Abstract
N-methyl-N-nitrosourea (MNU), a highly potent carginogen, is widely used to generate mammary tumours in murine species. In a model of MNU-induced mammary carcinogenesis using immature female Sprague-Dawley rats, large mammary tumours (largest dimension ≥0.5 cm) were obtained within a very short period of time. In addition, in the rats bearing MNU-induced mammary carcinomas, there were a number of tumours whose origins were not from mammary tissue but from several different tissues and from mammary non-epithelial tissue. The tumours were of mesenchymal or epithelial origin and they were located in the inguinal region. These tumours were diagnosed as fibroadenoma, combined tubular adenoma and fibroadenoma, hyperkeratotic papilloma, keratinous cyst and malignant peripheral nerve sheath tumour (MPNST) with smooth muscle differentiation. The occurrence of these other tumours in addition to the development of the mammary carcinomas may be attributed to a direct local effect of the intraperitoneal administration of MNU during the sexual development of the immature rats. In the MNU-induced mammary tumour model, coexistence of tumourigenesis in various non-mammary tissues should be considered an important factor that may interfere with experimental procedures and results and also the quality of life of the tumour-bearing animals.
Chemically-induced carcinogenesis in laboratory animals is widely used for conducting research on the biology of cancer. 1,2 In this regard, many chemical substances have been used to induce carcinogenesis in laboratory animals. One such chemical is N-methyl-N-nitrosourea (MNU), a highly potent carcinogen; and has been widely used for inducing carcinogenesis in murines. 3,4 The mechanism of its carcinogenic action is direct alkylation of DNA, thereby causing mutations. 5 Some strains of rats such as Sprague-Dawley, Long-Evans and Fischer 334 develop mammary gland tumours following exposure to MNU. 6
In animal models of chemically-induced tumourigenesis, the latency and the incidence of tumours vary according to the dose and the route of carcinogen administration. 3 In addition, age and stage of development are important factors for mammary tumourigenesis. 7,8 The tissue specificity of MNU depends on the age at which the development of mammary glands occurs. 9 Furthermore, after systemic exposure to the carcinogen, the animals may display different tissue tumourigenesis. 8,10
A large mammary tumour can provide many samples for numerous experimental analyses and can be used to study tumour progression. Therefore, we designed a study in which we proposed that large mammary tumours (diameter ≥0.5 cm) can be obtained in a short period of time following four once-weekly intraperitoneal injections of 50 mg/kg MNU to immature female Sprague-Dawley rats. This number and frequency of injections and dose were selected after careful analysis of the existing literature on the mechanism of action of MNU. 6,8
Material and methods
Animals
This project was approved by the Institutional Animal Care and Use Committee of Hacettepe University, Ankara, Turkey (approval number: 2004/34-5) before its commencement. Ten outbred, 21-day-old female Sprague-Dawley rats were obtained from the Experimental Animals Breeding Unit of Hacettepe University and housed in the Laboratory Animal Husbandry Facility, Department of Pharmacology, Faculty of Medicine, Hacettepe University. The rats were housed under environmentally controlled conditions (temperature 21°C; relative humidity 30–70%) with a 12/12 h light/dark cycle. The rats were provided with food pellets (standard pellet dairy chow, Dokuz Tug Yem Sanayii, Ankara, Turkey) and tap water ad libitum. The Guiding Principles in the Care and Use of Laboratory Animals together with those described in the Declaration of Helsinki were strictly adhered to in the conduct of all the experimental procedures. 11
Preparation and administration of the chemical carcinogen
A 10 mg/mL solution of MNU (Sigma, St Louis, MO, USA) was prepared by dissolving the chemical in physiological saline (0.9%, w/v). The pH of the solution was adjusted to 4.0 with hydrochloric acid (1 N HCl) (Carlo Erba, Milan, Italy) in order to increase the stability of the solution. The MNU solution was injected intraperitoneally (i.p.) at a dose of 50 mg/kg alternatively into the left and right regions of the peritoneal cavity when the rats were 21, 28, 35 and 42 days old.
