Abstract
Microsurgical autotransplantation of tissues is employed clinically to reconstruct defects following burns, trauma and surgical cancer ablation, and to correct congenital abnormalities. Transplant vessels of <3 mm are anastomosed by hand under the microscope. Experimentally, anastomotic patency rates decrease with increasing vessel diameter mismatch, and clinically, ratios of 3:1 or greater lead to unacceptably low arterial patency rates. A number of surgical techniques for dealing with size mismatch are described, but no one method has found favour, and few controlled studies of technique are reported. In this report, a rodent superficial caudal epigastric artery (SCEA)/femoral artery (FA) model for the study of these techniques is described in detail. The diameter ratio between these vessels lies in the clinically relevant range of 1:1.5–1:2.5. In the male Wistar rat, external vessel diameters were not found to increase markedly in size between animal weights of 300 and 500 g. The length of FA distal to the origin of the SCEA, which is important in allowing undisturbed distal run-off, was found to be negatively associated with animal body weight, implying that a smaller animal would be better for this model. Mean femoral arterial flow rate, measured by transit-time ultrasound, was noted to be statistically and physiologically significantly higher in the right artery when compared with the left. This model has advantages over interposition vein graft models in that it minimizes vessel compliance mismatch, and avoids the need for a second anastomosis.
Clinical reconstruction of tissue defects arising from ablative surgery, trauma or congenital abnormality has been greatly advanced over the last 40 years by the ability to autotransplant tissue composites using microvascular surgical techniques. Success rates in the order of 95% are enjoyed in many centres. 1
The corollary of a 95% success rate, however, is that one transplant in 20 fails. Anastomotic thrombosis remains the most common cause of transplant failure, and one factor in the development of thrombosis is vessel size mismatch. Monsivais 2 proved this experimentally using a rodent femoral axis model, interposing vein grafts of varying diameter mismatches. Clinically, Godina 3 attributed his early high failure rate in microvascular transplantation to the large-to-small diameter mismatch encountered when transplanting groin flaps to the lower limb.
End-to-end anastomotic techniques to manage diameter discrepancy can be divided into those that increase the circumference of the cut end of the smaller vessel, and those that decrease the circumference of the cut end of the larger vessel.
Techniques to increase the circumference of the smaller vessel include a fish-mouth incision, 4,5 or oblique section. 6 Alternatively, a wedge of the larger vessel may be removed to reduce its circumference to approximate it to that of the smaller vessel. 4,7,8 Where the upstream vessel is smaller, the mismatch may also be dealt with by invaginating it inside the larger downstream vessel to a varying degree. 9–11
Few studies of sufficient sample size are available to direct a choice of anastomotic method. Rickard et al. 12 attempted to answer the question of best method by computational modelling of haemodynamics across four different small-to-large anastomotic constructs. The authors showed that removing a wedge from the larger vessel produced least flow separation. However, these models used idealized geometries in non-compliant vessels and their direct clinical applicability is uncertain. An animal model is needed to validate these results.
We have used a superficial caudal epigastric artery (SCEA)/femoral artery (FA) model in the rat to study techniques of anastomosing arteries where the vessel diameter mismatch is small-to-large. The aim of this report is to describe the anatomic and flow characteristics of this model in detail. To date, we have used the model in a number of studies, performing over 300 microanastomoses.
Materials and methods
The study was carried out in two parts. Anatomic data were collected at the Department of Surgical Research, University of Cape Town, Cape Town, South Africa. Flow characteristics were determined during a second study carried out at Biological Services and the Wellcome Surgical Institute, University of Glasgow, Glasgow, UK.
Animals
Outbred male Wistar strain rats were used in both studies. In Cape Town, animals were obtained from the breeding colony of the Central Research Facility, University of Stellenbosch. This colony was established in 2001 from HsdOla:WI rats, and maintained as an outbred colony. Those used in the flow study were HsdHan™: WIST rats from Harlan UK Ltd, Bicester, Oxfordshire, UK. All animals were in the region of 350–550 g in body weight. Animal care in South Africa was practised according to University of Cape Town protocols, and followed the guidance given in EC Directive 91/507/EEC in the UK.
Husbandry
Husbandry in Cape Town and Glasgow was similar. Information on animal health status was obtained prior to arrival and all animals underwent an acclimatization period of at least seven days prior to commencing the study. The rat colonies were group housed in polypropylene solid floor cages with stainless-steel grid lids on racks. Wood shavings were used as bedding with nesting material and cardboard tubes provided as cage enrichment. The environmental temperature was maintained at 20 ± 2°C and relative humidity at 55 ± 10%. The lighting schedule was 12 h light and 12 h dark and there were 15–20 air changes per hour. A non-sterile pelleted diet (Rat Cubes, Epol [Pty] Ltd, Rustenberg, South Africa in Cape Town, and BEEKAY Rat and Mouse Standard Diet (expanded), B&K Universal Ltd, Hull, UK in Glasgow) and domestic mains water were offered ad libitum.
Anaesthesia
Parenteral ketamine HCl (Anaket-V®, Centaur Laboratories, Bryanston, South Africa) and xylazine HCl (Rompun®, Bayer [Pty] Ltd, Isando, South Africa,
In both studies, buprenorphine HCl (Temgesic®, Schering-Plough, Johannesburg, South Africa, in Cape Town; Vetergesic®, Reckitt Benckiser Healthcare [UK] Ltd, Hull, UK in Glasgow), and either bupivacaine HCl (Marcaine®, Adcock Ingram, Bryanston, South Africa) or levobupivacaine HCl (Chirocaine®, Abbott Laboratories Ltd, Queensborough, Kent, UK) were administered during the procedure. Hydration was maintained by the administration of normal saline by either the intravenous (Cape Town) or subcutaneous route (Glasgow).
Model description
The animal is placed in a supine position on a warming mat, and its hindlimbs are extended using loose, un-constricting elastic limb restraints pinned into a cork board. Bilateral caudally-concave incisions are made over the groins. The groin fat pad is dissected free from underlying external oblique muscle and spermatic cord, taking the SCEA with it. With the aid of soft retractors, the FA is found as it emerges from under the inguinal ligament and is dissected free from surrounding tissues. The dissection is completed from the inguinal ligament to a few millimetres distal to the bifurcation of the FA into the saphenous (SA) and popliteal (PA) arteries. The SCEA is similarly dissected free from surrounding tissues for a distance of 8 mm. The utmost delicacy must be employed in dissecting free this vessel because it is very easily damaged and prone to spasm. Small muscular branches of both the FA and SCEA are tied off close to their origin using 10-O monofilament nylon suture. A few drops of vessel plegic solution (2% lidocaine) are applied after the dissection is complete, and the vessels are kept moist at all times.
