Abstract
Piglets are considered to be suitable animal models for predicting the pharmacokinetics and pharmacodynamics (PK/PD) of test drugs for potential use in the paediatric population. Such PK/PD studies require multiple blood and urine samplings. The goal of the present study was to determine a suitable blood collection strategy applicable in the youngest age categories of six days, four weeks and eight weeks of age, as well as a urine collection technique for male piglets in the same age categories. Blood was collected either by a surgically-placed jugular vein catheter (six days old [n = 4] and four weeks old [n = 2] piglets) or by direct venepuncture of the jugular vein (four weeks old [n = 2] and eight weeks old [n = 4] piglets). A non-invasive method for total volume urine collection in male piglets was also developed using a urine pouch. No specific complications were encountered during anaesthesia or surgery for jugular catheter placement. After a 24 h recovery period, urine and blood were easily collected without technical complications. One piglet was humanely killed at week 2 because of septicaemia. Histological analysis of both veins in all four piglets revealed negligible damage to the blood vessel wall. In conclusion, the presented techniques for blood (jugular catheter and direct venepuncture) and urine collection (pouches) are suitable for PK/PD studies in piglets.
The human paediatric population is often neglected during the process of drug development. This omission is due to a variety of factors, including biological challenges resulting from physiological changes and maturation. Moreover, ethical concerns of experimenting within this population result in a lack of complete pharmacokinetic (PK) and pharmacodynamic (PD) data. 1
The piglet has been proposed as a suitable paediatric animal model for PK/PD research because of the anatomical and physiological resemblances between pigs and humans.2–4 Complete PK/PD modelling requires multiple blood and urine samplings over a short period of time, which demand appropriate collection techniques. Several techniques for placing intravenous catheters in pigs have been described in the scientific literature.5–12 Although intravenous catheterization of 8- and 10-week-old pigs is routinely conducted in different research facilities, such catheterization in neonatal and younger pigs is challenging. Moreover, most research papers reporting the use of intravenous catheters in these age categories do not provide adequate details of the catheterization protocol, limiting the ability of other researchers to adopt these techniques.13–18 Palmisano et al. 19 described intravenous catheterization in nine 3–4-day-old piglets using vascular access ports that were surgically placed three days after birth and were maintained over the following 54 days. It was possible to collect blood from all piglets at different time points, but during the study by Palmisano et al., 19 technical complications occurred with four out of nine catheters. Caroll et al. 8 performed a non-surgical cannulation technique of the jugular vein in two-week-old piglets. The intravenous catheters remained in place for 2–5 days. This technique was successful in all animals but was not evaluated in piglets aged less than two weeks. Flournoy and Mani 12 developed a method for percutaneous external jugular vein catheterization in four-week-old piglets using a triangulation technique that uses palpable landmarks to facilitate the percutaneous insertion of the catheter. However, the determination of anatomical palpable landmarks in younger piglets may be difficult due to their immature musculoskeletal development, as also reported in human infants. 20 Overall, most of these intravenous catheterization procedures were performed in animals in various age categories and technical complications were frequently encountered. Moreover, the reuse of piglets for blood sampling at different ages was not described.
In addition to blood sampling, urine collection is often of interest for both PK and PD studies. Wykes et al., 13 Bauer et al. 15 and Kansagara et al. 17 described the insertion of a urinary catheter in male and female piglets, aged 1–3 days, for 12–27 h and for 12 h, respectively. Wijtten et al. 21 used non-invasive urine pouches to collect the voided urine (at 2, 8 and 18 h post-administration for one or more days) at the age of 3–5 weeks. In these studies, urine collection was successful, but the use of these techniques at different ages was not investigated, and the usefulness of these techniques in age-related PD studies remains to be determined. Nguyen et al. 14 and Chin et al. 16 performed cystostomy in 7–10-day-old piglets, but no details about their procedures were provided. Shoveller et al. 22 and John et al. 18 also described urine sampling in piglets but did not mention the exact procedures used. The major reason for the lack of a straightforward and easy-to-apply urine collection technique is the anatomy of the urogenital system of the pig; 23 both the glans (corkscrew shape) and the corpus (sigmoid flexure) of the pig penis makes it difficult to catheterize the male urethra. 24 The female urethra can be catheterized with a Foley catheter, but localization of the external urethral orifice can be challenging.25,26 Additionally, the suburethral diverticulum in female piglets renders catheter placement even more difficult. 26 Furthermore, long-term catheterization of the urethra can lead to complications such as urinary tract infections, urinary tract damage, and catheter defects. The method of urine sampling depends on the PK or PD parameters needed for the PK/PD modelling. Certain studies require quantitative urine collection (e.g. to determine the cumulative urinary drug excretion), whereas others only need spot samples (e.g. to investigate the effect of drugs targeting the kidneys and urinary tract). Bacteriological and faecal contamination may also affect the choice of a particular collection method. Both invasive (cystocentesis, catheterization and suprapubic cystostomy) and non-invasive techniques (pouches, collecting spontaneous urination, manual emptying of the bladder and metabolic cages) can be used to determine PK/PD parameters in urine. Detailed reports about a standardized, easy-to-apply method for urine collection in newborn and growing piglets are scarce. As a result, there is a need for safe, reproducible and easy-to-perform techniques for collecting blood and urine in piglets.