Evaluation of the tumours
Rats bearing mammary tumours with the largest dimension, e.g. ≥0.5 cm, were sacrificed by cervical dislocation. If the animals showed signs of lameness, paralysis or weakness, they were pre-emptively euthanized. Animals were necropsied and tumours were photographed to document their location and gross morphology. The tumours were excised, fixed in 10% formalin and then embedded in paraffin. Histopathological evaluation of the paraffin sections was performed under conventional light microscopy after haematoxylin–eosin staining. 12
Immunohistochemical analysis
The origin of the intraperitoneal tumour was investigated immunohistochemically using a panel of antibody markers against desmin, smooth muscle actin (SMA), CD10, CD34, CD117, S-100 protein, vimentin and high- and low-molecular-weight cytokeratins (HMWCK and LMWCK). 13 The antibodies against desmin and SMA are used as basic markers for identifying smooth muscle tumours. Antibodies against CD10 are particularly useful for the diagnosis of endometrial stromal neoplasia. CD34 stains normal and neoplastic endothelial cells, as well as a variety of soft tissue tumours. In addition to being expressed on the cell membrane of various normal cells, CD117 is expressed in gastrointestinal stromal tumours. S-100 protein is present in the neoplastic cells of neuroectodermal origin and the main use of the antibody is in the evaluation of peripheral nerve sheath tumours. Vimentin is characteristic of cells of mesenchymal nature and is expressed in tumours of neural origin. Cytokeratins are excellent markers for epithelial differentiation. 13
The paraffin-embedded tissue sections (4 µm thick) were stained using primary antibodies against vimentin (clone V9, dilution 1/200), HMWCK (clone AE3, dilution 1/100), LMWCK (clone AE1, dilution 1/100), S-100 protein (clone 4C4.9, dilution 1/200), CD10 (clone 56C6, dilution 1/60), CD34 (clone QBend10, dilution 1/200), SMA (clone 1A4, dilution 1/300) and CD117 (clone Rpoly, dilution 1/400), all of which were obtained from Neomarkers (Fremont, CA, USA), and the antibody against desmin (clone DE-R-11, dilution 1/150), which was obtained from Novocastra (Newcastle, UK). Binding of the primary antibodies was detected with polyvalent secondary antibodies using a biotin/streptavidin/horseradish peroxidase (HRP) detection system that was used according to the manufacturer's protocol (UltraVision Polyvalent HRP-diaminobenzidine (DAB) chromogen; Lab Vision, Fremont, CA, USA). Antibody binding was visualized using DAB tetrahydrochloride substrate. Immunostaining was evaluated by the intensity of staining (weak, moderate and strong) and by staining pattern (<5%, rare; 5–50%, focal and >50%, diffuse).
Results
Following the injections of MNU, no signs of toxicity, namely no loss of hair or any obvious change in behaviour, were observed in the rats. Each rat developed at least one mammary tumour (0.5 cm diameter) 9–12 weeks after completing the last of the four MNU injections. Mammary tumours were located in the thoracic and/or abdominal and/or inguinal subcutaneous regions, and were diagnosed histopathologically as ductal carcinomas (data not shown).
In five rats, eight tumours of different morphologies that were not mammary carcinoma and that originated from tissues other than mammary tissue and also from mammary epithelium were found in various locations in the inguinal region (Table 1). There was no correlation between the number of mammary carcinomas and the coexistence of other lesions (data not shown). They were mostly found subcutaneously, intradermally, in the epidermis, and one was located in the intraperitoneal cavity. The intraperitoneal tumour was located in the left ovarian lodge of the peritoneal cavity and caused dislocation of the intestines and other abdominal organs. This animal had developed three different types of tumours: a non-neoplastic tumour, a benign neoplasm and a malignant neoplasm in the peritoneal cavity (Figures 1A and B).
