Abstract
Oral administration of buprenorphine is becoming a popular method of providing analgesia for laboratory rodents. The mixing of buprenorphine with flavoured jello, which rodents find palatable, is becoming a commonly used method as it is thought to improve the efficacy of oral buprenorphine by increasing the time available for it to be absorbed via the oral mucosa. The aim of this study was to assess the effect of various methods of buprenorphine administration (subcutaneous saline, subcutaneous buprenorphine [0.05 mg/kg], buprenorphine gavage [0.5 mg/kg], buprenorphine in jello [0.5 mg/kg] and buprenorphine in golden syrup [0.5 mg/kg]) on thermal antinociceptive thresholds in laboratory rats. Buprenorphine administered subcutaneously, by gavage, in jello and in syrup induced significant increases in thermal antinociceptive thresholds compared with saline. This effect was observed up to 5 h postadministration for buprenorphine administered subcutaneously and by gavage, but only for one hour postadministration for buprenorphine administered in jello and in syrup.
Buprenorphine, a semi-synthetic partial opioid mu receptor agonist and kappa receptor antagonist, is a widely administered analgesic for research animals.1,2 The mixing of buprenorphine with flavoured jello is becoming an increasingly recommended means of providing analgesia for laboratory rodents.3–5 This method can be considered to provide a number of improvements over ‘more traditional’ routes of administration. Analgesia can be provided without the potential discomfort and distress associated with the additional handling and administration involved with other routes of administration, such as subcutaneous injection or gavage. The use of flavoured jello may increase the bioavailability of buprenorphine over other methods of oral administration. The bioavailability of buprenorphine administered orally without a palatable vehicle (via gavage or directly into the mouth) has been estimated as approximately 5-10%; 6 therefore an oral dose of 0.5 mg/kg would be expected to provide analgesia equivalent to 0.025-0.05 mg/kg subcutaneously.2,7 This low bioavailability is thought to be due to first-pass hepatic and gastric metabolism of the drug and in humans have been circumvented by use of a sublingual tablet formulation. 8 This allows the drug to pass through the buccal mucosa and therefore avoid hepatic and gastric metabolism. A similar approach has been successfully demonstrated in cats,9–11 with oral transmucosal administration providing equivalent analgesia to intravenous administration. The use of flavoured jello that rats find palatable3–5 may increase the bioavailability of buprenorphine by lengthening the transit time through the mouth and therefore increasing absorption via the oral mucosa.
Although an oral dose of 0.5 mg/kg in jello has become widely accepted for provision of analgesia in rats, there remains conflicting evidence concerning the efficacy of buprenorphine administered to rodents in this way. Well-controlled studies using antinociceptive testing have demonstrated an almost complete lack of efficacy of buprenorphine administered either by gavage or as jello at this dose.12,13 However, oral buprenorphine at 0.5 mg/kg in jello did appear effective in preventing the adverse effects of surgery on body weight7,14 and was effective in reducing postsurgical pain scores in rats following laporatomy. 15 There are two potential explanations for this disparity between studies. Firstly, the studies used different measures to determine the efficacy of oral buprenorphine, which could lead to different conclusions being drawn. Martin et al. 12 and Thompson et al. 13 used the tail flick antinociceptive test compared with postsurgical pain assessment used by Flecknell et al. 7 and Liles et al. 14 Secondly, differences in the palatable vehicles used between studies could lead to variable transit time in the mouth and so variable quantities of buprenorphine being absorbed across the buccal mucosa. If so, then the efficacy analgesia should be increased by delivering the buprenorphine in a material that maximizes the time available for oral transmucosal absorption. The aim of this study was to use changes in peripheral thermal antinociception as a simple preliminary method of comparing the effectiveness of buprenorphine administered orally using foodstuffs as novel vehicles with more ‘traditional’ administration routes in rats. This involved comparing buprenorphine administered in foodstuffs thought to increase the time available for oral transmucosal absorption (by remaining in the mouth for longer), with subcutaneous and gavage administration (completely bypassing the oral mucosa).
