Abstract
The effects of streptozotocin (STZ) were studied in eight high-health herd-certified Yorkshire × Swedish Landrace pigs (32.5 ± 2.6 kg initial body weight [BW]), and an insulin treatment protocol was developed to re-establish their metabolisms. A single intravenous dose of 150 mg STZ/kg BW successfully induced hyperglycaemia and alterations in their fat and protein metabolisms. Within 13 h post-STZ treatment blood glucose concentration had fallen to a range of 1.3 to 4.7 mmol/L. Hypoglycaemia was promptly treated with 0.5 g glucose/kg BW intravenously. All the pigs became hyperglycaemic with blood glucose concentrations >23 mmol/L within 48 h post-STZ. Two days post-STZ serum C-peptide concentrations fell below 60 ρmol/L in all the pigs and remained below 96 ρmol/L for five weeks until the end of the study. The pigs were left untreated for one week after STZ injection. At the end of this week 13-fold and nine-fold increases in serum concentrations of triglycerides and non-esterified fatty acids, respectively, were observed. Also, at this time-point a three-fold increase in the concentration of branched-chain amino acids (BCAA) was observed, and alanine and taurine were decreased by approximately 70% and 40%, respectively. During the week when the pigs were untreated, a reduced weight gain was observed, but after the onset of insulin treatment the daily weight gain was at least as good as that of conventional high-health pigs. Then a subcutaneous treatment with short-acting insulin was initiated. The initial dose of 2/3 IU/kg BW daily, divided between two doses, was gradually increased to 1 IU/kg BW. Within three weeks, the insulin treatment restored the metabolic changes in carbohydrate, fat and protein metabolisms produced by the STZ. In conclusion, the results underscore the usefulness of this animal model in translational research as insulin treatment re-establishes the changes in carbohydrate, fat and amino acid metabolisms observed in STZ-diabetic pigs and resolves clinical signs of disease similar to those in humans.
Animal models for diabetes mellitus are of great importance for gaining more information about the disease and for being able to find new and improved treatments. However, it is debatable as to why the results of animal experiments are often not reproducible in clinical human trials. One explanation is that animal models have a varying comparability to the human condition. The pig is widely used in preclinical diabetes studies, 1 and more data are needed to ensure the translational value of the porcine model for humans. Although spontaneous diabetes in the pig has been described, 2 it is extremely rare and therefore the disease must be induced to create porcine diabetes models. Streptozotocin (STZ) is a diabetogenic substance3,4 that can be used safely to induce a type-1 diabetes-like syndrome in conventional Landrace pigs at a single dose of 100–150 mg/kg body weight (BW).5,6 The diabetic state does not resolve spontaneously for at least 7.5 months. 5
In humans, type-1 diabetes mellitus (T1DM) is caused by an autoimmune destruction of the β-cells resulting in declined insulin production. Insulin promotes glucose uptake in tissues and ensures that blood glucose is kept at a relatively constant level. 7 Deficiency of insulin in T1DM causes uninhibited hepatic release of glucose and decreased uptake of glucose by peripheral tissues, causing hyperglycaemia.
Metabolically, T1DM can be compared with starvation since in the presence of insulin deficiency, tissues cannot utilize glucose to the same extent as in healthy individuals. In the fasting state in both healthy individuals and diabetics, serum concentrations of BCAA are increased and serum concentrations of gluconeogenic amino acids, predominantly alanine, are decreased.8–10
Insulin deficiency causes a rapid mobilization of fatty acids from adipose tissue and subsequently a rise in the plasma concentration of non-esterified fatty acids (NEFA). Elevated NEFA concentrations stimulate glycogenolysis and gluconeogenesis 11 as well as inhibiting insulin secretion, 12 both of which contribute to augmented hyperglycaemia.
Insulin acts as an antilipolytic and promotes storage of triglycerides (TG) in adipose tissue and inhibits the release of fatty acids into the circulation. 13 In diabetic patients with good glycaemic control, serum TG concentrations are within the reference range 14 or slightly decreased due to augmented plasma insulin levels. 15 Hypertriglyceridaemia is seen in patients with untreated or poorly controlled T1DM.14,16
Pigs with STZ-induced diabetes show clinical signs such as emaciation, polyuria, polydipsia, hyperglycaemia, increased levels of BCAA and decreases of alanine and taurine. 6 Thus, clinical signs of disease and metabolic alterations resemble that of non-insulin-treated diabetic humans. Successful insulin treatments of STZ-diabetic pigs have been given to outbred pigs at a dosage of 0.7–0.9 IU/kg BW/day. 17
In the present study STZ diabetes was induced in growing Yorkshire × Swedish Landrace pigs. The aim of the study was to (1) establish an insulin treatment protocol, and (2) further develop the animal model by examining whether the metabolic changes to carbohydrate, protein and fat metabolisms could be reversed.
Materials and methods
Animals
Eight high-health pigs (Yorkshire × Swedish Landrace, Serogrisen, Ransta, Sweden; Wallgren et al. 18 ) of both sexes were used. After an acclimatization period of two weeks, i.e. they were 10 weeks old; the pigs weighed 32.5 ± 2.6 kg. They were housed at the Department of Clinical Sciences in individual pens measuring approximately 3 m2, within sight and sound of one another. Straw and wood shavings (University Farm, SLU, Uppsala, Sweden) were used as bedding. A 12:12 h light/dark schedule (lights on at 07:00 h) was used and an infrared lamp (24 h) was provided in each pen. The room temperature was 18 ± 2℃. The pigs were fed a commercial finisher diet without growth promoters (SOLO 330 P SK, Lantmännen, Sweden) twice daily (07:00 and 18:00 h), the amount depending on BW and according to the Swedish University of Agricultural Sciences (SLU) regimen for growing pigs. 19 Water was provided ad libitum.
