Abstract
Previous studies have established that 7,12-dimethylbenz(a)anthracene (DMBA) can initiate skin tumourigenesis in conventional furred mouse models by acting on hair follicle stem cells. However, further cancer progression depends on repeated applications of tumour promoter agents. This study evaluated the timeline involved in skin tumourigenesis and progression in immunocompetent hairless SKH1-hr mice with dysfunctional hair follicles using only DMBA with no additional tumour promoter agents. The results showed that topical application of 30 µg (117 nmol) of DMBA over the back and flank regions of the mouse once a week and 15 µg (58.5 nmol) twice a week produced skin tumours after 7–8 weeks. However, by week 14 a heavy benign tumour load required the mice to be euthanized. Lowering the DMBA dose to 15 µg (58.5 nmol) once a week produced tumours more slowly and allowed the mice to be studied for a longer period to week 23. This low-dose DMBA regimen yielded a high percentage of malignant tumours (58.8%) after 23 weekly applications. Additionally DMBA-treated skin showed an increase in mean epidermal thickness in comparison to untreated and acetone-treated skin. Despite the aberrant hair follicles in SKH1-hr mice, this chemically driven skin cancer model in hairless mice can serve as a suitable alternative to the ultraviolet-induced skin cancer models and can be reliably replicated as demonstrated by both the pilot and main experiments.
In the past few decades, the global incidence of cutaneous squamous cell carcinoma (cSCC) has been on the rise.
1
The progression of cSCCs to terminal or morbid stages occurs – at least in part – because of genetic instability and a cumulative build up of irreversible genetic mutations in the skin. This sequential, multistage nature of skin cancer has been demonstrated in mouse skin cancer models using a two-stage chemical carcinogenesis protocol.
2
These stages involve:
Initiation: the first somatic irreversible mutation that is generally accomplished within two weeks following a single exposure to the ‘initiator’ agent. Promotion: this next stage requires repeated and chronic exposure lasting 10–40 weeks to a ‘promoter’, a non-mutagenic and hyperproliferation inducing agent, if prematurely discontinued tumours will not develop. Malignant progression: this stage of malignant transformation can be enhanced by an additional post-promotion application of an initiator agent.
3
Thus by emulating the tumour initiation and progression stages in mouse models of skin cancer, fundamental insight into the skin carcinogenesis process can be obtained. This could lead on to the potential discoveries for preventive or therapeutic interventions in cSCCs.
While furred mouse strains have commonly been used as skin cancer models, hairless mouse strains have proved to be more beneficial in skin cancer research,
4
mainly because hair depilation is not required, saving time and avoiding inflammatory skin reactions. The outbred albino SKH1-hr mouse strain is the most widely used hairless strain in dermatological research.
4
The popularity of the SKH1-hr mice for experimental carcinogenesis is predominantly due to:
The sensitivity of the mice. Skin tumours that resemble ultraviolet (UV)-induced cSCCs in humans at the morphological and molecular levels.
5
The readily observable non-pigmented skin facilitates the spotting of early cutaneous responses to carcinogens. SKH1-hr mice are euthymic and have a normal immune system that reacts to cSCC development in a manner comparable with humans, unlike athymic hairless mouse strains that are immunocompromised.
6
The two-stage chemical carcinogenesis protocol that mainly uses 7,12-dimethylbenz(a)anthracene (DMBA) as the initiator and 12-O-tetradecanoylphorbol-13-acetate (TPA) as the promoter, can produce skin tumours as early as six weeks with malignant transformation occurring at 18 weeks in furred SENCAR mouse strains.
7
However the efficiency and speed of the DMBA–TPA carcinogenesis varies highly between the mouse strains.
2
One limitation of the DMBA–TPA protocol is that it tends to produce predominantly benign papillomas and hyperplastic lesions that regress upon TPA discontinuation. On the other hand, studies have also shown that DMBA can serve as both the tumour initiator and promoter, i.e. a complete carcinogen.8,9 However this property of DMBA needs to be evaluated in SKH1-hr mouse strains for the following reasons:
Barring UV irradiation, no other agents have been evaluated as a complete carcinogen in skin for SKH1-hr mice.
5
DMBA initiates skin carcinogenesis by mainly inducing mutations in the quiescent stem cells
10
present in the hair follicle bulge region. Since SKH1-hr mice possess dysfunctional hair follicles, the tumourigenic response to DMBA may differ. Compared with immunocompromised hairless mice, the presence of fully functional innate immunity in SKH1-hr mice may also influence skin carcinogenesis in response to DMBA.
Due to these variability factors associated with SKH1-hr mice, the timeline involved in skin tumourigenesis and progression in response to DMBA need to be investigated.
In this experiment, we aimed to study the timeline involved in skin tumourigenesis and malignant progression in SKH1-hr mouse strains in response to treatments with DMBA with no additional promoter agents, in order to establish a useful experimental skin cancer model. This was achieved by first evaluating the sensitivity of SKH1-hr mice to carcinogenesis by DMBA at different dosing regimens in a pilot study. The DMBA ‘dose of choice’ was then adopted for a main study wherein parameters such as tumour sizes, malignant conversion and epidermal proliferation were studied as a function of time, to understand how DMBA acts as a carcinogen in a hairless, immunocompetent mouse strain such as SKH1-hr.
Materials and methods
Animals
This experimental animal protocol was approved by the Animal Research Committee of Erasmus University, Rotterdam. Female albino SKH1-hr hairless mice aged 6–7 weeks were ordered in two sequential phases: for the pilot study (n = 25 mice); and for the main study (n = 56 mice). The mice were purchased from a colony that was certified ‘specific pathogen free (SPF)’ according to the Federation of European Laboratory Animal Science Associations (FELASA) guidelines (Charles River Laboratories, Someren, The Netherlands). These mice were housed under SPF conditions at Erasmus Medical Centre Animal House. The mice were given an acclimatization period of two weeks upon arrival at the animal house.