The aim of the present pilot study was to optimize blood and urine collection techniques to adequately perform repetitive PK/PD studies in piglets. Therefore, a longitudinal study was done in four piglets starting when they were six days old until they were eight weeks old. The age groups tested (six days, four and eight weeks) correspond to the same important age categories of the human paediatric subgroups (neonate, infant and child). 27
Materials and methods
Animals
This study was approved by the ethical committee of the Faculties of Veterinary Medicine and Bioscience Engineering of Ghent University (EC 2014/47). Care and use of animals was in full compliance with the most recent national legislation (Belgian Royal Decree of 29 May 2013) 28 and European Directive (2010/63/EU). 29 The study comprised four homozygote stress-resistant piglets with a common health status (less sensitive to malignant hyperthermia, landrace × large white), two females (piglets 1 and 2) and two intact males (piglets 3 and 4) which were purchased from Seghers Hybrid (Wuustwezel, Belgium) when they were five days old (2.10 ± 0.07 kg body weight [BW]). Before surgical procedures were performed, the animals were group-housed in rescue decks (0.9 m × 1.40 m) (Provimi, Rotterdam, The Netherlands) (five days to three weeks old) and standard pig stables with partially slatted floors (2.30 m × 2.40 m) (three to eight weeks old). The piglets were housed individually after catheterization to prevent catheter removal. Group housing was reintroduced after removal of the intravenous catheters. The piglets had ad libitum access to milk at 5–21 days of age (RescueMilk®; Provimi). At day 21, milk was gradually reduced simultaneously with the introduction of pig starter feed and drinking water (HeavyPig 1®; Provimi). At 28 days of age, the piglets had ad libitum access to the pig starter feed and drinking water. Natural light was provided by translucent windows, and the stable temperature was 23.3 ± 1.1℃. Higher temperatures were obtained in the rescue decks using heating lamps. One day prior to surgery, a cotton towel was added to the decks and stables. The piglets were given the towel after surgery to mimic the smell of the other piglets when they were singly housed. The piglets could also see (Plexiglas®), hear and smell each other. The stables were enriched with hanging chains, rubber toys and balls of different sizes. The environmental enrichment was rotated daily.
Experimental design
The general overview of the experimental design is presented in Supplementary Figure 1 (see online at http://journals.sagepub.com/home/lan). Briefly, a double-lumen catheter was surgically placed in the jugular vein of the four six-day-old piglets (see Catheterization section below), and a urine pouch was additionally placed around the prepuce of the two male piglets (see Urine collection section below). After one day of acclimatization, multiple urine and blood collections were performed within a 12 h time period. The intravenous catheters and urine pouches were removed after the last collection point. At four weeks of age, the catheterization procedure was repeated in two of the four piglets (one male and one female; randomly selected) in the opposite vein. Blood collection from the remaining piglet was performed by direct puncturing of the opposite jugular vein (see Venepuncture section below). Urine was also collected in urine pouches from the same male piglets. At eight weeks of age, blood collection was performed by venepuncture in all animals instead of via catheters, and urine was again collected in pouches. After the last sampling point, the piglets were killed, and the jugular veins were collected for histological evaluation (see Histological parameters section below).
Anaesthetic protocol
The six-day-old piglets were not fasted before the surgical intervention to reduce the risk of hypoglycaemia. The four-week-old piglets were fasted 12 h prior to surgery to prevent food regurgitation. After intramuscular premedication in the lumbar muscles with morphine (0.1 mg/kg BW) (Morphine HCl®; Sterop, Anderlecht, Belgium), midazolam (0.5 mg/kg BW) (Dormicum®; Roche, Vilvoorde, Belgium) and ketamine (10 mg/kg BW) (Nimatek®; Dechra, Lille, Belgium), a catheter was placed in the marginal ear vein (24 standard wire gauge (SWG) in the six-day-old piglets and 22 SWG in the four-week-old piglets). General anaesthesia was then induced using propofol (Propovet®; Abbott Animal Health, Maidenhead, UK), administered slowly intravenously until endotracheal intubation was possible (cuffed endotracheal tube, 2.5–3 mm internal diameter (ID) in six-day-old piglets and 6–6.5 mm ID in four-week-old piglets). Anaesthesia was maintained with sevoflurane in an oxygen/air mixture (inspired oxygen fraction 60%) administered in a semi-closed circle system (AV-1; Dräger, Lübeck, Germany). Monitoring included pulse oximetry, inspired and expired gas analyses, electrocardiography and measurement of body temperature and blood pressure (oscillometry) using a multiparameter monitor (S/5, D-LCC15-03; Datex Ohmeda, OR, USA). During surgery, the body temperature was stabilized between 37℃ and 39℃ using a resistive polymer warming device (Hot Dog®; Augustine Biomedical, Eden Prairie, MN, USA).