Materials and Methods
Animals and husbandry
Male and female Wistar and Lewis rats (5 male and 5 female of each strain, n = 20) were obtained from a commercial supplier (Charles River, Kent, UK) and weighed between 150 and 200 g on arrival. The inclusion of more than one sex and strain in each treatment allowed for greater generalization of results without any loss in statistical power. 16 The rats were housed in groups of five in polypropylene cages (RC1, 40 × 17 × 25 cm, NKP, UK) with wood shavings (‘Aspen’, BS and S Ltd, Edinburgh, UK) and a diet of commercial pellet (No. 3, SDS Ltd, Witham, Essex, UK) and water ad libitum. Room temperature and humidity were controlled at 19 ± 2°C and 50 ± 10%, respectively, with a 12 h:12 h light-dark cycle (light: 07:00 to 19:00).
Rats were acclimatized to this environment for a period of 10 days before the studies began. These rats were used in both the pilot and main studies. Body weights were recorded daily between 09:00 and 10:00 h. The animals were free from any common pathogens according to the FELASA Health Monitoring Recommendations. 17 All procedures were carried out in accordance with the Animals (Scientific Procedures) Act 1986 and approved by the local ethical review process. On completion of all experiments, rats were humanely euthanized with slow rising concentration of CO2.
Palatability pilot testing
A pilot study was undertaken to establish which of the palatable vehicles would be most suitable for use in the main study. This included four foodstuffs that were tested, that were thought to be highly favoured by rats, therefore encouraging rapid and complete consummation of buprenorphine: golden syrup (Lyle's Golden Syrup, Tate & Lyle Sugars Ltd, Cheshire, UK), strawberry syrup (Askey's Strawberry Treat, The Silver Spoon Company, Peterborough, UK), sugar-free blackcurrent jello (Hartley's, Spalding, UK) and marmite (Marmite Yeast Extract, Unilever Bestfoods UK, Crawley, UK). Approximately 3 mL of each foodstuff was placed into the animal's home cages in small feeding bowls for five nights to allow the animals to habituate to the food and prevent neophobia. 18 The palatability of each foodstuff was assessed in 20 rats by separating them into four treatment groups (n = 5 group), each of which was fed the four foodstuffs according to a crossover design. This involved the animals being temporarily housed singly in individual cages (RB3 cages, NKP, 45 cm × 38 cm, height 20 cm) for a period of 60 min and given approximately 2 mL of each of the foodstuffs in a random order over a period of five days. An assessment of the approximate quantity of food consumed was made at 0, 30 and 60 min. The foodstuffs were either placed in a small food bowl or fed via a spatula through the cage bars. For those foodstuffs found to be palatable, the palatability with the addition of buprenorphine was then tested to ensure that this did not alter the consumption of foodstuffs.
Buprenorphine administration
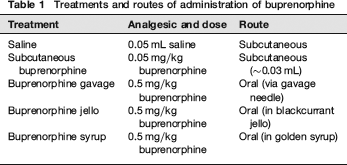
Buprenorphine (0.05 mg/kg: Temgesic®, Reckitt and Coleman, Hull, UK) was administered to rats in this study by various routes. Subcutaneous injections were made into the skin overlying the flank, using a 1 mL insulin syringe (BD PlastipakTM, Becton Dickinson SA, S Agustin del Guadalix, Madrid, Spain) while the animal was restrained by an assistant. Gavage was carried out using a 19-gauge stainless-steel gavage needle and a 1 mL syringe while the animal was restrained by an assistant. Palatable vehicles were administered in 2 mL portions using small food bowls in the cage or hand fed with a spatula (102 mm stainless-steel chattaway micro pattern spatula, VWR International Ltd, Leicestershire, UK) through the cage bars. Two millilitres of foodstuff were chosen based on previous experience that has shown rats (of various strains) will consistently consume this volume. With the exception of the blackcurrant jello, the required dose of buprenorphine was simply added to and then mixed with the foodstuff using a spatula, just before administration. Blackcurrant jello was prepared as directed by the manufacturer, using only half the directed quantity of water. To prepare the jello containing buprenorphine, it was cooled to around 20°C before adding the buprenorphine according to the method of Pekow. 3 It was then allowed to set in cubes of roughly 2 mL and kept in the refrigerator (3-5°C) overnight.