Mice were housed in type 1 Blueline cages (Tecniplast, Milan, Italy) with 3–4 mice per cage. The cage was lined with Lignocel BK 8/15 bedding (Tecnilab-BMI BV, Someren, The Netherlands) with a thickness layer of 1.5 cm. Tissues were provided in each cage as enrichment. Animal cages and beddings were changed every week according to the standard protocols. The mice were provided with a 12 h light/dark cycle (08:00/20:00 h in the summer and 07:00/19:00 h in the winter). Temperature (23 ± 1℃) and humidity (55 ± 10%) were monitored daily. Standard food pellets (Hope Farms BV, Woerden, The Netherlands) and acidified water were provided ad libitum during the entire study period. All procedures were carried out with care to avoid and minimize animal discomfort.
Pilot study
Twenty-five female albino SKH1-hr mice were randomly and equally distributed over five groups. The first two groups served as controls: untreated mice and mice treated topically with acetone once a week. In the third and fourth group, mice were treated once a week with a dose of 15 µg (58.5 nmol) and 30 µg (117 nmol) of DMBA (Sigma Aldrich Chemie, Zwijndrecht, The Netherlands), respectively. The mice in the fifth group were treated with 15 µg of DMBA twice a week. The topical application on mice was performed inside a Class II, type B2 biological safety hood (Nuaire, Plymouth, USA). The floor of the hood was covered with absorbent pads and lined with masking tape. The topical reagent mixtures used in the experiment comprised of acetone solution (control), and DMBA dissolved in acetone at concentrations of 0.15 mg/mL and 0.30 mg/mL. For topical application of the reagent, each mouse was taken from its cage and gently restrained by its tail. A 20–200 µL pipette was then used to apply 100 µL of the reagent mixture evenly over the back and flank regions of the mouse. The area painted with the reagent was approximately 30 cm2 which included a rectangular region of 5 cm (length) × 3 cm (width) = 15 cm2 on each side covering the flanks and the corresponding half of the back of the mouse. The mouse spine, shoulders, fore and hindlimbs were used as anatomical landmarks to keep the area painted with reagents constant between mice. Following the application, the mouse was further restrained for 15–20 s in order for the reagent mixture to be absorbed topically, after which the mouse was gently released into a new cage with fresh bedding. This step was repeated for the remaining mice, except for the control mice which were to remain untreated. The mice were examined every week after the first topical application, keeping track of body weight, skin changes, onset of tumour appearance, size and number of tumours. All tumours with a diameter greater than one millimetre were included for the total tumour count per mouse group at each time point. Increased tumour load (exceeding 2 cm3 per mouse), weight loss of more than 20% from the starting weight, signs of respiratory distress (rapid and shallow breathing) or circulatory failure (generalized swelling/oedema) were criteria for euthanasia. Euthanasia was performed by cervical dislocation of the mice which were anaesthetized with 5% isoflurane inhalation. After euthanasia, samples of dorsal skins and smaller tumours were collected using a 4 mm punch biopsy, while larger tumours were collected by simple excision. Haematoxylin and eosin (H&E) slides were obtained from these skin samples (as described in Section A of the supplementary material, see http://lan.sagepub.com) and evaluated histopathologically. The results of the tumour count in the different groups were computed statistically using two-sided Wilcoxon rank sum test, with P < 0.05 being considered significant.
Main study
After determining the optimal DMBA dosing regimen, 56 SKH1-hr hairless female mice were randomly divided into three groups: group 1 untreated, group 2 acetone-treated and group 3 treated with DMBA (dosage determined from the pilot study). After 4, 7, 11, 14, 17, 20 and 23 weeks of topical applications, two untreated, two acetone-treated and four DMBA-treated mice were sacrificed at each time point. Following euthanasia, tumours formed in the DMBA-treated mice at each time point were counted, and grouped by tumour diameter: 1–2 mm; 2–4 mm; and >4 mm. These values were then plotted against the number of weekly DMBA applications. As described in the pilot study, skin samples were collected, processed and sent for histopathological evaluation by a certified veterinarian pathologist. The lesions were histopathologically identified based on standardized diagnostic criteria and nomenclature for proliferative and non-proliferative lesions in the laboratory rodent skin. 11 Following diagnosis, the lesions were categorised into benign, premalignant and malignant lesions. Malignant lesions were further categorized as pre-existing benign lesions progressing to invasive carcinomas; or de novo carcinomas – lesions that were directly present as cSCCs in histopathology. The details involved in the histopathological classification of skin lesions and the diagnostic criteria involved are described in Section B of the supplementary material (see http://lan.sagepub.com).
In order to measure the epidermal thickness, H&E slides were digitally scanned using a Nanozoomer 2.0 HT instrument (Hamamatsu Photonics, Hamamatsu, Japan). Epidermal thickness was measured at a minimum of five random sites per tissue section from the digital scans of the H&E slides obtained from untreated, acetone-treated and DMBA-treated mouse skin using the NDP 2.0 software. The mean epidermal thickness was then compared between each group and studied with respect to weeks of DMBA application. Comparison of mean epidermal thickness between the different groups was statistically analysed using unpaired two-tailed Student’s t-test of unequal variance, with P < 0.05 being considered significant.
Results
Pilot study
The mice from groups 1 and 2 (untreated and acetone-treated) did not show any skin changes or tumour formation throughout the pilot study (as shown in Figure 1). The first DMBA-induced tumour was visible at week 7 for mice from group 5 (15 µg or 58.5 nmol DMBA/mice twice a week) and week 8 for mice from group 4 (30 µg or 117 nmol DMBA/mice once a week). All mice from groups 4 and 5 developed tumours by week 10. Mice from group 3 (15 µg or 58.5 nmol DMBA/mice once a week) were the last to develop skin tumours at week 10 and were all tumour-bearing by week 14. The total tumour counts for group 5 and group 4 were significantly higher than group 3 (P < 0.05, see Figure 1) until week 14. There were no significant differences observed in the rate of tumour formation between groups 4 and 5 (P = 0.49).