Experimental design
All procedures were approved by the Ethics Committee for Animal Experimentation, Uppsala, Sweden.
The animals were handled frequently during the acclimatization period and they became very tame so that blood could be easily collected from the indwelling catheter without stress, which could otherwise have influenced the blood analyses. Pigs of this age group often suffer from gastrointestinal diseases, 20 e.g. swine dysentery which alters the protein metabolism. 21 A two-week acclimatization period should cover the incubation periods of most infectious diseases for growing pigs, which was another reason for choosing this length of time. None of the pigs showed any clinical signs of disease during habituation.
The protocol ran for a total of seven weeks (Figure 1). During the two-week acclimatization period the pigs were tamed and handled frequently, and trained to step onto an electronic scale. After acclimatization, the pigs underwent surgery and an indwelling silicon catheter (SIL-C70 with round tip; Instec Solomon, Plymouth Meeting, PA, USA) was inserted into their vena jugularis dexter under aseptic conditions. STZ was given intravenously according to Jensen-Waern et al.
6
In brief, anaesthesia was induced with a combination of medetomidine (Domitor® vet, 1 mg/mL; Orion Pharma Animal Health, Sollentuna, Sweden) at a dose of 0.05 mg/kg BW, and tiletamine and zolazepam (Zoletil®, 250 mg/mL; Virbac, Reading, Carros, France) at a dose of 5 mg/kg BW intramuscularly. Buprenorphine (Temgesic®, 0.3 mg/mL; Schering Plough, Brussels, Belgium) was administered intramuscularly for additional analgesia at a dose of 0.01 mg/kg BW. Ceftiofur (Excenel vet®, 50 mg/mL; Pfizer Oy, Helsinki, Finland) was given preoperatively at a dose of 1 mg/kg BW. The airways were intubated and general anaesthesia was maintained with isoflurane (IsoFlo® vet; Orion Pharma Animal Health) vaporized in air and oxygen. The concentration of isoflurane (1–2.5%) was continuously adjusted to achieve an adequate depth of anaesthesia. The airways were mechanically ventilated to maintain an end-tidal carbon dioxide (EtCO2) of approximately 5 kPa. During anaesthesia, non-invasive blood pressure, O2 saturation, electrocardiogram (ECG), Fi/EtCO2, Fi/EtO2, Fi/Et isoflurane, heart rate, respiratory rate, rectal body temperature, tidal volume, minute volume and positive end-expiratory pressure were monitored. The silicone catheter was tunnelled subcutaneously and exteriorized on the back between the scapulas, where it was placed in a canvas pocket which was sutured onto the skin. The surgical procedure lasted approximately one hour.
Experimental design. The study lasted for seven weeks, acclimatization and social training (two weeks), streptozotocin (STZ) induction followed by one week of untreated diabetes and four weeks of insulin treatment.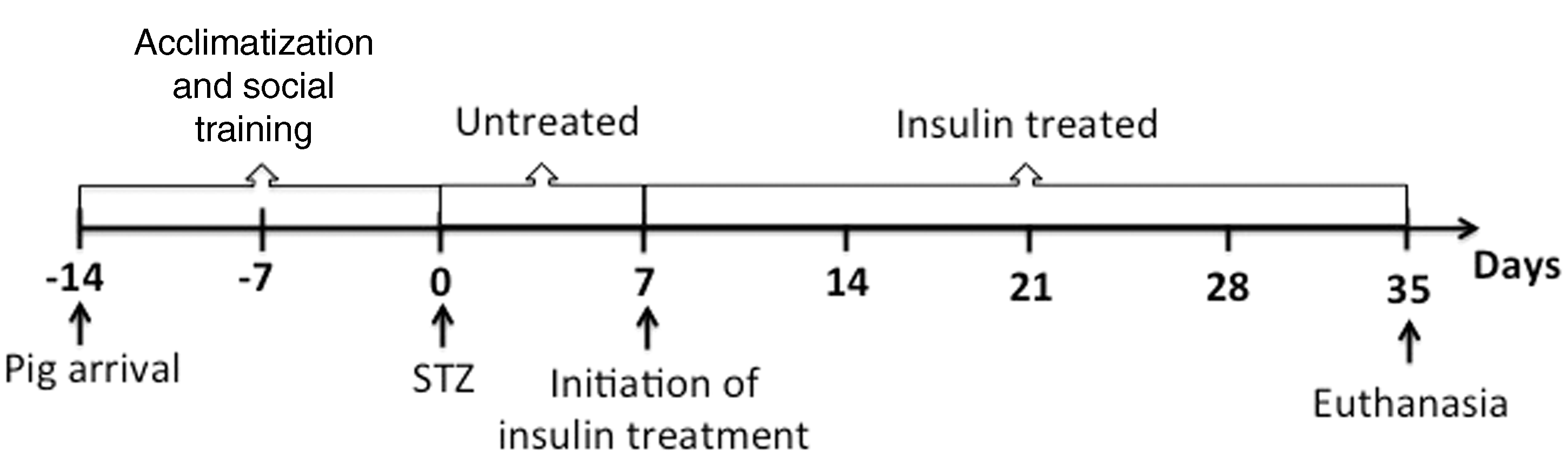
The catheter was used to facilitate frequent blood sampling under non-stressful conditions and intravenous medication. Flushing of the catheter was carried out once daily with 2‰ heparinized saline (5000 IU/mL; Leo, Ballerup, Denmark). During the first week post-surgery, blood samples were taken at least once daily for measurements of glucose concentration. After one week, insulin treatment was initiated and blood glucose concentrations were measured at least four times daily. Additional blood samples were taken at 2, 7, 14, 21, 28 and 35 days post-STZ for the other blood analyses. Clinical examination was carried out at least once daily throughout the study. BW was measured three times a week.
