Abstract
Streptozotocin (STZ) given intravenously destroys pancreatic beta cells and is widely used in animal models to mimic type 1 diabetes. The effects of STZ on the clinical state of health and metabolism were studied in six high health certified domestic pigs weighing 19 ± 1.3 kg at the start of the experiment. A single STZ dose of 150 mg/kg of body weight successfully induced hyperglycaemia and alterations in amino acid metabolism. Within 9 h after STZ administration, the blood glucose values fell from 5.4–7.5 mmol/L to 0.8–2.2 mmol/L. Hypoglycaemia was treated with 0.5 g glucose/kg body weight. In all pigs, hyperglycaemia was produced 24 h after STZ treatment, and 3 days after STZ injection, the glucose concentration was >25 mmol/L. Mean C-peptide concentration was 0.25 ± 0.16 μg/L since 2 days after STZ injection until the end of the study. The serum concentration of the branched-chain amino acids (BCAA) increased four-fold, and alanine and taurine decreased by approximately 70% and 50%, respectively, after STZ treatment. All but one pig remained brisk and the physical examination was normal except for a retarded growth rate and a reduction of the skeletal muscle. At the end of the study, the pigs were moderately emaciated. Postmortem examination confirmed muscle wasting and a reduction of abdominal and subcutaneous fat. In conclusion, STZ-induced diabetes in pigs fulfils the requirements for a good animal model for type 1 diabetes with respect to clinical signs of the disease and alterations in the carbohydrate and amino acid metabolism.
Reliable and reproducible animal models are of great importance in diabetes research. Rodent models are widely used, 1 and pigs are suitable when large animal models are required. 2 A pig model can be valuable when repeated biopsies and blood samples are required, and in studies concerning possible therapeutic perspectives such as transplantation of islets.
Since spontaneous diabetes is extremely rare in pigs, the disease has to be induced either surgically by pancreatectomy 3,4 or chemically. 5 In Swedish Landrace pigs, i.v. injection of a single dose of 100–150 mg streptozotocin (STZ) has been shown to be a safe procedure for inducing type 1 diabetes. 3
Diabetic illness not only affects the carbohydrate and lipid metabolism (reviewed by Taylor and Agius 6 ) but also alters the metabolism of protein. In rats, STZ-induced diabetes has been found to result in a decrease in the rate of protein synthesis and an increase in its degradation. Further, elevations of branched-chain amino acids (BCAAs, i.e. leucine, isoleucine and valine) and decreases in the concentrations of glutamate, glutamine, alanine, glycine, proline, serine and threonine were observed in the blood. 7
Alanine is the main gluconeogenic amino acid and is reported to contribute to the formation of glucose in the liver of rats 8 and man. 9 As in the case of experimentally-induced diabetes in rats, the amino acid catabolism is increased in diabetic man in the fasting state, as reflected by an increased uptake of alanine by the liver and accelerated BCAA catabolism in muscles. 10 The blood concentrations of BCAA are approximately twice as high in type 1 diabetic patients under poor metabolic control as in non-diabetic subjects. 11 Also in other wasting disorders, such as cancer and injury, the plasma concentrations of BCAAs are often elevated and their turnover rates altered. 12 The free amino acid-like compound taurine has been shown to be associated with diabetes. In both type 1 and type 2 diabetic patients, the plasma taurine concentrations have been found to be lowered. 13,14 Taurine supplementation has a beneficial effect on the blood glucose and lipid levels, as shown in streptozotocin-nicotinamide (STZ-NA) diabetic rats. 15
The aim of the present study was to further investigate the clinical signs and metabolic changes in STZ-induced diabetes in growing domestic pigs. In particular, the amino acid metabolism was addressed.
Materials and methods
Animals
Six high health certified SPF pigs (Yorkshire × Landrace, Serogrisen, Ransta; Wallgren et al. 16 ) weighing 19 ± 1.3 kg at arrival were used. They were housed at the Department of Clinical Sciences in individual pens measuring approximately 3 m 2 , with straw as bedding and within sight and sound of one other. A 12:12 h light/dark schedule (lights on at 07:00 h) was used and an infrared lamp (24 h) was provided in a corner of each pen, the temperature was 20 ± 2ºC. The pigs were fed ad libitum on a commercial finisher diet without growth promoters (Singel Flex, Odal, Sweden) and had free access to water.