Hargreaves procedure (foot withdrawal latency)
Antinociceptive thresholds were determined using the Hargreaves thermal nociceptive hind-paw withdrawal assay (UGO Basile Ltd, Biological Research Apparatus, Comerio, Italy) before (baseline) and at 1, 3 and 5 h after administration with buprenorphine. The thermal stimulus was maintained at an intensity of 80%, increasing in temperature to a maximum of 55°C in 33.1 s, after which the thermal stimulus was cut off if the rat failed to withdraw its hind-paw, to prevent tissue injury. 19 Rats were habituated to the Hargreaves chamber for 15 min prior to testing and withdrawal latency was measured three times, using opposite paws with 5 min intervals between tests. A mean of the three recordings was then calculated to give a single measure for each time point. The stimulus intensity and cut-off time were selected, so changes in withdrawal latency would be easy to detect while no tissue damage was induced.
Main study
In the main study five treatments (see Table 1) were administered to 20 rats in a crossover randomized block design, with a 42 h ‘wash-out’ period between treatments. This washout period was chosen based on a short pilot study that demonstrated that foot withdrawal latencies to a thermal stimulus had returned to baseline levels in animals receiving buprenorphine. For the ‘jello’ treatment group, the buprenorphine was administered via 2 mL cubes of jello in food bowls placed into the cages housing individual rats (as in the pilot study). For the ‘syrup’ group, the buprenorphine was administered in 2 mL of syrup via a spatula to singly-housed rats (as in the pilot study). All treatment administrations were carried out between the hours of 10:00 and 11:00 in the morning. Thermal antinociceptive thresholds were measured using the Hargreaves apparatus as described earlier prior to drug administration (baseline) and at 1, 3 and 5 h after dosing.
Treatments and routes of administration of buprenorphine
Statistical analysis
The data were tested (Kolmogorov-Smirnov test) and were normally distributed with homogeneous variance. Therefore the data were analysed using parametric analysis with SPSS (Version 14, SPSS Inc, Chicago, IL, USA) software. A repeated-measures analysis of variance was conducted on the paw withdrawal latencies. This included treatment, sex, strain and cage number as between-subject factors and the recording times (pre- and postadministration) as within-subject factors. Post hoc analysis of the withinsubject factors was conducted using both simple and repeated contrasts and between-subject using ‘Tukey's’ post hoc tests. A significant value was considered at P < 0.05.
Results
Overall no adverse effects (e.g. abnormal behaviour, excitation, agitation, etc.) were observed at any of the time points with any of the administrations of buprenorphine. Some of the animals receiving buprenorphine subcutaneously and by gavage exhibited signs of mild sedation (decreased activity, etc.); however, this did not appear to alter foot withdrawal latencies.
Pilot study
Syrup and blackcurrant jello were found to be the most preferred foodstuffs of those tested as all of the rats consumed up to 2 mL of each. The rats seemed unwilling/incapable of eating quantities of foodstuffs larger than approximately 2 mL. Feeding the syrup via a spatula through the bars of the cage and jello in cubes via small food bowls was acceptable to all animals. Therefore, 2 mL portions of syrup via a spatula through the bars of the cage or 2 mL cubes of jello in food bowls were used to deliver the oral buprenorphine throughout the main study.