One week post-STZ, subcutaneous treatment with short-acting porcine insulin (Caninsulin® vet, 40 IU/mL; Intervet, Boxmeer, The Netherlands) was initiated. The initial dose of 2/3 IU/kg BW was gradually increased to 1 IU/kg BW, divided equally between two doses per day. The pigs were injected with insulin during feeding.
At the end of the experiment, the pigs were euthanized with an intravenous overdose of pentobarbital sodium (Pentobarbital vet, 100 mg/mL, Apoteksbolaget, Stockholm, Sweden). The pigs underwent gross examination post-mortem.
Induction of diabetes
Due to the short half-life of STZ (Sigma S0130, Stockholm, Sweden) it was dissolved in 100 mmol/L disodium citrate buffer solution, pH 4.5, at a concentration of 80 mg/mL, and administered intravenously within 5 min, while the animals were under general anaesthesia. The total amount of STZ administered per individual corresponded to 150 mg/kg BW.
Postoperative care
The animals were carefully monitored during recovery from anaesthesia and for 13 h post-STZ injection. Blood glucose concentrations were measured with test strips (Accu-Chek, Roche Diagnostics, Basel, Switzerland; validated for porcine blood at the Department of Clinical Chemistry, SLU) before and 1, 3, 7, 9, 11 and 13 h after the administration of STZ. Blood glucose levels <3 mmol/L were promptly treated with an intravenous bolus of glucose (Glukos APL, 500 mg/mL; Apoteksbolaget) corresponding to 0.5 g/kg BW.
Blood analyses
The blood glucose concentrations were measured as described above. Concentrations ranging from 0.2 mmol/L to 33.3 mmol/L could be measured by the Accu-Chek device. EDTA-preserved blood was analysed at SLU for total and differential white blood cell counts, and haematology (haemoglobin [Hb], red blood cell concentration [RBC], erythrocyte volume fraction [EVF], mean corpuscular volume [MCV], mean corpuscular haemoglobin concentration [MCHC], reticulocytes, morphology) was performed with an electronic cell counter validated for porcine blood (Advia 2120, Siemens, Erlangen, Germany). The serum samples were analysed for enzyme activities of aspartate amino transferase (ASAT), alanine amino transferase (ALAT), γ-glutamyltransferase (GT) and glutamate dehydrogenase (GLDH) and creatinine (CREA), total bilirubin, magnesium (Mg), sodium (Na), potassium (K) and chloride (Cl) at SLU using automated equipment (Architect C4000, Abott Diagnostics, North Ryde, Australia).
C-peptide, NEFA, TG and ketone body measurements
Serum C-peptide concentration was determined at the Department of Immunology, Genetics and Pathology, Uppsala University, Sweden using Porcine C-peptide ELISA (Mercodia, Uppsala, Sweden). Serum NEFA concentrations were measured at SLU using a 96-well Serum/Plasma Fatty Acid Kit Non-Esterified Fatty Acids Detection 100 point kit (Zen-bio, Durham, NC, USA). Serum TG concentrations were analysed at SLU using automated equipment (EKTACHEM DT60II; Medinor, Stockholm, Sweden) capable of measuring TG concentrations ranging from 0.17 mmol/L to 4.52 mmol/L. Blood ketone bodies concentrations were measured with test strips (FeeStyle Precision β-ketone, Abbot/ADC, Chicago, IL, USA) when clinically indicated and at the time of euthanasia.
Amino acid assay
The free amino acids and the amino acid-like compound taurine were measured in serum samples after precipitation of the proteins with a 1:10 dilution of 5% trichloroacetic acid followed by centrifugation at 2700 g. The supernatants were collected and stored at –80℃ until the assay was performed. The amino acid concentration (µmol/L) was measured with reverse-phase high-performance liquid chromatography using a 5 µm 150 × 3.9 mm C18 column (Syncronis C18 150 × 4.0 5 µm Syncronis Column; Västra Frölunda, Gothenburg, Sweden) according to the method of Pfeifer et al. 22 with some modifications.6,21
Statistical analyses
All values are expressed as mean ± SD and comparisons were made using repeated measures one-way analysis of variance (ANOVA). The statistical significance of differences was determined by Wilcoxon matched-pair signed rank test. P values <0.05 were considered to be statistically significant.
Results
The general appearance of all the pigs, except for No. 3, was unremarkable throughout the study. Before the onset of insulin treatment the diabetic pigs showed clinical signs of polyuria/polydipsia, and their daily weight gain (DWG) was suppressed and some pigs showed a weight loss. After the onset of insulin treatment, the pigs had a DWG of 532 ± 96 g. This corresponds to the weight gain of conventional high-health herd pigs of the same age, which is approximately 500 g (A Wallenbeck, SLU, personal communication). Pig No. 6, which did not receive any insulin treatment during the entire study period, had a DWG of 186 g, which corresponds to 37% of the weight gain of healthy conventional pigs.
All the animals recovered from the surgical procedure and STZ injection within two days. Pre-STZ blood glucose concentrations were within 2.2–7.6 mmol/L. In six out of eight pigs the nadir blood glucose concentrations (2.5–4.7 mmol/L) were recorded within 9 h post-STZ injection. In two pigs the lowest levels were recorded after 11 and 13 h post-STZ, with blood glucose values of 4.7 and 1.3 mmol/L, respectively. All the pigs showed high blood glucose concentration (>23 mmol/L) within 48 h post-STZ. One week after STZ, pig No. 3 was removed from the study due to postoperative complications. The indwelling catheter of pig No. 6 stopped functioning before the initiation of insulin treatment and it was decided that this pig would act as a non-treated control.
