 3 × 3 cm2 field between the sternal manubrium and the mandible was shaved, disinfected with iodine (Iobac®, Unitron Ltd, Tørring, Denmark) and hair was removed with a hand-held vacuum cleaner. In total, 500 IU/mL of heparin mixed with isotonic saline was used as the catheter lock solution. Aheatedmat with a thermometer was placed under the animal, and temperature was maintained at 38°C. In all three groups a 1-2 cm transverse median cutaneous incision was made with scissors approximately 1 cm cranially to the sternal manubrium. The subcutaneous tissues were bluntly dissected, and the artery was located and gently freed from the surrounding tissue and nerve and elevated and kept moist with isotonic saline. Two silk ligatures (Ethicon Sutupak 5-0) were placed around the artery, one cranially and one caudally. The cranial ligature was tightened and the caudal left loose. With iris scissors an incision of approximately 1/3 through the artery was made at a 45° angle between the two ligatures and bleeding prevented by elevating forceps. The catheter flushed with lock solution was inserted into the carotid artery and advanced towards the heart for a distance of 24 mm. The caudal ligature was tied without compromising catheter patency. The ends of the cranial ligature were then tied to a paper anchor on the catheter for fixation. Different approaches (see below) were used for dorsal fixation of the catheter. After fixation, a tether (Dacron button, DiLab, Lund, Sweden) was placed subcutaneously under the dorsal incision. Woundswere closed by simple interrupted sutures using Vicryl 4-0 (Ethicon, Somerville, NJ, USA). The catheterwas then put through a protective spring, which was fastened to the tether. Finally, the spring was secured to the swivel system. The catheter was checked for patency and was sealed with a piece of nylon thread inserted into the end of the catheter.
The conventional group (n = 10) was cannulated using a traditional non-aseptic procedure and uncoated PVC Tygon® catheter (outer diameter [OD] of 0.9 mm, inner diameter [ID] of 0.6 mm; DiLab) gamma-irradiated at a minimum dose of 25 kGy (Codan Steritex, Espergærde, Denmark), and subsequently stored in zip-lock bags. Between animals, the tips of the instruments were dipped in iodine (Iobac®, Unitron A/S). The gloves used by the surgeon were non-sterile latex gloves that were not put on until surgery began, and the instruments were manipulated with bare hands. The surgeon did not use a mask, cap or gown. The table was not disinfected prior to use and the lamp and microscope were not disinfected. Injection fluids were withdrawn from capped bottles. Surgical drapes were not used, and the instruments were placed on nonsterile tissue paper. Haemostasis was maintained during surgery by electrocauterization and compression. After having placed the catheter from the ventral side the animal was placed in ventral recumbency and the shaved field was disinfected using the iodine solution. An approximately 3 cm transverse, median cut was made above the scapulae using scissors, and the skin was loosened by blunt dissection 1-2 cm cranially and caudally to the incision. A needle holder was then tunnelled subcutaneously from the dorsal incision to the ventral incision to grab the catheter and brought through the tunnel to the dorsal incision. At the end of the procedure the tether was dipped in iodine and with the catheter led through, it was placed subcutaneously at the incision and sutured to the underlying muscle layer with two separate sutures (Vicryl 4-0; Ethicon) as far apart from each other as possible.