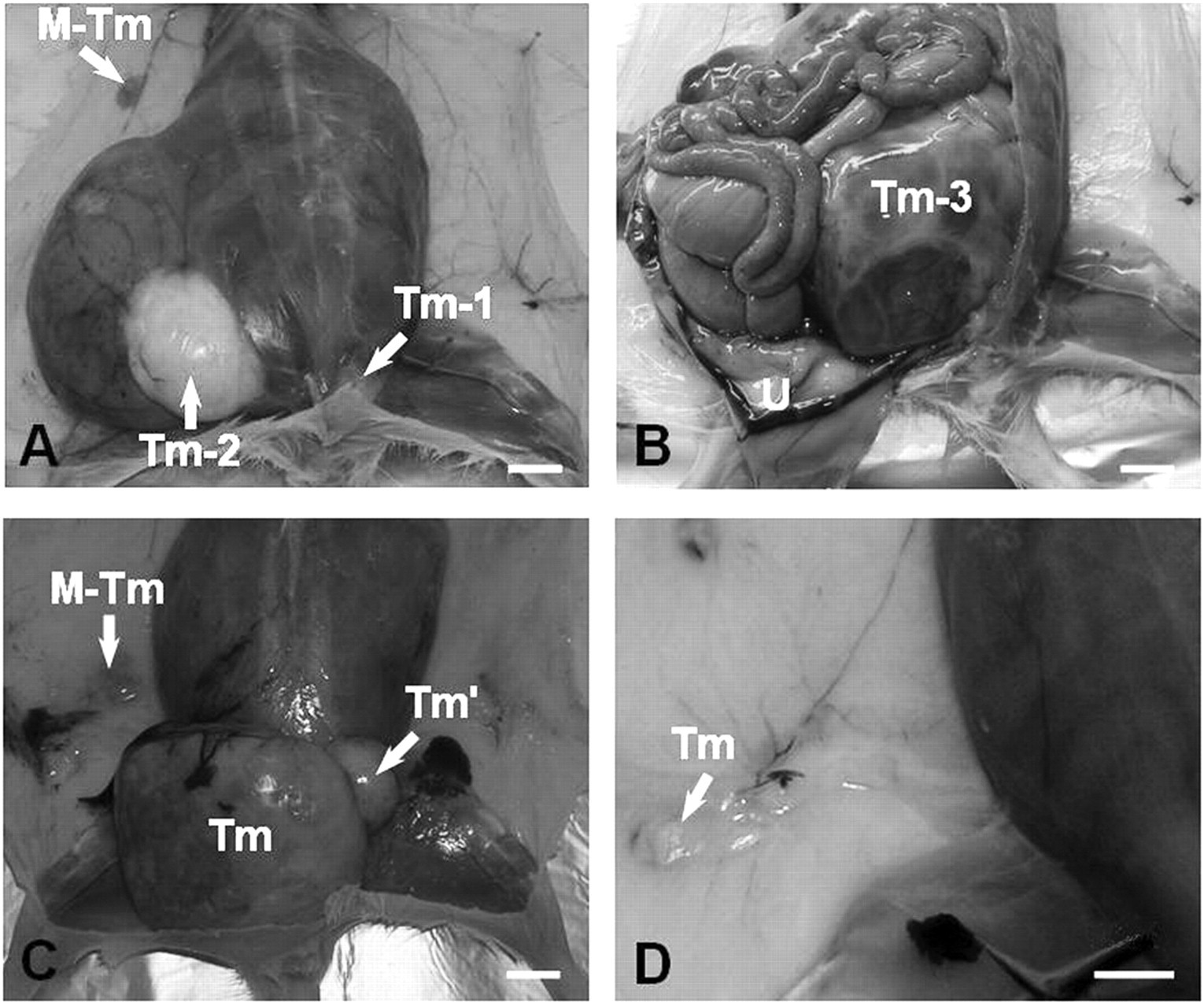
Location and gross morphology of the lesions detected in the mammary-carcinoma-bearing Sprague-Dawley rats, (bar, 1 cm). In addition to the development of the mammary tumour (M-Tm), one rat was found to have three tumours whose origins were from different tissues and were located (A) intradermally (Tm-1), subcutaneously (Tm-2) and (B) intraperitoneally (Tm-3) (U, uterus). (C) A bilobed (Tm and Tm') appearance of a large subcutaneous tumour mass was seen in one of the rats. (D) Small intradermal (Tm) tumours were observed in the inguinal region
Other tumours in addition to the mammary carcinomas found in N-methyl-N-nitrosourea injected immature Sprague-Dawley rats
s.c.: subcutaneous; i.p.: intraperitoneal; i.d.: intradermal; epid.: epidermal
*The largest dimension of the tumour
†§Tumours observed in one animal
We were able to categorize the non-mammary tumours into five classes according to their histopathology and location (Table 1). Except for the intraperitoneal tumour, all the lesions or tumours were benign. Those lesions located intradermally and in the epidermis were diagnosed as keratinous cysts (n = 3) and hyperkeratotic papillomas (n = 2), respectively (Figure 1D). Macroscopically, keratinous cysts could be distinguished from hyperkeratotic papillomas because they were capsulated cystic masses with greyish-white keratinous content, whereas hyperkeratotic papillomas were protruding skin lesions with a papillomatous appearance. Histopathologically, the cysts were epidermal in type and contained a distinct granular layer with numerous keratohyalin granules within the cyst wall (Figure 2A). Hyperkeratotic papillomas were elevated nodular lesions that were characterized by acanthosis, hyperkeratosis and papillomatosis (Figure 2B).
