Abstract
Chronic jugular vein or central venous cannulation is routinely performed in human and animal patients for access to blood circulation. In mature swine, chronic catheter placement techniques have typically involved venous isolation via extensive cut-down, blunt dissection and manipulation of ventral neck tissues prior to catheter placement. More recently, guide-wire-assisted percutaneous techniques have become standard practice in human and veterinary medicine due to the minimization of soft tissue and vessel damages. Laboratory animal piglets are becoming more popular research models because of their immature immunological system, ease of handling and costs. However, external jugular veins are very difficult to catheterize in paediatric animals including freshly weaned piglets. The objective of this study was to develop a simple, safe and efficient method for external jugular vein cannulation in young piglets. In total, 20 piglets were anaesthetized and percutaneously catheterized with a guide-wire technique using palpable anatomical landmarks and triangulation. With this minimally invasive catheterization, it has allowed our veterinarians and veterinary technicians to quickly and easily obtain central venous access in piglets undergoing operative procedures.
Swine models are commonly used in the study of human diseases because of their similar anatomical and physiological characteristics of the cardiopulmonary system. 1,2 Most cardiopulmonary studies involve multiple catheterizations of peripheral vasculature. Cannulation of the external jugular vein or the central venous line (CVL) access is essential for pulmonary artery catheterization and central venous pressure monitoring. 3,4 Clinical and research patients may require large volumes of fluids or other pharmacological substances prohibiting use of smaller peripheral veins. Chronic blood sampling via CVL eliminates periodic venepunctures, reduces anaesthesia administration and decreases unnecessary prolonged restraint. In addition, long-term CVL usage reduces or eliminates variations in the biochemical, haematological and hormonal parameters observed during jugular venepuncture in restrained swine. 5–8
In laboratory swine, CVL placement is often performed by surgical cut-down procedures, which have been described in detail. 9–11 These techniques have proven quite successful, although they do require prolonged general anaesthesia and occasional significant postoperative management. Percutaneous catheterizations have become a standard of care as physicians are gravitating towards less insidious ‘bedside’ procedures to include less invasive techniques for vascular access. Veterinary practitioners are also incorporating less invasive diagnostics and treatments for their animal patients. 12 The Guide for the Care and Use of Laboratory Animals also discusses the availability and appropriateness of the use of less invasive procedures while conducting laboratory animal research. 13 The need for less invasive techniques for vascular access in animal models is an important refinement to ensure valid comparisons among animal subjects for human research.
Percutaneous catheterization methods for central venous access have been well described in adult and paediatric patients. 14–17 In adults, a triangulation method is often used for inserting CVL catheters into the internal jugular vein (IJV) using visual and palpable landmarks. 17–20 In infants, anatomical features are smaller and landmarks are more challenging to palpate because of immature musculoskeletal development, making catheterizations more difficult. 21,22 Ultrasound-guided modalities have been incorporated during infant catheterizations to identify smaller vasculature and decrease the risks of carotid artery punctures. 20,23,24 A minimally invasive percutaneous technique for external jugular vein catheterization in mature pigs has been developed recently. 25 Venepuncture techniques described in that publication used anatomical landmarks involving cephalicus muscles, which are readily visualized and palpable on a mature pig. However, in young piglets, these muscles are not yet fully developed; thus, typical landmarks used for catheterization of the external jugular vein are not easily palpable or identified in the immature pig. Similarly, non-surgical techniques have been developed for external vein catheterization and venepuncture in young pigs and adult sows; however, the physical landmarks, angle of venepuncture, direction of needle and needle depth are vague or have not been mentioned by the authors. 26–28
The aim of this study was to develop a simple, safe and efficient technique using available commercial materials for percutaneous external jugular vein catheterization in the young piglet. We describe a minimally invasive procedure for gaining CVL access in piglets via catheterization of the external jugular vein using a triangulation method involving three easily palpable landmarks and a standard commercial, wire-guided central venous catheter kit.