Catheterization procedure of the left jugular vein at four weeks of age.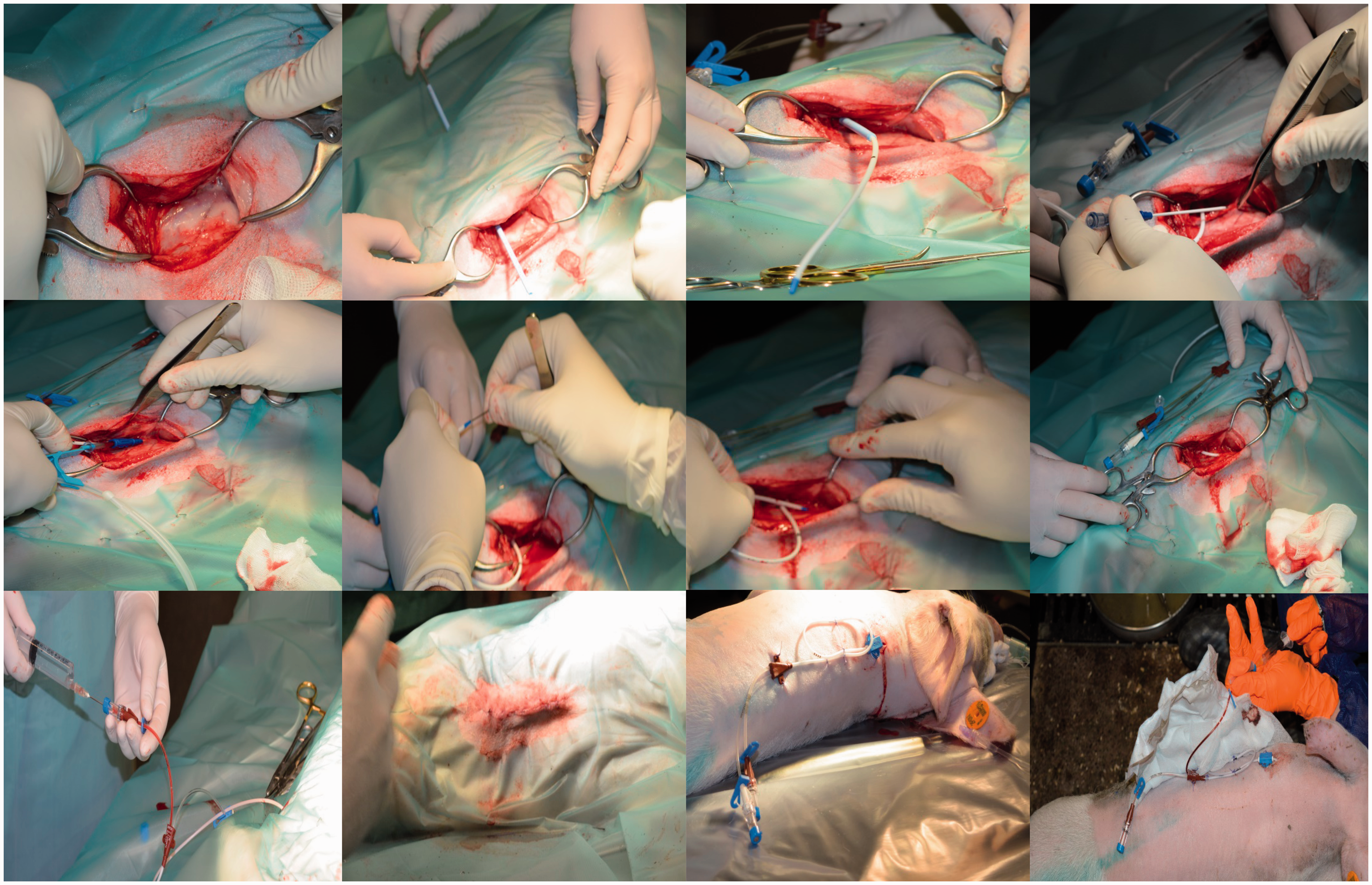
Blood collection
Catheterization
Catheterization of the right and left jugular veins was performed at six days (n = 4) and four weeks (n = 2) of age in the same piglets, respectively. The most optimal type and length of the intravenous catheter were first determined in cadavers. A double-lumen catheter (two-lumen central venous catheterization set; Arrow International, Cleveland, OH, USA – at six days: 4 Fr, 30 cm, at four weeks: 7 Fr, 60 cm) was surgically placed according to the procedure described by Gasthuys et al. 11 They performed the technique successfully in older and heavier pigs (approximately 8–10 weeks old and 20–25 kg BW) but did not test the procedure in younger piglets. In the current study, the external jugular vein was surgically exposed, and the heparinized catheter was introduced using the Seldinger technique. In brief, the jugular vein was punctured with a trocar, and a guide wire was advanced through the trocar towards the vena cava. The catheter was advanced over the guide wire, and the guide wire was then removed. The catheter was tunnelled towards the neck region, immediately cranial to the withers, and attached to the skin with sutures (Figure 1). After catheter placement, the incisions were closed in a three-layer continuous pattern, and adhesive dressings were applied over the wounds. Pre-operatively the catheters were flushed at regular time intervals with sterile diluted heparin in saline solution (six days: 0.04% v/v, four weeks: 1% v/v – Heparin Leo® 5000 IU/mL; LEO Pharma A/S, Ballerup, Denmark) to verify the functionality of both lumens. After one day of acclimatization, blood was sampled at 12 time points (5, 15 and 30 min; and 1, 1.5, 2, 3, 4, 6, 8, 10 and 12 h) within a 12 h time period, drawing one millilitre of blood each time from the distal lumen and flushing the catheters afterwards.