Graph showing mean number of skin tumours per mouse (tumours with diameters >1 mm) in the different groups at various time points (*P = 0.017 for group 5 compared with group 3 from week 7, **P = 0.039 for group 4 compared with group 3 from week 10, §euthanasia for mice from groups 4 and 5 after reaching the maximum allowed tumour load).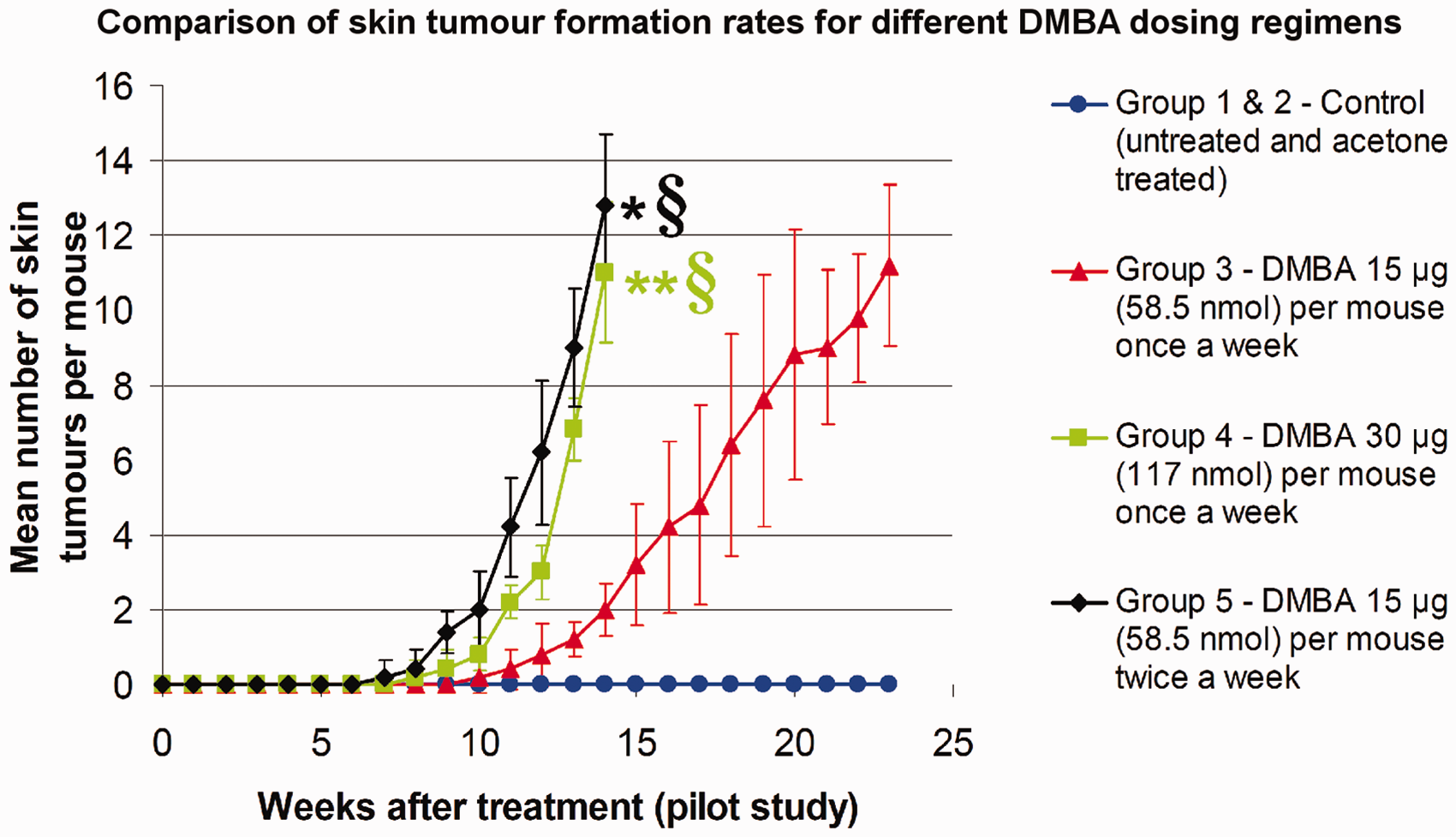
As seen in Figure 1, the tumour yield increased considerably for groups 4 and 5 leading to euthanasia by week 14. Due to the heavy tumour yield, the mice from these groups could not be followed beyond week 14. Tumours sampled at this stage were found to be mainly benign papillomas, with no evident malignant lesions. Therefore the DMBA dosages used for groups 4 and 5 were considered to be unsuitable for the main study, as these dosages produced a heavy benign skin tumour load much too quickly for the mice. By contrast, mice from group 3 could be followed up to week 23 after which they were euthanized. Skin samples obtained at that time point comprised of both benign papillomas and malignant lesions such as cSCCs. Therefore this particular DMBA dose regimen allowed the mouse models to be followed up to the malignant transformation endpoint, without resulting in a heavy tumour load for the mice. Consequently weekly topical application of 15 µg (58.5 nmol) of DMBA per mouse was selected as the standard carcinogenesis protocol for the main study.