Materials and methods
General procedures
All procedures were performed in strict accordance with the guidelines outlined in the Guide for the Care and Use of Laboratory Animals. 13 The animals were housed in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC). The swine (n = 20) were conventional Yorkshire farm pigs (Sus scrofa domestica) purchased through a USDA-approved vendor (ABI, Danboro, PA, USA). The piglets were weaned and approximately four weeks old at purchase. All animals were individually housed in 1.25 m × 2.75 m stainless-steel large animal runs with an expanded metal flooring. The room temperature was maintained at 22 ± 3°C, with 50 ± 20% relative humidity and 12–15 air exchanges hourly. Photoperiods were 12:12 h light:dark, with lights on from 06:00 to 18:00 h. The animals were fed AKEY 2000 porcine growing diet (AKEY, Lewisburg, OH, USA) twice daily, and water was available ad libitum by automatic watering systems. Recommendations for the insertion of vascular catheters were followed in accordance with the Centers for Disease Control's universal precautions. Prior to inserting catheters, veterinarians or veterinarian technicians washed their hands with an antimicrobial scrub (iodine or chlorhexidine-based). All personnel performing or assisting with catheter insertions wore sterile gloves and gowns, caps, masks, and other appropriate surgical attire.
Anaesthesia
Twenty (12 female, 8 male) small-sized Yorkshire piglets weighing 4.8 ± 0.84 kg were administered atropine (0.04 mg/kg) and buprenorphine (0.005 mg/kg) intramuscularly as a preanaesthetic. After being gently restrained in a Panepinto sling, a peripheral intravenous catheter was placed percutaneously into an auricular vein. Intravenous fluids (lactated Ringers) were administered at 10 mL/kg/h throughout the procedure and during recovery. Endotracheal intubation was conducted on all animals after induction using isoflurane (4–5%) in 100% oxygen via a face mask. Anaesthesia was maintained using isoflurane (1–2%), and physiological monitoring was performed in accordance with American Society of Anaesthesiologists' standards. The piglets were allowed to breathe spontaneously unless they developed respiration problems (apnoea, end tidal CO2 > 50 mmHg, saturation O2 < 91%) and were subsequently placed on a mechanical ventilator.
Anatomy and triangulation
The external jugular vein is formed at the junction of the maxillary and linguofacial veins and extends caudally toward the thoracic inlet. It converges with the subclavian vein to form the brachiocephalic vein, which subsequently becomes the cranial vena cava. The external jugular vein begins approximately ventrolateral to the angular process of the mandible (caudal ramus) and courses caudally toward a point just lateral to the manubrium, where it enters the thoracic inlet. Three easily identifiable landmarks were palpated on the piglet body while in the dorsal recumbent position to target the right external jugular vein. The landmarks are the (1) angular process or the caudal ramus of the right mandible, (2) the cranial manubrium, and (3) the cranial point of the right shoulder formed by the greater tubercle of the humerus. To correctly palpate the landmarks, the piglet's neck requires full extension, the right leg must be retracted caudally till the brachium is almost parallel with the table, and the short vertical axis of the body must be as close to perpendicular to the table as possible. We can illustrate the palpable landmarks, the triangulation technique and initial venepuncture site on the ventral neck by postmortem dissection of the neck (Figure 1). Initially, all three landmarks were identified and connected to form a large triangle. After locating approximately the midway point of the primary lines, secondary lines were drawn beginning from the landmarks and extended to the opposite side midway point to form another small triangle. The site for initial insertion of the catheter needle is at the centre of the smaller triangle (Figure 1).