Post-mortem examinations of insulin-treated pigs revealed a good body condition including normal amounts of muscle mass and fat deposition in all the animals. Signs of subacute thrombophlebitis in the vena jugularis dexter were observed in three pigs. Post-mortem examination of pig No. 6 revealed skeletal muscle wasting, subcutaneous fat reduced to a minimum, and with no visible fat reserves in the abdominal cavity.
Blood analyses
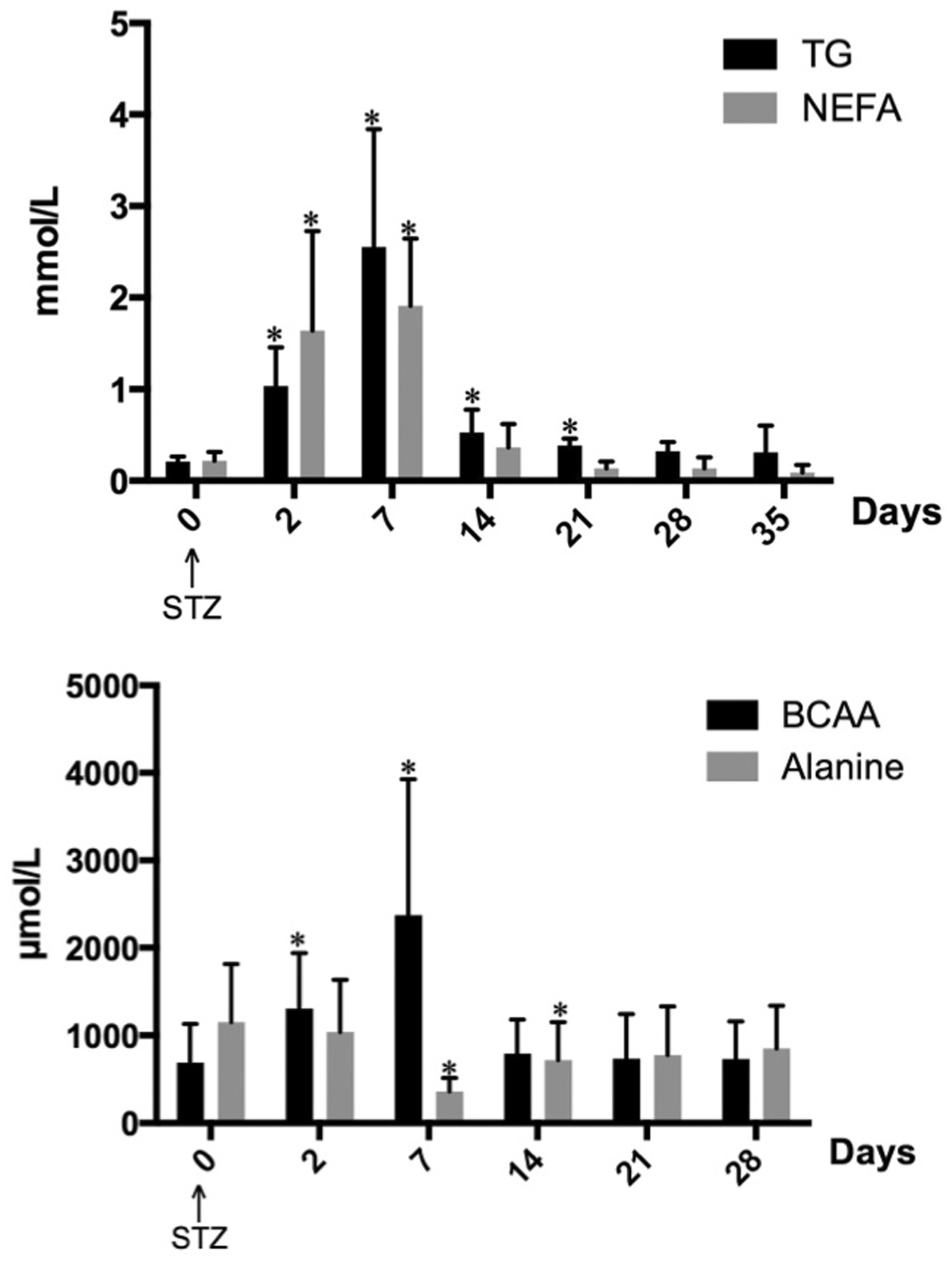
During the first week post-STZ injection, when the pigs did not receive any insulin treatment, fasting blood glucose concentrations were higher than 17 mmol/L in all of the pigs; i.e. they were hyperglycaemic. After initiation of insulin treatment, blood glucose concentrations were somewhat lowered and after nine days of treatment (with an insulin dose of 1 IU/kg BW daily) the fasting blood glucose concentrations ranged between 4 and 14 mmol/L. Even though the pigs responded to insulin treatment, fasting blood glucose concentrations were higher compared with fasting values before STZ treatment (Figure 2). As an illustration of how blood glucose concentrations were lowered over time with insulin treatment, fasting blood glucose concentrations of pig No. 4 are presented in Figure 3.