Experimental design
All procedures were approved by the Ethical Committee for Animal Experimentation, Uppsala, Sweden.
The protocol ran for a total of six weeks. During a two-week acclimatization period the pigs were handled frequently and trained to step onto an electronic scale. After acclimatization, the animals underwent surgery, which consisted of an indwelling silicon catheter into the jugular vein under aseptic conditions according to the method of Rodrigues and Kunavongkrit. 17 In brief, after an overnight fast atropine sulphate, 0.05 mg/kg body weight (b.w.) (Atropin®, Merck NM, Sweden), was given i.m. and anaesthesia was induced with a combination of medetomidine (Domitor® vet 1 mg/mL, Orion Pharma Animal Health, Sollentuna, Sweden), tiletamine and zolazepam (Zoletil® 250 mg of each compound/mL, Reading, Carros, France) at a dose of 0.03 mL/kg b.w. Intramuscular buprenorphine (Temgesic® 0.3 mg/mL, Schering-Plough, Brussels, Belgium) was provided for additional analgesia and administered i.m. preoperatively at a dosage of 0.1 mg/kg b.w. The animals were intubated and general anaesthesia was maintained with isoflurane vaporized in oxygen, with 25% nitrous oxygen added initially (5–10 min). The concentration of isoflurane (1–2.5%) was continuously adjusted to achieve an adequate depth of anaesthesia. The silicon catheter was brought through a subcutaneous tunnel and exteriorized on the back between the scapulas, and finally covered with a piece of canvas. Flushing was carried out twice daily with 2‰ heparinized saline (5000 IU/mL, Leo, Denmark, Apoteksbolaget, Sweden).
After the surgical procedure, penicillin (Penovet® 300 mg/mL, 0.7 mL/10 kg; Boehringer-Ingelheim, Sweden) was given i.m. for three consecutive days. The catheter was used for i.v. medication and blood sampling. Blood samples were taken at least twice daily for measurements of the glucose concentration, and every second day for the other blood analyses. Clinical examinations were performed at least daily throughout the study. Body weight was measured three times a week, and daily weight gain was calculated. At the end of the experiment, the pigs were killed with an overdose of pentobarbital sodium, and muscle biopsy specimens (1 cm3) from the midportion of M. longissimus dorsi and M. biceps femoris were taken and immediately frozen in liquid nitrogen and stored at −80ºC until analysed. The animals underwent gross examination postmortem and specimens from the liver and pancreas were fixed in 10% buffered formalin for measurement of the insulin contents.
Induction of diabetes
Because of the short half-life of STZ (Sigma S0130, Stockholm, Sweden), it was dissolved in 100 mmol/L disodium citrate buffer solution, pH 4.5, at a concentration of 80 mg/mL, and administered i.v. (approximately 1 mL/s) within 2 min, while the animal was under general anaesthesia. The total amount of STZ per individual corresponded to 150 mg/kg b.w.
Postoperative care
After recovery from anaesthesia, the animals were carefully monitored for 12 h by an experienced veterinarian. Blood glucose concentrations were repeatedly measured by test strips (Accu-Chek, Roche Diagnostics, Basel, Switzerland; this test was validated for porcine samples at the Department of Clinical Chemistry, SLU) before and 1, 3, 7 and 9 h after administration of the STZ dose. In all pigs, the glucose concentration fell below 2.3 mmol/L within 7–9 h after the injection of STZ, and the hypoglycaemia was promptly treated with an i.v. bolus of glucose (Apoteksbolaget, Sweden) corresponding to 0.5 g/kg b.w.
Blood analyses
Blood glucose concentrations were measured as described above or enzymatically (Konelab, Espoo, Finland). The serum C-peptide concentration (ng/mL) was determined with an ELISA kit (Mercodia, Uppsala, Sweden).