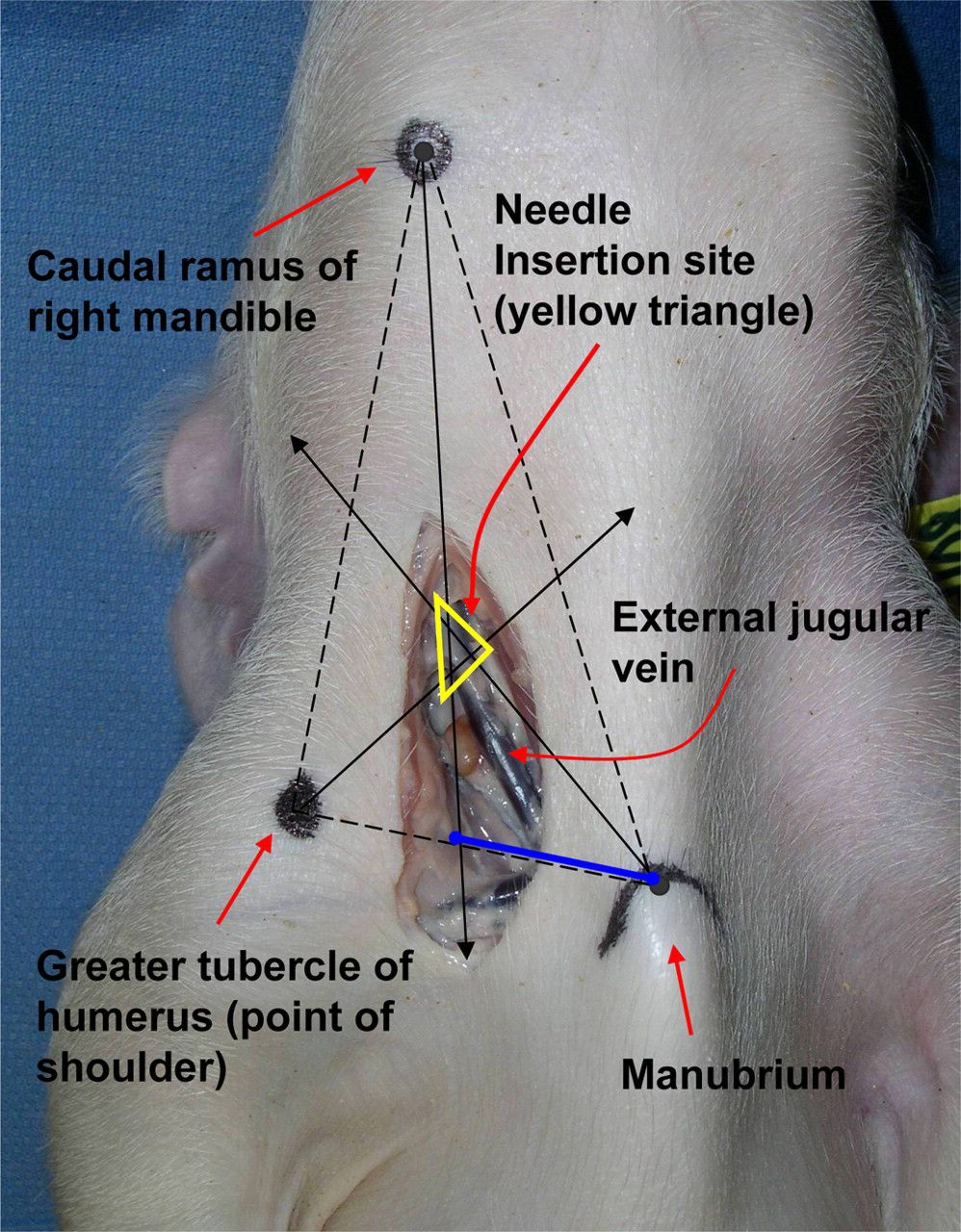
Photograph of a dissected piglet's neck in dorsal recumbency showing landmarks and the triangulation technique used to identify the external jugular vein. The primary lines (dashed) are drawn between the palpable landmarks to form a large triangle. The secondary lines (solid) are drawn from the landmarks to the midway points between the primary lines to form a smaller triangle. The smaller triangle (yellow) lies directly over the external jugular vein's expected path and provides an initial entry point for the introducer needle (blue line = approximate direction for needle after initial entrance)
Technique
The anaesthetized pigs were placed in dorsal recumbency. Hair was clipped from the insertion site and its surrounding area. The clipped region was scrubbed with chlorhexidine gluconate solution and isopropyl alcohol alternately for a total of three scrubs and a contact time of 5–10 min. The site was draped with a sterile surgical drape. After triangulation, the piglet's external jugular vein was manually occluded using previously described methods with minor modifications. 25 An assistant placed his/her thumb at the tip of the manubrium in the centre of the drawn curve (Figure 1). While pressing gently down on the manubrium, the thumb was rolled toward the ipsilateral shoulder cranial to the sternocostal articulation of the first rib, providing inward pressure to occlude the external jugular vein. The ipsilateral limb was retracted caudally with the brachium almost parallel to the table. All materials used in the catheterization process were supplied in a commercial 20-G × 12 cm single lumen central venous catheter kit (Arrow Int, Reading, PA, USA). The catheterization was performed by first inserting an 18- to 20-gauge introducer