A small-to-large arterial mismatch is created by tying off the FA immediately distal to the origin of the SCEA using 10-O monofilament nylon suture material or similar. This diverts all blood flow into the SCEA. The SCEA is then tied off 7 mm distal to its origin. Following the application of microvascular clamps, the FA is divided less than half a millimetre distal to the first tie. The SCEA is then divided 6 mm from its origin (Figure 1). This length is important because it allows blood flow characteristics to settle into a laminar pattern before reaching the anastomosis, and it permits a tension-free anastomosis.
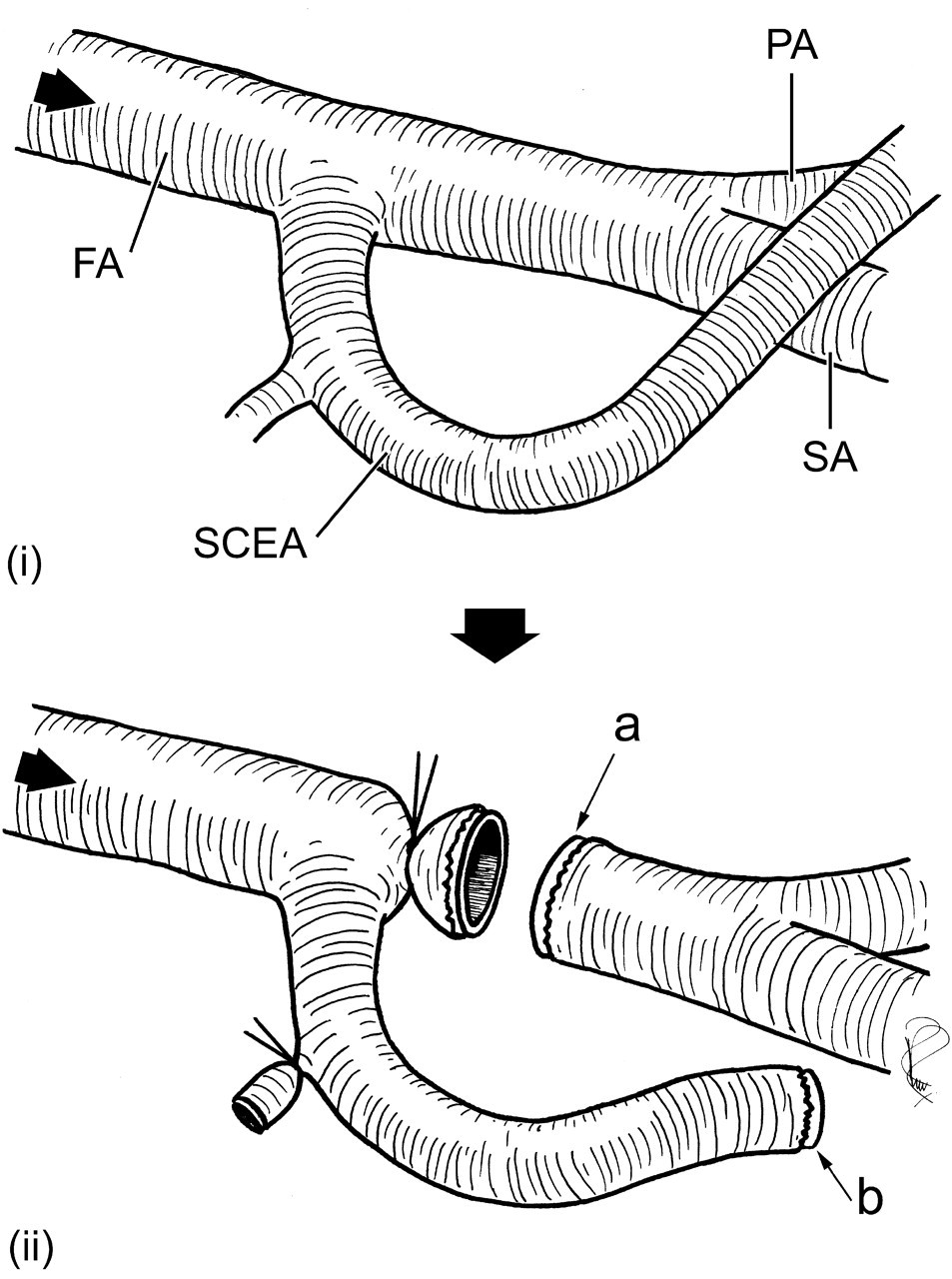
(i) Anatomy of the distal femoral artery. FA: femoral artery; SCEA: superficial caudal epigastric artery; PA: popliteal artery; SA: saphenous artery. (ii) The FA is tied off immediately distal to the origin of the SCEA, diverting all flow into the SCEA. The FA and SCEA are then divided, giving a large downstream vessel (a) to which a smaller upstream vessel (b) can be anastomosed
The SCEA may then be anastomosed to the distal FA, giving a clinically applicable small-to-large size mismatch in the region of 1:1.5–1:2.5 (Figure 2).