The aseptic group (n = 10) was cannulated using an aseptic procedure. The Tygon® PVC catheters (Saint-Gobain, Charny, France) and the instruments in their box were steam autoclaved (steam sterilizer type 25T, Thermo Electron LED GmbH, Langenselbold, Germany) at 121°C for 20 min. Instruments and materials needed for the operation were unpacked aseptically and placed on an aseptic area covered by a sterile drape, which was only touched at the very edge when placed. The table as well as the lamp and microscope were disinfected with ethanol, and the animal was anaesthetized and shaved at another table. Additionally, the left side of the neck was shaved to connect the two fields in a 1 cm broad field. Injectables were withdrawn from capped bottles. The bottle containing the heparin used for the lock solution was disinfected with ethanol. Haemostasis was maintained during surgery by compression using sterile gauze. The surgeon wore a cap and mask, scrubbed using soap (Kimcare General, Kimberly-Clark Professional Europe, Copenhagen, Denmark) and put on sterile gloves (Ansell, Brussels, Belgium). The surgeon prepared the lock solution and a syringe with isotonic saline to keep the artery moist when it was elevated for manipulation. A sterile drape with a hole large enough to perform the procedure (Buster® cover, 60 × 90 cm, Kruuse, Langeskov, Denmark) was placed over the animal. After placing the catheter as described above, the hole in the drape was extended laterally to the left side and the catheter flushed with the lock solution was tied to a blunt trochar, which was then tunnelled subcutaneously from the ventral incision to the dorsum of the animal, and an incision was made with a scalpel blade over the tip of the trochar, which was then pulled through the hole along with the catheter. The ventral incision was then sutured. The drape was removed and sterile gauze was used to manipulate the limbs and placed the animal in ventral recumbency. A hole was cut in the centre of a new drape, and the drape was placed over the animal with the hole centred on where the catheter exited, and the catheter was pulled through this hole and onto the drape. A cutaneous incision was made with scissors that extended from the hole made at the catheter exit and across the midline at the scapulae. The skin was loosened by blunt dissection 1-2 cm cranially and caudally to the incision. The tether was presterilized by the manufacturer.
The CBAS group (n = 10) was cannulated with a CBAS® C-30 heparin-coated polyurthane catheter (3F no luer, round tip, 60 cm 2 movable beads; Instech Solomon) using the same procedure as the conventional group.
Clinical examination
Baseline peripheral body temperature readings were performed in the afternoon (16:00-17:00) and the following morning (08:00-09:00) after implantation of the chip. After surgery the animals had their peripheral body temperature read each afternoon between 16:00 and 17:00 and each morning between 08:00 and 09:00 by a DAS-5007 Pocket Scanner (Plexx). Rats were weighed prior to surgery, and again after necropsy. Approximately 300 μL (15 drops) blood was sampled from each animal by tail vein puncture each morning for six days after surgery. The final sample on day 7 postsurgery was obtained at necropsy (see above). The sample was centrifuged at 3600 rpm for 10 min, and the serum was frozen at -20°C until further use. The concentration of haptoglobin in serum was quantified using a commercial rat haptoglobin ELISA test kit (E-25HPT-11, Dunn Labortechnik GmbH, Asbach, Germany). Absorbance was measured using a Multiscan EX microplate photometer (Thermo Electron Corporation, Waltham, MA, USA) and calculated using Ascent software for multiscan v2.6 (Thermo Electron Corporation).
Euthanasia and necropsy
Prior to euthanasia and necropsy, catheters were flushed with saline to check patency. All animals were euthanized under anaesthesia as described above. Euthanasiawas accomplished by exsanguination performed by cardiac puncture in the open thoracic cavity. One millilitre of blood was centrifuged at 3600 rpm for 10 min and sera were frozen at -20°C until all samples were to be analysed. The parameters alanine aminotransferase (ALAT), aspartate aminotransferase (ASAT), alkaline phosphatase (ALP), albumin, total protein, calcium, creatinine, glucose, urea, phosphate, sodium, potassium and chloride in the serum samples were measured using a Cobas Mira Plus CC (Roche ABX Diagnostics, Montpellier, France). Approximately 20 drops of blood were smeared onto a blood agar plate using a sterile cotton swab. The kidney swabs were also smeared onto a blood agar plate. Plates were incubated aerobically at 37°C for 48 h and examined for growth. The sonicated samples obtained at necropsywere vortexed for 15 s and 0.1 mLof the samplewas surface plated onto blood agar using an angled Pasteur glass pipette. Serial dilutions and cultivations were made until the original plus four progressive 1:10 dilutions had been plated. After aerobic incubation at 37°C for 48 h the number of organisms was quantified. Two plates were plated with only the saline used to make the dilutions. The first plate was plated after three days, while the second was plated just before the end of the study. The last part of the blood samplewas used for the terminal haptoglobin quantification.