Blood glucose concentration (mean ± SD) in pigs Nos. 1, 2, 3, 4, 5, 7, 8 after fasting overnight, the insulin treatment started one week after streptozotocin (STZ) injection. *Indicates a significant difference (P < 0.05) from before (STZ) treatment. Blood glucose concentration of pig No 4 after an overnight fast, after the initiation of insulin treatment.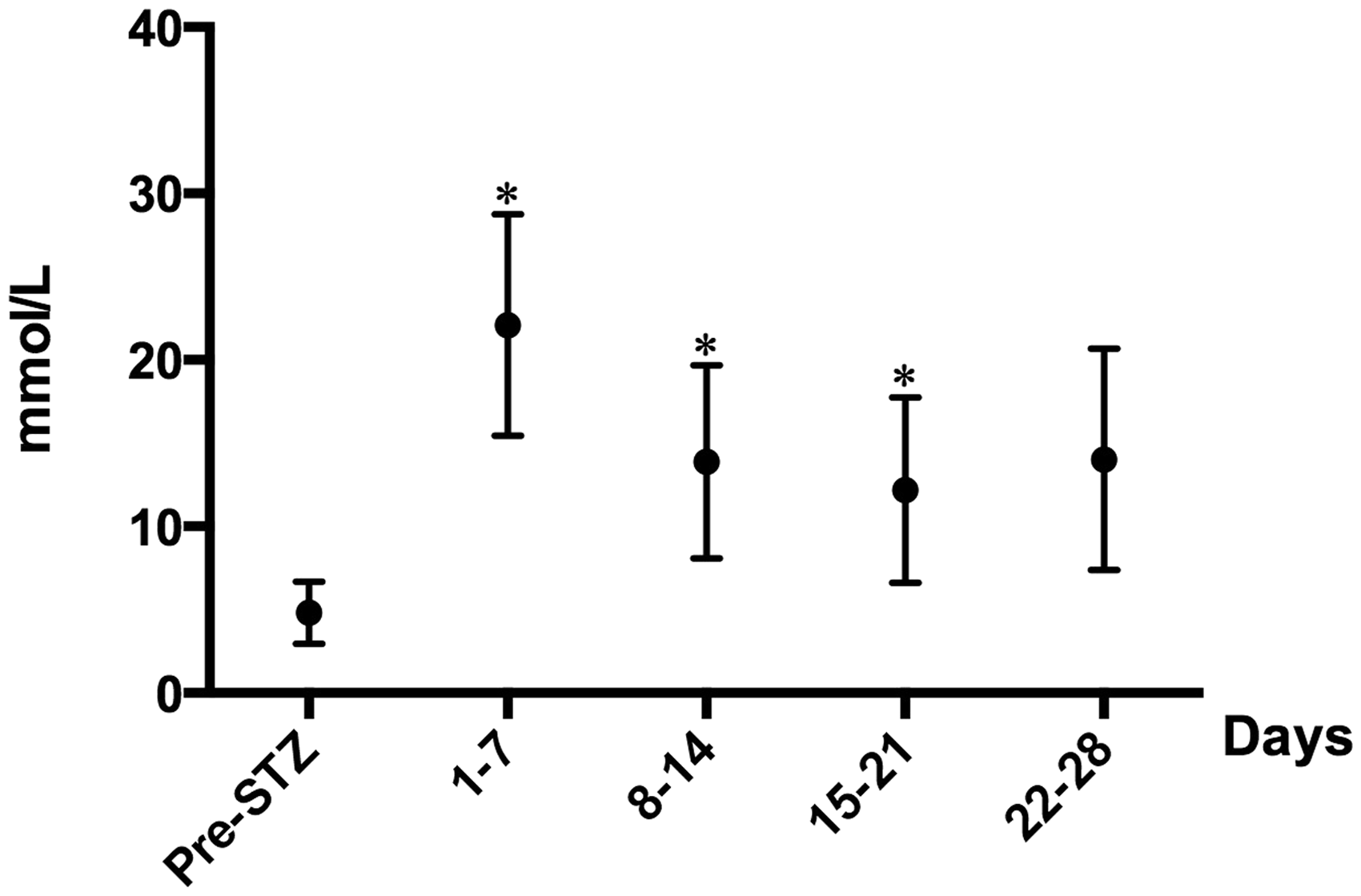
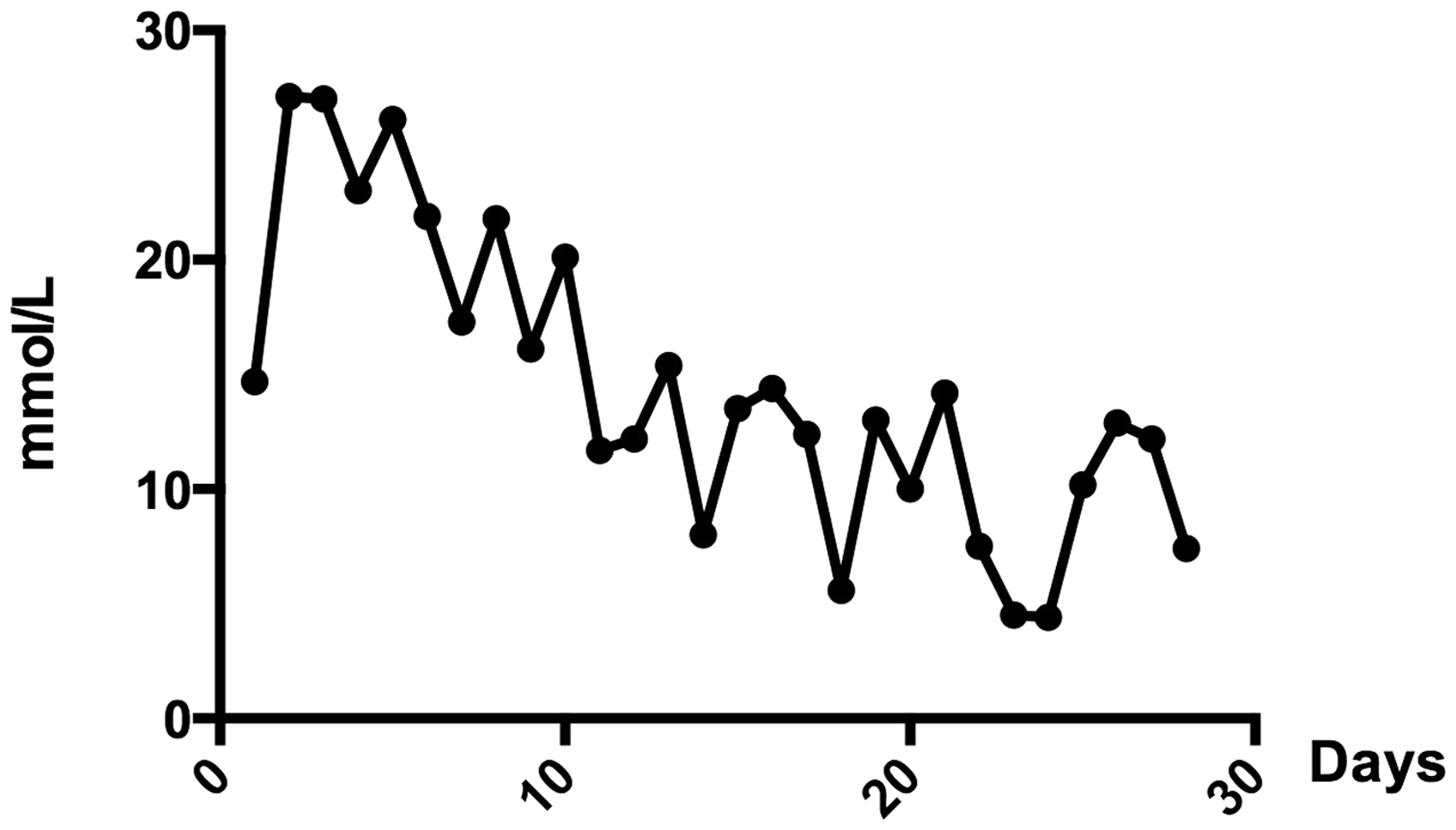
In each individual, blood glucose concentrations fluctuated considerably throughout the day with periods of both hyperglycaemia and hypoglycaemia. Hyperglycaemic peaks were seen 1.5 h after feeding when blood glucose concentrations were significantly higher compared with fasting values (P < 0.001; Figure 4). During the four weeks of insulin treatment, hypoglycaemia, with blood glucose concentrations <4 mmol/L, was detected on six sampling occasions. Pig No. 6, which did not receive any insulin treatment, had blood glucose concentrations above 24 mmol/L throughout the study period.
