Abstract
Significant advances have been made in our ability to assess pain and administer appropriate pain relief in laboratory animals. However, providing long-lasting analgesia using a route that does not involve animal restraint remains difficult. The objective of this study was to investigate whether oral administration of slow-release morphine or hydromorphone results in increased thermal nociception in laboratory rats. The results showed that 64 mg/kg morphine and 16 mg/kg hydromorphone induced comparable increases in foot withdrawal latencies for up to three hours postadministration; however, slow-release morphine increased response latencies for up to 11 hours. Whether these dose rates provide clinically effective pain relief has yet to be determined; however, these data suggest that using slow-release preparations could be an effective and highly practical method of elevating pain thresholds for a relatively prolonged period.
The reduction or elimination of pain in laboratory animals is a necessary and critical refinement for animal-based research and fulfils the 3Rs principles of Russell and Burch. 1 This should be a relatively simple refinement to implement because of the wide range of different analgesics currently available. 2 Despite significant progress in our ability to assess pain in rats and to administer appropriate doses of analgesics, a number of practical issues still need to be addressed. One such problem is the possibility that handling an animal to inject analgesics could be highly distressing and/or cause additional pain. This may be further exacerbated by the relatively short duration of action of many opioids, meaning that this procedure might have to be performed repeatedly. These problems could be addressed by administering analgesics through less stressful routes (e.g. orally) and by using long-acting preparations that reduce the need for repeated administration. Although a number of long-acting oral formulations of common analgesics are available for use in humans, very few have been tested in laboratory animals to determine whether they do actually possess prolonged antinociceptive properties.
Morphine and hydromorphone are μ-opioid (OP3) receptor agonists 3 that act directly on the central nervous system to relieve pain. Morphine is a naturally occurring opioid analgesic, whereas hydromorphone is a semi-synthetic drug that shares the same pharmacological properties as morphine. Both are routinely administered to laboratory and companion animals as injectable agents that provide relatively short periods (2-6 h) of pain relief.4–6
Oral morphine and hydromorphone slow-release preparations are widely used in humans to relieve postoperative and chronic pain over prolonged periods (up to 12 h). They are engineered to produce prolonged analgesia using different controlled-release technologies. The prolonged effects of hydromorphone (Palladone-SR®) are based on a controlled-release melt extrusion technology that uses granules containing hydromorphone hydrochloride. The coating of these granules dissolves away in the gastrointestinal tract, releasing the active ingredient slowly over a prolonged period. 7 The long-acting analgesic properties of morphine (MST Continus®) are based on a cationic exchange resin technology in which morphine is bound to the resin, so that the drug is released gradually when morphine is displaced by ions in the gastrointestinal tract. 7 There is currently no information on the effects of these or other slow-release oral preparations in laboratory or companion animals. The aims of this study were to develop a practical method of orally administering these slow-release preparations and to use changes in peripheral thermal antinociception as a simple preliminary method of evaluating their potential effectiveness for alleviating clinical pain in rats.
Materials and Methods
Animals and husbandry
Male and female Wistar (n = 10) and Lewis (n = 10) rats (150-200 g) obtained from a commercial supplier (Charles River, Kent, UK) were used in the pilot and main studies. The inclusion of more than one sex and strain in each treatment group allowed for greater generalization of results without any loss in statistical power. 8 An additional 20 Lewis rats (10 male, 10 female) were used in a supplementary study. On arrival, all rats were housed in standard RC1 cages (Tecniplast, London, UK) in groups of five according to sex and strain. Room temperature and humidity were controlled at 19 ± 2°C and 50 ± 10%, respectively, with a 12 h:12 h light-dark cycle (light: 07:00 to 19:00). Animals were given a commercial pellet diet (No. 3, SDS Ltd, Witham, Essex, UK) and water ad libitum. Wood shavings (‘Aspen’, BS and S Ltd, Edinburgh, UK) were used as bedding. Rats were acclimatized to this environment for a period of 10 days before the studies began. Body weights were recorded daily between 09:00 and 10:00 h. The animals were free from any common pathogens according to the FELASA Health Monitoring Recommendations. 9 All procedures were carried out in accordance with the Animals (Scientific Procedures) Act 1986 and approved by the local ethical review process. On completion of all experiments, rats were humanely euthanized with slow rising concentration of CO2 (in accordance with the recommendations of the report of the Newcastle CO2 consensus meeting 10 ).