EDTA-preserved blood was analysed for total and differential white blood cell counts with an electronic cell counter validated for porcine blood (Cell-Dyn 3500, Abbott, Wiesbaden, Germany). Serum amyloid A (SAA; μg/mL) was measured in serum samples diluted 1:500, using a commercially available ELISA (Tridelta Phase range SAA kit, Tridelta Development Ltd, Greystones, Ireland). Serum samples were analysed for activities of aspartate amino transferase (ASAT), alanine aminotransferase (ALAT), γ-glutamyltransferase (γ-GT) (μkat/L) and glutamate dehydrogenase (GLDH; nkat/L) using automated equipment (Cobas MIRA, Roche Diagnostics, Basel, Switzerland).
Amino acid assay
Free amino acids and the amino acid-like compound taurine were measured in serum samples after precipitation of the proteins with a 1:10 dilution with 5% trichloroacetic acid followed by centrifugation at 2700
Tissue analyses
Cross-sectional areas (μm2) of approximately 200 muscle fibres were measured with an MOP digiplan analyzer (Kontron, Messgeräte GmbH) on photographs of myofibrillar ATPase-stained (pH 4.6) transverse sections. 20
The insulin content of biopsy samples from the liver and pancreas was determined by extracting insulin by mixing homogenates with acid–ethanol (0.18 mmol/L HCl in 96% ethanol) overnight at 4°C. Insulin was measured by enzyme-linked immunosorbent assay (Porcine Insulin ELISA kit, Mercodia, Uppsala, Sweden). DNA was measured using a Quant-iT PicoGreen dsDNA assay kit (Molecular Probe, Eugene, OR, USA). The results are expressed as (ng insulin/ng DNA) × 10E3.
Statistical analyses
All values are expressed as mean ± SD and comparisons were made using one-way factorial analysis of variance (ANOVA, Holm-Sidak method). Linear relationships between variables were tested by correlation analysis (Pearson's correlation coefficients). P values < 0.05 were considered statistically significant.
Results
All animals recovered from the surgical procedure and STZ injection within two days. However, pig no. 6 showed a decreased general appearance, and was euthanized with an overdose of pentobarbital sodium 6 days after the surgery. Postmortem examination of the pig revealed a pale kidney and liver with multiple haemorrhages. The remaining five pigs had a good appetite but did not gain any weight during the first two weeks after surgery. During the third week after the STZ dose their mean weight rose from 22.3 ± 2.0 kg to 25.7 ± 2.9 kg, and when the experiment was terminated, the mean weight was 30.5 ± 1.7 kg. All pigs had a large, distended abdomen relative to the size of the chest and hindquarters after about two weeks, but no undulation was palpable, and ascites was thus not suspected. In general, the skeletal muscle mass was reduced and on palpation, the biceps muscle, for example, was approximately one-third smaller than that of a non-diabetic pig. At the end of the study, all pigs were moderately emaciated, but their demeanour remained normal and no other clinical signs of disease than those described above were noted. The postmortem examinations revealed skeletal muscle wasting, including a thin abdominal wall, in all animals. The subcutaneous fat was reduced to a minimum and there were no visible fat reserves in the abdominal cavity. The liver and kidney were yellowish in colour.
Blood analyses
Before the STZ injection, the glucose values, measured at approximately 11:00 h, ranged from 5.4 mmol/L to 7.5 mmol/L. Within 9 h after the administration of STZ these values fell to 0.8–2.2 mmol/L. Hypoglycaemia was treated as described above and 24 h after the STZ dose, the glucose values had risen and ranged from 9.2 mmol/L to 15.1 mmol/L. Three days after the STZ injection, the glucose concentration was >25 mmol/L on all measurement occasions.
The highest concentrations of serum C-peptide were observed one day after STZ administration (range from 0.24 μg/L to 1.0 μg/L) and the mean serum C-peptide concentration since two days after the STZ injection until the end of the study was 0.25 ± 0.16 μg/L.
In all pigs, the total WBC count (range 8.1–18.6 × 109/L), the differential counts and the SAA concentrations (<9.8 mg/L) were within the reference values throughout the experiment.