Venepuncture
Blood was also collected by direct venepuncture of the jugular vein in the four-week-old (n = 1) and eight-week-old (n = 3) piglets. One millilitre of blood was sampled with a Venoject system (Venoject® multisampling needle; Terumo, Leuven, Belgium) at 12 time points (5, 15 and 30 min; and 1, 1.5, 2, 3, 4, 6, 8, 10 and 12 h) within a 12 h time period.
Urine collection
A non-invasive urine collection technique, based on the method described by Wijtten et al.,
21
was tested in male piglets at eight days and four and eight weeks of age (Figure 2). Urine collection in both studies was performed using human stoma rings and pouches (Esteem Synergy Uro®, 48 mm, kindly donated by ConvaTec, Braine-l’Alleud, Belgium). At the end of anaesthesia, the piglets were shaved around the preputial opening and an adhesive spray (Medical Adhesive 7730®; Hollister, Waver, Belgium) was applied. The stoma rings were then placed around the prepuce. After 3 min, the rings were fixed on the preputial skin, and Leukoplast® (BSN Medical, Hamburg, Germany) was used to tape around the collar. The pouches were attached to the ring during the urine sampling period according to the manufacturer’s instructions. The urine pouches were additionally fixed to the skin with Hypafix® (BSN Medical) bandages. Multiple urine collections (four time points, with renewal of the urine pouches at 0, 3, 6 and 9 h) were performed within a 12 h time period.
Urine collection technique in male piglets at four weeks of age.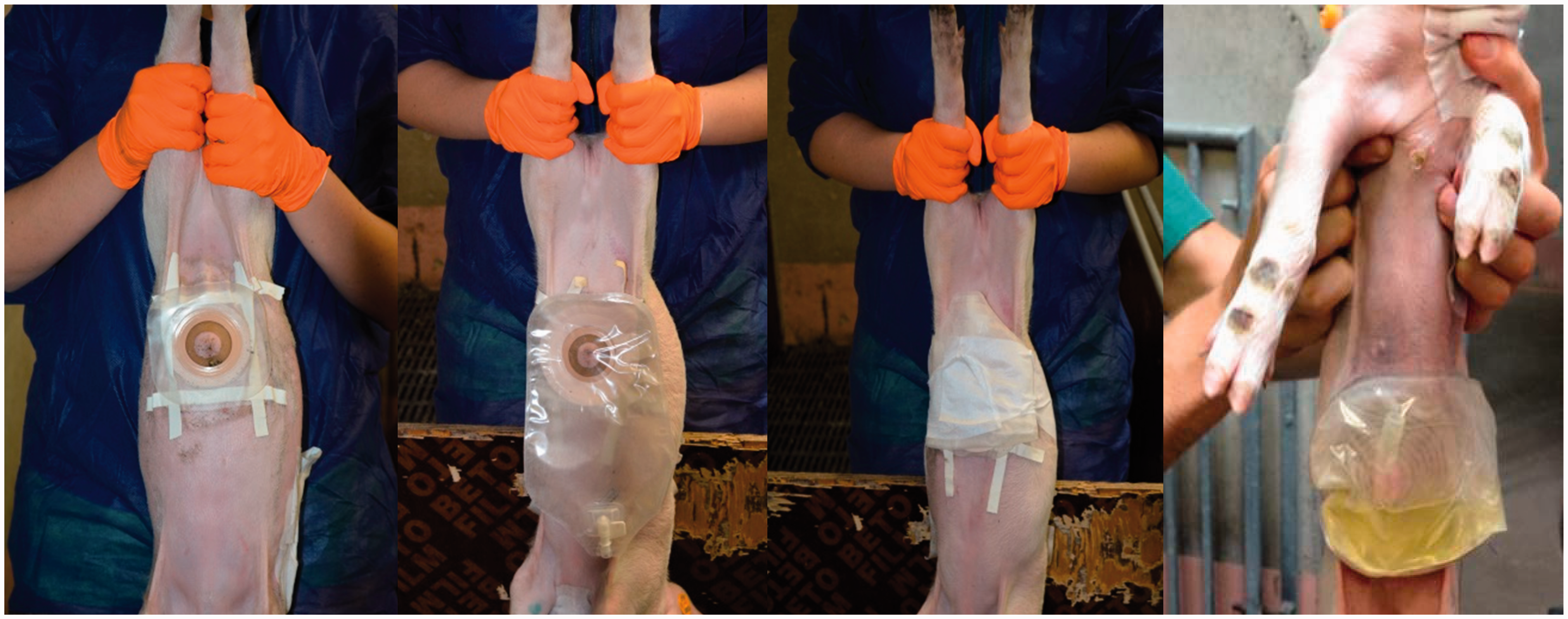
Bacteriological, clinical and histological parameters
On arrival at our research facility, the piglets’ noses and perinea were swabbed for bacteriological screening (blood agar and methicillin-resistant Staphylococcus aureus [MRSA] selective plate). A susceptibility test according to the Clinical and Laboratory Standards Institute (CLSI) standard regulation was performed to determine the antimicrobial sensitivity of the bacteria. 