Main study
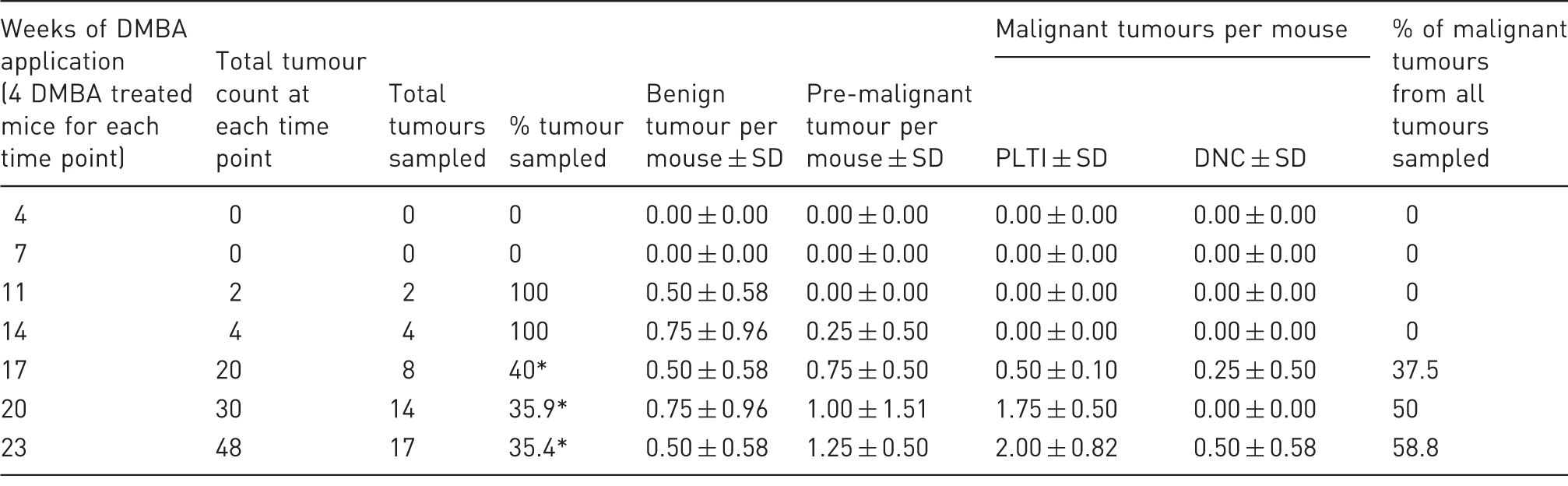
In the main study, mice were subjected to weekly topical application of 15 µg of DMBA per mouse as optimized in the pilot study. As seen in Figure 2a, tumours were first detected when they reached a size of >1 mm diameter at the 11th week of DMBA application. The total number of tumours counted at each time point for DMBA-treated mice increased with subsequent weeks of DMBA application. Majority of the tumours observed from week 11 to week 17 of DMBA applications measured 1–2 mm in diameter. From week 20 onwards, slightly bigger tumours with 2–4 mm diameters constituted the majority of the visible lesions. This trend was observed till the last time point of the study, i.e. week 23. Large tumours with >4 mm diameters initially developed at week 17 and then a gradual upward trend with subsequent weeks of DMBA application. As seen in Table 1, all counted tumours were histopathologically evaluated at weeks 11 and 14. Due to the high tumour yield from week 17 to week 23, all tumours counted in mice could not be sampled and diagnosed. Therefore, where there were three or more tumours of a similar gross appearance and size, only one tumour was randomly sampled as a representative of these three tumours. Therefore percentages of tumours sampled and diagnosed were 40% at 17 weeks, 35.9% at 20 weeks and 35.4% at 23 weeks.
(a) Mean number of tumours per mouse according to tumour diameters in DMBA-treated mice at each time point of DMBA application. (b) Distribution of skin tumour diagnosed as benign, premalignant, malignant progression of pre-existing lesions and de novo malignant lesions (malignant tumours arising in normal skin without a pre-existing lesion) at each time point of DMBA application. SCC: squamous cell carcinoma. Total tumour counts and distribution of benign, premalignant and malignant tumours (consisting of pre-existing lesions turning invasive (PLTI) and de novo carcinomas (DNC)) per individual mouse. Where there were three or more tumours of similar gross appearance and size in a mouse, only one tumour was randomly sampled as a representative lesion in that particular mouse. SD: standard deviation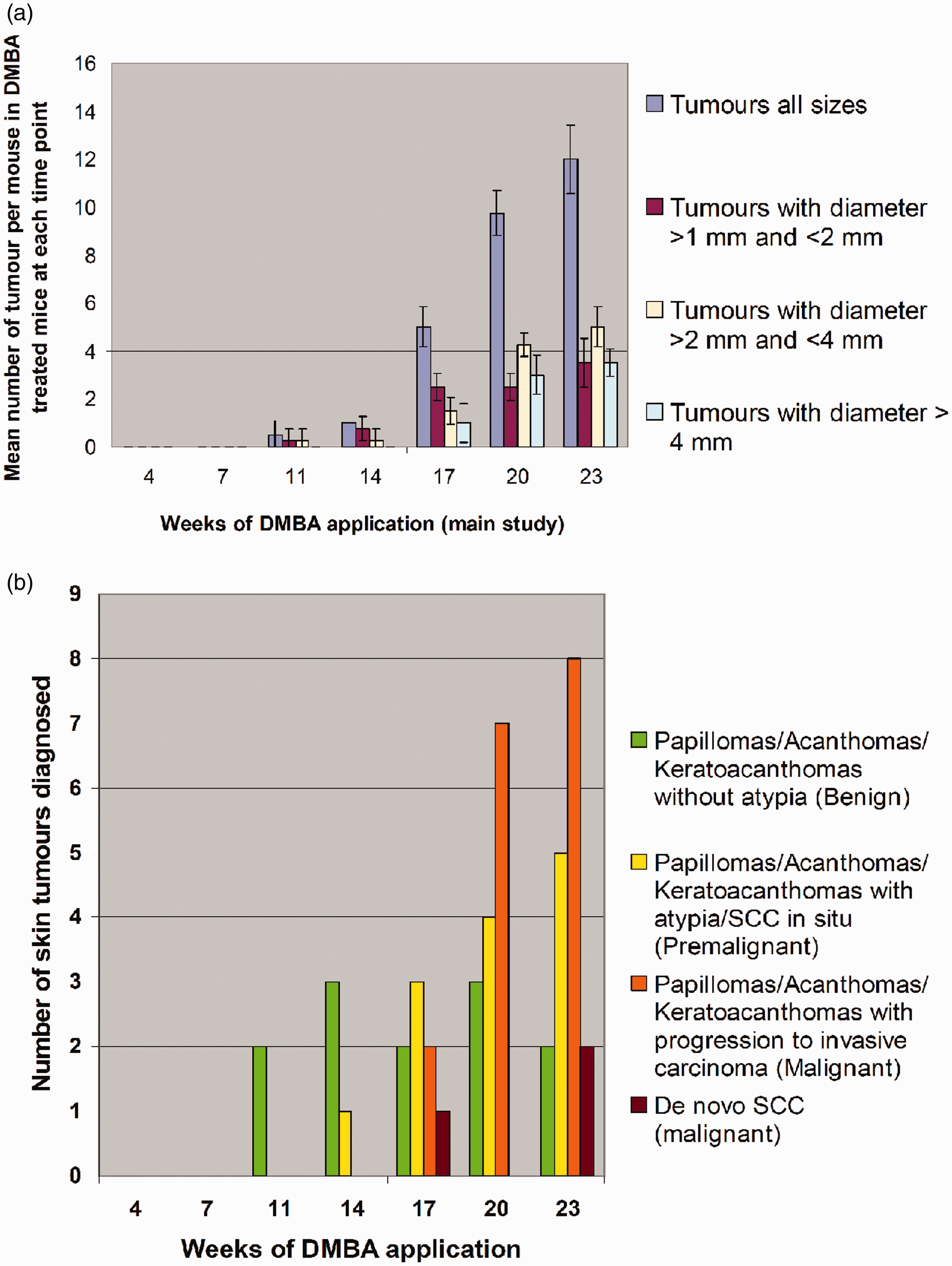
The histopathological evaluation of clinically normal skin obtained from DMBA-treated mice either showed (i) no changes when compared with control mouse skin (Figures 3a and 3b) or (ii) they exhibited mild to moderate epidermal hyperplasia (acanthosis) as seen in Figures 3c and 3d. Sebaceous hyperplasia was observed in untreated, acetone-treated and DMBA-treated mice as well, as this trait is characteristically observed in hairless mice such as SKH1-hr mice.