The enzyme activities were within the reference range in pigs nos 1, 2 and 4 in all samples measured. In pigs 3 and 5 increased activities of ALAT, γ-GT and GLDH were observed one to three weeks after the STZ injection.
Amino acid concentrations
The total amino acid concentration in the blood did not change during the study period but the concentrations of the BCAAs leucine, isoleucine and valine were increased from two days after the STZ injection and onwards (Figure 1).
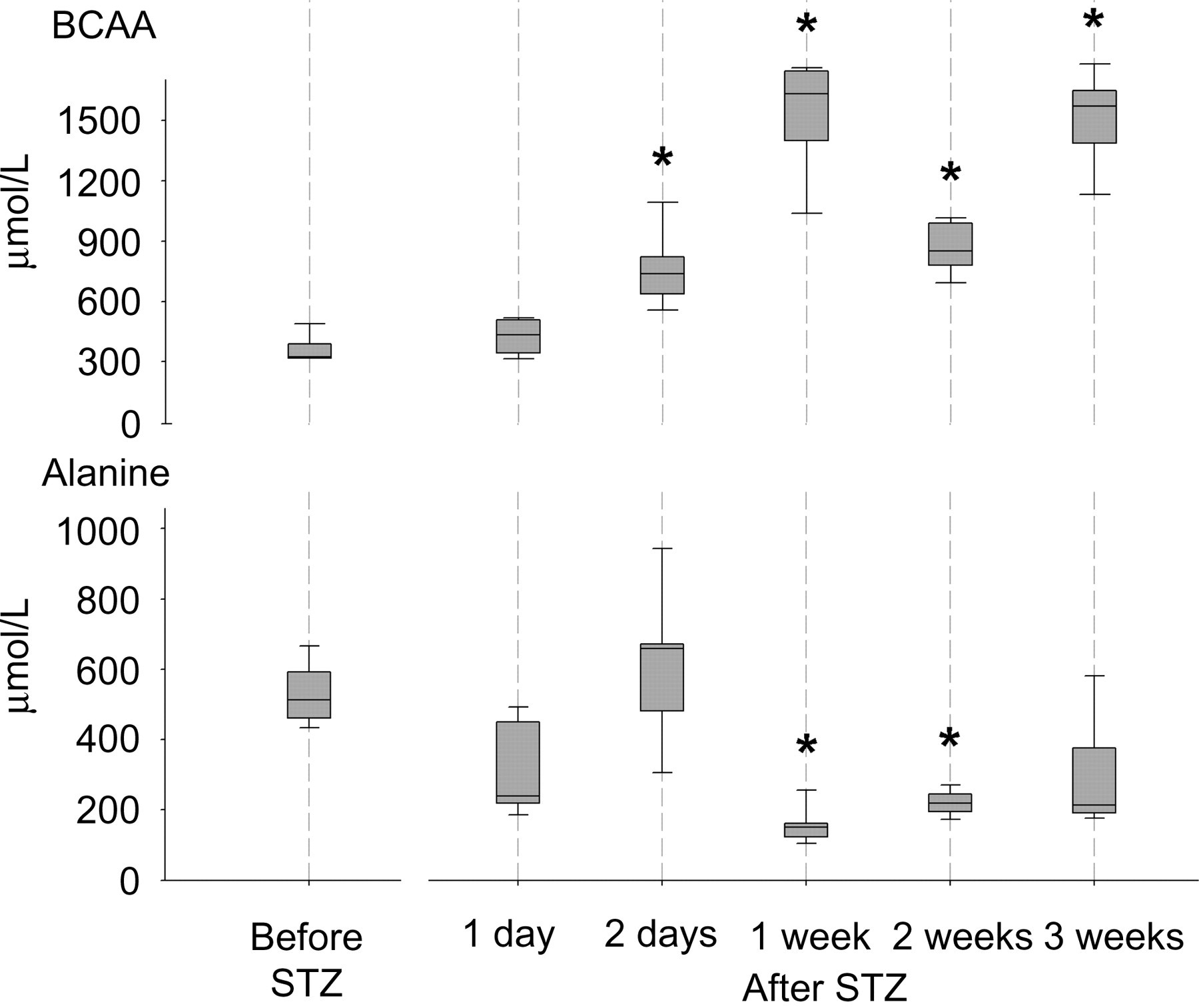
Concentrations of branched-chain amino acids (BCAA) (leucine, isoleucine and valine) and alanine in serum samples taken from pigs before and after a streptozotocin (STZ) injection. The boundaries of the box indicate the 25th percentile, the median value and the 75th percentile. Whiskers indicate the 95th and 5th percentiles. *Significantly different from value before STZ injection (P < 0.05)
A marked decrease (by approximately 70%) in the alanine level one week after the STZ injection was observed (Figure 1), but no significant change was noted in the total concentration of gluconeogenic amino acids, i.e. all the measured amino acids except lysine, leucine and taurine (Table 1). The taurine concentration fell one week after induction of hyperglycaemia and remained at a lower level throughout the study. A correlation (r = −0.72, P < 0.01) between the concentrations of glucose and taurine was found two days after the STZ injection.