30 Once the intravenous catheters were in place, the health and condition of the animals were monitored daily. Clinical parameters were assessed, including rectal body temperature, white blood cell (WBC) count (catheterized piglets, flow cytometry), BW, behaviour (feed and water consumption, clinical appearance) and wound healing. The intravenous catheters were flushed once daily with a sterile diluted heparin solution (six days: 0.04% v/v, four weeks: 1% v/v), and sealing caps and bandages were changed when needed. After the catheters were removed, the piglets were monitored daily by assessing their clinical appearance, observing their behaviour (feed and water consumption) and measuring their rectal body temperature. Euthanasia was performed by intramuscular injection with a mixture (0.22 mL/kg) of xylazine hydrochloride (Xyl-M 2%®; VMD, Arendonk, Belgium) and tiletamine–zolazepam (Zoletil 100®; Virbac, Barneveld, The Netherlands), followed by intracardiac injection with an overdose of sodium pentobarbital (sodium pentobarbital 20%®; Kela, Hoogstraten, Belgium). The jugular vein tissue of the four piglets was sampled, fixed immediately in 4% formaldehyde for 48 h, and dehydrated in an ethanol series before embedding in paraffin using a Microm tissue processor STP 420D (Prosan, Merelbeke, Belgium) and Microm embedding station EC 350-1 (Prosan). Sections (8 µm thick) were cut using a Microm H360 microtome (Prosan) and then stained with haematoxylin and eosin (HE) and Van Gieson’s stain. Subsequently, light microscopy (Olympus BX61, Olympus DP50 camera, Olympus Cell software series) was used to determine whether there was any inflammation of the jugular vein at the site of intravenous catheter insertion and the surrounding tissues.
Results
The nasal cavities of all the piglets and the perinea of three of the four piglets were MRSA positive. All recovered MRSA strains were resistant to penicillin G, ceftiofur, fluoroquinolones, tetracyclines and streptomycin, and sensitive to neomycin, gentamicin and potentiated sulphonamides.