needle through the skin at the centre of the triangulation (Figure 2). The needle was then directed at a 30° angle downward and medial-caudally toward the tip of the assistant's thumb occluding the external jugular vein. Flashback of dark blood indicated successful puncture of the external jugular vein. A 5 mL syringe was occasionally used to aspirate the needle to verify the location and remove clots. After venepuncture, a flexible guide wire (j-wire length, 60 cm) was inserted into the lumen of the external jugular vein via the introducer needle. A portion of the guide wire was left in place and the introducer needle was withdrawn. The skin puncture site was slightly enlarged using a #11 surgical blade. A rigid vessel dilator was threaded over the guide wire, advanced through the skin and subcutaneous tissues, and withdrawn. The central venous catheter filled with heparinized saline (10 U/mL) was advanced over the guide wire into the vessel to approximately the cranial right atrium. Once the catheter was in place, the guide wire was removed and the catheter was flushed with heparinized saline (10 U/mL) to confirm patency and a luer-lock catheter cap was placed on the catheter hub. After the catheter was temporarily secured to the skin using tape, placement was verified using fluoroscopy. If necessary, the catheter was adjusted to ensure that the tip was correctly positioned at the entrance to the cranial right atrium. After adjustments the catheter was secured to the skin using a Statlock device (Venetec Int, San Diego, CA, USA). All piglets received buprenorphine (0.01 mg/kg, 0.3 mg/mL, intramuscularly) before extubation and bupivicaine (0.5%, 1.0 mL) was infused subcutaneously approximately 1–2 cm caudal of the catheter insertion. All necks were wrapped using rolled