Morphine and hydromorphone preparation and administration
Analgesics
Prolonged action morphine suspensions (MST Continus®, 30 mg sachets) and prolonged action hydromorphone capsules (Palladone-SR®, 8 mg capsules) were obtained from a commercial supplier (Napp Pharmaceuticals Ltd, Cambridge, UK). Morphine suspensions were prepared with 1 mL of water for injection to obtain doses of morphine in appropriate volumes for administration. Hydromorphone capsules were opened and the internal granules were weighed out according to the required doses. During the investigation, suitable oral doses (for rats) of morphine and hydromorphone slow-release preparations were calculated using data from Zheng et al. 11 Anderson et al. 12 and Souter and Fitzgibbon. 13
Gavage
Gavage was carried out using a 19-gauge stainless-steel gavage needle and a 1 mL syringe while each rat was held and the drug infused by the same person.
Syrup vehicle
Drugs administered in syrup vehicles were presented in 2 mL portions of syrup (Lyle's Golden Syrup, Tate & Lyle Sugars Ltd, Cheshire, UK), fed to the animals using a spatula (102 mm stainless-steel chattaway micro pattern spatula; VWR International Ltd, Leicestershire, UK) while they were temporarily housed in individual cages (RB3 cages, NKP, 45 cm × 38 cm, height 20 cm).
Hargreaves procedure (foot withdrawal latency)
Foot withdrawal latencies in response to noxious thermal stimulus were determined using the Hargreaves thermal nociceptive hind-paw withdrawal assay (UGO Basile Ltd, Biological Research Apparatus, Comerio, Italy) before (baseline) and at 1, 3 and 5 h after administration with drugs. The thermal stimulus was maintained at an intensity of 80%, increasing in temperature to a maximum of 55°C in 33.1 s. The thermal stimulus was cut off if the rat failed to withdraw its hind-paw by this time, as a safety measure. 14 Rats were habituated to the Hargreaves chamber for 15 min before testing and withdrawal latency was measured three times, using opposite paws with 5 min between tests. A mean of these three recordings was then calculated to give a single measure for each time point. The stimulus intensity and cut-off time were selected so that no tissue damage was induced in any animal during the procedure.
Pilot studies
A pilot study was undertaken to establish the most palatable vehicle for administration of these oral preparations. Five foodstuffs were tested that were thought to be highly favoured by rats with a sufficiently strong flavour to disguise the presence of the drug including syrup (Lyle's Golden Syrup), strawberry syrup (Askey's Strawberry Treat, The Silver Spoon Company, Peterborough, UK), raspberry jelly (Hartley's, Spalding, UK), marmite (Marmite Yeast Extract, Unilever Bestfoods UK, Crawley, UK) and chocolate spread (Nutella, Ferrero UK Ltd, Watford, UK). Approximately 3 mL of each foodstuff was placed into the animal's home cage in small feeding bowls for five nights to allow the animals to develop a taste and perhaps preference for these novel foodstuffs. The palatability of each foodstuff was assessed in 12 rats (6 Lewis, 6 Wistar; 3 male and 3 female of each strain). Each individual was placed into a small cage alone for 10 min and given approximately 2 mL of each of the foodstuffs in a random order over a period of five days. The foodstuffs were either placed in a small food bowl or fed via a spatula through the cage bars. Gavage was not considered an appropriate route of administration due to potential distress caused by the necessary restraint and the administration. For those foodstuffs found to be palatable, the palatability with the addition of either morphine or hydromorphone was then tested to ensure that their inclusion did not alter consumption of the foodstuffs. 15
A second pilot study was undertaken to establish likely effective oral doses of morphine and hydromorphone. Twelve rats (6 Lewis, 6 Wistar; 3 male and 3 female of each strain) were divided into three treatment groups. The positive control group (n = 4) was given a subcutaneous injection of 5 mg/kg morphine. This dose has been shown to be effective in thermal antinociceptive tests. 16 The morphine group (n = 4) received an oral dose of 16 mg/kg by gavage to ensure that the correct dose was achieved. The hydromorphone group (n = 4) received an oral dose of 4 mg/kg by placing the granules in syrup, which was then fed to the rats as the granules were too large to be administered by gavage. All drugs were administered between 10:00 and 11:00 h. If no change in withdrawal latencies was observed with the oral doses of morphine and hydromorphone, then the doses were doubled and administered after a 48 h washout period. This procedure was repeated until significant changes in withdrawal latencies were observed.