Main study
Time (baseline and 1, 3 and 5 h postadministration) had a significant effect on the withdrawal latencies of the rats when they received buprenorphine subcutaneously, by gavage, in jello and in syrup (P = 0.007, P = 0.001, P = 0.023, P = 0.025, respectively). Foot withdrawal latencies exhibited with subcutaneous saline were not significantly affected by time. Compared with baseline, withdrawal latencies were significantly longer at 1, 3 and 5 h postadministration when receiving buprenorphine by the subcutaneous route (P = 0.026, P = 0.005, P = 0.035, respectively) and by gavage (P = 0.015, P = 0.003, P = 0.02, respectively). Withdrawal latencies were significantly longer at one-hour postadministration compared with baseline when receiving buprenorphine in jello and in syrup (P = 0.021, P = 0.025 respectively), but not at 3 and 5 h postadministration. Although non-significant increases in withdrawal latencies were observed compared with baseline for both buprenorphine in syrup and jello (see Figure 1), none of the other factors tested (e.g. sex, strain and cage) had a significant effect on foot withdrawal latencies either alone or through interactions.
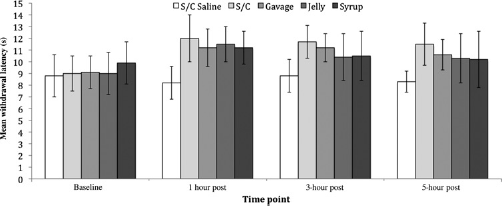
Mean foot withdrawal latencies at baseline and the 1, 3 and 5 h postadministration for subcutaneous saline, subcutaneous buprenorphine, gavage buprenorphine, buprenorphine in jello and buprenorphine in syrup (±1 SD). S/C = subcutaneous
There was a significant difference in foot withdrawal latencies between the five treatments postadministration (P = 0.001), with significantly shorter latencies being observed when saline was given at one hour postadministration compared with buprenorphine administered subcutaneously (P < 0.000), by gavage (P = 0.012), in jello (P = 0.005) and in syrup (P = 0.007). However, only buprenorphine administered subcutaneously and by gavage induced significantly longer latencies compared with saline at 3 and 5 h postadministration (P = 0.000 and P = 0.01, respectively). Longer withdrawal latencies were observed compared with saline for both buprenorphine in syrup and jello; however, these were substantial but not significant (see Figure 1). There was no significant difference in foot withdrawal latencies between the five different routes of administering buprenorphine via any route tested.
For both the buprenorphine in syrup and jello treatments, there was variation between individuals over whether all of the foodstuffs containing buprenorphine were consumed during the feeding period. Thirteen rats out of the 20 consumed all of their syrup containing buprenorphine and 15 rats out of the 20 consumed all of their jello containing buprenorphine. All rats received complete dose of buprenorphine when it was administered subcutaneously and by gavage.
Discussion
The aim of this study was to use peripheral thermal antinociceptive thresholds in laboratory rats to compare buprenorphine administered orally using foodstuffs thought to prolong transmucosal absorption with subcutaneous and gavage administration. The foodstuffs were hypothesized to increase the time available for oral transmucosal absorption by prolonging the time available for the absorption of buprenorphine in the mouth and thus increase its effectiveness and duration of action in peripheral antinociceptive tests. The foodstuffs chosen for oral administration of buprenorphine were golden syrup and sugar-free blackcurrant jello. Both of these vehicles were consumed by all rats and were easily and rapidly administered by a spatula through the cage bars (syrup) or placed into food bowls inside the home cage (jello). This eliminates any potential distress and/or discomfort associated with subcutaneous or gavage administration.
The results clearly demonstrate that all rats receiving buprenorphine subcutaneously (0.05 mg/kg), by gavage (0.5 mg/kg) or orally (0.5 mg/kg) via jello or syrup exhibited comparable increases in foot withdrawal at one hour postadministration. However, only buprenorphine administered subcutaneously and by gavage induced comparable elevations in all rats at 3 and 5 h postadministration. Although the mean withdrawal latencies induced by buprenorphine in syrup and jello were not significantly different from either baseline or saline at 3 and 5 h postadministration, both treatments did nevertheless induce longer mean withdrawal latencies (see Figure 1). This lack of significance is due to the variation in withdrawal latencies between individuals observed at 3 and 5 h postadministration of buprenorphine in syrup and jello.