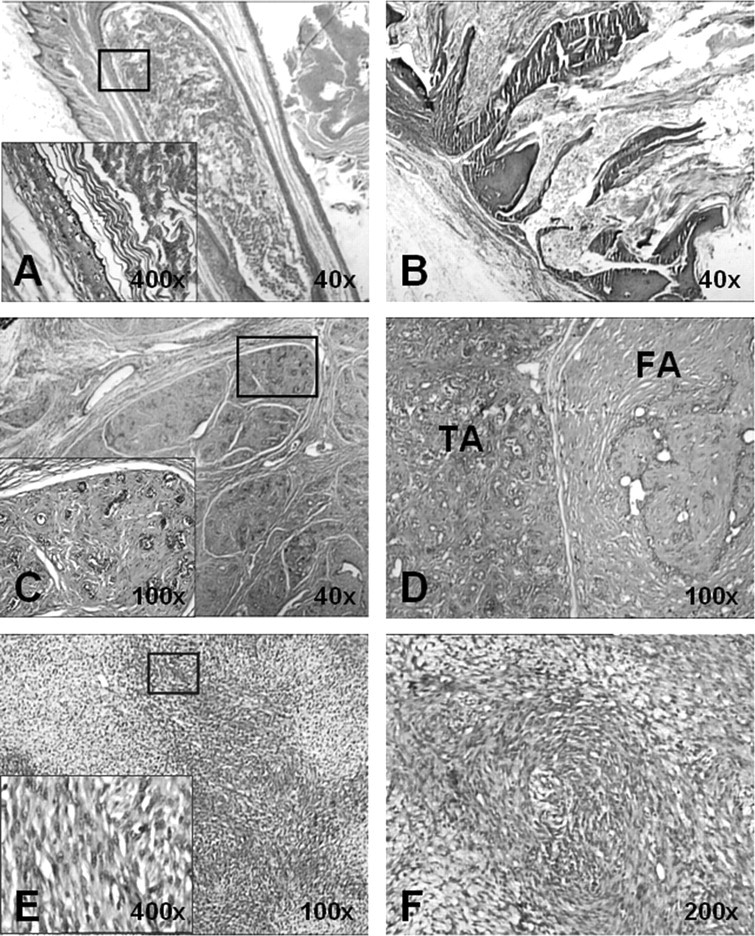
Histopathological evaluation of representative sections prepared from the tumours was done by haematoxylin–eosin (HE) staining. (A) Keratinous cyst of the epidermal type had a granular layer and numerous keratohyalin granules in the cyst wall (×40). Higher magnification of the area in the square is shown in the left-hand corner (×400). (B) A hyperkeratotic papilloma displaying an elevated nodular lesion (×40). (C) Fibroadenoma was easily distinguished with its stromal component and composed of loose connective tissue surrounding the ductal structures (×40). Higher magnification of the area in the square is shown in the left-hand corner, and the ductal structures (epithelial component) can be clearly seen (×100). (D) The combined tubular adenoma (TA) and fibroadenoma (FA) tissues is shown. The TA area was rich in closely packed tubules in the stroma, which consisted of loose connective tissue (×100). (E) The intraperitoneal tumour was organized into hypocellular and hypercellular areas (×100). The tumour cells have an atypical spindle shape and are shown at a higher magnification in the left-hand corner (×400). (F) The spindle cells in the hypercellular areas of the intraperitoneal tumour were arranged in whorls (×200)
The subcutaneous tumours developed as tubular adenomas or fibroadenomas. One such tumour was of a mixed type and contained tissues that were typical of tubular adenoma and fibroadenoma (Figures 1A and C). This tumour was diagnosed as a combined tubular adenoma and fibroadenoma. Macroscopically, the fibroadenoma was defined as a sharply demarcated firm mass whose cut surface displayed whorl-like structures with slit-like spaces. Histopathologically, the fibroadenoma was composed of epithelial and stromal components. The epithelial component consisted of ductal