Blood glucose concentration (mean ± SD) in pigs Nos. 1, 2, 4, 5, 7, 8 before and 2 h after feeding, after the initiation of insulin treatment. *Indicates a significant difference (P < 0.05) from before feeding. Serum triglyceride (TG), non-esterified fatty acids (NEFA, upper panel), branched-chain amino acids (BCAA) and alanine (lower panel) concentrations in pig Nos. 1, 2, 3, 4, 5, 7, 8 before and after streptozotocin (STZ) treatment. From day 7 onwards, the animals were treated with insulin twice daily. *Indicates a significant difference (P < 0.05) and ***Indicates a significant difference (P < 0.001) from before streptozotocin treatment.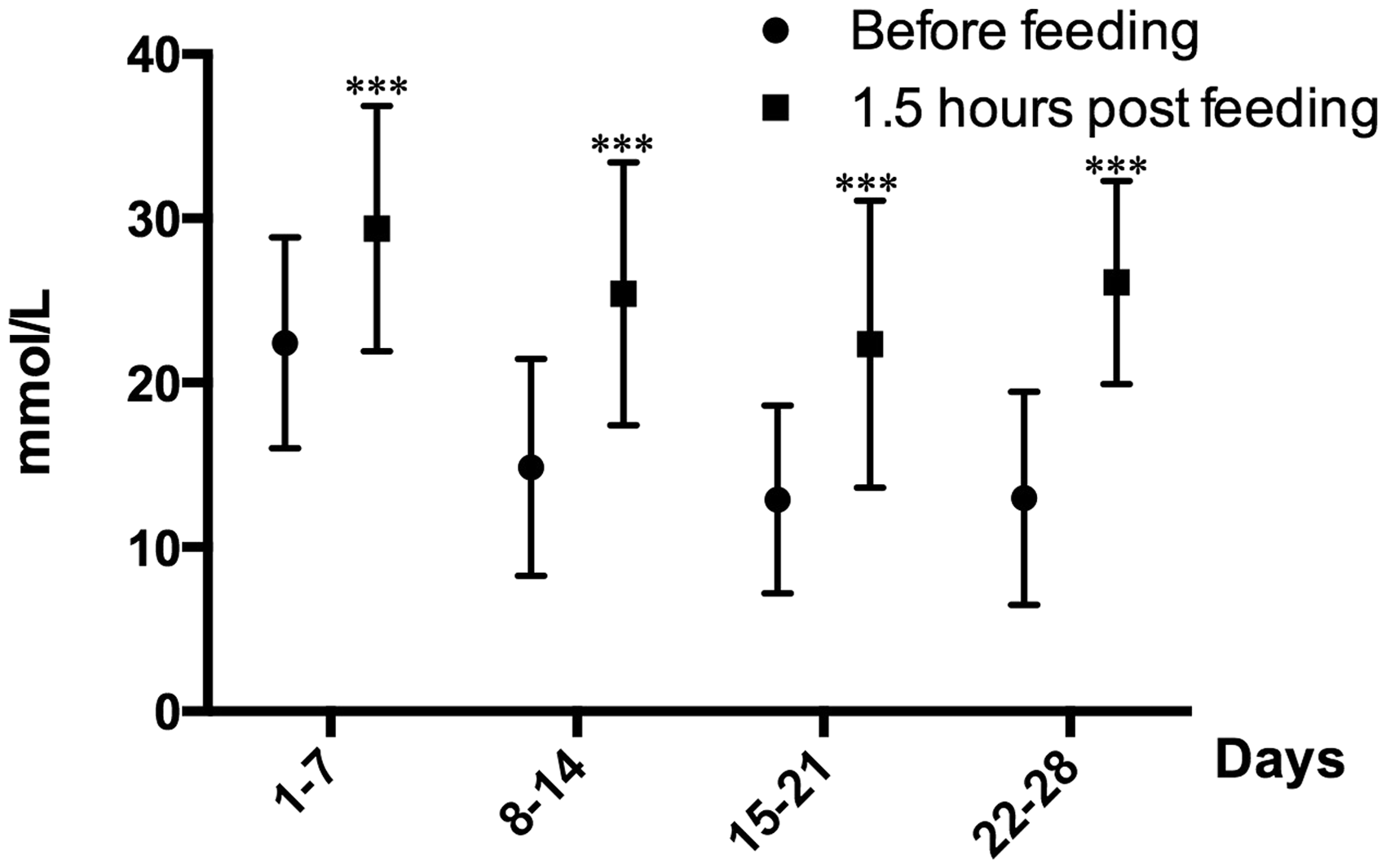
Hb, RBC, EVF, MCV, MCHC, reticulocyte particle count and erythrocyte morphology were within reference values in all the pigs on all sampling occasions throughout the study (data not shown). In five pigs, slight neutrophilia was seen on one sampling occasion, either at two or three weeks post-STZ. Other than that, total and differential white blood cell counts were within reference range for all the pigs during the study period.