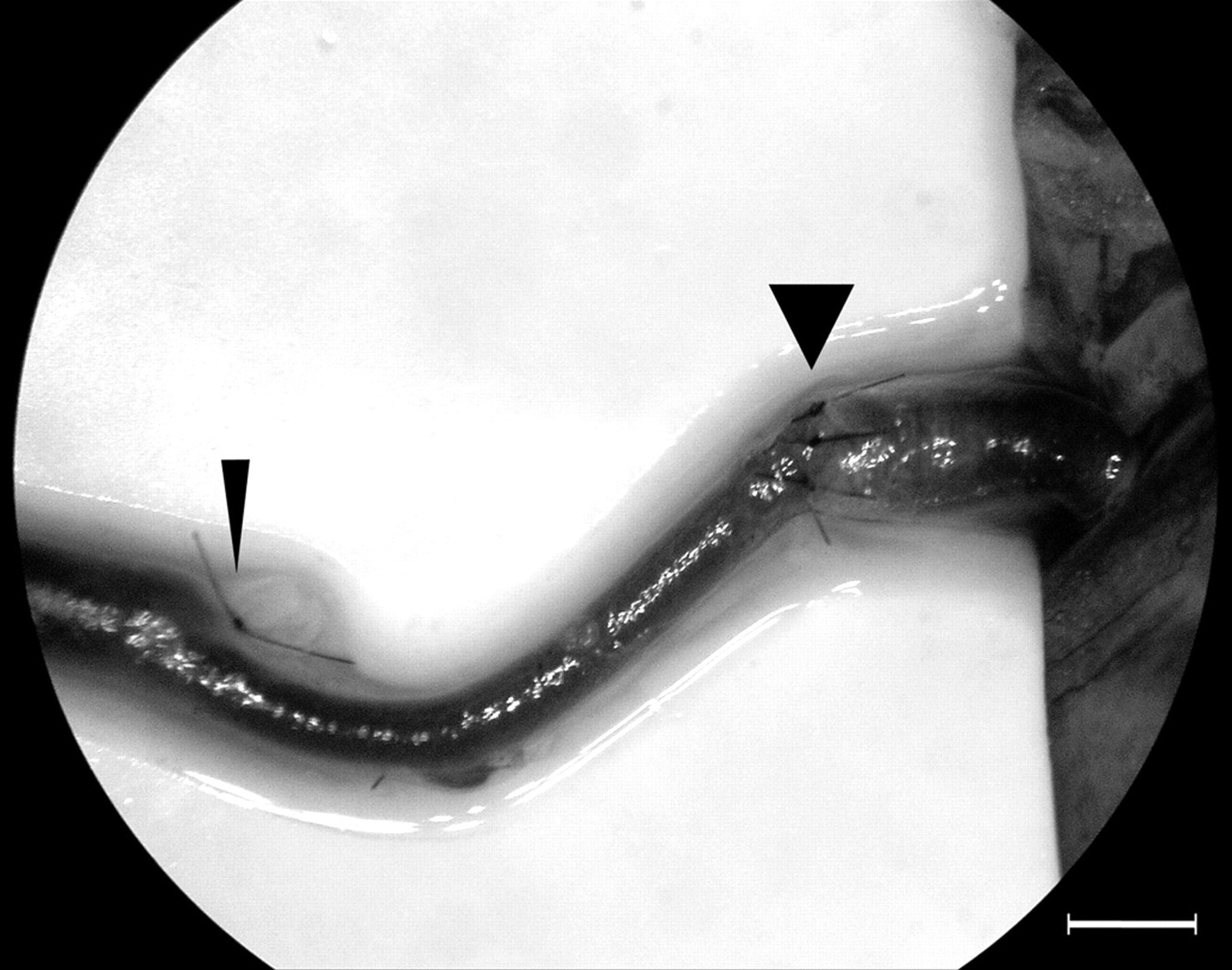
An oblique-section anastomosis using the model. Scale bar = 1 mm. Flow is from left to right. Narrow arrowhead = tie around the femoral artery (FA) distal to the superficial caudal epigastric artery (SCEA) origin, wide arrowhead = sutured anastomosis between the small, upstream SCEA and larger downstream FA
Anatomic characterization
Forty outbred male Wistar rats were included in the anatomic characterization. Ethical approval for this part of the work was obtained from the University of Cape Town Animal Ethics Committee (Rec. Ref. 03/041). Animals were weighed and anaesthetized using parenteral ketamine and xylazine. The FA and SCEA were dissected free as described above. Following topical application of 2% lidocaine, digital images of these vessels were obtained using a 4.0 MP consumer digital camera (Nikon Coolpix® 4500, Nikon Corporation, Tokyo, Japan,
In each animal and on each side, three replicate measurements were taken of each of the following (Figure 3):
External diameter of the FA immediately distal to the SCEA origin; External diameter of the SCEA at a point 6 mm from its origin from the FA; Length of FA from origin of SCEA to its bifurcation into PA and SA.
SCEA/FA diameter ratio was calculated from (1) and (2).
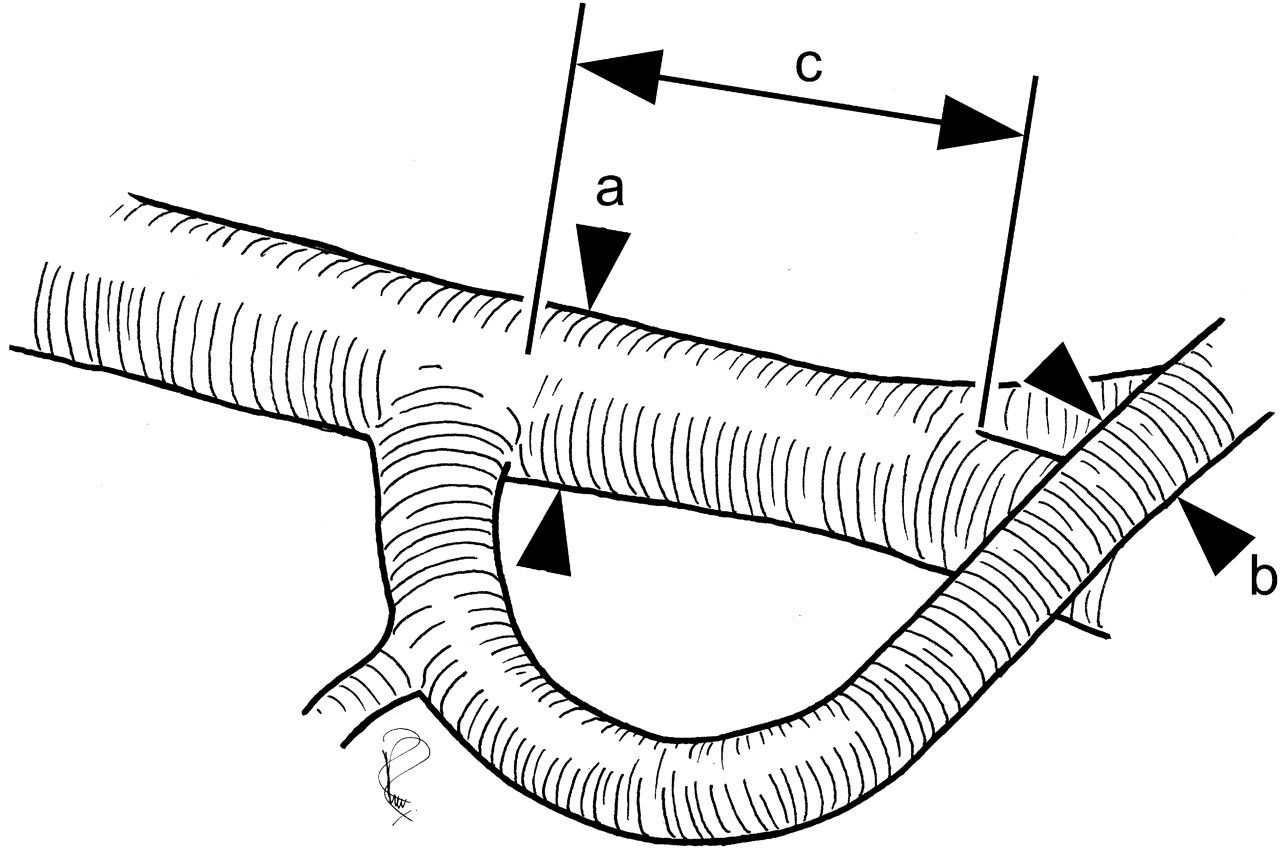
Anatomical measurements taken. (a) Femoral artery (FA) external diameter distal to the superficial caudal epigastric artery (SCEA) origin. (b) SCEA external diameter 6 mm from its origin. (c) Length of FA between the origin of the SCEA and its bifurcation into the popliteal (PA) and saphenous (SA) arteries
Flow characterization