structures that were usually lined by myoepithelial cells and cuboidal or flattened epithelial cells. These ductal structures were surrounded and compressed by proliferating stroma that was composed of loose connective tissue (Figure 2C). In the combined tubular adenoma and fibroadenoma, particularly in the tubular adenoma component, the tubules were closely packed and lined by cuboidal or low columnar cells that contained round uniform nuclei, and that rested on a myoepithelial cell layer in the stroma formed of loose connective tissue (Figure 2D).
The intraperitoneal tumour was a large irregularly lobulated solid mass whose cut surface was greyish-white in colour, and which exposed areas of focal haemorrhage. On histopathological examination, the tumour was found to be that of an extremely cellular sarcomatous tumour with regions of hypocellularity (Figure 2E). The tumour was composed of hyperchromatic spindle cells that were arranged in whorls, mostly in the hypercellular areas (Figure 2F), and that had high mitotic activity (Figure 2E). The tumour also had areas of focal necrosis in the absence of significant pleomorphism. To determine the origin and type of this sarcomatous tumour, it was further analysed with immunohistochemical staining using a panel of antibodies. The tumour cells showed diffuse and strong cytoplasmic staining for vimentin and S-100 protein (Figures 3A and B), whereas their staining for SMA and desmin was moderate and focal (Figures 3C and D). Staining using the other markers, namely HMWCK, LMWCK, CD34, CD117 and CD10, was negative (data not shown). Based on its morphological features and the results of the immunohistochemical analysis, we concluded that this sarcomatous tumour was a malignant peripheral nerve sheath tumour (MPNST) with smooth muscle differentiation.