gauze, cast padding and elastic self-adhesive bandage (Vetrap™, 3M™, 3M Inc, St Paul, MN, USA) The piglets recovered in a padded stall with thermal support, fluids and supplemental oxygen. Prior to and after use, the catheters were flushed with heparinized saline (10 U/mL) to confirm patency and maintain heparin lock, respectively.
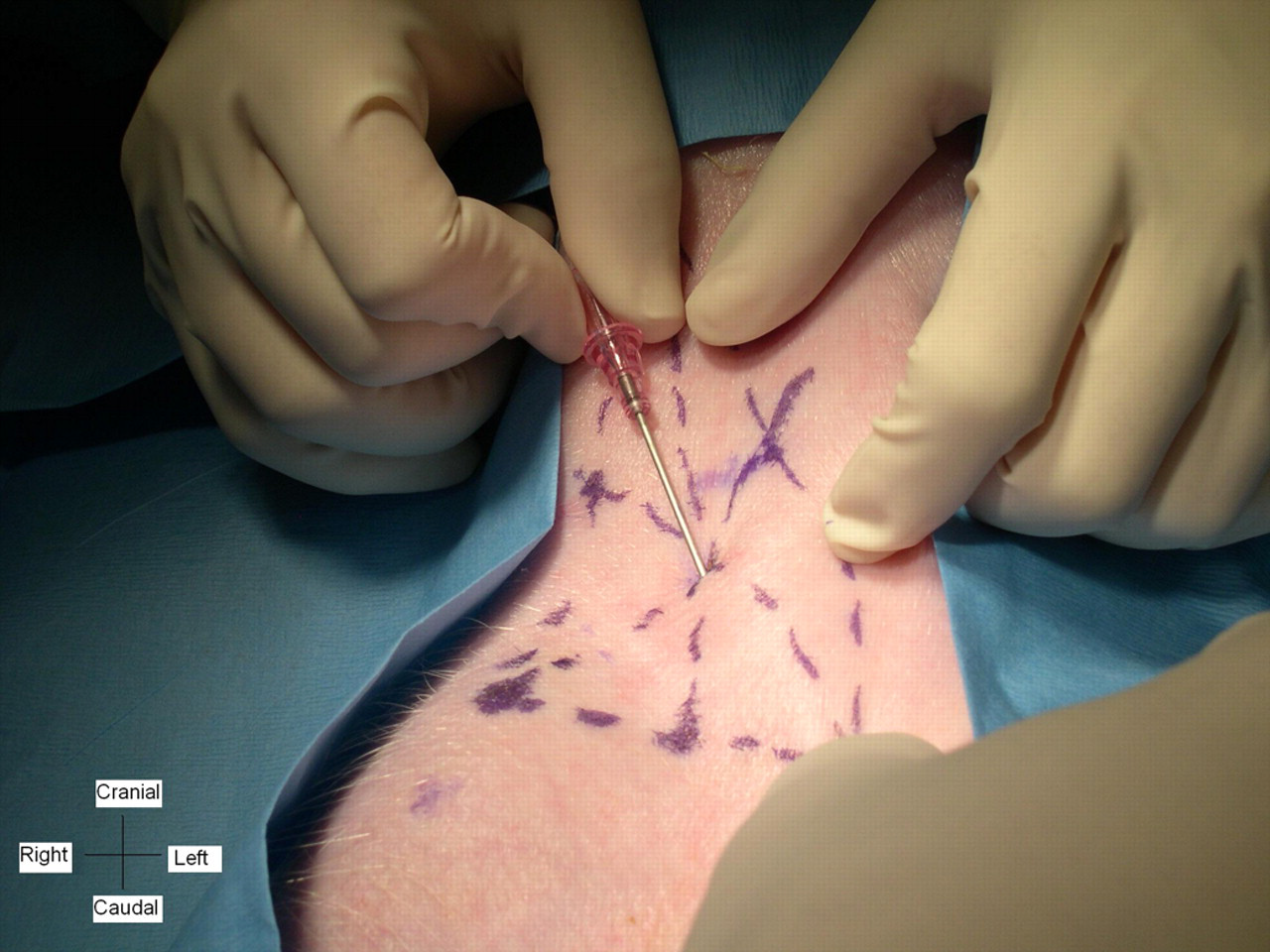
Photograph of a piglet in dorsal recumbency demonstrating the entrance of the introducer needle into the triangle drawn on the skin surface. The needle is then directed toward the assistant's thumb manually occluding the external jugular vein on the ipsilateral side of the manubrium
Results
Catheters were placed in piglets (n = 20) by one of three experienced veterinary technicians or one veterinarian. Our technicians commonly place jugular venous catheters in mature or adult swine to gain central venous access. However, no individuals in our surgery department had ever placed a CVL in young piglets. The median time required for the procedure was 52.5 min (range 25–120 min; n = 10), which included anaesthesia induction, surgery preparation, catheterization, placement verification with fluoroscopy, neck wrapping and endotracheal tube extubation. These placement times did not include postoperative periods, although recoveries were smooth and uneventful. Eighteen catheters were placed in the right external jugular vein and two were placed in the left one. Left-sided CVL catheters were inserted due to inadvertent puncture of the right carotid artery. The catheters remained in place for an average of seven days postoperatively. Catheter tip placement in the cranial vena cava just cranial to the right atrium was verified using fluoroscopy (Figure 3). Catheters were flushed once daily with heparinized saline (10 U/mL).