Anaesthesia and surgery were well tolerated by all the piglets, although catheterization of the ear veins before induction of anaesthesia was challenging in the six-day-old piglets due to vasoconstriction and the small size of the ear veins. The mean total (range) anaesthetic time was 45 min (30 to 60 min) for the six-day-old and 37.5 min (30 to 45 min) for the four-week-old piglets. During surgery, the jugular veins were easily accessible for catheter placement, and no complications were encountered throughout the entire surgical procedure. The mean total surgical time (range) was 35 min (20 to 50 min) for the six-day-old and 30 min (25 to 35 min) for the four-week-old piglets. Recovery from sevoflurane anaesthesia was quick and quite smooth in all the piglets, although paddling was regularly observed before they regained sternal recumbency.
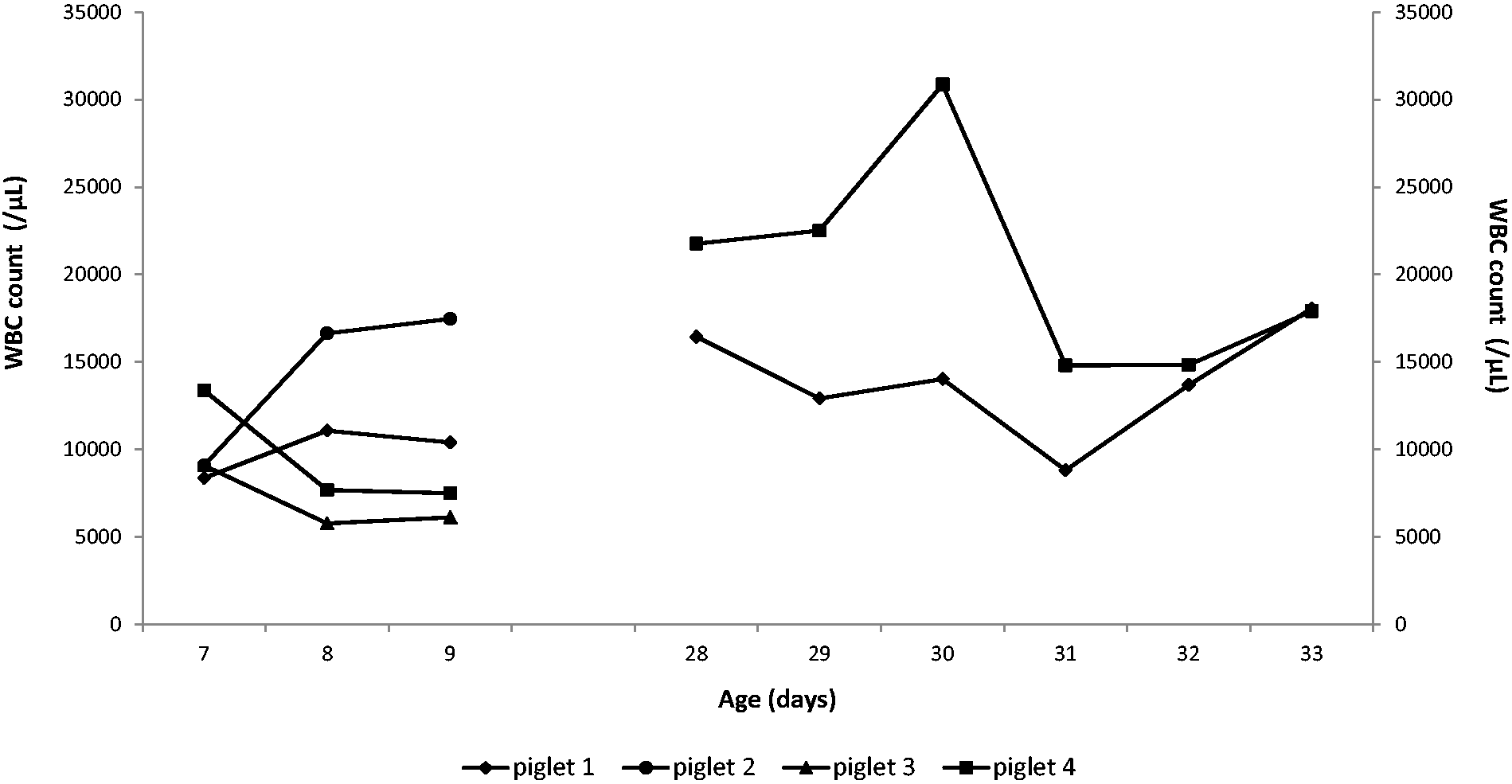
Figures 3a and b show the rectal body temperature and BW, respectively. The daily analysis of the WBC count of the catheterized piglets is shown in Figure 4. The growth rate of the male piglets (piglets 3 and 4) was consistent with the normal growth rate curve described by Swindle,
31
starting from 2.13 ± 0.02 kg at arrival and increasing to 8.24 ± 0.48 kg at four weeks and 21.80 ± 2.30 kg at eight weeks of age. One female piglet (piglet 1), however, was 3 and 8 kg heavier than the male pigs at four and eight weeks of age, respectively.
Evolution of rectal body temperature (a) and body weight (b) after surgical procedures in the six-day-old (n = 4) and four-week-old (n = 2) piglets. Evolution of white blood cell (WBC) count after surgical procedures in the six-day-old (n = 4) and four-week-old (n = 2) piglets.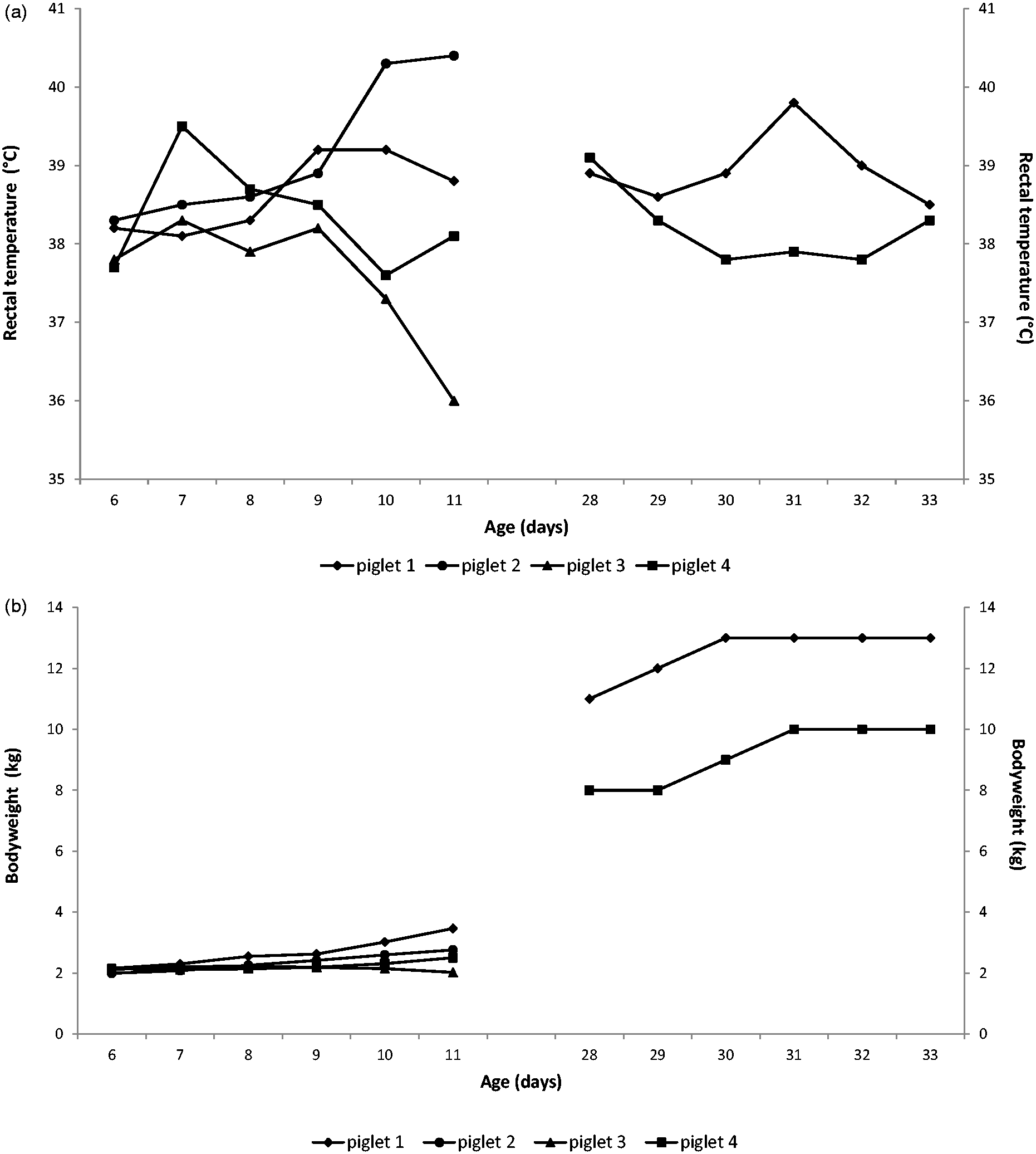
One piglet (piglet 2, catheterized only at six days of age) showed clinical signs of an infection three days after catheter removal (fever [>40℃], lameness and swollen joints). After unsuccessful antimicrobial treatment with sulphadiazine–trimethoprim (Borgal®; Virbac, Leuven, Belgium – intramuscular, five days, 15 mg/kg BW/day), the piglet was killed, and both jugular veins were collected for histological examination. The three remaining piglets (piglets 1, 3 and 4) showed no clinical signs of infection; with normal behaviour, including a good appetite and gain in BW during the entire experiment. Mild local swelling of the surgical wound was observed and disappeared over the course of the experiment.