Main study
Morphine (64 mg/kg) and hydromorphone (16 mg/kg) were administered to 10 rats (Wistar: 3 male and 3 female; Lewis: 2 male and 2 female) in a crossover design so that all rats received both drugs over two treatment days separated by a 48 h washout period. This washout period was chosen based on a short pilot study that demonstrated that foot withdrawal latencies to a thermal stimulus had returned to baseline levels in animals receiving both morphine and hydromorphone. The treatments were administered according to a randomized block design. Drugs were administered in 2 mL syrup vehicles via spatulas to singly housed rats between the hours of 10:00 and 11:00 in the morning. Thermal nociception was measured using the Hargreaves apparatus as described earlier.
Further investigation of slow-release morphine
In order to further investigate the duration of the effect of the slow-release morphine preparation (64 mg/kg), an additional group of Lewis rats (n = 20: 10 male and 10 female) were administered either morphine (n = 10) or saline (n = 10) with equal number of males and females in each treatment group. The treatments were administered according to a randomized design. Drugs were administered in 2 mL syrup vehicles via spatulas to singly housed rats between the hours of 10:00 and 11:00. Thermal nociception was measured using the Hargreaves apparatus before (baseline) and 1, 3, 5, 7, 9 and 11 h postadministration.
Statistical analysis
The data were normally distributed according to a Shapiro-Wilks test and analysed using parametric analysis with SPSS (Version 14, SPSS Inc, Chicago, IL, USA) software. A repeated-measures analysis of variance (ANOVA) was conducted on the paw withdrawal latencies. This included treatment, sex, strain and cage number as between-subject factors and the recording times (pre- and postadministration) as within-subject factors. Post hoc analysis of the withinsubject factors was conducted using both simple and repeated contrasts. Comparisons of morphine and hydromorphone treatment data were made using area-under-curve (AUC). AUC was calculated for each individual from the postadministration time points (e.g. 1, 3, 5, 7, 9 and 11 h) and compared between the treatments. AUC data were analysed using a one-way ANOVA, with treatment, sex, and strain being included as factors.
Results
Overall no adverse effects (e.g. abnormal behaviour, excitation, agitation, etc.) were observed at any of the time points with either the morphine or hydromorphone preparations. Some of the morphine- and hydromorphonetreated animals exhibited signs of mild sedation (decreased activity, etc.); however, this did not appear to alter foot withdrawal latencies.
Pilot studies
Syrup was found to be the most preferred foodstuff as all of the rats consumed up to 2 mLof syrup when itwas presented. The rats seemed unwilling/incapable of eating quantities of foodstuffs larger than approximately 2 mL. Feeding the foodstuffs via a spatula through the bars of the cage was the only method accepted by all animals. Therefore, 2 mL portions of syrup via a spatula through the bars of the cage were used to deliver the analgesics throughout the main study.
It was established that the effective doses were 64 mg/kg morphine and 16 mg/kg of hydromorphone. These doses generated detectable and significantly increased withdrawal latencies among all rats. These doses were therefore used throughout the main study.
Main study
Rats administered morphine showed significant differences in withdrawal latencies compared with baseline at 1 h (P = 0.001), 3 h (P = 0.000) and 5 h (P = 0.002) after administration (see Table 1). No other factors tested (e.g. sex, strain) had a significant effect on foot withdrawal latencies either alone or through interactions (Figure 1).