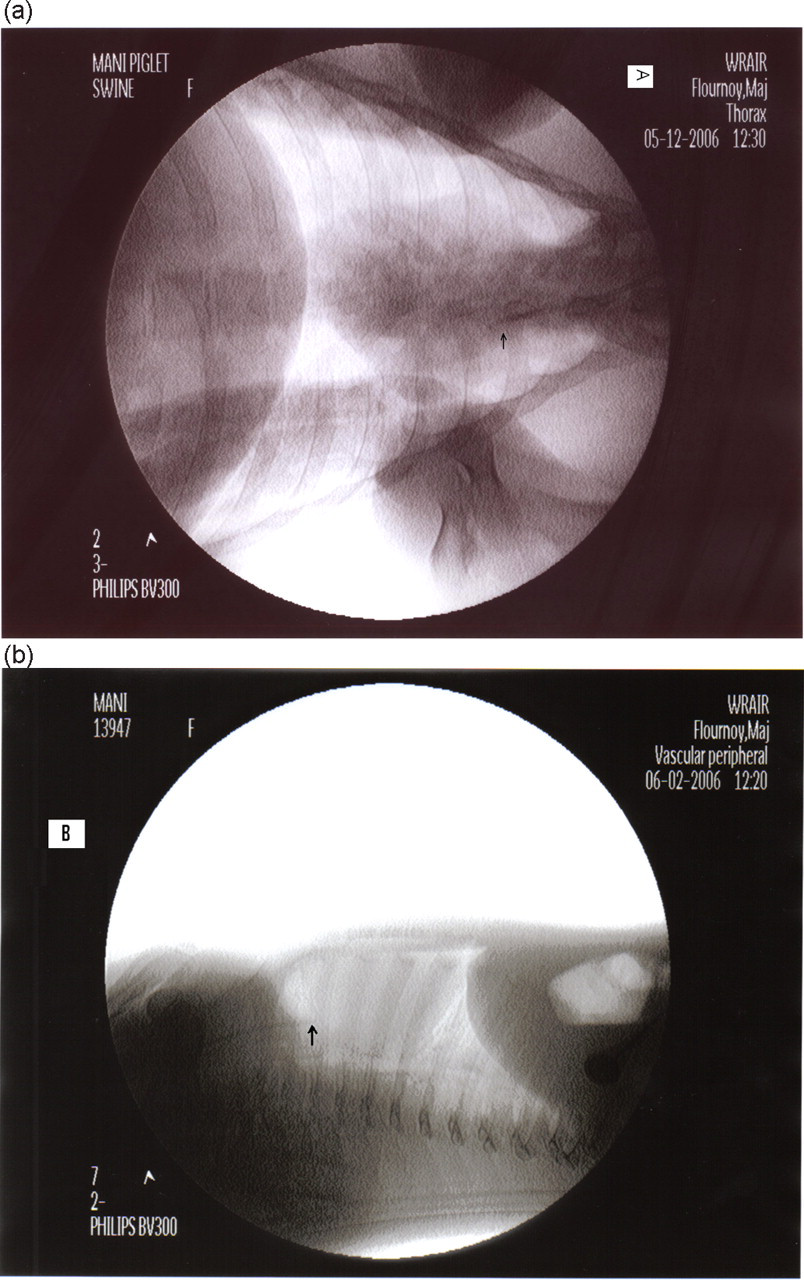
Postplacement radiographs used to confirm the location and placement of central venous line (CVL) catheters in the external jugular vein. (a) Ventral dorsal thoracic radiograph showing the tip of the CVL catheter just cranial to the base of the heart or the right atrium. (b) Right lateral thoracic radiograph showing the tip of the CVL catheter just cranial to the base of the heart or the right atrium
Minor complications that occurred during the catheterization procedure were carotid artery puncture, haematoma and premature clotting. Unintentional puncture of the carotid artery occurred twice in two separate piglets. Manual pressure was held on the venepuncture site both times and no adverse clinical signs were observed. Mild hematomas occurred in several of the pigs because of premature needle withdrawal; however, they produced no significant abnormalities or prevented subsequent successful catheterizations. About half of the numbers of pigs developed precipitous clotting prior to the insertion of the guide wire. Clotting mostly prevented the guide wire from passing easily into the lumen of the external jugular vein. During unexpected clotting, the needle was aspirated and flushed with a 10 mL syringe filled with heparinized saline (10 U/mL) to assist in removing clots and to prevent further clotting. After flushing, the guide wire was reinserted and the catheterization procedure continued. In addition, prothrombin time (PT) and activated partial thromboplastin time (aPTT) clotting times were conducted on all piglets preoperatively and no abnormalities were noted.