4
The epidermal hyperplasia observed in DMBA-treated skin was multi-focal in nature within the treated area and not diffuse. However epidermal hyperplasia seen at this stage was not associated with atypia. All tumours sampled at 11 weeks of DMBA application were benign and non-invasive in nature (Figure 2b and Table 1) and consisted mainly of benign papillomas (Figures 3e and 3f) and acanthomas with no atypia.
Haematoxylin and eosin (H&E) images of mouse skin samples – left and right columns corresponding to low and high magnifications, respectively; while black dashed rectangles indicate the region from where the higher magnification was obtained. The histopathological diagnoses were given as: (a,b) normal mouse skin, (c,d) mouse skin with hyperplasia, (e,f) benign papilloma without atypia, (g,h) squamous cell carcinoma in situ, (i,j) keratoacanthoma with progression to invasive carcinoma and (k,l) moderately differentiated cutaneous squamous cell carcinoma. Normal skin regions (a,b) were observed in controls (untreated or acetone-treated) and DMBA-treated mice, while the pathological regions (c to l) were seen only in DMBA-treated mice. Black arrowheads in 3j (invasive keratoacanthoma) shows islands of tumour cells infiltrating into the dermis indicating malignant transformation, while tumour cells are invading deeply in the subcutis in 3l (squamous cell carcinoma).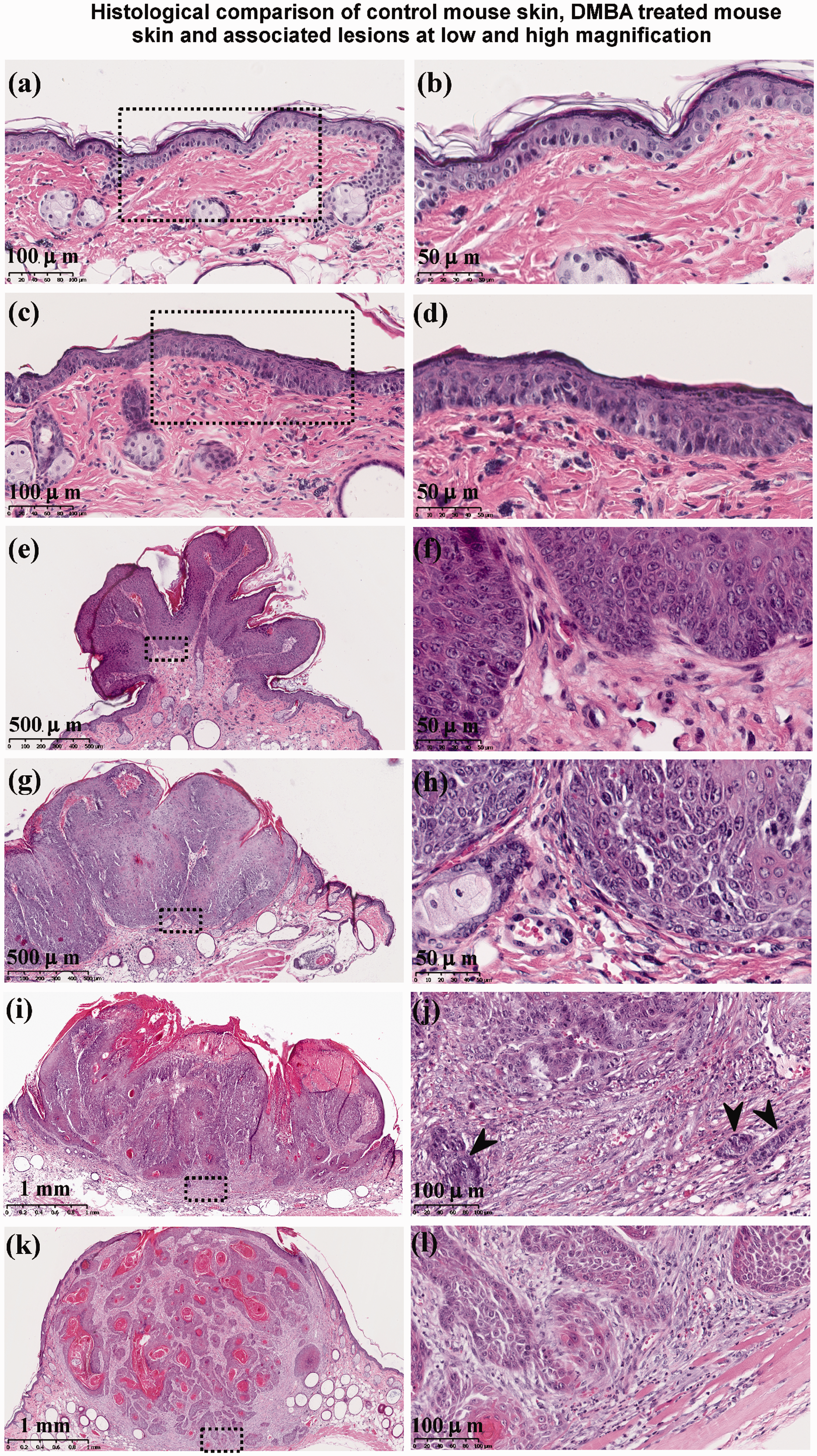
At 14 weeks, lesions are comprised of benign and premalignant skin tumours (Figure 2b and Table 1). The former included keratoacanthomas and papillomas with no atypia, while the latter included squamous cell carcinoma in situ (SCCIS) (Figures 3g and 3h), papillomas and acanthomas with atypia. Atypia is typically characterised by increased nucleo-cytoplasmic ratio; densely stained nuclei; increased variability in cellular and nuclear shapes and sizes; and increased mitotic figures. However until this time point, there was no invasion of the basement membrane or the dermis as clearly seen in Figures 3d, 3f and 3h. Malignant tumours were first observed only after 17 weeks of DMBA application, clearly exhibiting dermal invasion. These included keratoacanthomas (Figures 3i and 3j), acanthomas and papillomas progressing to invasive carcinomas or de novo cSCCs (Figures 3k and 3l). The results further show (as seen in Figure 2b and Table 1) that malignant tumours induced by DMBA in this animal model comprised mainly of pre-existing lesions progressing to invasive carcinomas while de novo carcinomas constituted a minority of the malignant lesions. However it is uncertain if all the de novo carcinomas truly arose without a precursor lesion, since it could not be clinically confirmed if the lesion was originally malignant or benign at the very onset as histopathological confirmation occurred only at later time points.