The intravenous catheters were functional during the experiment (i.e. tested for 48 h after placement), and all blood collections were easily performed in all the animals. Sealing caps and bandages were changed when needed. The piglets did not show signs of pain or distress during the removal of the catheters. The wounds around the catheter entrance healed within days after the removal of the catheters. All blood collections by venepuncture of the jugular vein were performed with no complications, and only minimal signs of distress were observed.
The attachment and removal of the urine pouches were easily done, and no leaks were observed. Moreover, no skin irritation was noted during and after detachment of the ring system. This non-invasive technique allowed urine to be collected at different time points in the male piglets.
Histological analysis of the left and right jugular veins of the eight-week-old piglets, which were catheterized at the age of six days and four weeks, respectively, did not reveal any signs of inflammation. One of four right jugular veins was damaged, but remodelling of the blood vessel endothelial layer had occurred (piglet 3) (Figure 5).
Histological analysis of the left (a) and right (b) jugular veins. Remodelling (arrow) of the right jugular vein (c) and (d) (haematoxylin–eosin and Van Gieson staining) in a piglet after catheterization and venepuncture of the jugular vein.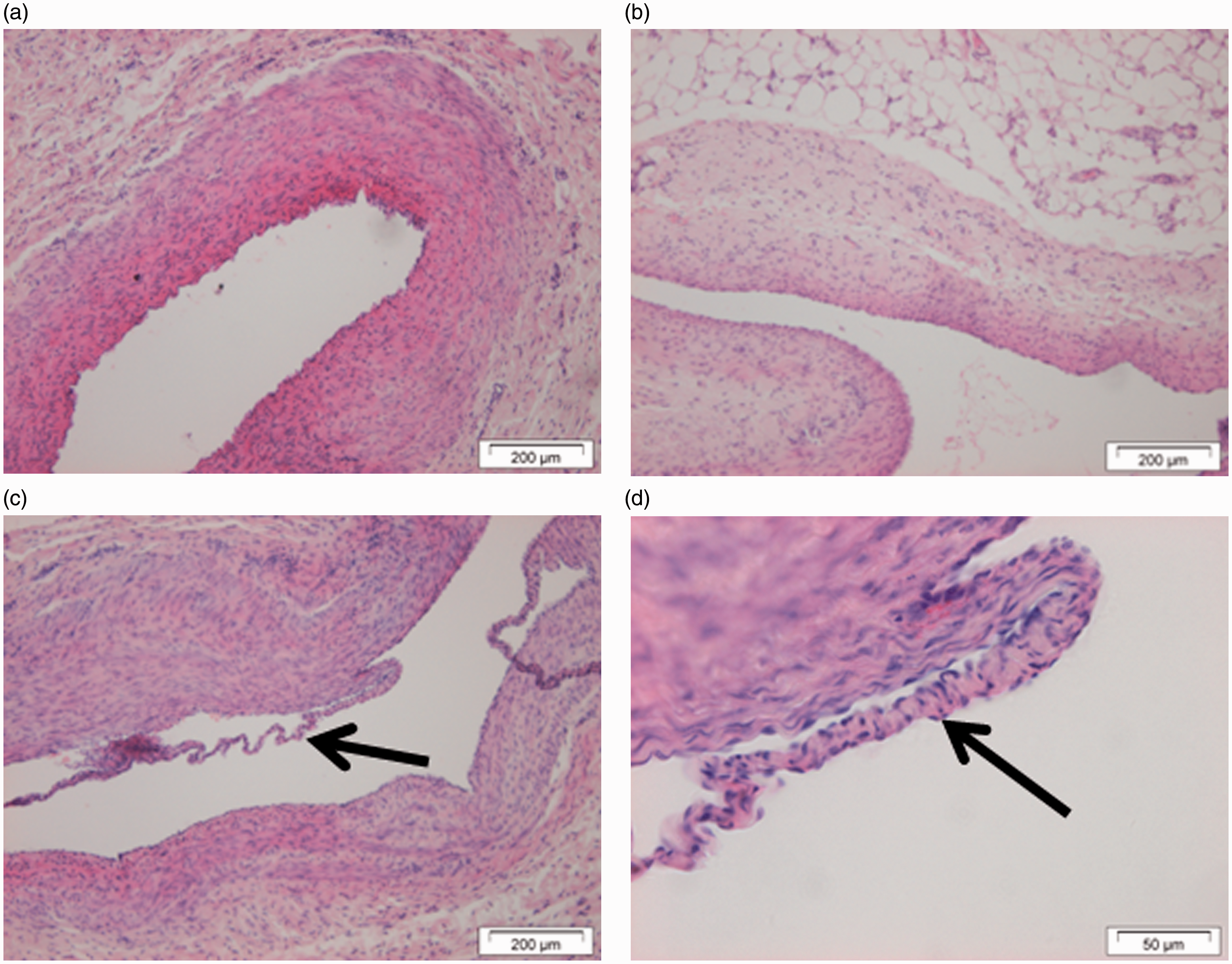
Discussion
The aim of the current study was to determine optimal blood and urine collection techniques for repetitive sampling in piglets.