Seventeen additional outbred male Wistar strain rats were included in the flow study, which formed part of a larger project authorized by the UK Home Office under the terms and conditions of the Animals (Scientific Procedures) Act 1986.
To ensure physiological stability, gaseous anaesthesia was used as described. Heart rate and arterial oxygen saturation were monitored using a Nonin Model No. 8500AV pulse oximeter attached to a forelimb (Nonin Medical, Inc, Plymouth, MN, USA,
The FA and SCEA were dissected out bilaterally as described. The deep FA and small muscular branches were tied off. A few drops of lidocaine 2% solution were applied. FA flow rate was recorded by placing a transit-time ultrasound flow probe (1RB, Transonic Systems Inc, Ithaca, NY, USA,
Statistical analysis
Statistical analysis of results was performed in Minitab® 15 for Windows® (Minitab Inc, State College, PA, USA,
Results
Anatomic characterization
One digital image was technically inadequate for diameter measurement, leaving 39 pairs available for analysis. One further image was inadequate for measurement of FA length distal to the SCEA origin, leaving 38 pairs available for this analysis. Mean animal weight was 423.1 g (range 343–505 g).
(a) FA external diameter (Figure 4). Mean FA external diameter (±standard deviation [SD]) was 0.9725 ± 0.0073 mm on the left and 0.9749 ± 0.0073 mm on the right. Regression analysis showed a significant association between diameter and animal body weight (P = 0.0030, R 2 = 3.8%). The regression equation was FA external diameter = 0.777 + (0.000464 × weight), predicting an FA external diameter of 0.9166 ± 0.0327 mm at an animal weight