Mean epidermal thickness in clinically normal skin from control and DMBA-treated mice
The mean epidermal thickness was assessed in terms of weekly topical applications received by mice as shown in Figures 4(a–f). Mean epidermal thickness in control mice did not differ significantly from week 4 to week 23. In addition, there were no significant differences between the mean epidermal thickness in the two control groups of untreated mice and acetone-treated mice. In contrast to the control mice, clinically normal skin in DMBA-treated mice showed a gradual increase in epidermal thickness with successive weekly DMBA applications as seen in Figure 4g. It is notable that the mean epidermal thickness in DMBA-treated mice was significantly higher by about 40% compared with control mice after just four weekly DMBA applications. The increase was even higher at 92% and 135% after 11 and 23 weeks of DMBA applications, respectively.
Epidermis in clinically normal skin of SKH1-hr mice after 4, 11 and 23 weeks of treatment with acetone (a,c,e) and DMBA (b,d,f). (a,c,e) shows normal hairless mouse skin, (b,d) shows mild acanthosis and (f) shows moderate acanthosis. 4(g) shows the variation of mean epidermal thickness in time (weeks of topical application) for clinically normal untreated, acetone-treated and DMBA-treated mouse skin (* and ** signifies P < 0.05 for DMBA-treated skin compared with untreated and acetone-treated skin, respectively).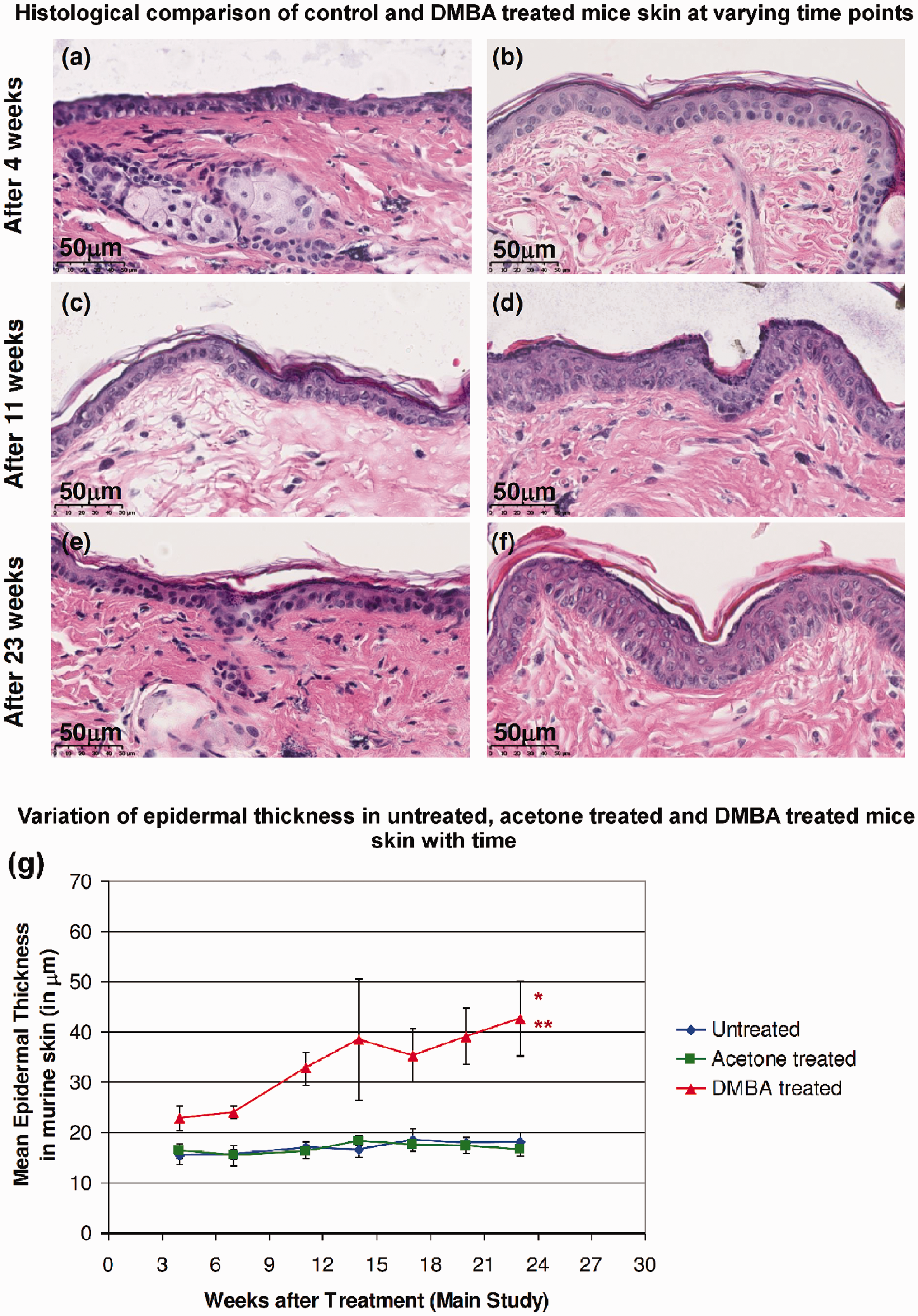
Discussion
Sensitivity of SKH1-hr mice to carcinogenesis by DMBA
The immunocompetent hairless SKH1-hr mice were highly prone to skin carcinogenesis with only DMBA applications in all three test groups as demonstrated in the pilot study. It may be that despite the scarcity of active hair follicles or the presence of abnormal hair follicles in SKH1-hr mice, DMBA can still initiate carcinogenesis from the interfollicular epidermis 2 or the rudimentary pilosebaceous appendages. 12 Giovanella et al. had earlier suggested that hairless mice were susceptible to chemical carcinogenesis, if treated with the carcinogen at a young age. 13 Their study implied that at a young age pilosebaceous appendages still retained functional epithelial remnants that could undergo carcinogenesis. This concurs with our findings as the SKH1-hr mice used in this study were initiated at the young age of 8–9 weeks (after two weeks of acclimatization when the mice were 6–7 weeks old), and were therefore susceptible to DMBA-induced carcinogenesis.