The abdomen was opened and after examination for gross pathological lesions the kidneys were split down the middle using a sterile scalpel, and a sterile swab was used to collect a sample from the pelvis, the surrounding medulla and the cortex of each kidney. The catheter tip was checked for correct placement at the aortic arch and the tip along with a piece from the section at the height of the scapula (approximately one cm of each) were removed aseptically, placed in 1 mL of sterile phosphate-buffered saline (PBS) and sonicated for 30 s at 42 kHz (Bransonic 3510 E-MT, Branson UL Transonics Corporation, Danbury, CT, USA) based on the method used by Sherertz et al. 32 After sonication, the catheter piece was immediately removed from the PBS and discarded. A glass bead sterilizer (Keller Glassbead sterilizer, Simon Keller AG, Burgdorf, Switzerland) operating at 250°C for 10 s was used to sterilize instruments that were used for the aseptic removal of the catheter pieces.
Statistics
Statistical analysis was performed using Minitab 15 for windows (Minitab Inc, State College, PA, USA). Qualitative fractions were compared by χ2 test or Fisher's exact test, while quantitative data were tested for normal distribution by Anderson-Darling test. Normally distributed data were compared overall by one-way analysis of variance and subsequently group-to-group by Student's t-test, while other quantitative data were compared similarly by Kruskall-Wallis test and Mann-Whitney U test. For comparison of paired measurements, the paired Student's t-test was applied for normally distributed data, while the Wilcoxon signed-rank test was applied for non-normally distributed data.
Results
There were no significant differences in the weight gain between the three groups of animals. All animals but one from the aseptic group having some problems with its drinking nipple gained weight during the study. In all three groups, the body temperature significantly rose in the afternoon postsurgery (P < 0.0019), while there were no significant differences compared with baseline the next morning. Hereafter there was a significant drop in temperature during day 1 and a further decrease from day 1 to day 3 after which the temperature remained stable until the final significant drop was observed between day 6 and day 7. This final drop was significantly smaller in the CBAS group compared with the conventional group (Table 1).
Morning (08:00–09:00 h) and afternoon (16:00–17:00 h) peripheral body temperature (ratio °C/°C) relative to the baseline value of rats cannulated in the common carotid artery
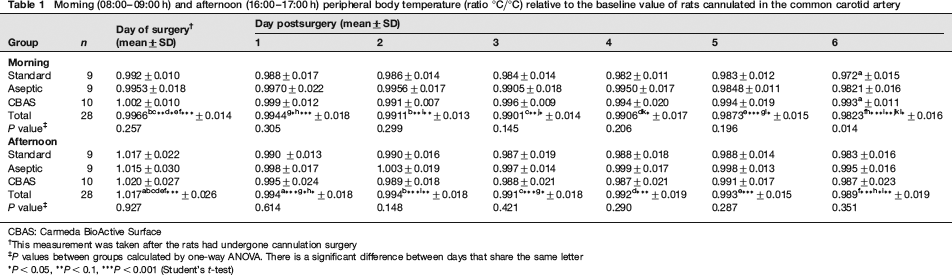
CBAS: Carmeda BioActive Surface
This measurement was taken after the rats had undergone cannulation surgery
P values between groups calculated by one-way ANOVA. There is a significant difference between days that share the same letter
P < 0.05,
P < 0.1,
P < 0.001 (Student's t-test)
On day 7 postsurgery two catheters, one from the aseptic group and one from the CBAS group, were unflushable. More catheters from the CBAS group were found to be hard to flush compared with the conventional and aseptic groups (Table 2).