Blood glucose (mmol/L), serum C-peptide (μg/L) and amino acid (μmol/L) concentrations in samples taken from pigs before and after a streptozotocin (STZ) injection
Data are presented as mean ± SD (n = 5). Total amino acid (AA) includes taurine; gluconeogenic AA = all measured AAs except lysine, leucine and taurine; Branched-chain amino acid (BCAA) = leucine, isoleucine and valine
*Significantly different from values before STZ injection (P < 0.05)
Tissue analyses
In the muscle biopsy samples taken from the diabetic pigs, the mean cross-sectional areas of fibres of M. longissimus dorsi and M. biceps femoris were similar. The areas of type I, IIA and IIB fibres were 1253 ± 200, 1134 ± 119 and 1482 ± 272 μm2, respectively. Fibre areas were also measured in muscles from a control pig obtained from the same SPF farm and of similar weight, and the corresponding mean fibre areas were 1545, 1350 and 3010 μm2.
The insulin content was low in all tissue sample studies, ranging from 0.348 to 0.694 ng/ng DNA × 10E3 in the pancreas, and from 0.603 to 1.081 ng /ng DNA × 10E3 in the liver.
Discussion
A single STZ injection successfully induced alterations in the amino acid metabolism and hyperglycaemia. One week after this injection, the concentrations of BCAAs were increased and the alanine level was lowered. Within three days of the STZ injection, the glucose values were >25 mmol/L in all animals, i.e. of a magnitude similar to that in untreated human patients with type 1 diabetes. The increased levels of C-peptide on the first day after administration of STZ indicate the onset of beta cell destruction, and the low content of insulin in pancreatic tissue samples taken postmortem further confirms the annihilation of beta cells.
The first day after the STZ treatment is critical because of the hypoglycaemia, and the animals have to be carefully monitored and given i.v. glucose promptly. All pigs survived this vulnerable time period, and all except one remained brisk, displayed haematological data within reference values, and showed a good appetite throughout the study. Neither did any pig display signs of ketoacidosis. Pigs of this age group and from the same high health herd have previously been raised at our department, and during a similar period of time those animals showed an average daily weight gain of >400 g. In the present study, there was no weight gain at all during the first two weeks after the STZ injection. This resembles the stunted growth of the juvenile human diabetic patient observed in the pre-insulin era. The basal energy expenditure in uncontrolled adult diabetic patients has also been found to be significantly higher (approximately 15%) than predicted from their age, sex and body surface area. Insulin treatment successfully reduced the metabolic rate to matched predicted values. 21 During the last period of the experiment the pigs gained weight again, which may have been due to some uptake of glucose, since the C-peptide values rose to up to 0.3 μg/L. Similar C-peptide data, indicating that their studied pigs were not fully diabetic after administration of 150 mg STZ/kg b.w., were recently reported by Hara et al. 22
Alanine plays a key role in the glucose–alanine cycle; the amino acid is transported in the blood from the muscle to the liver and glucose is formed through gluconeogenesis. In a normal man, alanine availability is the rate-limiting factor in gluconeogenesis in starvation. 9 In human diabetics, 24 h after withdrawal of insulin, the splanchnic uptake of alanine and other glyconeogenic amino acids was 1.5–2 times greater than in healthy controls, and in arterial blood the levels of alanine, glycine and threonine were reduced. 23 Amino acids are also important energy sources during an infection; for instance in pigs experimentally infected with Brachyspira hyodysenteriae, a significant decrease in the serum alanine concentration was observed in the presence of clinical signs of disease. 19 The BCAAs, i.e. leucine, isoleucine and valine, constitute the major substrate for repletion of muscle nitrogen and are a source of energy both in the fasting state and after protein feeding. 10