Mean withdrawal latencies after administration of oral analgesics (±;1 SD)

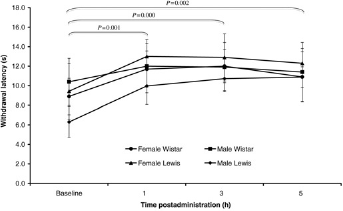
Comparison of withdrawal latencies of Wistar and Lewis rats receiving morphine 64 mg/kg orally (±1 SD). Sex and strain did not have a significant effect on withdrawal latencies
Rats receiving hydromorphone showed significant differences in withdrawal latencies compared with baseline at 1 h (P = 0.000), 3 h (P = 0.02), but not at 5 h after administration. The effect of hydromorphone appeared to peak at 1 h (P = 0.000), began to tail off at 3 h (P = 0.02) and was insignificant at 5 h (Table 1). In addition to the effect of hydromorphone administration on foot withdrawal latencies, the sex of the rats (P = 0.03) and sex and strain interaction (P = 0.003) also had a significant effect on foot withdrawal latencies (Figure 2). Female Lewis rats had higher withdrawal times over every time point compared with male Lewis rats and both male and female Wistar rats. The comparison between morphine and hydromorphone showed no significant difference in mean withdrawal latencies between the treatment groups in the postadministration observations for male and female rats (Figure 3). This is further demonstrated by a comparison of AUC as no significant difference was found between the treatment groups for the foot withdrawal latencies in the postadministration observations. However, strain (P = 0.03) and a sex-strain interaction (P = 0.012) had a significant effect on AUC of foot withdrawal latencies. Female Lewis rats had higher AUC of foot withdrawal latencies compared with male Lewis rats and both male and female Wistar rats (see Figure 4).
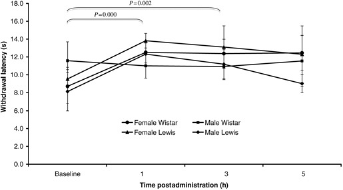
Comparison of withdrawal latencies of male and female Wistar and Lewis rats receiving hydromorphone 16 mg/kg orally (±1 SD). Sex and a sex-strain interaction had a significant effect on withdrawal latencies (P = 0.03 and P = 0.003, respectively)
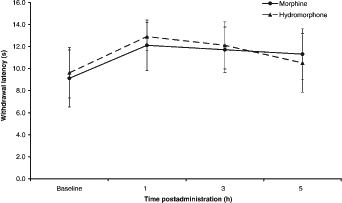
Comparison of mean withdrawal latencies from baseline over time after administration in male and female rats with either 64 mg/kg morphine or 16 mg/kg hydromorphone (±1 SD)
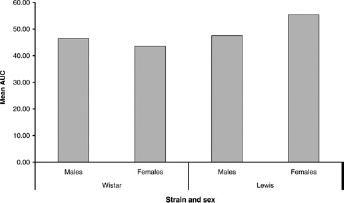
Comparison of mean area-under-curve (AUC) for foot latencies postadministration of 16 mg/kg hydromorphone between sex and strain. However, a sex and a sex-strain interaction had a significant effect on AUC (P = 0.03 and P = 0.012, respectively)
Further investigation of slow-release morphine
There was a significant difference in foot withdrawal latency between the two treatment groups, with the significantly longer latencies being observed in the animals receiving the slow-release morphine compared with saline (P = 0.000). There was no significant effect of either sex or cage on the foot withdrawal latencies between the treatments. The significantly longer foot withdrawal latencies associated with slow-release morphine compared with saline occurred at 3, 5, 7, 9 and 11 h postadministration (P = 0.001, P = 0.000, P = 0.000, P = 0.000, P = 0.000 and P = 0.000, respectively) (see Figure 5). This significant treatment effect is further demonstrated by a significant difference in the AUC of foot withdrawal latencies for the postadministration observations between the two treatment groups, with the AUC being significantly greater in those animals receiving the slow-release morphine compared with saline (P = 0.000) (see Figure 6). However, postadministration time point also had a significant effect on foot withdrawal latencies in both the saline- and morphine-treated animals (P = 0.000, P = 0.001, respectively). For the saline-treated group, the latencies were significantly lower than baseline at 7, 9 and 11 h postadministration (P = 0.001, P = 0.01 and P = 0.001, respectively) but not significantly different from baseline at 1, 3 and 5 h postadministration. For the morphine-treated group, the latencies were significantly higher than baseline at 3, 5 and 7 h postadministration (P = 0.02, P = 0.004 and P = 0.01, respectively) but not significantly different from baseline at 1, 9 or 11 h postadministration.