of 300 g, and a diameter of 1.0094 ± 0.0216 mm at 500 g. The predictive interval (PI), i.e. how accurately it is possible to predict FA diameter in a given animal of a known body weight, however, was wide (95% PI = 0.7514–1.0818 mm at 300 g and 0.8517–1.1671 mm at 500 g). There was no significant difference in FA external diameter between left and right-sided vessels (P = 0.8211, ANOVA).
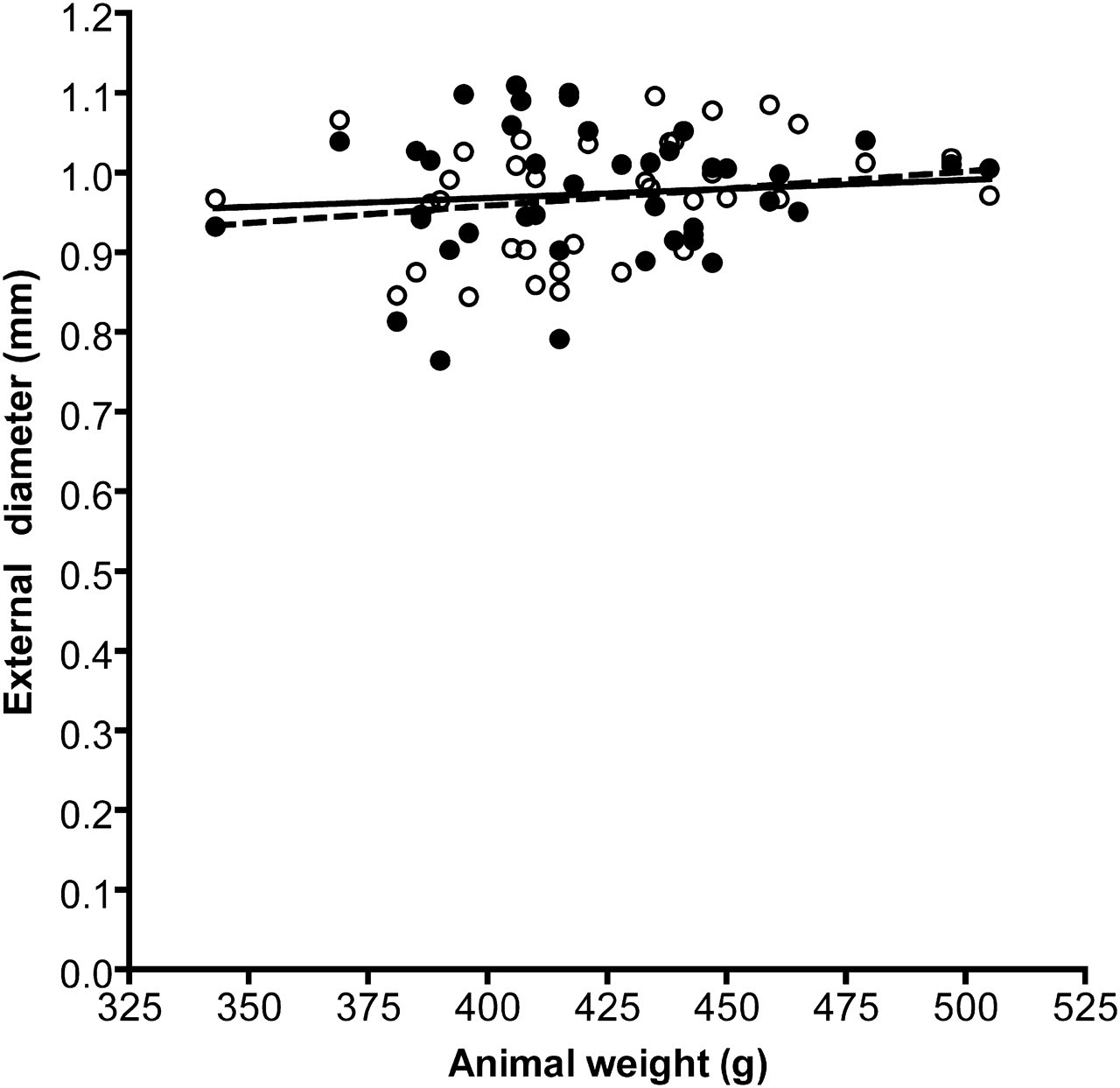
Scatterplot of the femoral artery (FA) external diameter versus weight. Values are means of three replicate measurements. Open circles and interrupted line: left FA, closed circles and solid line: right FA
(b) SCEA external diameter (Figure 5). Mean SCEA external diameter (±SD) was 0.5621 ± 0.0052 mm on the left side and 0.5430 ± 0.0051 mm on the right. Regression analysis of both sides together showed a significant association between vessel size and animal body weight (P < 0.0001, R 2 = 5.9%). The regression equation was SCEA external diameter = 0.376 + (0.000417 × weight), predicting an SCEA external diameter of 0.5012 ± 0.0226 mm at an animal weight of 300 g (95% PI = 0.3875–0.6150 mm), and a diameter of 0.5846 ± 0.0148 mm at 500 g (95% PI = 0.4760–0.6932 mm).
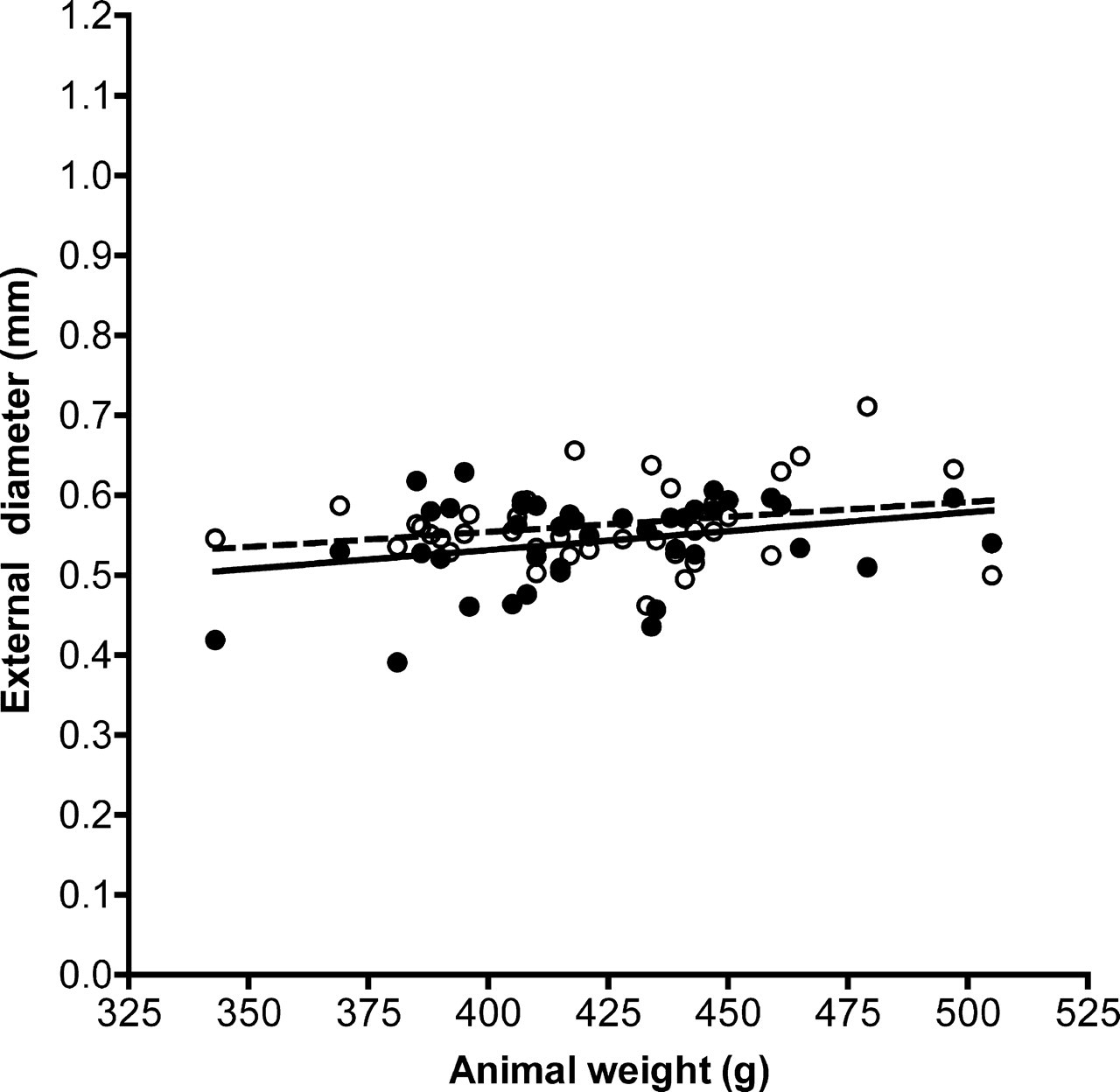
Scatterplot of the superficial caudal epigastric artery (SCEA) external diameter versus weight. Values are means of three replicate measurements. Open circles and interrupted line: left SCEA, closed circles and solid line: right SCEA
There was a significant difference in SCEA external diameter between sides (P = 0.0095, ANOVA). The difference in mean diameters was 0.0190 mm, with the left SCEA being larger.