This shorter duration of antinociceptive effects of buprenorphine administered in jello or syrup is counter to our hypothesis that these foodstuffs would increase in effectiveness and duration of action of oral buprenorphine in antinociceptive tests. There are a number of potential explanations for this finding. Firstly, the foodstuffs did not maximize the time available for oral transmucosal absorption, by not remaining in the mouth for long enough to ensure sufficient absorption. Consequently, the majority of the buprenorphine would have been swallowed and undergone first-pass hepatic and gastric metabolism reducing bioavailability of oral buprenorphine. 8 Secondly, some of the rats did not completely consume all of the buprenorphine-containing syrup and jello that they were given, which was likely to have resulted in a suboptimal dose being given to those individuals. Although this is likely to have contributed to the shorter than expected duration of effect observed with buprenorphine in syrup and jello, we feel that this is unlikely to be the sole reason as the vast majority of animals consumed both the syrup and jello completely. The variation in consumption of syrup and jello between individuals may also account for the non-significant increases in withdrawal latencies observed over baseline and saline at 3 and 5 h postadministration with buprenorphine in syrup and jello. Alternatively, the presence of additives in the vehicles used to orally administer buprenorphine could have altered antinociceptive effects. For example, both the blackcurrant jello and golden syrup contain the sugars, glucose and sucrose that have been suggested to have analgesic properties in humans and rodents.20,21 Ultimately, to establish the effectiveness of foodstuffs at increasing the bioavailability of orally administered buprenorphine will require further studies to identify foodstuffs that are consistently and entirely consumed by rats irrespective of age, sex and strain, and pharmacokinetic studies to identify the actual bioavailability produced by such routes.
The significant peripheral antinociceptive effects observed following gavage is in contrast to previous studies,12,13 which found no detectable levels of analgesia after oral administration. Although this might be argued to be due to the differences in the antinociceptive test used (Hargeaves verses tail flick), we would suggest that this is unlikely as both utilize a noxious thermal stimulus. A more probable explanation is the variation due to strain and sex differences between this study (male and female Wistar and Lewis rats) and other studies (male Long Evans and Sprague-Dawley rats) in either the bioavailability of buprenorphine administered by gavage or the responses to the antinociceptive tests used. Although no such differences in the antinociceptive effects of buprenorphine were observed in this study, strain and sex differences in responses to antinociceptive testing have clearly been demonstrated in a range of mouse strains.22,23 No such strain or sex differences in the antinociceptive effects of buprenorphine were observed in this study. This may be because rat strains do not vary in their responses to antinociceptive tests in the same way as mouse strains or that the two strains tested in this study do not vary in their responses to antinociceptive testing but both differ from other strains. Further testing of other rat strains following administration with buprenorphine by gavage will be required to answer this question.
This study demonstrates that peripheral thermal antinociceptive effects can be provided by orally administered buprenorphine. This supports the findings of previous studies employing behavioural-based pain scoring that demonstrated the efficacy of buprenorphine administered in jello in controlling postsurgical pain.2,15 However, the lower efficacy of buprenorphine in jello and syrup in the present study and the complete lack of efficacy at currently used dose rates in previous studies12,13 potentially indicates that there is considerable variation in effectiveness between individuals, institutions and strains. We suggest that further studies employing pharmacokinetic assessments are carried out to identify the quantity of buprenorphine that is absorbed via the buccal mucosa. In the interim if oral buprenorphine is used for postoperative analgesia, then we suggest that a reliable method of pain assessment is also used. Animals that have inadequate analgesia could then be given an additional dose of buprenorphine by subcutaneous injection.
Footnotes
Acknowledgements
We would like to thank all of the staff at the Comparative Biology Centre for all of their help and support throughout my project and Charles River UK for supplying the animals.