Catheter patency on day 7 postcarotid artery cannulation surgery in three groups of rats

A catheter was easy to flush if only a light pressure on a 3 mL syringe was required to inject saline into the animal. It was hard to flush if a heavy pressure on a 3 mL syringe was required to inject saline into the animal, and unflushable if it was not possible to inject saline into the animal with a 3 mL syringe using full force
†Significantly different, P < 0.01 in Fisher's exact test (easy to flush versus unflushable + hard to flush [stacked data])
There was no significant difference between the groups with respect to the incidence of kidney gross lesions, i.e. focal necrosis observed as white spots in the cortex similar to infarcts (Figure 1), i.e. 4/10 rats in the standard group, 4/10 rats in the aseptic group and 1/10 in the CBAS group. Approximately 80% of the kidney swabs from all three groups were culture positive, and 10%, 22% and 30% of the blood cultures were also positive. The number of quantifiable colony forming units (CFUs) cultured from both the catheter tip and the subcutaneous catheter segment was not differing significantly between the groups (Table 3).
Culture-positive kidney swabs, blood cultures and quantitative cultures of the catheter tip and subcutaneous catheter segment from the three groups of rats cannulated in the carotid artery

CBAS: Carmeda BioActive Surface, CFU: colony-forming units
†Pearsons χ2 test
‡Kruskal-Wallis test

Kidney lesions in rats subjected to cannulation of the carotid artery. The image on the left shows the extent of the lesion on the surface of the kidney, while the image on the right shows the extension of the lesions into the cortex and medulla
There were no significant differences in serum haptoglobin concentrations between the three groups. In all groups, haptoglobin rose significantly to a median of 5.462 times that of the baseline value (P < 0.001 Wilcoxon signed-rank test) at day 1 postsurgery, then once again rose significantly from day 1 to day 2 postsurgery (Table 4). The relative level compared with the baseline values started declining after day 2 until the level became significantly lower than day 2 at day 5 postsurgery. A second significant increase was observed from day 6 to day 7 postsurgery (Table 4).
Haptoglobin values at the different days postsurgery relative to the measured baseline value (baseline = 1) of rats cannulated in the common carotid artery
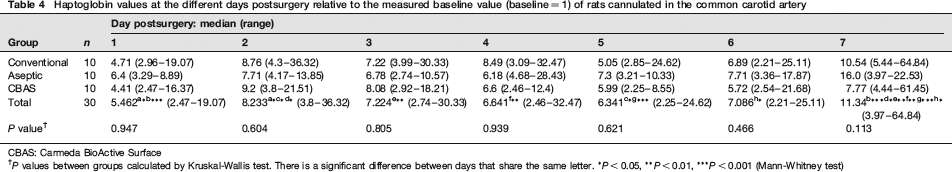
CBAS: Carmeda BioActive Surface
P values between groups calculated by Kruskal-Wallis test. There is a significant difference between days that share the same letter.
P < 0.05,
P < 0.01,
P < 0.001 (Mann-Whitney test)
The aseptic group had significantly lower serum concentration of ASAT than both the conventional group and the CBAS group, and the aseptic group also had a significantly lower concentration of serum creatinine than the CBAS group (Table 5). Although there were significant differences between the groups when testing albumin, total protein and phosphate, this could not be confirmed by individual t-tests (Table 5). There were no significant differences between the groups when testing ALAT, ALP, calcium, glucose, urea, sodium, potassium or chloride.
Biochemical parameters in the serum of rats cannulated in the carotid artery on day 7 postsurgery
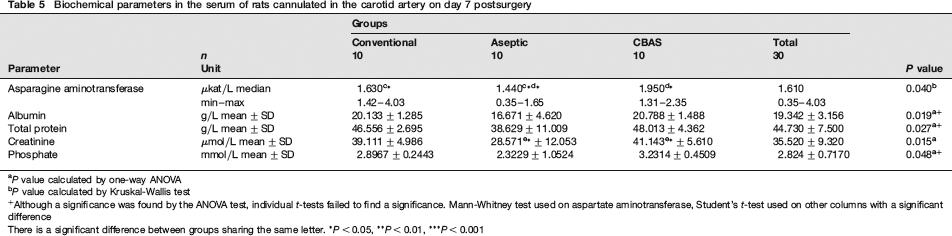
P value calculated by one-way ANOVA
P value calculated by Kruskal-Wallis test
Although a significance was found by the ANOVA test, individual t-tests failed to find a significance. Mann-Whitney test used on aspartate aminotransferase, Student's t-test used on other columns with a significant difference
There is a significant difference between groups sharing the same letter.