Serum creatinine, electrolytes (Na, K, Cl, Mg) and total bilirubin were within reference ranges during the entire study period in all the pigs. In pig No. 6 a slight increase in serum GLDH activity (39 nkat/L) was seen at the time of euthanasia (five weeks post-STZ). The other enzyme activities (ALAT, GT) were within reference ranges on all sampling occasions for all the pigs. In pig No. 8 a seven-fold increase in ASAT was seen at the time of euthanasia (five weeks post-STZ). Other than that, ASAT was within the reference range on all sampling occasions for all the pigs.
C-peptide, NEFA, TG and ketone body measurements
Two days after STZ injection the serum C-peptide concentrations fell below 60 ρmol/L in all the pigs. Five out of eight pigs had serum C-peptide concentrations below the assay range, i.e. <20 ρmol/L, throughout the study period. In three pigs serum C-peptide concentrations ranged from <20 to 96 ρmol/L.
Serum NEFA concentrations before STZ treatment were 0.23 ± 0.09 mmol/L. Two days post-STZ treatment serum NEFA concentrations were significantly higher than before treatment. Fourteen days post-STZ treatment, i.e. seven days after the initiation of insulin treatment, there were no significant differences in serum NEFA concentrations before and after STZ treatment (Figure 5). In pig No. 6, which did not receive any insulin treatment, an elevation of serum NEFA concentration was seen on day 2 (1.4 mmol/L). The concentration was then decreased during the study, but was higher than the NEFA concentration of the insulin-treated pigs from day 14 onwards.
The mean serum TG concentration before STZ treatment was 0.22 ± 0.06 mmol/L. Two days post-STZ treatment, serum TG concentrations were significantly higher than before treatment, and peak TG values were observed seven days post-STZ treatment. Twenty-eight days post-STZ treatment, i.e. 21 days after the initiation of insulin treatment, there were no significant differences in serum TG concentration before and after STZ treatment (Figure 2). In pig No. 6, an elevation of serum TG concentration was seen on day 2. The concentration was somewhat lowered subsequently but remained higher than those of the insulin-treated pigs throughout the study.
Five days post-STZ treatment pig No. 5 showed inappetence. Blood ketone bodies were measured and indicated hyperketonaemia with a concentration of 4 mmol/L beta-hydroxybutyrate. Another STZ-treated pig was used as a reference and had a blood ketone concentration of 0.4 mmol/L. Pig No. 5 was given 2.5 IU of rapid-acting insulin (Actrapid®, 100 IU/mL; Novo Nordisk A/S, Bagsvaerd, Denmark) and treatment with Caninsulin® vet was initiated. Two days later, appetite and general appearance were normal.
At the time of euthanasia the mean blood ketone concentrations were 0.04 ± 0.05 mmol/L for all the pigs.
Amino acid assay
The total serum amino acid concentration did not change during the study period but a 2–3-fold increase in the BCAA levels were seen two and seven days after the STZ treatment. After one week on insulin treatment the concentration of BCAA was similar to that before the STZ injection (Figure 5). The alanine concentration fell after the induction of diabetes and serum values decreased significantly compared with those before the STZ treatment at seven and 14 days. Two weeks after the initiation of insulin treatment, there were no significant differences in serum alanine concentration before and after STZ treatment (Figure 5). The arginine concentration was significantly increased by 60% seven days after STZ, and taurine was decreased by 40%. At termination of the study (i.e. after four weeks of insulin treatment) there were no differences in serum amino acid composition compared to before the induction of diabetes. However, the serum concentration of alanine was decreased in pig No. 6 seven days post-STZ, and the concentration was somewhat increased during the study period but was lower than that of the insulin-treated pigs. Also, in this pig an increase in BCAA was seen on day 7, although the concentration subsequently decreased slightly but remained above that of the insulin-treated pigs throughout the study period.
Discussion
Diabetes mellitus was successfully induced in all the pigs by a single intravenous injection of STZ. All the pigs became hyperglycaemic and had serum C-peptide concentrations below 60 ρmol/L within 48 h after the STZ injection; values below 100 ρmol/L are seen in human patients with T1DM. It has been reported that insulin treatment and glycaemic control stimulates β-cell regeneration in STZ-diabetic mice. 23 In the present experiment the pigs remained diabetic throughout the study period with serum C-peptide concentrations below 100 ρmol/L even though the animals were treated with insulin and gained glycaemic control.