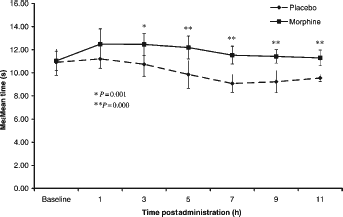
Comparison of mean latency of foot withdrawal pre- and postadministration (±1 SD) between the morphine- and saline-treated groups
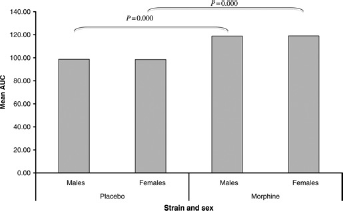
Comparison of area-under-curve (AUC) for postadministration period for male and female Lewis rats (±1 SD) between the morphine- and saline-treated groups
Discussion
One aim of this study was to investigate potentially simple and practical methods of administering slow-release oral preparations of morphine and hydromorphone to laboratory rats. The most practical vehicle for oral delivery for both preparations was syrup; the only foodstuff preferred by all of the rats tested and that could easily, rapidly and effectively be administered with a spatula through the cage bars, thus eliminating any distress and/or discomfort. The second aim was to use changes in peripheral thermal antinociception as a preliminary evaluation of potential effectiveness and duration of action of these preparations. Oral preparations of morphine (64 mg/kg) and hydromorphone (16 mg/kg) induced comparable increases in the withdrawal latencies of Lewis and Wistar rats up to 3 h postadministration. However, this increase over baseline was maintained only beyond 3 h postadministration with the oral preparation of morphine. 64 mg/kg of morphine appeared to increase withdrawal latencies for at least a period of 7 h postadministration. Although withdrawal latencies with morphine only remained significantly above baseline until 7 h postadministration, they remained significantly higher than the saline (control) latencies up to 11 h. The lack of significant differences compared with baseline beyond 7 h results from the withdrawal latencies decreasing with each successive time point. Despite the rest period between successive tests, this was most likely to result from the animals becoming progressively more sensitized to the application of the heat stimulus (hyperalgesia). They may also have responded earlier through learning that stimulus onset would eventually result in an uncomfortable or painful sensation and therefore respond more rapidly following each successive application of the heat stimulus. Further studies need to be carried out to determine which of these explanations accounts for the responses observed.
The extended duration of antinociception obtained with the morphine preparation suggests that this could also provide longer lasting effects on pain thresholds had a more clinically relevant pain model been used. Although this aspect was not addressed here, all opioids have broad ranging effects in both acute phasic tests and tonic efficacy tests that are thought to be more relevant to models of clinical pain.17,18 In support of this, the increased response latencies we observed with morphine were of a similar duration (6-14 s) to the effects seen in inflammatory assays using intra-articular injection of carrageenan or postoperative pain models such as the plantar incision test. 19 Also, it is likely that responses in both phasic and tonic antinociceptive assays actually overestimate the doses actually needed to elicit relief from clinically induced pain,20,21 so we would predict significant clinical efficacy at the dose rates tested here. To confirm this, we would require more extensive analyses of an invasive nature where rats would be exposed to pain. These would also have included a postsurgery dose ranging study including serial blood sampling to assay the plasma availability of the drug. As this study was intended as an initial test of the potential value of this new method of drug delivery, we needed to consider the 3Rs implication of conducting these additional tests. Due to the possibility that animals might suffer unnecessarily, we felt that these objectives were beyond the justifiable scope of the study and preferred to use a relatively non-invasive thermal assay.