P < 0.05,
P < 0.01,
P < 0.001
Discussion
Vascular cannulation is one of the most common experimental procedures in rats used for pharmacological studies. Approximately 80% of the rats in the present study, independent of the hygienic level of the procedure and heparin coating, developed kidney infection, which may interfere with the renal excretion of drugs and their metabolites. However, the values of creatinine and urea did not indicate reduced renal function, 33 although the aseptic group had significantly lower serum creatinine compared with the CBAS group. Thirty percent of the animals had pathological lesions in the kidneys resembling infarctions, and 20% of the animals had positive blood cultures. The bacteria seem to migrate down the external catheter surface and then proliferate to form a thrombus and fibrin sheath at the tip 34 as early as 24 h after catheterization. 35 Neither systemic36,37 nor lock solution administration 11 of anticoagulants seem to prevent this. The heparin concentration in the lock solution may need to be higher than the 500 IU/mL used in this study to prevent fibrosis, neointimal cellular proliferation and thrombus formation, but this must be balanced with the risk of sideeffects from overdosing heparin in the postsurgical phase. 38 Bacteria adhering to a catheter through a biofilm may effectively be protected from systemic antibiotics in concentrations as high as 200 × the minimal inhibitory concentration.23,39,40 In human medicine using maximal sterile barriers (sterile gloves, mask, cap, gown and large drape) versus standard asepsis (gloves and small drape) when inserting catheters seem to protect against catheter-related infection, 41 but this is under hospitalized conditions where the catheter entry site can be kept clean at all times. In an animal facility, conditions are far more adverse as the rats are in contact with urine- and faecescontaminated bedding.
There were no significant differences between the groups in inflammatory or microbiological parameters, such as CFUs and haptoglobin. The levels of haptoglobin in serum were comparable to those obtained in the study by Giffen et al. 42 Serum haptoglobin may not be suitable as indicator of catheter-related infection, as a rise does not appear to be ‘dose-dependent’ in relation to the inflammatory factor, 42 and may increase solely due to tissue damage or the foreign body reaction to the catheter and not due to the presence of bacteria. The peripheral body temperature of the animals rose the afternoon after surgery in all the groups and then it fell steadily until the time of euthanasia. The antipyretic effect of carprofen may explain the temperature drop, but then a rise should have been observed after withdrawal. It is more likely that the reduction in temperature was due to a combination of shaving the animals, decreased activity levels and increased stress due to being housed alone and attached to the microdialysis system.
A larger proportion of the CBAS catheters were harder to flush, despite being coated with heparin which has been proven to reduce thrombus formation and increase catheter patency.11,43 This could be attributed to the catheter material and the catheter diameter. The CBAS catheters were larger in diameter, i.e. a 23 G needle was required to fill the lumen, while only a 25 G was needed for the Tygon PVC catheters, and made of polyurethane which is less flexible than PVC. Both of these factors have been shown to be detrimental to catheter patency 20 due to more vascular intimal irritation and damage, and less blood flow around the catheter. In conclusion, rats cannulated in the carotid artery are prone to develop kidney infection, which is very likely to be a confounding variable in pharmacological studies. The infection does not seem to be preventable by using aseptic procedures during surgery and, therefore, further precautions, such as the use of closed access ports, should be considered. Furthermore, heparin coating alone without current flushing does not seem to maintain catheter patency.
Footnotes
Acknowledgements
The author would like to thank Christian Jes Jensen for technical assistance and the staff of the H Lundbeck Ltd animal facilities for care and postoperative treatment of the animals, and for their cooperation.