Discussion
The ‘Seldinger’ technique, a guide-wire-assisted cannulation method, was developed initially to gain access to jugular veins percutaneously for angiography. 29 With few modifications, this technique has become widely used for cannulation techniques in human and veterinary medicine. The Seldinger percutaneous technique utilizes a flexible guide wire inserted into the vasculature to guide the catheter into the vein or the artery. Although the techniques are virtually the same, the main differences between humans and animals are the preferred access sites. In humans, the IJV is larger in diameter and is the preferred site whereas in most animals the external jugular vein is more often accessed due its size and more superficial location. 10,30–32
The advantage of these percutaneous methods is that it causes less damage to soft tissues and blood vessels and reduces postoperative pain. However, disadvantages of percutaneous techniques have been reported in the veterinary literature to include catheter cost and longer placement times. 32 The catheter kit used in this study included all necessary items for catheter placement and costs approximately US$18. More expensive catheter kits have been previously reported. 25,32 We did not compare the placement times of our percutaneous method with a cut-down method; however, as the technicians became more familiar with the anatomical landmarks and methodology, the placement times decreased.
The percutaneous technique described here represents an efficient and reliable non-invasive technique for placing a jugular venous catheter in the external jugular vein of a piglet. Non-surgical catheterization of the jugular veins in young pigs has been previously described. 26–28 Although these reports cite short cannulation times, the methodologies only describe catheter placement briefly as they do not sufficiently depict palpable landmarks to narrow down the initial entry of the introducer needle. The triangle formed by our landmark method allows an individual to focus on a specific area of the ventral neck and helps prevent ‘blind’ venepunctures. After initial entry beyond the skin, the direction of the needle is also key to successful venepuncture. Previous reports do not specifically give the orientation to which the needle should be directed once the needle has advanced beyond the skin. We found that if the needle is directed towards the base of the manubrium, the vein is easily punctured. The depth of needle puncture should not be greater than 4 cm to prevent entry into the chest cavity, resulting in an acute pneumothorax. Unlike previously published methods, our procedure was performed aseptically. After anaesthesia induction we surgically prepared the venepuncture site, draped off the area with sterile draping using sterile gloves, and maintained stringent aseptic control of all items contained in the CVL kit to include strict control of the long guide wire (j-wire). Guide wire contamination can be a problem as bacteria can be directly introduced into the bloodstream if the wire is allowed to touch non-sterile or contaminated surfaces. We also infused a sodium channel blocker (e.g. bupivicaine) solution at the periphery of the catheter for local analgesia.
Very few complications were encountered during the catheterization procedures, one of which was inadvertent catheterization of the carotid artery. Puncture of the carotid artery during placement of CVLs is commonly reported in the veterinary and human literature. 21,25,33,34 Upon puncture of the artery by the introducer needle in two pigs, the needle was immediately removed and direct pressure was applied for 5 min. The animals remained stable during the procedures and did not experience any intra- or postoperative complications (e.g. haemorrhage, anaemia, cardiopulmonary complications). Mild to moderate swelling did develop over the ventral neck region; however, we chose to place the CVLs in the contralateral external jugular vein. Another commonly reported complication associated with jugular vein cannulation is haematomas. 17,32 Although haematoma formation occurred, it was not a common problem with this procedure. As stated previously, the two incidents of carotid arterial puncture resulted in mild swellings on the neck but they did not interfere with subsequent bleedings or produce postoperative complications. Dissection of the ventral neck after euthanasia revealed haematomas; however, postmortem examinations of the ventral neck region were not performed on every piglet in this study to assess haematoma formation or tissue damage. Other reported complications include pneumothorax, haemothorax, nerve damage, thromboembolism and infections. 14,17,21,32 The use of antiseptic-impregnated catheters and antibiotics are common strategies to decrease the incidences of thromboembolism and infections. 35,36 We did not encounter any of these previously reported complications in our piglets nor did we administer antibiotics perioperatively for prevention of catheter-related infections after surgery. Antibiotics are unnecessary to prevent catheter-related infections in swine if meticulous attention is paid to aseptic handling of the catheters during the intraoperative period and during sampling postoperatively. 2,37,38 One complication that we noted was that successful entry into the jugular vein with the introducer needle depended on head positioning. The short axis (dorsal–ventral) of the head was required to be perpendicular to the table for optimum needle entrance with subsequent catheterization. During CVL placement in adults, patients are typically placed in a Trendelenburg position with their head turned 45º toward the contralateral side. Optimum head angles have been reported when relying on external landmarks for location of neck blood vessels during jugular vein cannulation. 39 In non-human primates and infants, jugular veins can easily obstruct by simply turning the head to one side. 40,41 We did not measure head angles to obtain an optimum position for successful venepunctures; however, when the head was not perpendicular to the table, there was a trend toward multiple attempts prior to successful venepuncture. On the smaller weight piglets, occasionally, the j-wire would not pass easily into the external jugular vein. This problem was likely due to the size of the vasculature. To facilitate guide wire insertion in our smaller piglets, we simply reversed the j-wire and inserted the straight end through the introducer needle.