In the first week post-STZ injection the pigs did not receive any insulin treatment and were markedly hyperglycaemic. The ketonaemia observed in one pig five days post-STZ injection shows that ketone production in the liver can be accelerated in STZ-diabetic pigs, which is also the case for diabetic humans with untreated or poorly controlled T1DM. Diabetic ketoacidosis (DKA) develops in patients with severe insulin deficiency due to activation of hormone-sensitive lipase which causes breakdown of TG and release of NEFA into the circulation.13,24 The highest levels of plasma concentrations of NEFA are seen in patients with the most severe ketoacidosis. 16 Ketogenesis in the liver is stimulated by high glucagon levels25,26 and in DKA an exaggerated production of the ketone bodies β-hydroxybutyric acid and acetoacetic acid is seen causing arterial pH to decrease. In the present study, the pig which developed ketonaemia did not develop DKA since the ketonaemia was discovered at an early stage. This underscores the importance of frequent clinical examinations. The ketonaemia could be reversed by insulin treatment, similarly to the condition in human patients.
Apart from the incidence with the pig that developed ketonaemia, the general appearance of the pigs was unremarkable throughout the study period. After STZ injection the pigs showed polyuria/polydipsia, which was resolved by insulin treatment. Non-insulin-treated STZ-diabetic pigs in the present study showed low DWG, in accordance with previous observations by Jensen-Waern et al. 6 However, after the onset of insulin treatment, the DWG was at least as good as that of conventional high-health pigs.
Insulin treatment was initiated seven days post-STZ treatment. The dose was gradually increased to 1 IU/kg BW daily and blood glucose concentrations of 4–14 mmol/L were reached. The blood glucose concentrations fluctuated throughout the day with postprandial glycaemia (Figure 4). Hypoglycaemia was only measured on a few occasions and no additional treatment of these pigs was needed as none showed any clinical signs of hypoglycaemia. Since hypoglycaemia occasionally occurred it was not possible to give a higher dosage of short-acting insulin twice daily without the risk of inducing a life-threatening condition.
The STZ treatment produced alterations in the fat metabolism. A significant increase in serum concentrations of NEFA and TG were seen within two days and TG peak values were observed seven days post-STZ injection. This mimicked the fat metabolism of untreated-diabetic humans.16,27 Hypertriglyceridaemia and increased NEFA concentrations in the pigs were reversed by insulin treatment. There were no significant differences in serum NEFA and TG concentrations from days 7 and 21 after the initiation of insulin treatment, respectively, compared with the pre-STZ injection levels.
In humans, serum TG levels vary with age, from 0.3 ± 0.02 mmol/L to 1.1 ± 0.02 mmol/L. 28 In the present experiment, the serum TG concentrations seen in the pigs prior to the STZ injection and from day 21 after the initiation of insulin treatment correspond to those of healthy children and adolescents. At the time of euthanasia the pigs were about five months old, i.e. in the transition to sexual maturity.
Serum NEFA concentrations also vary with age in humans. Healthy adults have NEFA levels of approximately 0.5–0.6 mmol/L. 29 Young children, <8 years old, have higher NEFA levels. During the adolescent years, NEFA levels decrease and reach around 0.4 mmol/L at the age of 18 years. 30 In the present experiment, the serum NEFA concentrations of the pigs before STZ treatment and seven days after the initiation of insulin treatment correspond to the levels seen in healthy adult and older adolescent humans.
After the induction of diabetes BCAA were used as fuel, and glucose was formed through gluconeogenesis, mainly from alanine (Figure 5). These metabolic changes are also seen in humans during starvation or with untreated diabetes.8–10 The changes in amino acid metabolism were successfully restored by insulin to the levels seen before STZ treatment. The increase of arginine concentrations during the hyperglycaemic period is interesting. Arginine is an essential nutrient for T-lymphocytes but myeloid suppressor cells also use two enzymes involved in arginine metabolism to control T-cell responses. 31 Thus, arginine levels may have a role in the pathogenesis of diabetes. The taurine concentration fell one week after the induction of hyperglycaemia, which is in accordance with our previous results in STZ-induced diabetic pigs. 6 The magnitude of this decrease is similar to that described in human T1DM. 32 Rodents have a high biosynthetic capacity of taurine, whereas this capacity is very low in humans. 33 Thus, the porcine diabetes model has an advantage over rodent models in this respect. Taurine is involved in a number of biological actions, e.g. osmoregulation, antioxidation, and modulation of neurotransmitters. Beneficial effects of taurine supplementation on diabetes and diabetic complications have been reported but its clinical usefulness has to be further evaluated. 34
At an insulin dosage of 1 IU/kg BW the metabolisms of fat and amino acids were normalized and clinical signs of disease were resolved, i.e. it is a satisfactory treatment regimen. However, by dividing the insulin dose and feeding three times daily, the pigs might gain even better glycaemic control with less severe fluctuations throughout the day, which may be desirable in studies investigating the long-term effects of glycaemic control. Another approach would be to combine short-acting insulin with long-acting insulin. However, porcine long-acting insulin is unfortunately not readily available.
In conclusion, the metabolism in non- or insulin-treated Yorkshire × Landrace pigs with STZ-induced diabetes has been described in depth. Short-acting insulin at a dose of 1 IU/kg BW daily re-establishes the changes in carbohydrate, fat and amino acid metabolisms and no clinical signs of disease are observable after the onset of treatment. The fat metabolism is of particular interest as it has not been studied in insulin-treated diabetic pigs. The animal model should be valuable in preclinical islet transplantation research since purification of islets of Langerhans is cumbersome but islets from Landrace pigs are readily available.
The translation of animal data to humans relies on an evidence-based choice of animal models. In diabetes research many thousands of animals are used every year, thus well-documented models are of great value for both ethical and economic reasons.
Footnotes
Acknowledgements
Financial support by the Swedish Research Council FORMAS is gratefully acknowledged. Drs Bidda Rolin and Birgitta Andrén are acknowledged for valuable information on insulin treatment in pigs and Mr Leo Waern for skilful animal handling.