One suggested limitation of using antinociceptive tests when screening opioids is the effect of opioid-induced sedation on an animal's responses to the noxious stimulus, e.g. foot withdrawal latencies. 22 However, there is a growing body of evidence to suggest that although sedation is associated with common opioid analgesics, this does not appear to have a significant effect on responses to the noxious stimuli used in common antinociceptive tests. Petersen et al. 23 found no significant increase in latency to respond to a thermal noxious stimulus following administration of a range of sedatives in rats. In addition, the results of an unpublished study 24 demonstrated that only buprenorphine (analgesic) and not acepromazine (sedative) had a significant effect on rat foot withdrawal latencies from a noxious thermal stimulus. The findings of Wegner et al. 25 further support this as they demonstrated that opioid analgesics induced a similar degree of sedation as acepromazine, but only the former induced significant analgesia in dogs. A study carried out in cats showed similar findings. 26
The difference in the duration of action between morphine and hydromorphone is likely to be related to differences in their slow-release characteristics. The limited duration of action of hydromorphone (peak: one hour) is likely to be due to its slow-release technology being compromised during preparation and administration, as it could not be administered orally in its encapsulated form. The capsules containing the slow-release granules have to be broken open in order to provide an appropriate dose in a form that would be consumed voluntarily by the rats. As a consequence, the controlled-release melt extrusion coating on the granule could have been damaged either due to chewing of the granules during consumption and/or by being placed into the syrup. It has been suggested that disrupting this granule coating could cause a bolus of hydromorphone to be released more rapidly than expected, leading to a considerably shorter duration of action. This is based upon human clinical trails using Palladone-SR® granules removed from their capsules for administration through feeding tubes. This showed similar results to this study, with 70% of the hydromorphone being released at two hours compared with only 40% when it remains in its encapsulated form (personal communication, 2007). This may offer an explanation of the shorter than expected duration observed in this study. In addition, absorption of hydromorphone through the buccal mucosa may also explain such a rapid peak effect; particularly as the thick syrup used as a vehicle for the granules would have prolonged the amount of time the granules were in the mouth and further encouraged the rats to chew them. However, to identify the reason for the shorter than expected duration of action observed with hydromorphone would require pharmacokinetic studies to be carried out.
This shorter than expected duration of action was not observed with the morphine preparation, which uses a different formulation and controlled-release technology. It is formulated in a suspension rather than granular form and uses a cationic exchange resin to bind the morphine, which is less likely to be damaged by placing it into syrup for administration. The duration of efficacy of morphine was observed to be at least in excess of 7 h postadministration. The duration of an orally administered analgesic is dependent on the gastrointestinal transit time, as in mammals the small intestine is the prime location of orally administered drug absorption. Although the gastrointestinal transit time of a healthy untreated rat is approximately 6 h, 27 the slowing of gastric motility that has been observed to be associated with opioid administration 28 may account for the extended length of analgesia with this morphine preparation.
The effect of hydromorphone on foot withdrawal latencies seems somewhat dependent on the sex and strain of the rat, with female Lewis showing longer latencies than male Lewis or Wistar rats of either sex. This sex and strain effect is not seen with the morphine preparation. There was a difference in the onset of action with morphine between the first and second studies, with increases in withdrawal latencies over baseline occurring at one hour after administration in the first study, but not until between 1 and 3 h postadministration in the second study. We are unsure why there would be a discrepancy in the onset of action, but a likely explanation is slight batch differences between the rats used in each study, as the drug preparations, study method, drug administration and strains were identical in both studies. These findings further highlight the importance of considering sex, strain and potentially batch when analysing animal-derived data such as foot withdrawal latencies.
This study suggests that administering a slow-release preparation of morphine (64 mg/kg) via palatable foodstuff may offer a potential method of achieving longer lasting analgesia in rats. However, further investigation will be required to test the efficacy of this preparation for treating pain such as those that occurs following surgery. The results of this study, although only a primary screen, suggest that this could be developed into an effective analgesic regimen. Developing non-intrusive methods such as oral drug administration is an obvious and extremely desirable welfare refinement since this could reduce the likelihood of the treatment method inducing any further distress and/or pain. Such refinements are not only important for maximizing welfare, but could also have a significant positive impact on the validity and integrity of research results.
Footnotes
ACKNOWLEDGEMENTS
The authors would like to thank Charles River UK for supplying the animals for this study and the staff of the Comparative Biology Centre at Newcastle University for their assistance throughout this study.