(c) Distal FA length (Figure 6). Mean length (±SD) of FA distal to the origin of the SCEA was 3.457 ± 0.0572 mm on the left side and 3.298 ± 0.0572 mm on the right. Regression analysis of both sides together revealed a negative association between this length and animal body weight (P = 0.0040, R 2 = 3.6%). The regression equation was distal FA length = 4.86 − (0.00350 × weight). This predicts a length of 3.8076 ± 0.2643 mm (95% PI = 2.4748–5.1404 mm) at 300 g and a length of 3.1083 ± 0.1744 mm (95% PI = 1.8356–4.3810 mm) at 500 g.
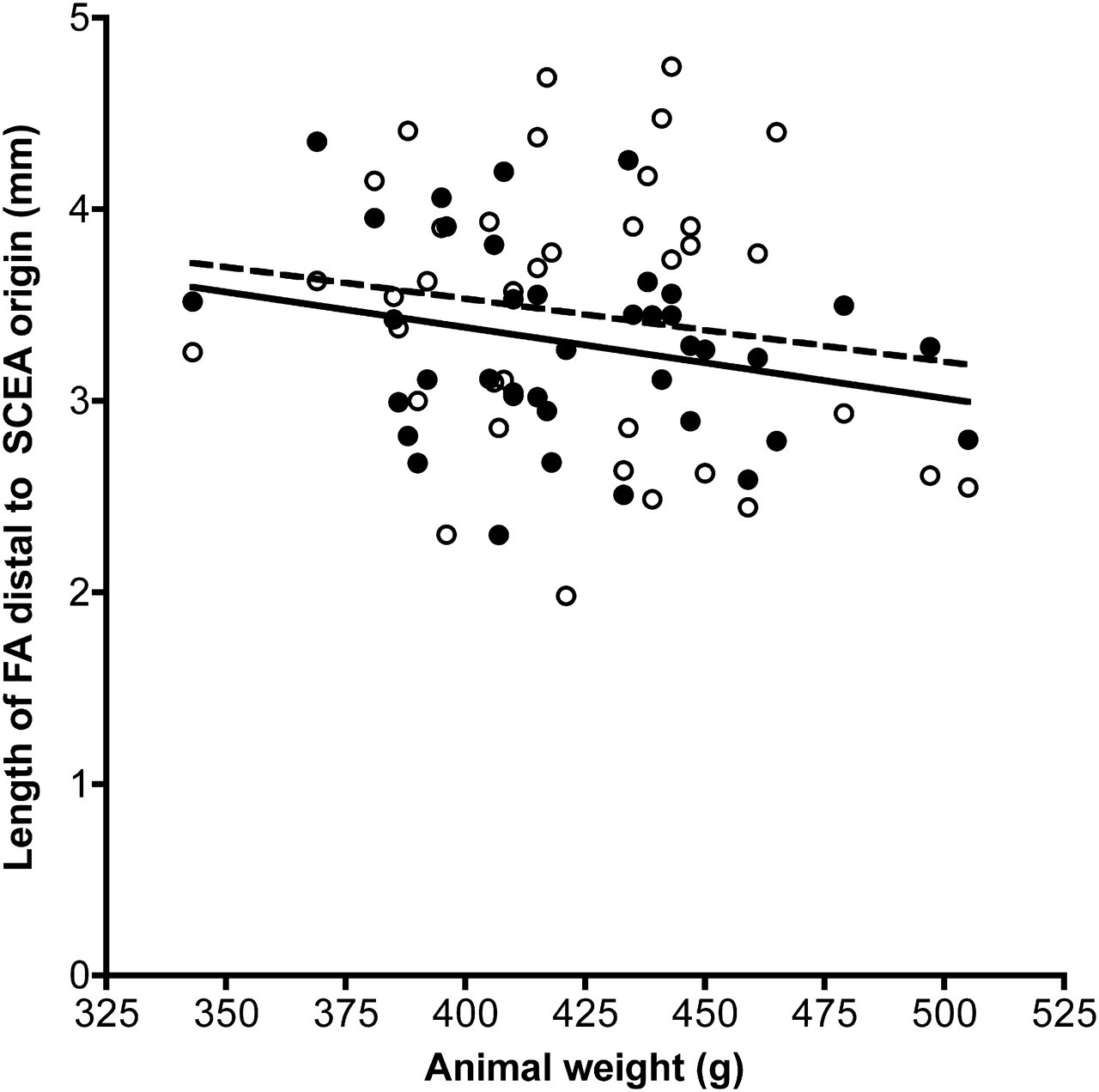
Scatterplot of distal femoral artery (FA) length versus weight. Values are means of three replicate measurements. Open circles and interrupted line: left distal FA length, closed circles and solid line: right distal FA length
There was a significant difference in distal FA length between sides (P = 0.0496, ANOVA). The difference in mean lengths was 0.3189 mm, with the left distal FA being longer.
(d) SCEA:FA diameter ratio (Figure 7). This ratio was calculated on each side in each animal, giving a total of 39 paired calculations. Mean ratios were left side, 1:1.741 and right side, 1:1.811. There was no significant association between diameter ratio and animal body weight (P = 0.4710, linear regression analysis). There was no significant difference in diameter ratio between left and right sides (P = 0.1580, ANOVA).