Knowledge of anatomy is essential for efficient and skilful placement of central venous catheters, especially in seriously ill and young patients. However, anatomical knowledge and technical skills vary among veterinary medical personnel. Bony prominences or structures are easily palpated and identified with minimal training. These prominent landmarks can be used as reference points for imaginary lines, for two-dimensional drawing on the surface of the skin, and ‘spatial mapping’ of the physiological structures of interest. These drawing or mapping techniques (e.g. triangulation) are commonly used in human and veterinary medicine to identify subdermal structures (e.g. nerve, vasculature and epidural space) that are invisible or not easily palpable. 42–45 In veterinary medicine, the jugular furrow or vascular pulsations are most often used to locate the external jugular vein for CVL placement or venepuncture in mature animals. In our young piglets undergoing general anaesthesia, we found pulse and furrow identification difficult. Thus, we used three prominent and easily identifiable bony landmarks to develop a technique for CVL placement in our young piglets, one of which was the manubrium or the broad part of the sternum, which is very prominent and easy to palpate in swine. The other bony landmarks in our technique were the ramus of the mandible and the point of the shoulder. The mandible (or caudle mandibular angle) is commonly used to identify the external jugular vein for catheterizations in dogs and cats. 31
Although we were successful in catheterizing the external jugular vein in all the piglets in a reasonable amount of time, we did not measure the specific amount of time from initial needle entry until the catheter was secured to the skin with the Statlock device. Short but different timeframes would have resulted for each pig as several technicians participated in catheterizations and skill levels varied among individuals. Also, we did count the number of ‘failed’ venepunctures before the external jugular vein was successfully accessed in order to obtain a first attempt rate that is commonly reported in the human literature. This rate may not be as critical to report in veterinary medicine as most laboratory animals are anaesthetized for CVL placement compared with humans, who may be awake or only sedated for IJV catheterization. Another limitation of the study was that the timeframe catheters remained in the piglets. There were no reported infections or premature dislodgements in any of the animals during that week. It is possible that the catheters could have lasted longer than a week or infections may have developed if the catheters remained longer; however, this study supported another ongoing project that limited the number of piglets and the length of time CVL catheters remained in the animals.
In summary, we used a reliable and efficient triangulation methodology to percutaneously catheterize the external jugular vein in young piglets using three easily identified and palpable landmarks. The catheters (CVL) were inserted aseptically by several of our veterinary technicians with minimal training and anatomical familiarization. Catheters were left in the external jugular vein approximately a week without any infections, occlusions or dislodgements.
NOTE
The views of the authors do not purport to reflect the position of the Department of the Army or the Department of Defense (AR 360-5, Para 4-3).
Footnotes
ACKNOWLEDGEMENTS
We would like to express our sincere thanks to Dr Marti Jett, Chief of Division of Molecular Pathology, for her constant support in this project. We also thank the veterinary technicians of the Department of Veterinary Surgery for helping us complete this project. This research was supported by grants (USARMC) and seed funding (ILIR).