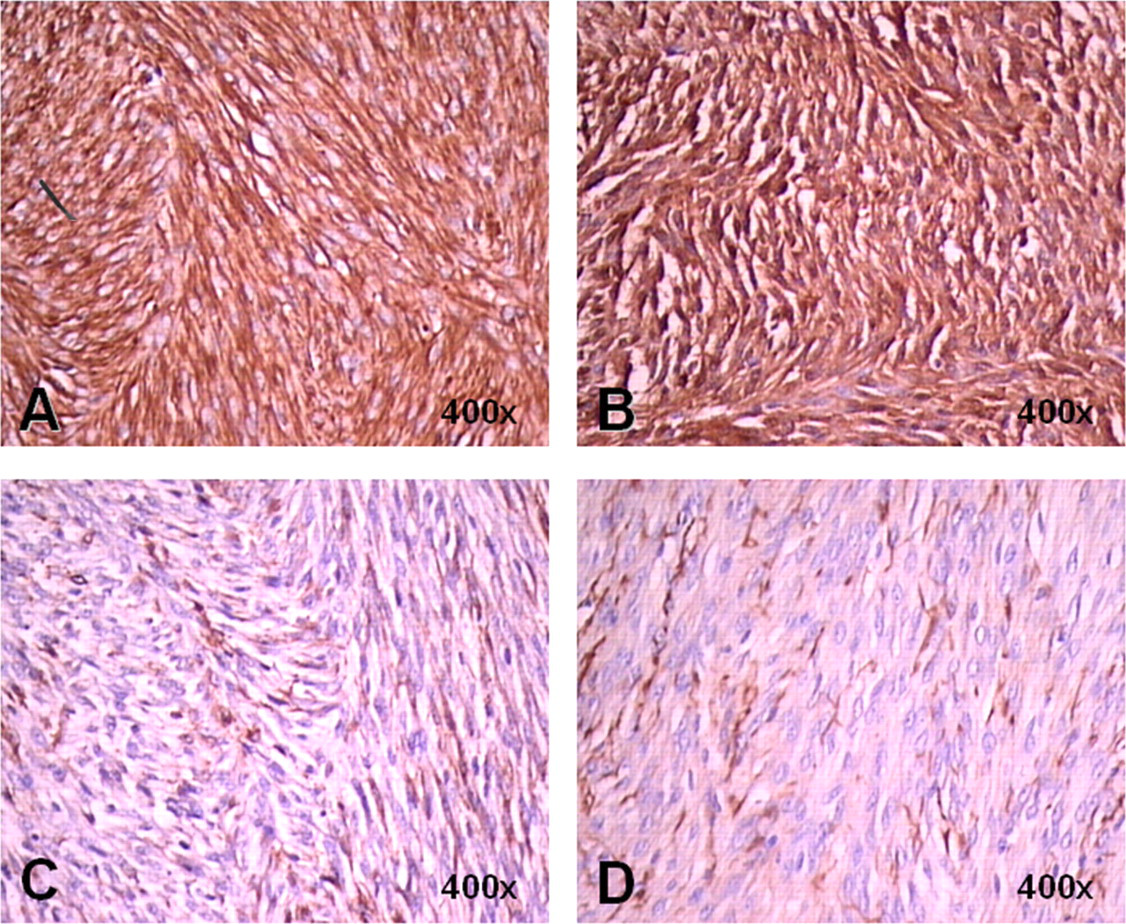
Differential diagnosis of the intraperitoneal tumour was performed by immunohistochemical staining using antibodies against (A) vimentin and (B) S-100 protein. The tumour showed diffuse and strong immunoreactivity to both markers (DAB, ×400). There were also focal areas with moderate positive staining using antibodies against (C) desmin and (D) SMA (DAB, ×400)
Discussion
Although the systemic administration of chemical carcinogens into laboratory animals may cause non-specific mutations, only certain tissues are selectively affected and transform into neoplasia. Sprague-Dawley rats are widely used in MNU-induced carcinogenesis studies to generate mammary tumours. Herein, we report the coexistence of different tissue tumourigenesis that may interfere with the outcome of experiments in a group of Sprague-Dawley rats that developed large mammary carcinomas following a series of injections of MNU.
N-methyl-N-nitrosourea has been administered to different murine species for inducing carcinogenesis in several organs, such as the ovary, 4 skin, 10 endometrium, 14 thymus 15 and colon. 16 In these studies, the resident cells were induced to proliferate if the target tissue was in a quiescent state, and the sensitized tissue was then exposed to MNU.