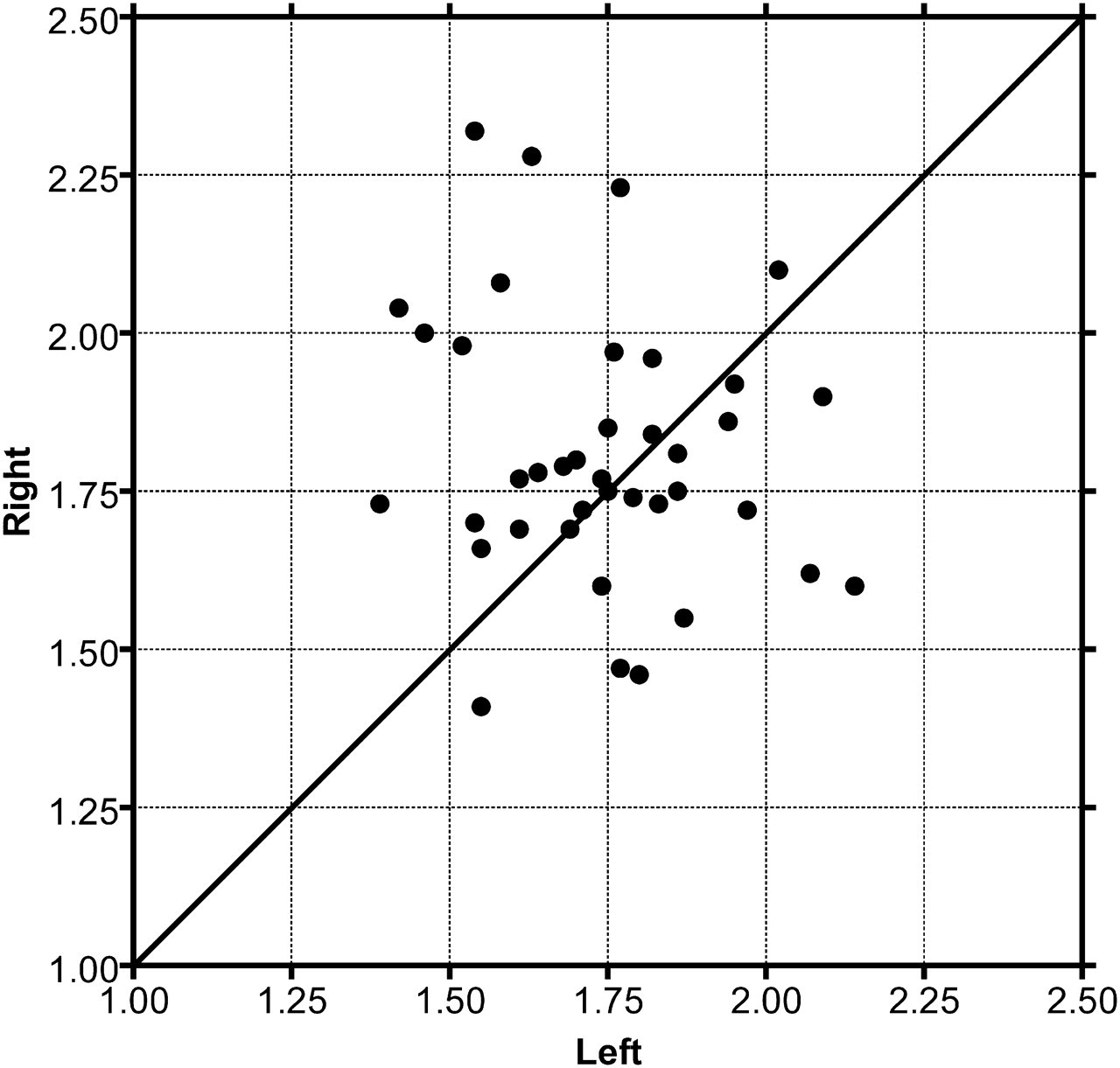
X/Y plot of paired superficial caudal epigastric artery:femoral artery (SCEA:FA) mean diameter ratios. N = 39. Diagonal line marks equal left and right ratios
Flow characterization
Paired results were available in all 17 animals (Figure 8). Mean animal weight was 448.9 g (range 322–564 g). Mean core body temperature at time of measurement was 37.509 ± 0.361°C. There was no significant association between flow rate and core temperature of the animal at the time of measurement (P = 0.079, linear regression analysis).
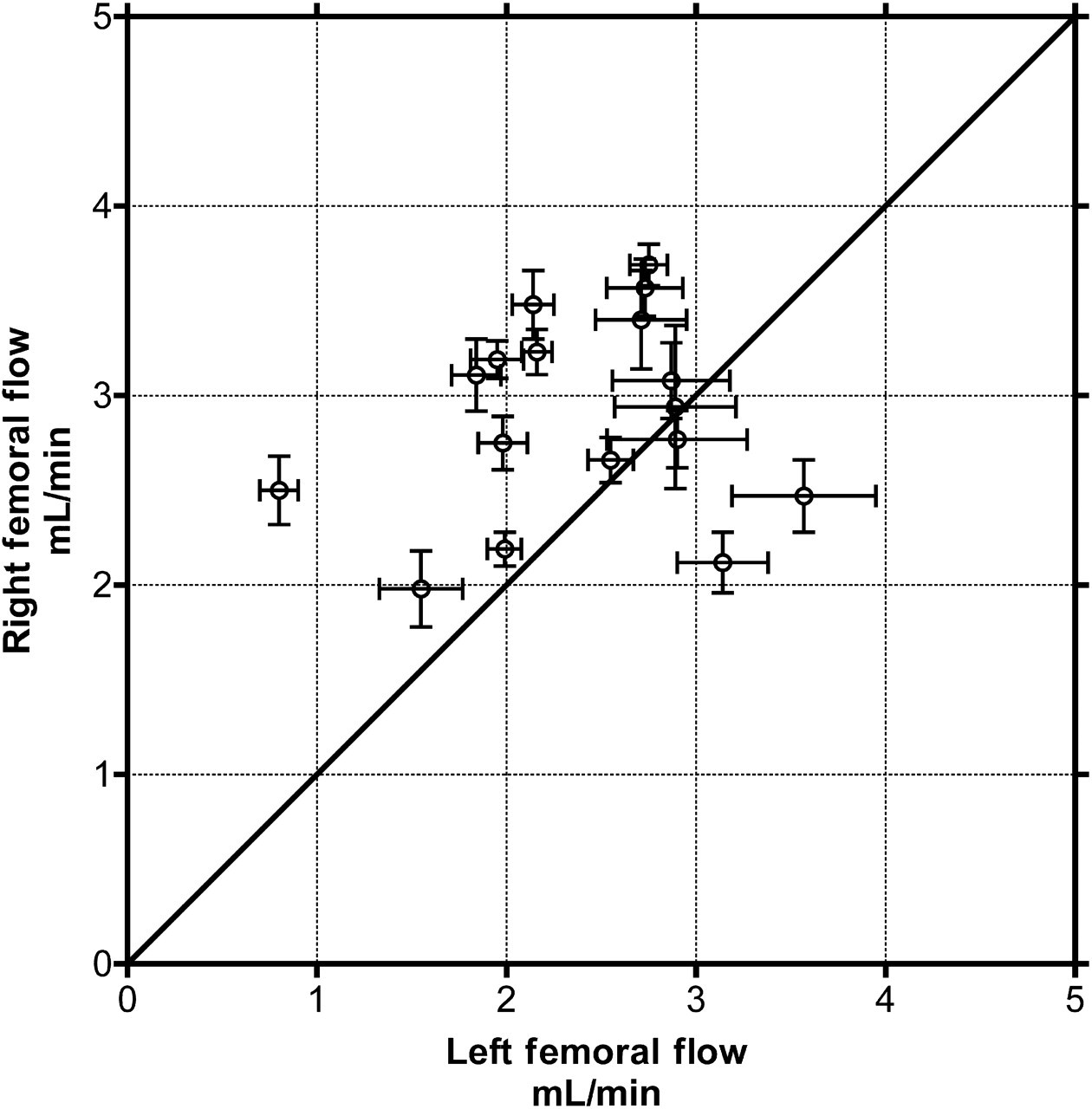
X/Y plot of paired femoral artery (FA) volume flow rates (mean ± SD). N = 17. Diagonal line marks equal left and right volume flow rates
Mean flow rates were left side, 2.391 mL/min and right side, 2.882 mL/min, a difference of 0.491 ± 0.1849 mL/min. This difference was statistically significant (P = 0.0179, ANOVA). There was no association between flow rate and the side order in which measurements were taken (P = 0.1801, ANOVA).