Higher frequencies of DMBA application increased skin tumour formation significantly in SKH1-hr mice, as shown in Figure 1. This finding implies that besides the carcinogen concentration, the frequency of carcinogen application has a major influence on tumour formation rates during chemical-induced skin carcinogenesis. On the other hand, no significant differences were observed between group 4 (30 µg of DMBA per mice once a week) and group 5 (15 µg of DMBA per mice twice a week) in this study as the total dose exposure of DMBA in groups 4 and 5 was the same, i.e. 420 µg of DMBA. This obviously implies that a weekly DMBA application at a particular dose is just as effective in tumour induction as a twice weekly DMBA application at half the dose. DMBA application protocols can thus be customized to minimize animal discomfort by limiting DMBA applications to just once a week, instead of twice weekly. However this finding differs from what was observed by Iversen et al. where a higher frequency of DMBA application at a constant dose led to an increased tumour incidence. 14 This may be attributed to the different dosing regimens adopted in that study.
Although higher dosing frequency of DMBA or higher DMBA concentration per application (as shown in Figure 1) produced tumours more quickly, these tumours were all benign. The mice were euthanized prematurely as a result of the heavy tumour load which excluded them for further study through to the later stages of malignant transformation. Therefore for an optimal skin cancer model with the ability to show all the stages of cancer progression, parameters such as carcinogen concentration per application, frequency of carcinogen application, and total carcinogen dose received by the animal need to be modulated accurately. A more suitable route to establish an experimental skin cancer model using SKH1-hr mice would be to adopt weekly DMBA applications at a lower concentration range of 10 to 15 µg of DMBA per mouse. However, the same protocol may need to be modified for other hairless mouse strains depending upon their sensitivity to DMBA.
High carcinoma yield in SKH1-hr mouse skin with repeated applications of only DMBA
The early visible tumours (1–2 mm diameter) that were formed after 11 weeks of DMBA applications consisted mainly of papillomas and acanthomas, which concurs with earlier studies of UV or DMBA–TPA-induced skin carcinogenesis.2,5 Malignant tumours were first observed after 17 weekly DMBA applications and 58.8% of the sampled tumours were diagnosed as malignant after 23 weekly DMBA applications as seen in Table 1. However it should be noted that although keratoacanthomas regularly progressed to cSCCs in mice, these lesions rarely become malignant in humans.