In the present study, the total gluconeogenic and BCAA concentrations before STZ treatment were of a magnitude similar to those reported previously in domestic crossbred pigs. 19,24 A marked decrease in serum alanine, and a four- to five-fold increase in BCAA, was observed one week after the chemical treatment and throughout the study. Accordingly, STZ-induced diabetes in rats has resulted in decreased serum levels not only of alanine but also of glutamate, glutamine, glycine, proline, serine and threonine; 7,25 in addition a marked increase in the activities of liver key enzymes of gluconeogenesis has been shown by Wimhurst and Manchester. 26 Analogous to the data obtained in the present study, BCAA concentrations also rose significantly in those studies on rats.
That taurine is associated with STZ-induced diabetes was shown by the lowered serum levels observed one to three weeks after STZ injection. Data on the individual pigs showed that the lowest concentrations of taurine were observed in pigs with the highest serum glucose levels. The decrease in serum taurine was similar in magnitude to that reported in earlier studies on human diabetic patients in comparison with healthy controls. 13,14 It is notable that taurine is reported to have insulin-like properties and to be involved in osmoregulation and antioxidation. These features may be related to the findings that taurine supplementation has beneficial effects on several of the complications often seen in human diabetes, such as cardiomyopathy, retinopathy and nephropathy (for reference see Kim et al. 27 ).
The pig that was euthanized six days after surgery displayed increased activity of GLDH, a marker for centrolobular liver necrosis, multiple liver haemorrhages and a pale kidney. These adverse effects are most likely due to the toxicity of STZ. 28,29 Increased enzyme activities were also observed occasionally in two other pigs, but their demeanour remained normal and they showed no other clinical signs of disease than those described in the Result section. Approximately two weeks after the STZ treatment all pigs had a distended and pear-shaped abdomen. The appearance resembled that seen in pigs with ascites, but no signs of fluid were evident. This was confirmed at autopsy that revealed weakened and reduced abdominal musculature, explaining the abdominal malformation. Likewise, the clinically observed reduction of the locomotor muscles was corroborated both at the gross pathological and histological examinations. The assumption that skeletal muscle had been used as energy substrate was supported by the fact that the cross-sectional areas of all muscle fibres were diminished. Of specific interest is that the type IIB fibres were about half the size of those measured in healthy pigs. The porcine skeletal muscle is unique among the animal kingdom as it contains up to 90% anaerobic type IIB fibres. 30 In comparison, type IIB fibres constitute approximately 15% of human skeletal muscle. 31 In Svalbard reindeers, it has been shown that during starvation it is primarily IIB fibres that contribute to the energy supply achieved by protein degradation. 32 Further, in adolescent children suffering from anorexia nervosa, no type IIB fibres were found at all and the mean cross-sectional area of type IIA fibres was significantly smaller than that in healthy subjects. 33 The low daily weight gain and the clinical symptoms described above show that the energy demand could not be met even if the pigs were fed ad libitum, and that the carbohydrate, fat and protein metabolism were altered.
Taken together, STZ induction of diabetes in SPF pigs of Yorkshire × Landrace origin, fulfils the requirement of a good animal model for type 1 diabetes, not only with respect to symptoms of disease and alterations in the carbohydrate metabolism but also to changes in the amino acid metabolism. Thus, the STZ pig model can be valuable in studies concerning possible therapeutic perspectives such as transplantation of islets in combination with stem cells.
Footnotes
Acknowledgements
The financial support from the Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning, and from Corline Systems AB, is gratefully acknowledged.