Discussion
The majority of studies using animals to examine the geometric design of arterial microanastomoses to overcome diameter mismatch have used an interposition vein graft to create the diameter discrepancy. 2,13–16 The use of a vein graft introduces two further variables into these studies: those of a significant compliance mismatch and the requirement for a second anastomosis. In addition, the use of large vein grafts from the same animal is a potential source of morbidity, either resulting from their harvest or from any consequent disruption of venous drainage.
The model described in this paper was originally suggested by de la Peña-Salcedo et al. 11 and has been modified by us. These animal studies have been conducted in order to validate and complement our in silico work. 12 The model keeps any compliance mismatch within an arterial range, and avoids the need for a second anastomosis. Furthermore, it permits the paired study of technique in one animal, reducing the number of animals that need to be used to comprehensively answer this research question.
The vessel diameter mismatch provided by this model is in the range of 1:1.5–1:2.5. This ratio is clinically applicable – it is beyond a (small) mismatch that might be managed by judicious mechanical dilation of the smaller vessel, but not so large as to be seldom encountered in clinical practice. There did not appear to be any association of this ratio with animal body weight, and no significant ratio difference was found between left and right sides.
The association between FA and SCEA external diameters and body weight of the outbred Wistar rat is to be expected. While there was a statistically significant difference in SCEA external diameter between left and right sides, the actual difference in mean diameters was very small in this sample population and not, in the authors' opinion, technically significant. It is also noteworthy that the size of the vessels, as predicted by the regression equations, does not increase markedly with animal weight. Together, these observations imply that it is unnecessary to house and feed animals until they are very heavy and a younger 300 g animal would suffice.
The length of FA distal to the SCEA origin is important in the study of anastomotic techniques because of the need for undisturbed distal run-off. A length of 1–2 mm would appear to be sufficient. 17 This distance is particularly important in the study of an invagination or ‘sleeve’ technique, where the SCEA is invaginated inside the distal FA. Regression analysis showed a negative correlation of this length with animal weight, and with the desire for as long a distal FA as possible, a smaller animal would appear to be better in this regard. The predictive interval, however, was very wide.
We have used this model in one large study of two microanastomotic techniques, attaining a patency rate of 95% at one hour. No animals died as a direct result of the model. However, while we do not have precise incidence figures, an estimated 2% of animals in this larger study exhibited an anatomical anomaly where the SCEA originates with the PA and SA as part of an FA trifurcation. This occurred in none of the 40 animals included in the anatomical study, but in one animal used in the flow study. This unfortunate finding makes the animal unusable in the study of anastomoses between vessels of disparate diameters.
Mean FA volume flow rates in these Wistar rats were found to approximate those found in perforating arteries in man. 18 In this relatively small population sample, a statistically significant difference in mean flow rate between left and right femoral arteries was found. This difference is unexplained by small variations in animal core temperature, or by the order in which consecutive measurements were taken. With a difference in mean arterial flow rates of 0.491 ± 0.1849 mL/min, this difference is physiologically significant. This finding has not, to our knowledge, been reported previously. In the light of mammalian anatomical asymmetry, it is perhaps logical that this should be so. The implication of this finding is that in a paired study of techniques, blood flow rates following anastomosis must be compared with preanastomotic baseline measurements, and the assignment of an anastomotic technique to one side or the other must be done randomly.
In conclusion, an animal model has been described, which allows the paired study of microanastomotic techniques between small upstream and larger downstream arteries, and provides a vessel diameter mismatch of clinical relevance. Counter-intuitively, we have found that the use of larger animals is unnecessary, and may be technically disadvantageous. Of secondary, but pragmatic importance, the use of smaller animals should lead to cost savings. In a paired study of blood flow rates through anastomoses performed using this model, the disparity in FA mean volume flow rates between left and right sides would necessitate randomization of a technique to one side or the other.
Footnotes
ACKNOWLEDGEMENTS
These studies were supported by grants from the Stephen Plumpton Trust and the Canniesburn Research Trust.
The authors wish to acknowledge the direct and indirect support provided by the following: Professor Delawir Kahn, Professor Anwar Mall, Mr Gert Engelbrecht and technical staff of the Department of Surgical Research, University of Cape Town; Mr Iain Mackay of the Canniesburn Unit for Plastic Surgery, Glasgow Royal Infirmary; Dr Joyce Ferguson, Mr David McLaughlin and animal care staff of the Veterinary Research Facility, Biological Services, University of Glasgow; Professor Mhairi Macrae and Mrs Lindsay Gallagher of the Department of Neurophysiology, University of Glasgow; and Professor George Gettinby of the Department of Statistics and Modelling Science, University of Strathclyde.