Before performing any interventions, the piglets were bacteriologically screened for MRSA. This screening was necessary because of the age of the animals on arrival and the inherent risk of bacteriological contamination of the intravenous catheters.11,32.33 Indeed, the immaturity of the immune system of piglets increases their susceptibility to opportunistic infections such as MRSA. Moreover, MRSA is present in most conventional pig farms in Belgium. 34 Therefore, it is important to screen piglets from a new farm before starting trials, and to determine the antimicrobial sensitivity of the bacteria. Both Gasthuys et al. 11 and Palmisano et al. 19 have reported bacteriological contamination of intravenous catheters. Despite the strict hygienic precautions used in this study, one piglet showed clinical signs of septicaemia five days after intravenous catheterization. This infection may have been caused by the young age of the animal, i.e. the immaturity of the piglet’s immune system, the catheterization procedure and/or the environment. The susceptibility of piglets to opportunistic MRSA infections, especially at the age of six days, made it even more challenging to optimize the catheterization technique.
Blood collection from the six-day-old piglets was only performed with a commercialized double-lumen jugular vein catheter because the collection of multiple blood samples using the classic venepuncture technique was considered to be difficult and ethically less feasible in neonatal piglets. Catheterization was successful, despite the relatively large diameter of the catheter compared with the diameter of the jugular veins. At the age of four weeks, catheterization and direct venepuncture were compared to determine the most appropriate technique for blood collection.
Both these procedures are suitable for multiple blood samplings, however, each method has its advantages and disadvantages. Venepuncture does not require anaesthesia and surgery of the animals, however restraining the animals can induce minimal stress responses. Furthermore, damage to the jugular veins is possible. Blood collection with intravenous catheters is easy to perform and requires minimal restraint of the piglets.
In the eight-week-old piglets, only the venepuncture technique was tested. This procedure was chosen instead of a new surgical intervention because two of the three piglets had already been anaesthetized twice and both veins had already been catheterized. The piglets had also become familiar with human contact, so minimal restraint was needed. Histological analysis of the jugular veins did not reveal inflammation around the catheterized veins, and the damage to both veins was negligible. Taking these observations into account, it is possible to catheterize the jugular vein without severe damage. Consequently, recatheterization of the vein may be possible.
The urine collection technique used in the study is non-invasive and has multiple advantages compared with invasive techniques.13–17 First, no surgery is needed. The pouches can be easily applied to the abdomen with minimal restraint of the animal. Second, no urinary tract infections have been observed during the entire experiment. Finally, the technique can also be used in neonatal piglets. A disadvantage is the inability to attach the urine pouches to the perineum of female piglets, causing the pouches to be loosened, and consequently faecal contamination of the urine. Hence, a non-invasive technique for collecting urine from female piglets is still lacking.
In conclusion, the present study shows that the evaluated blood and urine collection (male piglets only) techniques can be used successfully in piglets. Both intravenous catheterization at the ages of six days and four weeks as well as direct venepuncture at the ages of four and eight weeks were successfully performed in piglets. However, catheterization of piglets is preferred since this is a more ethical method of collecting multiple blood samples. Consequently, porcine PK/PD studies can be easily performed in these different age categories.
Footnotes
Acknowledgements
This study was supported by the ‘Agency for Innovation by Science and Technology in Flanders (IWT)’ through the ‘SAFEPEDRUG’ project (IWT/SBO 130033). Assistance from L Gasthuys, A Devriendt, M De Veirman and the surgical staff during the animal experiments was gratefully appreciated. Special thanks to T Van Limbergen for assistance during the search for urine collection techniques in piglets. Also special thanks to J De Laat (Cargill) for demonstrating the urine collection technique.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the ‘Agency for Innovation by Science and Technology in Flanders (IWT)’ through the ‘SAFEPEDRUG’ project (IWT/SBO 130033).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