The results in this study show that tumour formation with weekly DMBA application in SKH1-hr mice was slower than that of UV carcinogenesis as demonstrated by de Gruijl et al. 15 However, this hairless immunocompetent skin cancer model relied only on low dose weekly DMBA applications and succeeded in producing a high malignant carcinoma yield (50% at 20 weeks and 58.8% at 23 weeks) which is comparable with UV carcinogenesis, 15 and also clearly higher than that produced by multistage chemical carcinogenesis using DMBA–TPA for the given time points. 16 This finding may be explained by the Iversen et al. study 8 where mice developed more carcinomas upon frequent applications with 10 nmol of DMBA when compared with those that received 10 nmol of TPA. The underlying mechanism hypothesized is that DMBA always acts as an initiator at low dose levels, but functions as a complete carcinogen at higher doses. 17 Other studies seem to indicate that repeated applications of DMBA induce chronic inflammation in the skin via mediators such as TNF-α, IL-1α and polymorphonuclear leucocytes,18,19 which is similar to the role of UV as a chronic inflammatory agent causing carcinogenesis. 20
Another notable finding observed in this study was that tumours categorized as malignant were predominantly benign tumours which became invasive rather than de novo cSCCs. As seen in Table 1, after 23 weeks of DMBA, the ratio of de novo cSCCs to benign lesions progressing to invasive carcinomas was 1:4. This observation suggests that malignant lesions induced by DMBA in this animal model arise predominantly from pre-existing benign lesions that undergo cumulative sequential mutations over time caused by chronic repetitive DMBA applications, although de novo cSCCs may arise occasionally. However this finding is limited and needs further validation as we were unable to determine the exact and absolute proportions between benign, premalignant and malignant tumours for the later time points, since all tumours could not be sampled from week 17 onwards due to increased tumour counts. Although since one tumour was sampled for every three or more tumours of a similar size and morphology, the results may still provide an insight into the relative proportions among benign, premalignant and malignant tumours. Furthermore it would be interesting to compare the rate of de novo cSCCs versus skin malignancies arising from pre-existing lesions between UV-irradiated and DMBA-only skin cancer mouse models. Future findings will be highly relevant in the fundamental understanding of how distinctively UV and DMBA function in triggering de novo cSCC formation without developing a precursor lesion. This may prove to be valuable in identifying potential therapy targets for treating de novo cSCCs which are more aggressive compared with skin malignancies arising from a pre-existing lesion. 21
DMBA induces epidermal hyperplasia in SKH1-hr mouse skin
Previous studies have reported epidermal hyperplasia induced by DMBA in the oral cavity of hamsters 22 and rat mammary tissues. 23 This study further confirmed that repeated application of only DMBA was equally capable of inducing epidermal hyperplasia in the unique skin type of SKH1-hr mice as well. DMBA-induced hyperplasia was observed in SKH1-hr mouse skin after only four weeks of DMBA applications and this was further enhanced after subsequent weekly applications. In a previous study Olsen et al. have observed early changes in cell division kinetics after low dose applications of DMBA on hairless mouse skin. 24 However these changes were transient and were not necessarily related to carcinogenicity, as the study was followed for up to three days only. A more detailed molecular insight is needed to understand how DMBA triggers epidermal proliferation via analysis of markers of cell proliferation such as Ki67; keratinocyte differentiation such as keratin 5 and 14; and inter-cellular adhesion such as e-cadherin.
DMBA-induced skin cancer model in mice as an alternative to skin cancer models induced by UV or DMBA–TPA
From this study, an experimental mouse skin cancer model has been established that can be reliably replicated for research related to diagnostic imaging, chemopreventive or chemotherapeutic studies. Additionally this model can demonstrate all the stages of skin carcinogenesis using low dose topical DMBA applications within 23 weeks, which is comparable with UV skin cancer models, and is faster than in DMBA–TPA skin cancer models. The pilot component in this study shows how DMBA itself (without the aid of tumour promoter agents) can successfully induce skin tumourigenesis and malignant progression in a hairless and immunocompetent mouse strain such as SKH1-hr. This eliminates the need for future pilot trials and thus reduces the number of mice required to establish cSCC models in SKH1-hr mice using chemical carcinogens. Furthermore, this skin cancer model has been established in a hairless and immunocompetent mouse strain making it more comparable with human subjects, unlike conventional hairy mice or nude immunocompromised mice.
The demonstrated protocol is simple and initiator/promoter applications can be easily performed in a conventional laminar flow cabinet. The model is a suitable alternative with comparable efficiency to UV skin cancer models. UV skin cancer models require installation of special UV lamps for mouse skin irradiation in the animal house, while the present established model only involves the purchase of DMBA and acetone which constitutes the carcinogen reagent mixture. In addition, due to the low dose weekly DMBA application, animal handling is restricted to just once a week which is less stressful for the mice compare with UV irradiation regimes which range from daily to thrice weekly. However it must be borne in mind that DMBA is extremely carcinogenic and thus extreme caution must be taken while handling DMBA or preparing DMBA solutions. Any surface or apparatus that comes in contact with DMBA mixture should be decontaminated by wiping with ethanol, while materials such as pipette tips, tissues and cage beddings of DMBA-treated mice should be disposed of as hazardous waste. 2
A deficiency encountered in this study was that SKH1-hr mice were followed through from the ages of 6–7 weeks, which roughly equates to a 13-year-old in human terms, 25 till the ages of 31–32 weeks (after two acclimatization weeks and 23 weekly DMBA applications) corresponding to a 20–22-year-old human. Since human skin tends to develop cSCC more quickly only after the age of 50 years, 26 the findings of this mouse model may not ideally translate to cSCC progression in aged human skin. Furthermore, the presence of aberrant hair follicles in SKH1-hr mouse complicates the extrapolation of findings in cSCC to human skin indicating the need for a more ‘human-like skin’ for translational research. Schmook et al. have suggested that pig skin resembles that of human skin and is therefore the most suitable model. 27 However, establishing an in vivo porcine cSCC model is complex and the timescale for observing skin tumour initiation and progression will be considerably longer. While the porcine model can be adopted for observing acute reactions to carcinogens like DMBA or UV, the hairless mouse skin is a more useful model for studying all the stages of skin carcinogenesis within a feasible timescale for translation to cancer diagnostics and therapeutics.
Conclusion
In this study a mouse skin cancer model with the complete spectrum of skin carcinogenesis was established in hairless and immunocompetent SKH1-hr mouse strains. This was accomplished using a simple carcinogenesis protocol that only utilized weekly low dose DMBA applications. The study also demonstrated that repeated DMBA applications can directly cause epidermal proliferation in mouse skin without using an additional tumour promoter. However additional studies are needed to understand how DMBA triggers epidermal changes as a tumour promoter agent. This developed skin cancer model would be extremely useful for the purposes of refining diagnostic imaging or improving chemotherapeutic and chemopreventive drug trials in skin cancer.
Footnotes
Acknowledgments
We are extremely indebted to Dr Pablo Secades and Dr Norman Sachs for their insight into developing the carcinogenesis protocol. We also appreciate Dr Daniela Salvatori, Dr Raoul Kuiper and Dr Senada Koljenović for their initial input into the histopathological assessment of the skin samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Dutch Technology Foundation (STW), which is part of The Netherlands Organisation for Scientific Research (NWO), and which is partly funded by the Ministry of Economic Affairs (Grant # UU 10322).