In our experiment, we proposed that immature 21-day-old Sprague-Dawley rats serially injected with MNU over 21–42 days could be used as a rapidly developing rodent model of mammary carcinogenesis. We proposed this method of inducing mammary carcinogenesis because the serial injection of MNU coincides with the hormone-mediated development of terminal end buds of mammary tissue. 17 MNU can be administered intravenously, subcutaneously or intraperitoneally as a single systemic dose. 3 The intraperitoneal route of administration is widely used as the standard procedure because of the ease of administration and reliability in inducing carcinogenesis. However, the inguinal location of the other non-mammary carcinomas may indicate a more potent carcinogenic effect when this route of administration is used.
In a previous study, Thompson et al. 18 reported that 60% of 21-day-old Sprague-Dawley rats, to which a single intraperitoneal injection of 50 mg/kg MNU was administered, developed ductal carcinomas with a diameter of ∼0.1 cm, 30 days later. On the other hand, Ariazi et al. 19 reported that 76% of immature Fischer 344 rats that underwent the identical treatment developed mammary carcinomas after 36 weeks and 17% of these tumours were diagnosed as fibroadenomas. However, Ariazi et al. did not report the coexistence of mammary carcinomas and fibroadenomas or any other neoplasms in the same animals.
Although MNU administration to immature or mature Sprague-Dawley rats has been reported to result in approximately the same incidence of mammary carcinomas, the use of immature rats and high doses of MNU may shorten the latency period. 18,20 In our study, we also observed shortening of the latency period.
Our finding that mammary-tumour-bearing rats can develop various neoplasms or non-neoplastic tumours indicates a drawback of this model. These tumours, such as fibroadenomas, originated from mesenchymal or epithelial tissues or both, i.e. MNU targeting of these tissues may be attributed to the immaturity of the rats. If MNU administration commences when the rats are 21 days old and are nearing puberty, 3,17 the genome of proliferating mesenchymal cells may be more accessible to MNU alkylation than the cells in quiescent state. In a previous study, transplacental exposure of MNU caused tumourigenesis in various organs, which includes mesenchymal tissue during fetal development. 8 On the other hand, the administration of MNU to mature rats may decrease the incidence of fibroadenoma. 19
The occurrence of a combined tubular adenoma and fibroadenoma and an MPNST is rare in laboratory rats, as well as in MNU-induced tumour models. 6,21 On the other hand, Bosland et al. 22 have reported that Sprague-Dawley rats are not susceptible to MNU-induced skin tumours. In our study, we found that the administration of MNU to immature Sprague-Dawley rats resulted in a high frequency of skin lesions.
The diffuse and strong staining of the neoplastic cells of the sarcomatous tumour in the peritoneal cavity for vimentin and S-100 protein indicated that this tumour was of mesenchymal and neuronal origin. Positivity for these two markers has been used to diagnose the MPNST in Fischer 334 rats. 21 Focal expression of desmin and SMA indicated that smooth muscle differentiation was taking place in the cells of this tumour. This phenomenon is also observed in tumours derived from a primary culture of rat MPNST cells 21 and in human MPNSTs. 23,24 The cells of this tumour were negative for the other markers (CD10, CD34, CD117, HMWCK and LMWCK), all of which were used for diagnosing spindle cell tumours. 13
The systemic administration of MNU may target all tissues, and MNU may selectively initiate transformation of those tissues in which differentiation is an ongoing process, or whose DNA repair mechanism is ineffective. In our MNU-induced carcinogenesis rat model, rapid development of mammary carcinomas is associated with the simultaneous development of tumourigenesis in various non-mammary tissues. We believe that this phenomenon may interfere with experimental procedures and results, and the quality of life of such tumour-bearing animals.
Footnotes
ACKNOWLEDGEMENTS
This study was supported by grants from the Eczacibasi Scientific Research and Award Fund and the Hacettepe University Scientific Research Unit (project no. 05DO3104001). We wish to thank Dr Sibel Percinel for her helpful supervision in the histopathological evaluations.
